Abstract
Titanium dioxide (TiO2) nanostructures have garnered significant attention for their exceptional photocatalytic and antibacterial properties in wastewater treatment applications. In this study, a simple, surfactant‐free hydrothermal method was developed to synthesize flower‐like and microrod‐like TiO2 architectures directly on fluorine‐doped tin oxide (FTO) substrates, aiming to enhance photocatalytic efficiency and antimicrobial performance. X‐ray diffraction (XRD) confirmed the formation of the anatase phase with distinct TiO2 and SnO2 crystallographic planes, indicating high phase purity and crystallinity. Field emission scanning electron microscopy (FESEM) revealed uniformly distributed microrods (diameter 40–60 nm, length 1–2 μm) forming three‐dimensional flower‐like assemblies, offering increased surface area and improved charge‐transport pathways. Fourier transform infrared (FTIR) spectra identified Ti–O–Ti and Ti–OH functional groups, crucial for hydroxyl radical formation and interfacial charge transfer. The antibacterial activity, evaluated using the agar well diffusion method against Escherichia coli, Staphylococcus epidermidis, Proteus vulgaris, and methicillin‐resistant Staphylococcus aureus (MRSA), exhibited inhibition zones ranging from 12 to 18.5 mm, with MRSA showing the highest susceptibility. The antibacterial mechanism is attributed to the combined effects of reactive oxygen species (ROS) generation and surface‐induced membrane disruption. Photoluminescence (PL) analysis displayed a strong UV emission at 330 nm, confirming suppressed electron–hole recombination and enhanced charge separation efficiency. The hydrophilic nature of the TiO2 surface (contact angle ≈ 35°) facilitates efficient pollutant adsorption and self‐cleaning behavior. Overall, the study demonstrates that the hierarchical, morphology‐controlled TiO2 microrod structures synthesized via this economical and eco‐friendly hydrothermal route exhibit superior photocatalytic degradation (∼92% of methylene blue within 120 min) and potent antibacterial efficacy. Despite limited visible‐light activity, the findings open avenues for band‐gap engineering and hybridization with graphene, Ag, Cu, or ZnO to extend solar utilization. The developed TiO2 films hold strong potential as scalable, reusable, and sustainable photocatalysts for integrated wastewater purification and microbial disinfection technologies.
1. Introduction
Water is an essential resource for sustaining human life and is indispensable across various industrial domains, including electrical and electronics manufacturing, pharmaceuticals, and nutrition [1–3]. However, freshwater availability is diminishing due to prolonged droughts, rapid population growth, and competing demands from diverse sectors. Reports indicate that only 3% of Earth’s water is freshwater, underscoring the critical need for alternative water sources. Seawater, though abundant, has become a primary focus for drinking water production and industrial applications in numerous regions worldwide [4]. Wastewater treatment employs physical, chemical, and biological methods to recycle and recover usable water. However, with the exponential growth of technological advancements, traditional methods are insufficient to meet the rising demand for freshwater. Advanced treatment technologies, particularly those leveraging nanotechnology, have emerged as a transformative solution. Nanotechnology‐based approaches demonstrate remarkable adsorption capacities, largely attributed to the high surface‐area‐to‐volume ratio of nanomaterials [5]. Nanomaterials exhibit versatile applications across environmental detection, cosmetics, nutrition, and optoelectronics. Specific nanomaterials, including carbon nanotubes, zero‐valent nanoparticles, metal oxide nanoparticles, and nanocomposites, have shown significant potential in diverse wastewater ecosystems. Among these, metal oxide nanoparticles are highly effective in biological wastewater treatment due to their chemical stability, exceptional adsorption properties, photochemical activity, and antibacterial effects [6, 7]. Yang et al. [8] explored the fate and implications