Abstract
Objective. Sialic acid-binding immunoglobulin-like lectin 15 (Siglec-15) is overexpressed in various cancers which has led to the development of therapeutic anti-Siglec-15 monoconal antibodies (mAbs). In these preclinical studies, the therapeutic mAb, NC318 (antihuman/murine Siglec-15 mAb), was labeled with zirconium-89 and evaluated in human Siglec-15 expressing cancer cells and mouse xenografts for potential use as a clinical diagnostic imaging agent. Methods. Desferrioxamine-conjugated NC318 was radiolabeled with zirconium-89 to synthesize [89Zr]Zr-DFO-NC318. Cancer cell lines expressing variable Siglec-15 levels were used for in vitro cell binding studies and tumor xenograft mouse models for biodistributions. [89Zr]Zr-DFO-NC318 biodistribution and PET imaging studies to determine tissue uptakes (tissue : muscle ratios, T : M) included pharmacokinetic evaluation in Siglec-15+tumor xenografts and immunocompetent mice, blocking with nonradioactive NC318 (20, 100, and 300 μg) and xenografts with low/negligible Siglec-15 expressing tumors. Results. [89Zr]Zr-DFO-NC318 exhibited high affinity (
1. Introduction
Sialic acid-binding immunoglobulin-like lectin 15 (Siglec-15) is a cell surface type-1 transmembrane protein that is overexpressed in a variety of tumor types as well as in selected immune cells such as tumor-associated macrophages and dendritic cells. Siglec-15 expression in the tumor microenvironment (TME) is thought to be associated with permissive host immunity conducive to disease progression [1–4]. In addition to modulating tumor immune responses, Siglec-15 is expressed on osteoclasts and plays a role in osteoclast regulation and bone remodeling [5]. These diverse modulatory functions are activated upon Siglec-15 binding to sialoglycan structures such as Neu5Acα2-6GalNAcα (sialyl Tn) which is overexpressed in many cancer types including gastric, breast, lung, and ovarian [6]. CD44, a glycoprotein overexpressed on human hepatoma cells, has modified sialoglycans that can also serve as ligands for Siglec-15 thereby promoting tumor cell migration [7, 8]. Hence, Siglec-15 has been identified as an important immunomodulator and potential target for the treatment of osteoporosis and cancer offering certain advantages over other immunotherapeutics. Although Siglec-15 shares structural homology with programmed cell death ligand-1 (PD-L1), the immune regulatory mechanism is distinct and may offer an alternative therapeutic option in cancer patients unresponsive to PD-L1 immunotherapies [9].
In preclinical osteoporosis and tumor mouse models, antibody blockade of Siglec-15 resulted in increasing bone density and tumor regression (by reversing the immunosuppression in the TME), respectively [9–12]. These findings have prompted further development of anti-Siglec-15 monoclonal antibodies (mAbs) for evaluation as immune checkpoint inhibitors in clinical trials which include NextCure’s NC318 for solid tumors, Medimmune’s mAb for acute myeloid leukemia, and Daichi Sankyo’s DS-1501 for osteoporosis [9]. NC318 in a phase I clinical trial which included patients with non-small-cell lung carcinoma (NSCLC), melanoma, ovarian, colorectal, breast, and other types of cancer demonstrated efficacy primarily in patients with NSCLC (20%). This phase I trial was conducted without any biomarker assessment, but the NSCLC patient response rate of 20% was found to correlate with the Siglec-15 positivity of 25.7% (immunohistochemistry, IHC) observed in tumor biopsy stained sections of NSCLC patients (241) from a subsequent study [4, 9]. These results would indicate that tumor Siglec-15 expression may influence the therapeutic response; however, the molecular interactions remain to be elucidated. Hence, a biomarker has yet to be identified that would be predictive of therapeutic responses in patients and aid in patient selection for anti-Siglec-15 mAb therapies.