of various nanoparticles in wastewater treatment and anaerobic digestion. The study evaluated the effects of nano zinc oxide (ZnO), silver nanoparticles (AgNPs), nano zero‐valent iron, and nano titanium dioxide (TiO2) on anaerobic sludge digestion and wastewater processes. Similarly, Singh et al. [9] provided an in‐depth analysis of TiO2, ZnO, and Fe2O3 nanoparticles for wastewater treatment, highlighting their mechanisms and effectiveness. Mousa et al. [10] synthesized titania (TiO2) nanoparticles using plant extracts and chemical methods, achieving particle sizes of 15–20 nm. Photodegradation experiments demonstrated that plant‐extracted TiO2 exhibited superior photocatalytic efficiency compared to chemically synthesized counterparts, primarily due to variations in particle size influenced by the type of plant extract used. Keshari et al. [11] synthesized TiO2 nanoparticles via the sol–gel method, varying precursor conditions and annealing temperatures (300°C, 600°C, and 900°C). The resulting anatase‐phase nanoparticles exhibited crystallite sizes of 58–86 nm. Ultraviolet (UV) irradiation experiments revealed a reduction in methylene orange (MO) concentration with increased UV exposure, validating TiO2’s efficacy in photocatalytic water treatment. Navidpour et al. [12] developed TiO2 flower‐like structures on fluorine‐doped tin oxide (FTO) substrates using physical vapor deposition for photocatalytic applications. Their findings highlighted that film thickness, determined by temperature and process parameters, is critical for enhancing photocatalytic performance. The study recommended further research to optimize this method for industrial scalability. Hamed et al. [13] fabricated Ag‐doped TiO2 films on FTO substrates via a hydrothermal process for wastewater treatment. The optimal silver loading (1.0 wt%) improved photocatalytic activity by reducing the TiO2 band gap. This occurred through electron trapping, which suppressed electron–hole recombination and prolonged the photogenerated charge carriers’ lifetimes. Increasing silver content beyond this threshold resulted in diminished X‐ray diffraction (XRD) peak intensity and increased FWHM values, reflecting reduced crystallinity. Paumo et al. [14] conducted a comprehensive review of TiO2 nanoparticles under UV, visible, and infrared (IR) radiation. The study emphasized the use of anatase‐phase TiO2 for enhanced catalytic activity and recommended its application for the effective removal of emerging organic pollutants (EOPs) from industrial effluents and sewage. The research suggests that TiO2‐based systems could overcome the limitations of conventional water treatment technologies, offering sustainable and eco‐friendly solutions.
This work aims to rapidly synthesize and fabricate a flower‐like structure of TiO2 on the FTO substrate specifically for wastewater applications. The second section of the paper provides a detailed account of the ingredients and procedures used to synthesize TiO2 gel and create flower‐like structures on the FTO substrate. The results and discussion part in Section 3 covered the findings of the TiO2 flower‐like structures. The conclusion of the study is presented in Section 4, summarizing the whole project.
2. Hydrothermal Synthesis of Flower‐ and Microrod‐like Structures
2.1. Chemicals Required
The chemicals used are as follows: distilled water, hydrochloric acid (HCl [38%], SRL Chemicals Pvt Ltd.), and tetrabutyl orthotitanate ([97%] Ti (OCH2CH2CH2CH3)4, Sigma‐Aldrich).
2.2. Preparing the Substrate
During the first stage of preparation, the FTO substrate underwent a meticulous washing process using a soap solution, followed by further cleaning with distilled water. The substrate was then dried in a hot air oven at a temperature of 100°C for a duration of 30 min. Subsequently, the desiccated substrate was subjected to ultrasonication for a duration of 30 min in a sanitized beaker containing a mixture of acetone and ethanol to eliminate any extraneous contaminants. The ultimate substrate was once again dehydrated in a hot air oven for a duration of 1 h at a temperature of 150°C in order to eliminate any remaining solvent residues.