Radiolabeling of these therapeutic anti-Siglec-15 mAbs for immuno-positron emission tomography (immuno-PET) imaging could prove useful in quantitating in vivo expression levels of Siglec-15 in tumors in real time potentially aiding in patient selection, monitoring changes in Siglec-15 over a treatment time course, and determining the relationship of Siglec-15 expression levels to patient therapeutic responses. Although these therapeutic mAbs possess the high affinity and specificity required for a successful PET imaging agent, long lived PET radionuclides such as zirconium-89 (
2. Materials and Methods
Humanized IgG1 monoclonal Ab, anti-Siglec-15 mAb (NC318), was kindly provided by Dr. Ido Weiss (NextCure, Beltsville, MD, USA). The p-isothiocyanatobenzyl-desferrioxamine (DFO-Bz-NCS) was purchased from Macrocyclics, Inc. (Plano, TX, USA). Sodium acetate and Tris-HCl were purchased from Thermo Fisher Scientific (Waltham, MA, USA). The lyophilized whole human serum was obtained from MP Biomedicals, LLC (Solon, OH, USA) and dissolved in 2 mL saline. This serum solution was directly used without inactivation for the stability study of [89Zr]Zr-DFO-NC318. All other chemicals and reagents were purchased from Sigma-Aldrich (St. Louis, MO, USA) and used without further purification. PD-10 desalting columns were obtained from GE Healthcare Biosciences (Pittsburgh, PA, USA). Zirconium-89 oxalate was obtained from 3D Imaging (Little Rock, Arkansas, USA). Analytical high-performance liquid chromatography (HPLC) analyses were performed on an Agilent 1200 Series instrument equipped with a multiwavelength UV detector connected in series with a Bioscan flow count radiodetector. HPLC was performed using a size exclusion column (SE,
2.1. Synthesis of the DFO-NC318 Conjugate and Zirconium-89-Labeled NC318 ([89Zr]Zr-DFO-NC318)
The DFO-NC318 conjugate was prepared following the literature method using a 5-fold molar excess of DFO-Bz-NCS [13]. The purity of DFO-NC318 was determined by HPLC using a size exclusion column (SE-HPLC). The concentration (5.7 mg/mL) of the conjugates was measured using the BCA assay. The chelator to antibody ratio was 1.3, as determined by Liquid Chromatography Electro Spray Ionization Mass Spectrometry (LC-ESI-MS) using an Acquity UPLC H-Class coupled to a Xevo G2-XS QTof (Waters Corporation). The sample was deglycosylated using Rapid PNGase F (New England BioLabs) prior to analysis and desalted online using an XBridge Protein BEH C4 column (Waters Corporation) with a water/acetonitrile gradient containing 0.1% formic acid. Data processing was performed using UNIFI software (Waters Corporation), and peaks were assigned based on the unmodified antibody as a reference. Zirconium-89 conjugate ([89Zr]Zr-DFO-NC318) was prepared according to the literature method with minor modifications [13].
Briefly, a stock solution of zirconium-89 oxalate (~440 MBq) was diluted with 300 μL of HEPES buffer (0.5 M, pH 7.1-7.3). From this stock solution, ~150 MBq of zirconium-89 was used per radiolabeling reaction. The aliquot of zirconium-89 (~150 MBq, 110 μL) was further diluted with HEPES buffer (0.5 M, 800 μL, pH 7.1-7.3). 2,5-Dihydroxybenzoic acid (20 μL, ~5 mg/mL in water, pH adjusted to 7 with 2 M Na2CO3 solution) was added followed by a solution of DFO-NC318 (0.4 mg, 5.7 mg/mL, 70 μL). The reaction mixture was incubated for 1 h at room temperature and challenged with DTPA (5 μL, 0.1 M, pH 7) for an additional 10 min. The radiolabeled conjugate was purified by PD-10 column using 0.9% NaCl (pH 7). The molar activity and the purity of the radiolabeled conjugate were determined by SE-HPLC (
2.2. Storage and In Vitro Serum Stability
Following radiosynthesis, a saline solution of [89Zr]Zr-DFO-NC318 was stored at 4°C for 48 h and analyzed every 24 h by SE-HPLC to determine storage stability (Supplementary Information Figure S1 and Table S1). To determine serum stability, whole human serum (500 μL) was added to a solution of [89Zr]Zr-DFO-NC318 (~500 μCi in 500 μL of saline, pH 7.0) and kept at 37°C for up to 7 d. The radiochemical stability was determined every 24 h by directly injecting an aliquot of the solution into the HPLC and by iTLC (Supplementary Information Figure S2, Figure S3, and Table S2).
2.3. Cell Lines
Cell lines were grown at 37°C in 5% CO2 in DMEM (624-MEL WT/624-MEL+S15 human melanoma wild type or transduced with Siglec-15, provided by Dr. Ido Weiss (NextCure, Beltsville, MD, USA)), RPMI-1640 (LOX-IMVI (human melanoma); ATCC), and RPMI-1640 ATCC modified with 10 mM HEPES and 4500 mg/L glucose (HCC-827 (human non-small-cell lung carcinoma); ATCC). All media were supplemented with 10% FBS, 2 mM L-glutamine, and Pen/Strep.
2.4. In Vitro Studies
Saturation binding studies were performed to determine the
The biological specific activity or immunoreactive fraction (% immunoreactivity) of the [89Zr]Zr-DFO-NC318 was determined by a self-displacement method described by Choi et al. and Morris in which the % immunoreactivity was derived from a [89Zr]Zr-DFO-NC318 saturation curve and competition curve using as the competitor, nonradioactive NC318 (described above) [14, 15].