2.3. Hydrothermal Synthesis of Flower‐Like Microrod Structures
After ensuring the beaker is completely clean, it is carefully placed on a magnetic stirrer. 100 mL of distilled water is added to the beaker, followed by the gradual addition of 100 mL of HCl catalyst. After thorough mixing, 5 mL of tetrabutyl titanate (TBOT) precursor is introduced dropwise into the solution and stirred continuously for 15 min until it completely dissolves, as illustrated in Figure 1. The prepared solution is then transferred into a Teflon‐lined autoclave, and a precleaned FTO substrate is inserted with precision. The autoclave is sealed and maintained in a hot‐air oven at 200°C for 24 h under controlled conditions. After completion of the reaction, the system is allowed to cool naturally to room temperature. The FTO substrate is then removed, thoroughly rinsed with distilled water to remove unreacted residues, and dried in an oven at 150°C for 30 min to eliminate any residual moisture or solvent traces. Hydrothermal synthesis parameters such as precursor type, reaction temperature, and duration play a decisive role in achieving well‐defined TiO2 morphologies, including microrods and flower‐like structures. Lekphet et al. [15] demonstrated that surfactant‐assisted hydrothermal conditions enable precise control over TiO2 microshapes by tuning nucleation and growth kinetics. Zong et al. [16] showed that the formation of three‐dimensional flower‐like TiO2 architectures strongly depends on hydrothermal duration, influencing hierarchical assembly. Similarly, Mali et al. [17] reported a surfactant‐free TBOT‐based hydrothermal route, in which temperature variation induces a morphological transition from one‐dimensional nanorods to three‐dimensional flower‐like TiO2 structures. Therefore, the present choice of TBOT precursor, 200°C reaction temperature, and 24 h incubation is based on these established morphological‐control insights from the literature.

Hydrothermal synthesis of titania‐based flower‐like microrod structures.
2.4. Analysis of Flower‐Like Microrod Structures
The flower structures were analyzed after being dried at an oven temperature. Investigations were conducted on the crystal structure of the flowers using XRD (Rigaku, SmartLab). The functional bonds of the flower structures were studied using FTIR (PerkinElmer Spectrum), and the morphology was examined using FESEM (MIRA3 TESCAN). An investigation was conducted on the flower‐like structures to study their antibacterial properties. The bacteria used for the investigation included Escherichia coli, Staphylococcus epidermidis, Proteus vulgaris, and Enterococcus spp. Finally, the fluorescence activity of the flower‐like structures was examined using the Cary Eclipse fluorescence spectrophotometer by Varian.
3. Results and Discussion
Figure 2(a) shows the XRD analysis of the TiO2‐based flower‐like microrod structures. The analysis measures the intensity of X‐rays against the Bragg angle (2θ) and covers a range of 20–80° to investigate the structural properties.

XRD (a) and FTIR (b) spectra of titania (TiO2) flower‐like microrod structures.
(b)

Figure 2(a) illustrates the XRD patterns of TiO2 microrod formations with a flower‐like morphology on the FTO substrate. The investigation determined that the TiO2/FTO substrate consisted of SnO2 and TiO2. The sample contained a total of eight distinct peaks. These peaks corresponded to the orientation planes of (110), (101), (200), (211), (220), (310), (112), and (301) and were detected at Bragg angles of 26.71, 33.67, 38.74, 52.61, 54.34, 62.59, 63.45, and 65.45°, respectively. These values were determined based on the International Centre for Diffraction Data, ICDD (98‐003‐9177) [18, 19]. The existence of these peaks verifies that the FTO substrate has truly been covered with a layer of SnO2. Similarly, the peaks corresponding to TiO2 were detected at the crystallographic planes of (101), (200), and (220) with angles of 25.28°, 38.74°, and 54.04°, respectively. These findings are consistent with the anatase and tetragonal pattern, as specified by the ICDD (98‐017‐2914) [20, 21]. Given the lack of any noticeable peaks, it indicates that there are no contaminants present in the sample. Figure 2(b) displays the FTIR spectrum of the TiO2 microrod formations with a flower‐like morphology on the FTO substrate. The intense absorption band in the IR spectrum, ranging from 2250 to 3000 cm−1, is attributable to the stretching vibration of O‐H groups. This absorption is caused by the presence of water molecules during synthesis and the interaction of the surface area with organic additives, as well as the small particle size [22, 23]. There is another peak observed at a wavenumber of 1244 cm−1, which is associated with the stretching and symmetric vibration of Ti–OH bonds. The absorption band at 750 cm−1 is associated with the characteristic vibration of TiO2 [22, 24].
The morphological analysis of TiO2 flower‐like structures synthesized by a hydrothermal approach was investigated using field emission scanning electron microscopy (FESEM) and is shown in Figures 3(a), 3(c), and 3(d), along with the elemental analysis observed using energy dispersive X‐ray spectroscopy (EDAX) as an inset in Figure 3(b).

FESEM (a, c, and d) and EDAX (b) spectra of titania (TiO2) flower‐like microrod structures.
Figures 3(a), 3(c), and 3(d) present the FESEM micrographs of the hydrothermally synthesized flower‐like TiO2 microrod structures, obtained using 5 mL of titanium butoxide (TBOT) precursor. The images reveal the formation of well‐oriented, densely packed rod‐like microstructures radiating outward from central nucleation points to form a distinct flower‐like architecture. Each microrod exhibits a diameter of approximately 40–60 nm and a length of 1‐2 μm, consistent with the optimized precursor concentration and synthesis conditions. The uniform distribution and compactness of the rods across the FTO substrate demonstrate excellent film uniformity, adhesion, and structural integrity, with no observable microcracks or surface defects. The rods are predominantly aligned perpendicular to the substrate, showing minor angular variations among neighboring structures, indicative of anisotropic crystal growth under hydrothermal conditions. Such orientation enhances charge‐transport pathways and maximizes surface exposure for interfacial redox reactions. The high aspect ratio and hierarchical arrangement of the rods substantially increase the density of active sites, improving photon absorption, charge‐carrier separation, and surface reactivity key attributes for efficient photocatalytic and antibacterial performance. EDAX spectroscopy (Figure 3 (b)) confirmed the presence of only titanium (Ti) and oxygen (O), with atomic percentages corresponding to stoichiometric TiO2. The absence of any foreign elements verified the purity and compositional homogeneity of the synthesized films, underscoring the effectiveness of the single‐step hydrothermal synthesis in producing defect‐free, stoichiometrically balanced TiO2 layers. The formation of this flower‐like microrod architecture originates from a carefully optimized balance between precursor hydrolysis, condensation, and crystal growth kinetics during the hydrothermal process. Using 5 mL of TBOT under acidic conditions (HCl catalyst) at 200°C for 24 h enables a controlled hydrolysis rate of Ti (OH)4, favoring anisotropic growth along the c‐axis of the anatase lattice. The FTO substrate acts as a heterogeneous nucleation platform that directs vertical rod growth and suppresses random aggregation. As the reaction progresses, variations in surface energy among growing crystallites drive self‐assembly into three‐dimensional flower‐like clusters, minimizing interfacial strain and maximizing stability. Unlike conventional sol–gel or template‐assisted hydrothermal methods, this single‐step, surfactant‐free route eliminates secondary nucleation barriers, ensuring uniform crystallite orientation and high surface coherence. The resulting TiO2 structure exhibits excellent crystallinity, compactness, and surface smoothness, combining 1D electron‐transport channels (along the rods) with 3D light‐scattering interfaces (from the flower‐like geometry). This synergistic morphology directly enhances the photocatalytic and antibacterial efficiencies of the material. In summary, the optimized hydrothermal route represents a novel and environmentally benign strategy for engineering hierarchical, morphology‐controlled TiO2 architectures with superior structural quality, functional performance, and scalability for next‐generation photocatalytic and antimicrobial applications.