2.5. Mouse Models
Female athymic nude mice (Ncr-nu/nu, NCI-Frederick, MD) were injected subcutaneously in the right shoulder with 624-MEL WT/+S15 cells (
2.6. Biodistribution Studies
Tumor-bearing mice (tumor weights: 0.1-0.8 g) were injected intravenously (tail vein) with [89Zr]Zr-DFO-NC318 (0.37–0.74 MBq (10 to 30 μCi, 2 to 10 pmol)) and euthanized (via CO2 inhalation) at selected times. Blood samples and tissues were excised from each animal and weighed, and radioactivity content was determined (PerkinElmer 2480 Wizard3). For the NC318 blocking studies, mice were injected with [89Zr]Zr-DFO-NC318 only or coinjected with nonradioactive NC318 (20 μg (24x; 133 pmol), 100 μg (113x; 667 pmol), and 300 μg (309x; 2001 pmol). Blood and tissues were excised from each animal and weighed, and radioactivity content was determined (PerkinElmer 2480 Wizard3). Radioactivity content in the blood and each tissue was expressed as % injected dose per gram of tissue (%ID/g; (1)) and then normalized for body weight to a 20 g mouse (2) from which tissue : muscle ratios (T : M; (3)) were determined as follows:
Statistical analysis of the differences between the 2 groups was assessed using the Student
2.7. PET/CT Imaging Studies
Tumor-bearing mice were anesthetized with isoflurane/O2 (1.5%-3%
2.8. Human Siglec-15 Dosimetry Estimation
Human dosimetry estimates extrapolated from the mouse biodistribution studies were calculated using OLINDA V1.1 (Vanderbilt University, TN) with the mouse to human fractional organ extrapolation of the mean residence times of the ligand measured by the biodistribution described above. The %ID/g values (determined from the biodistribution studies described above) for a set of organs determined over a 7 d time course were used to extrapolate human dosimetry in the same organs. The whole organ was dissected from the carcass and counted to measure the organ’s radioactive content. For the bone, skin, muscles, and blood samples, a sample was dissected, weighed, and counted in the gamma counter. Because of the relatively small uptake in the skin, muscle, and blood, these tissues were not included in the kinetic input form of the OLINDA dosimetry estimation software.
Biodistribution data showed radioconjugate uptake in the mouse skeleton above the background. To account for this, a special case was made for bone dosimetry estimation. Instead of including the bone activity in the body remainder volume, the whole bone activity was estimated and entered into the trabecula bone input field in OLINDA. The bone tissue %ID/g and the %ID/organ were estimated using a murine bone fraction model of 53.3 (g/kg) [16]. This calculation gives an estimated bone mass of 1.07 g for a 20 g mouse. The %ID/g at each time point for each mouse was multiplied by the bone mass fractional estimate for each mouse.
From the %ID/organ, time activity curves (TAC) were generated from PET images and residence times were calculated in units of hours. Between the time points of the TAC, a trapezoidal model was used to estimate the area under the curve. For the last time point, an exponential decay curve with the half-life of zirconium-89 was used to extrapolate the tail of the TAC. Since the %ID/organ of the whole intestine was measured (including the contents), the absorbed activity between the large and small intestines was estimated by the MOBY fractional mass model for a 25 g mouse [17]. The result was that 75% of the activity was assigned to the small intestine and 25% to the large intestine. The large intestine was further separated using the ICRP 80 standard in which 57% of the radioactivity was assigned to the upper and the remaining 43% assigned to the lower large intestine.
2.9. Histopathology and Immunohistochemistry
All tissues were fixed in 10% neutral buffered formalin, routinely processed and sectioned at 5 μm for automated H&E staining, and digitized with an Aperio ScanScope XT (Leica) at 200x in a single
Whole slide imaging (WSI) was performed with an Aperio ScanScope XT (Leica) at 200x in a single
3. Results
3.1. Radiochemistry
Zirconium-89-labeled DFO-NC318 conjugate was prepared following the literature method with minor modifications [13]. The isolated radiochemical yields were in the range of 85-95% (

A representative HPLC of [89Zr]Zr-DFO-NC318. HPLC condition: eluent, 0.1 M sodium phosphate, 0.1 M sodium sulfate, 0.05% sodium azide, 10% isopropyl alcohol (pH 6.8), and flow rate 0.3 mL/min.