Flower‐like microrod structures of TiO2 demonstrate strong antibacterial capabilities through a blend of physical and chemical mechanisms that undermine bacterial cell integrity. These microstructures directly engage with bacterial cell walls, causing pit formation and localized deformation. This physical interaction enhances membrane porosity and destabilizes permeability, ultimately leading to cell death. Concurrently, TiO2 promotes the production of reactive oxygen species (ROS), particularly hydroxyl radicals (•OH) and superoxide anions (O2 −), which induce oxidative stress in bacterial cells. The ROS‐induced damage encompasses lipid peroxidation, protein denaturation, and nucleic acid breakdown, amplifying the antimicrobial impact. The TiO2microstructures inactivate crucial bacterial enzymes by blocking thiol (‐SH) groups, inhibiting enzymatic functions vital for cellular respiration and energy metabolism. Moreover, ROS disrupt respiratory chain enzymes by oxidizing iron–sulfur clusters, further compromising bacterial survival [25, 26]. Titanium ions interact with nucleic acids, resulting in bacterial DNA degradation through oxidative cleavage of the phosphodiester backbone and base modifications [27, 28]. ROS‐mediated disruption of phosphate groups on tyrosine residues impairs bacterial signal transduction pathways, halting proliferation. Positively charged TiO2 ions are electrostatically attracted to negatively charged bacterial membranes, accumulating and disrupting membrane potential, leading to cytoplasmic leakage and collapse of ionic gradients essential for homeostasis. The antimicrobial efficacy of TiO2 microstructures is heavily influenced by their physical and chemical attributes, including particle size, surface charge, pH, ionic strength, and the presence of capping agents. Nanoparticles with customized morphologies and optimized surface functionalities enhance bacterial cell interaction while minimizing host cell toxicity. The antibacterial activity of TiO2 flower‐like microrod structures was evaluated against pathogenic bacterial strains, including methicillin‐resistant Staphylococcus aureus (MRSA), Escherichia coli, S. epidermidis, P. vulgaris, and Enterococcus spp. Inhibition zones (Table 1) ranged from 12 to 18.5 mm, demonstrating significant antibacterial potential. Notably, MRSA exhibited the largest inhibition zone, indicating heightened sensitivity to TiO2. At a minimal dose of 10 μg/well, both Gram‐positive (MRSA) and Gram‐negative (E. coli) bacteria showed marked growth suppression, as illustrated in Figures 4(a), 4(b), 4(c), and 4(d). Despite their potent antibacterial properties, TiO2 flower‐like microrods exhibit low cytotoxicity to mammalian cells, making them an attractive candidate for biomedical applications. This low toxicity, combined with their efficacy against resistant bacterial strains, positions TiO2 as a promising agent for developing advanced antimicrobial therapies [29, 30]. The ability to synthesize nanoparticles with precise size, morphology, and functionalization holds significant potential for improving infection control strategies in clinical and environmental settings.
Zones of inhibition (mm) obtained with titania (TiO2) flower‐like microrod structures using the hydrothermal process.

(a–d) Zones of inhibition obtained with the titania (TiO2) flower like microrod structures.
The antibacterial activity of the hydrothermally synthesized flower‐like TiO2 microrod structures can be attributed to a synergistic interplay between physicochemical and photochemical mechanisms that collectively induce bacterial cell inactivation. Firstly, the hierarchical microrod morphology and surface roughness of the TiO2 film significantly enhance bacterial adhesion and subsequent physical disruption of the cell envelope. The sharp, high‐aspect‐ratio edges of the rods can mechanically rupture bacterial membranes upon contact, leading to leakage of intracellular contents and loss of cell integrity. This topography‐induced mechanical damage plays a critical role, especially against Gram‐positive bacteria with relatively thick peptidoglycan layers, as observed in S. aureus and Enterococcus spp. Secondly, under UV illumination, TiO2 generates ROS through photogenerated charge‐carrier reactions. The electrons (e−) excited to the conduction band react with the adsorbed O2 molecules to form superoxide radicals (O2 −•), while valence band holes (h+) oxidize surface hydroxyl groups and water molecules to produce hydroxyl radicals (•OH) and hydrogen peroxide (H2O2). These ROS are highly oxidative and can attack multiple cellular components simultaneously—oxidizing membrane lipids, damaging DNA and proteins, and denaturing cytoplasmic enzymes—ultimately leading to bacterial cell death. Thirdly, the hydrophilic nature of the TiO2 surface (water contact angle ≈ 35°) promotes intimate contact between the bacterial membrane and the catalyst interface, facilitating electron transfer and ROS exposure at the cell–surface boundary. The enhanced wettability ensures efficient diffusion of oxygen and water molecules to active sites, further improving ROS generation and bacterial degradation efficiency. The combined mechanical interaction (surface morphology) and oxidative stress (ROS formation) thus define a dual‐mode antibacterial mechanism. The flower‐like hierarchical design enhances light absorption, prolongs charge‐carrier lifetimes by reducing recombination (as evidenced by the photoluminescence [PL] analysis), and increases the density of surface‐active sites—all of which intensify ROS production and bacterial inactivation. This dual mechanism accounts for the large inhibition zones observed (12–18.5 mm), with MRSA showing the highest susceptibility, likely due to its cell wall composition and higher affinity for surface adsorption. In summary, the antibacterial mechanism of the TiO2 microrods is governed by (i) direct physical disruption of bacterial membranes from the sharp, rough surface topology and (ii) indirect oxidative degradation via photogenerated ROS, together providing an effective and sustainable antimicrobial action suitable for environmental and biomedical applications.