3.2. In Vitro Cell Binding Studies
High specific binding and affinity (

(a) Representative plot from an in vitro [89Zr]Zr-DFO-NC318 saturation binding assay using 624-MEL+S15 cells with each point representing the average of duplicates; for this plot:

From [89Zr]Zr-DFO-NC318 competition studies, the
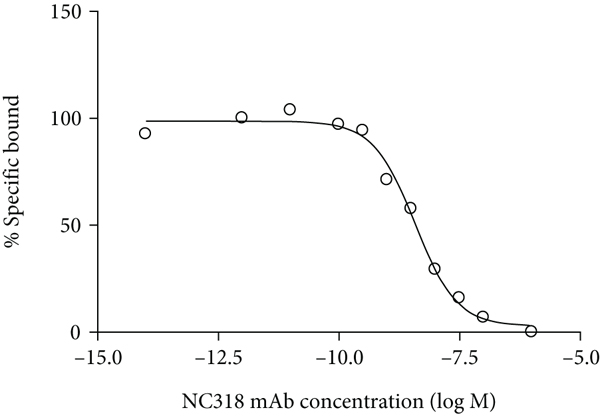
(a) Representative plot from an in vitro [89Zr]Zr-DFO-NC318 competition-binding assay using NC318 (self-displacement (Morris method)) with 624-MEL+S15 cells. Each point (average of duplicates) represents % specific bound (

3.3. In Vivo Biodistribution Studies
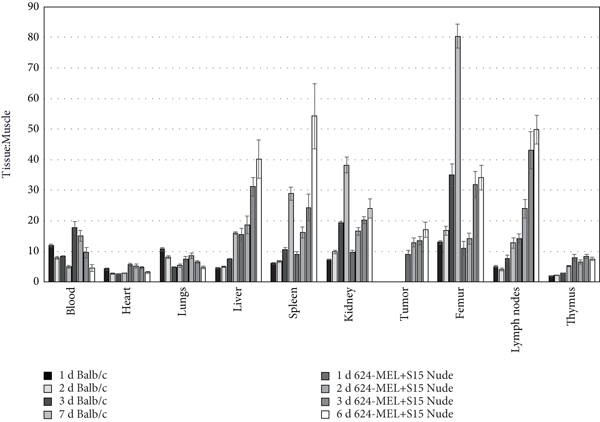
In normal immunocompetent mice (Balb/c) and 624-MEL+S15 xenograft nude mice, [89Zr]Zr-DFO-NC318 biodistributions were determined at 1, 2, 3, and 6 or 7 d post injection (Figures 4(a) and 4(b)). In the Balb/c and 624-MEL+S15 xenograft mice, [89Zr]Zr-DFO-NC318 distribution was rapid with clearance from the blood and most nontarget tissues with the exceptions of the liver, kidney, and femur which most likely were not decreasing over the time course due to the contribution of radioactive metabolites and released zirconium-89 (Figure 4(a)). Although clearance from the blood was comparable for the Balb/c and 624-MEL+S15 xenograft mice, other differences were observed in the targeting and pharmacokinetics of [89Zr]Zr-DFO-NC318. The highest tissue uptakes (%ID/g) were observed in the femur (14.0-16.3%ID/g), kidney (6.8-9.1%ID/g), and spleen (5.0-7.0%ID/g) in the Balb/c mice whereas in the 624-MEL+S15 xenograft mice, the highest uptakes were observed in the lymph nodes (9.4-16.9%ID/g), liver (9.7-11.0%ID/g), and spleen (7.5-11.0%ID/g). The high uptakes in the lymph nodes and spleen as well as the retention of [89Zr]Zr-DFO-NC318 over the time course would be consistent with targeted binding since these tissues are known to have Siglec-15+ cell populations (macrophages). However, in the case of the liver and the femur, only part of the radioactive uptake may represent specific Siglec-15-targeted uptake by hepatic macrophages and bone myeloid cells (osteoclasts), respectively. Previous findings have demonstrated that zirconium-89 released from the DFO chelate tends to be deposited in the epiphysis of the bone [20]. The tumors of the 624-MEL+S15 xenograft mice exhibited stable [89Zr]Zr-DFO-NC318 uptakes from 1 d (6.7%ID/g) to 7 d (4.9%ID/g) with a statistically insignificant loss of 28%. This lower 624-MEL+S15 tumor uptake indicates lower levels of Siglec-15 compared to the spleen and lymph nodes which is in concordance with the modest expression levels found in vitro for this cell line. In the immunocompetent Balb/c mice, the highest tissue to muscle ratios (T : M) occurred at 3 d and 7 d in the femur (35 : 1 T : M (3 d) and 80 : 1 T : M (7 d)), kidney (19 : 1 T : M (3 d) and 38 : 1 T : M (7 d)), and spleen (11 : 1 T : M (3 d) and 29 : 1 T : M (7 d)) which progressively increased over the time course (Figure 4(b)). This differed from the 624-MEL+S15 xenograft mice in which peak T : Ms were observed at 3 d and 7 d in the lymph nodes (43 : 1 T : M (3 d) and 50 : 1 T : M (7 d)) and liver (31 : 1 T : M (3 d) and 40 : 1 T : M (7 d)). In addition, high T : Ms for the 624-MEL+S15 xenograft mice were observed in the spleen (24 : 1 T : M (3 d) and 54 : 1 T : M (7 d)) and femur (32 : 1 T : M (3 d) and 34 : 1 T : M (7 d)) at the later times. The high T : Ms in the spleen and lymph nodes with the subsequent increases over the time course most likely occurred due to a faster clearance from the muscle, a nontarget tissue, compared to a target tissue in which [89Zr]Zr-DFO-NC318 is retained as a result of specific Siglec-15 binding. However, in the case of the T : M increases in the liver and femur at later times, the total radioactive content may only in part represent specific Siglec-15 uptake but radioactive metabolites (including released zirconium-89) which would be expected to increase at later time points as well. Similarly, the tumor T : Ms steadily increased over time from 9 : 1 to 17 : 1 indicating Siglec-15 targeting but lower expression levels compared to the lymph nodes and spleen of the 624-MEL+S15 xenograft mice. Over the time course, the Balb/c mice had lower blood and liver T : Ms with higher kidney and femur T : Ms at 7 d compared to the 624-MEL+S15 xenografts indicating that the Balb/c mice had faster clearance and metabolism of [89Zr]Zr-DFO-NC318. The Balb/c mice were 8 weeks younger than the 624-MEL+S15 mice which may in part account for the faster pharmacokinetics in nontarget tissues, but the other differences in the T : M values of the blood and Siglec-15+ tissues may be representative of compensatory alterations in immune cell populations in the athymic 624-MEL+S15 xenograft mice. In the 624-MEL+S15 xenograft mice, the lymph node T : Ms were significantly increased 3- to 6-fold (

(a) Biodistribution (%ID/g (normalized to 20 g mouse)) of [89Zr]Zr-DFO-NC318 in MEL-624+S15 nude xenograft and Balb/c mice (immunocompetent) from 1 d to 7 d. Each bar represents the mean
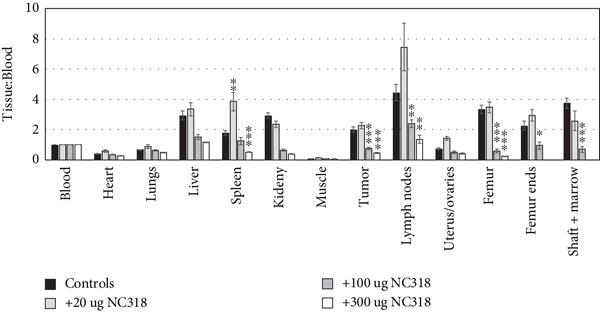
Other blocking biodistributions with unlabeled NC318 were carried out after 3 d of uptake using 624-MEL+S15 xenograft mouse groups injected with [89Zr]Zr-DFO-NC318 or coinjected with [89Zr]Zr-DFO-NC318 +unlabeled NC318 (20, 100, and 300 μg) (Figures 5(a)–5(c)). With the lowest NC318 dose of 20 μg (24x), blood and tissue uptakes (%ID/g) were comparable to the [89Zr]Zr-DFO-NC318 control group (~ 0.8 μg associated mass) except for a significant increase of 1.5-fold in the uterus/ovaries (3.85%ID/g;

(a) Biodistribution (%ID/g) of [89Zr]Zr-DFO-NC318 in 624-MEL+S15 xenografts at 3 d after receiving coinjections of [89Zr]Zr-DFO-NC318 (~0.8 μg) + NC318 (0 (control), 20, 100, and 300 μg). Each bar represents the mean


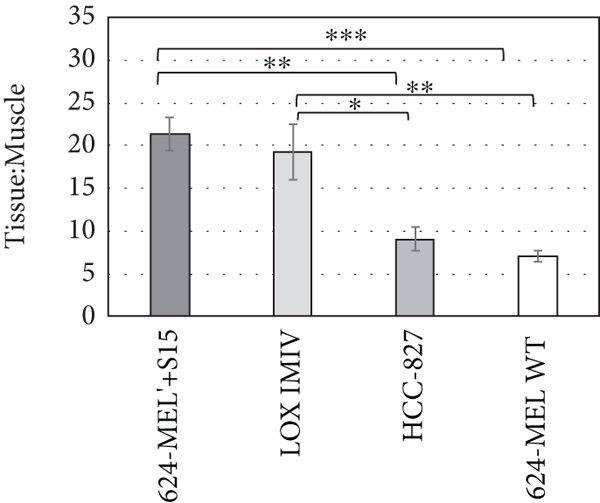
Biodistribution studies after 3 d of uptake were performed with other tumor xenograft models using tumor cell lines with moderate (624-MEL+S15 and LOX IMIV) and low/negligible (624-MEL WT and HCC-827) Siglec-15 expression levels to assess the ability of [89Zr]Zr-DFO-NC318 to distinguish quantitative changes in Siglec-15 levels in vivo (Figures 5(d) and 5(e)). The highest [89Zr]Zr-DFO-NC318 uptake was observed in the 624-MEL+S15 tumor (6.75%ID/g) which was not significantly different from the LOX IMIV tumor uptake (5.20%ID/g). In contrast, the HCC-827 (4.49%ID/g;
3.4. Small Animal PET Imaging Studies
[89Zr]Zr-DFO-NC318 (3.7 MBq (100 μCi), associated mass~ 20 μg)) was injected into 624-MEL+S15 xenograft mice which were imaged at 1, 3, and 6 d post injection (Figure 6(a)). In the PET images at 1 d post injection, 624-MEL+S15 tumors and spleens were visible but were more easily delineated at 3 d along with lymph nodes due to greater clearance from some nontarget tissues (Figure 6(a)). From PET images obtained at 3 d post injection of [89Zr]Zr-DFO-NC318 into xenograft mice, 624-MEL+S15 and LOX IMIV tumors were easily visualized with tumor T : Ms of ~20 : 1 whereas HCC-827 and 624-MEL WT had lower tumor T : Ms ranging from 5 : 1 to 8 : 1 and not as easily distinguished (Figure 6(b)). These PET imaging results were in agreement with the biodistribution results and further demonstrating that tumors with low to negligible Siglec-15 expression levels may be detectable with [89Zr]Zr-DFO-NC318 imaging due to the contribution of Siglec-15+ immune cells in the TME.