The recombination of holes and electrons within a material typically results in a PL peak. The PL excitation spectrum of the hydrothermally synthesized flower‐like TiO2 microrod structures is shown in Figure 5(a). The prominent emission around 330 nm corresponds to a direct band‐to‐band transition between X1b ⟶ X1a at the X‐edge of the Brillouin zone (BZ). The moderate intensity of this emission indicates suppressed electron–hole recombination, reflecting efficient charge separation and migration within the TiO2 lattice. This lower recombination tendency can be attributed to the enhanced crystallinity, high aspect ratio, and well‐ordered microrod morphology of the hydrothermally synthesized structure. Although only the flower‐like TiO2 sample was analyzed in this study, similar morphology‐driven reductions in PL intensity have been widely reported for hydrothermally synthesized TiO2 nanostructures when compared to pristine TiO2 films [31–33]. The enhanced charge‐carrier dynamics facilitate photocatalytic degradation via ROS, such as hydroxyl radicals (•OH), that oxidize complex organic pollutants into simple, nontoxic molecules like CO2 and H2O.

Photoluminescence spectra (a) and water contact angle of titania (TiO2) flower‐like microrod structures (b).
(b)

Figure 5(b) illustrates the water contact angle measurement performed on the hydrothermally synthesized flower‐like TiO2 microrod structures. The surface exhibited a contact angle of approximately 35°, confirming its hydrophilic character. This strong wettability is attributed to the presence of abundant surface hydroxyl (−OH) groups generated during the hydrothermal and photoactivation processes. The photo‐induced activation of TiO2 promotes the formation of Ti3+ defect sites and oxygen vacancies, which subsequently undergo hydroxylation when exposed to moisture or air. These polar hydroxyl groups form hydrogen bonds with incoming water molecules, significantly reducing surface tension and enhancing water spreading across the surface. The hydrophilic nature of the TiO2 flower‐like microrods facilitates improved interfacial interactions between the catalyst surface and aqueous pollutants, thereby enhancing photocatalytic degradation efficiency. Increased wettability allows better diffusion of water and dissolved oxygen to reactive sites, accelerating the generation of ROS such as hydroxyl (•OH) and superoxide (O2 −•) radicals during illumination. These radicals are responsible for the oxidative breakdown of organic contaminants. Moreover, hydrophilic surfaces prevent biofouling and support uniform bacterial adsorption, contributing to the material’s antibacterial efficacy by promoting closer bacterial–surface contact and enhancing ROS exposure. Consequently, the combined effect of surface hydroxylation and microrod topology not only improves photocatalytic pollutant removal (reducing total dissolved solids, ammonia, and chemical oxygen demand) but also reinforces the antimicrobial functionality of the TiO2 films. Thus, the 35° contact angle observed for the TiO2 microrod structures confirms that the developed films possess photo‐induced hydrophilicity, which plays a pivotal role in facilitating efficient water purification and bacterial inactivation processes [34].
The photocatalytic activity of the hydrothermally synthesized flower‐like TiO2 microrod catalyst was evaluated through the degradation of methylene blue (MB) under UV irradiation, as shown in Figure 6.