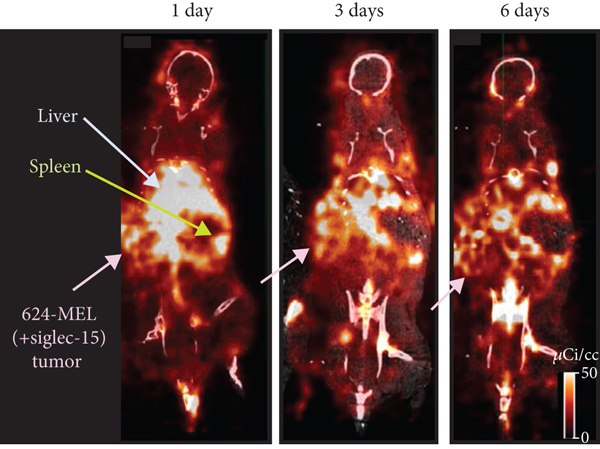
(a) Coronal PET/CT images of 624-MEL+S15 mouse xenografts at 1, 3, and 6 d post injection of [89Zr]Zr-DFO-NC318 (3.7 MBq (100 μCi), associated mass~ 20 μg). (b) PET images of 624-MEL+S15, LOX IMIV, HCC-827, and 624-MEL WT mouse xenografts at 3 d post injection of [89Zr]Zr-DFO-NC318 (3.7 MBq (100 μCi), associated mass~ 20 μg).

3.5. Histology and Immunohistochemistry
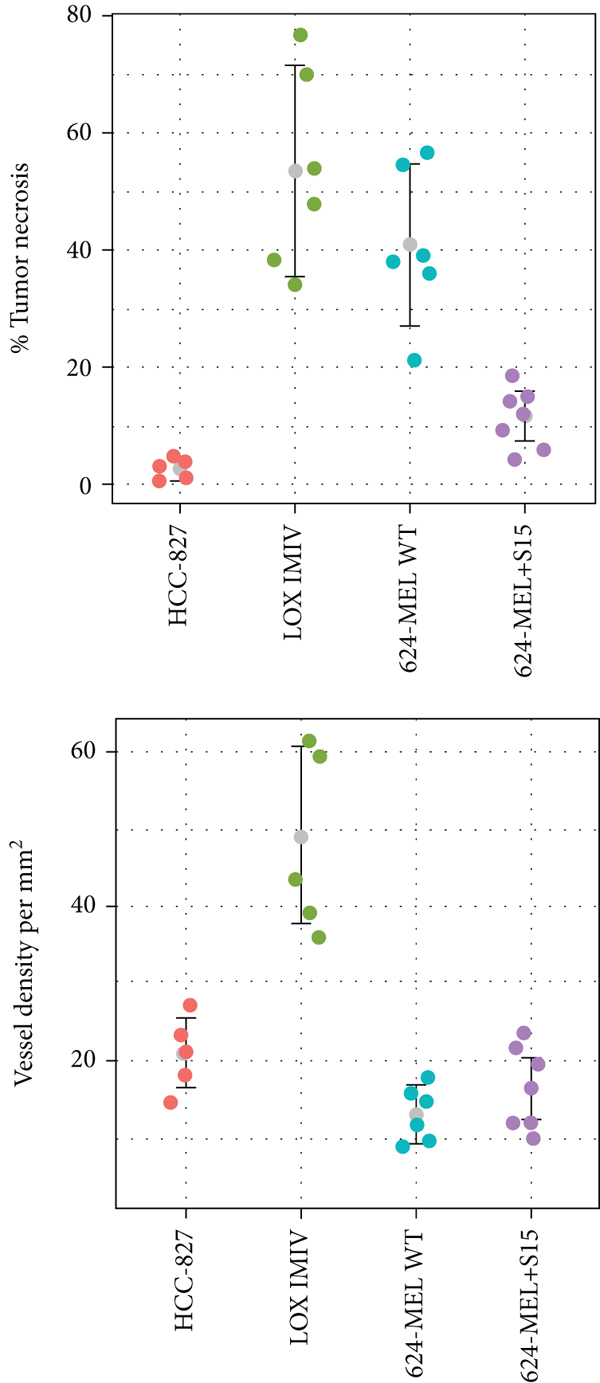
From the biodistribution studies, 624-MEL+S15, LOX IMIV, HCC-827, and 624-MEL WT tumors were prepared for H&E and IHC staining for Siglec-15, CD31 (vessel density), CD45 (hematopoietic cells including leukocytes, dendritic cells, NK cells, stem cells, and macrophage/monocyte), and Iba1 (macrophages) to confirm Siglec-15 targeting and identify mouse immune cells in the TME (Figure 7(a)). All tumor types were found to have varying degrees of tissue necrosis from H&E staining which were excluded from the quantitative scoring of the Siglec-15 and CD31 IHC results (Figures
7(b) and 7(c)). The LOX IMIV and 624-MEL WT tumor types had the greatest degree of necrosis which may in part be related to the tumor size in which the majority of these tumors ranged from 0.8 to 1.5 g whereas the HCC-827 tumors were smaller <0.8 g. The vascular density determined from CD31 IHC staining was found to differ among the tumor types with LOX IMVI (

(a) Representative images of H&E and IHC stained sections (CD45, Iba1, and Siglec-15) from 624-MEL+S15, LOX IMIV, HCC-827, and 624-MEL WT tumors. (b) Comparison of individual tumor necrosis (%; estimated from H&E staining) and vessel density (estimated from CD-31 IHC staining) for the 4 tumor types. (c) IHC quantitative analysis of Siglec-15 expression levels (staining intensity score and
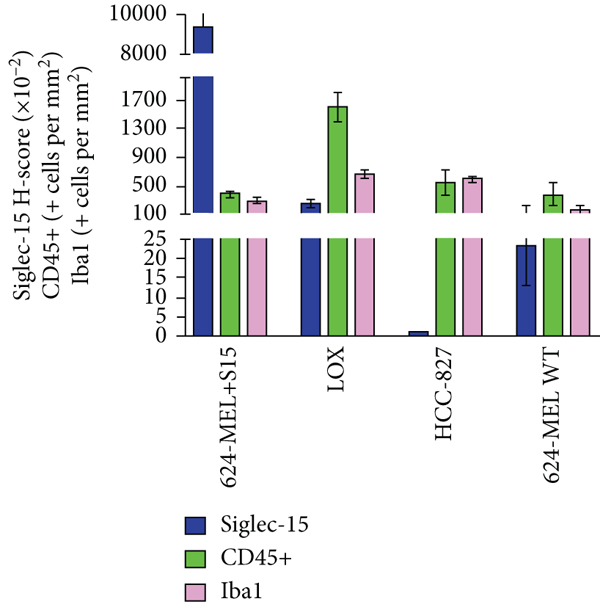
Using dual immunofluorescence staining to distinguish human Siglec-15+ tumor cells from Siglec-15+ murine cells, 624-MEL+S15 was found to have the highest human Siglec-15 expression levels (16.4% NUMA(+)) which was 5- to 6-fold greater than the LOX IMIV (3.1% NUMA(+)) tumor type (Figures 8(a) and 8(b)). These results for the % NUMA(+) cells which represent Siglec-15+ human tumor cells are comparable to the rank order of the Siglec-15 expression levels (

(a) Representative images of Siglec-15 (S15) IHC and dual immunofluorescence staining (NUMA1&S15) of 624-MEL+S15, LOX IMIV, HCC-827, and 624-MEL WT tumors from xenograft mice to distinguish positive Siglec-15 (+S15) human tumor cells (green (NUMA1, human nuclear marker) and pink (+S15)) from mouse +S15 immune cells in the TME (blue (mouse) and pink (+S15)); white arrows identify human tumor cells (NUMA(+)) positive for S15. (b) Quantification of human tumor cells positive for S15 (% NUMA(+)) and murine +S15 cells (% NUMA(-)) of each tumor type from dual immunofluorescence stained images; each bar represents the

In addition to the presence of murine Siglec-15+ positive cells in the TME, murine Siglec-15+ positive cells were generally observed in the spleen and lymph nodes in IHC sections from 624-MEL+S15 xenografts further confirming the specific uptake of [89Zr]Zr-DFO-NC318 in these tissues (Figure 9).
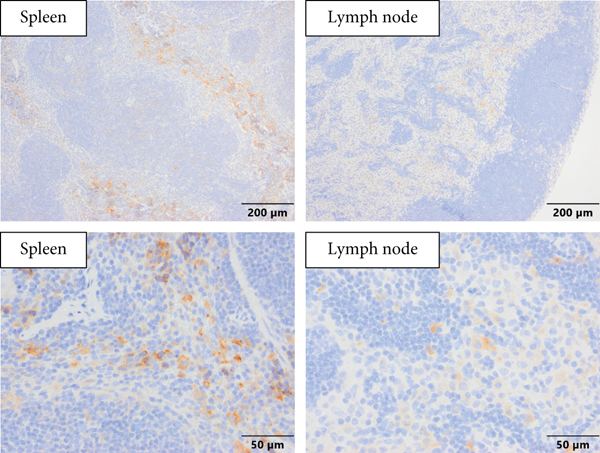
Representative Siglec-15 immunohistochemistry stained sections of the spleen and lymph nodes from 624-MEL+S15 xenografts. Spleen: Siglec-15+ cells were commonly observed in the splenic red pulp with a cytoplasmic to membranous staining pattern; lymph node: faint Siglec-15+ cells were commonly observed in the medulla but occasionally were present in the paracortex or cortical regions of the node.