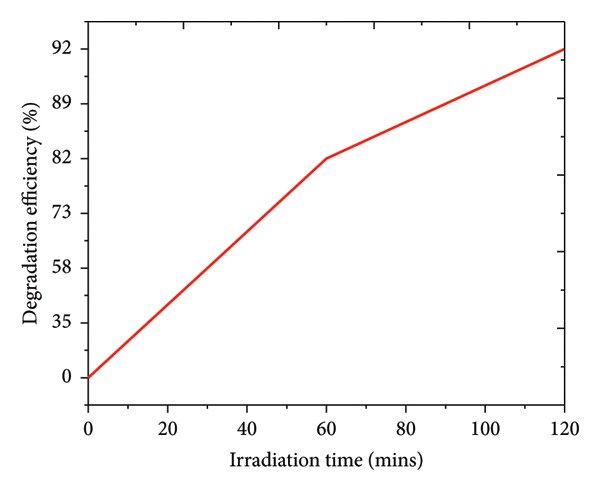
Photocatalytic degradation efficiency of methylene blue (MB) under UV irradiation using hydrothermally synthesized flower‐like TiO2 microrod catalyst.
The photocatalytic degradation efficiency of MB increased rapidly during the first 60 min of UV illumination and reached approximately 92% after 120 min, confirming the exceptional photocatalytic capability of the hydrothermally synthesized flower‐like TiO2 microrod structures. The pronounced activity arises from the hierarchical flower‐like morphology, which offers an extensive surface area for photon absorption and pollutant adsorption, while the anisotropic rod arrangement facilitates efficient charge‐carrier migration along the one‐dimensional growth axis. The lower electron–hole recombination rate, previously confirmed by PL analysis (Figure 5(a)), directly contributes to the observed enhancement in photocatalytic kinetics.
The temporal degradation behavior follows a pseudo‐first‐order kinetic model, expressed as
4. Conclusion
A hydrothermal synthesis method was successfully employed to fabricate flower‐like TiO2 microrod structures directly on FTO substrates. The optimized structures exhibited diameters of approximately 40–60 nm and lengths of 1–2 μm, as confirmed by SEM and EDX analyses. XRD patterns verified the anatase phase of TiO2, while FTIR spectra revealed characteristic Ti–O and Ti–OH vibrations, confirming hydroxylated surface groups that enhance interfacial reactivity. The synthesized TiO2 microrods demonstrated strong antibacterial activity against both Gram‐positive and Gram‐negative pathogens, with inhibition zones ranging from 12 to 18.5 mm, the highest being observed for MRSA. The enhanced antibacterial effect is attributed to the dual mechanism of surface‐induced mechanical disruption and ROS‐mediated oxidative stress. PL analysis showed a UV emission peak at 330 nm, associated with a direct band‐to‐band transition (X1b ⟶ X1a), indicating suppressed electron–hole recombination and efficient charge separation. The photocatalytic degradation of MB under UV irradiation confirmed the high functional performance of the material, achieving approximately 92% degradation within 120 min. This enhanced photocatalytic efficiency stems from the hierarchical flower‐like morphology, which improves light harvesting, surface area, and charge‐carrier transport. Furthermore, water contact angle measurements (∼35°) revealed the hydrophilic nature of the TiO2 surface, which facilitates pollutant adsorption, surface activation, and self‐cleaning behavior. Despite these promising results, challenges such as limited visible‐light absorption and long‐term stability under real environmental conditions remain. Future efforts will focus on band‐gap engineering and hybridization with materials such as graphene, Ag, Cu, or ZnO to extend photoresponse into the visible spectrum and enhance durability. In conclusion, the present work demonstrates that morphology‐controlled, surfactant‐free TiO2 microrod structures can simultaneously deliver high photocatalytic and antibacterial performance. These results establish the synthesized TiO2 films as reusable, eco‐friendly, and scalable photocatalysts for wastewater purification and disinfection, offering a sustainable pathway for next‐generation environmental remediation technologies.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request. Any additional datasets generated and analyzed during the current study can be provided upon request for noncommercial research purposes.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
No funding was received for this manuscript.