3.6. Dosimetry Estimation for [89Zr]Zr-DFO-NC318
The extrapolation of radioconjugate residence times in humans was determined from the radioactivity content of the organs and tissues using the [89Zr]Zr-DFO-NC318 biodistribution results with the Balb/c mice (Table 1). The five highest radiation absorbed doses were observed in the osteogenic cells (1.53 mSv/MBq, 5.67 rem/mCi), red marrow (0.868 mSv/MBq, 3.21 rem/mCi), kidney (0.562 mSv/MBq, 2.08 rem/mCi), adrenals (0.305 mSv/MBq, 1.13 rem/mCi), and spleen (0.278 mSv/MBq, 1.03 rem/mCi). The total body dose was 0.211 mSv/MBq or 0.779 rem/mCi, and the effective dose was 0.250 mSv/MBq or 0.926 rem/mCi.
[89Zr]Zr-DFO-NC318 human dosimetry extrapolated from Balb/c mouse biodistributions (includes the organs used in the kinetics input form of OLINDA, the mouse residence times, the extrapolated human residence times, and the dose the tissue received per unit injected activity).
4. Discussion
[89Zr]Zr-DFO-NC318 demonstrated specific binding to Siglec-15 with nM binding affinity in vitro and discriminated in human cancer cell lines low to moderate concentrations of Siglec-15. In vivo [89Zr]Zr-DFO-NC318 had sufficient stability to be highly retained in Siglec-15+ tumors as well as mouse lymph nodes, spleen, and bone which are known to have murine Siglec-15+ immune cell populations. From these preclinical studies, [89Zr]Zr-DFO-NC318 was found to exhibit appropriate targeting and dosimetry to be considered for clinical applications including patient selection for Siglec-15-targeted immunotherapeutics and then patient monitoring of treatment responses.
Siglec-15 has emerged as a novel immune inhibitor which utilizes a pathway that is distinct from the PD-1/PD-L1 immune checkpoint pathway and therefore may represent the next generation of immunotherapeutics. Preliminary results in mouse tumor models have shown tumor regression in both PD-1 sensitive and insensitive tumors suggesting that Siglec-15-targeted immunotherapeutics may offer an alternative to patients resistant to PD-1/PD-L1 therapies [22, 23]. In a NC318 clinical trial with NSCLC patients who were refractory for anti-PD-1 mAb therapy, 20% had complete or partial responses and 30% had stable disease [9]. Although the results from this clinical trial are encouraging, the patients were not selected based on their PD-1/PD-L1 or Siglec-15 expression levels, and therefore, more definitive results may be possible with selected populations. Development of diagnostic agents and a predictive biomarker are needed to select patients for Siglec-15-targeted therapeutics and to gain a better understanding of the relationship of the interactions between Siglec-15 positive tumor cells and immune cells in the tumor TME to therapeutic responses. An IHC assay has been developed for patient selection for Siglec-15-targeted therapies which detects Siglec-15+ tumor and immune cells in tumor biopsies [24]. These IHC results reflect the Siglec-15 positivity of the tumor at the time and location of the biopsy and may not reflect changes that have occurred in the TME from the time of the biopsy to the start of therapy or during the therapeutic time course. Since preclinical studies would suggest that the overexpression of Siglec-15 in the TME plays a role in the immunosuppression of the tumor and a therapeutic such as NC318 would act to reverse this immunosuppression in the TME, assessing the dynamic changes in the TME of Siglec-15 expression levels in response to therapy is needed [9]. Immuno-PET imaging with [89Zr]Zr-DFO-NC318 could provide a real-time readout of Siglec-15 expression levels in all lesions of the patient and surrounding tissues as well as monitor changes in expression levels with treatment.
Generally, the uptake of [89Zr]Zr-DFO-NC318 was higher in the lymphoid tissues of the athymic tumor-bearing mice compared to the immunocompetent Balb/c mice indicating that the athymic mice had increased Siglec-15+ expressing immune cells. Athymic mice are known to have impaired T-cell function with fewer circulating leukocytes and depletion in “thymus dependent” areas of the spleen, lymph nodes, and bone marrow compared to normal mice but have normal cytotoxic responses to T-independent antigens [25, 26]. In contrast, natural killer (NK) cell and macrophage cytotoxic activities have been reported to be enhanced in athymic mice compared to immunocompetent mice suggesting that these immune cell types may compensate for the T-cell deficiency [27]. In particular, athymic mice transplanted with human tumor cells have been found to have allograft cytotoxic responses mediated by macrophages and NK cells [27, 28]. As the tumor progresses, a state of chronic inflammation ensues resulting in the accumulation of myeloid-derived suppressor cells (MDSCs) which is a heterogeneous immature myeloid cell population with immunosuppressive functions that operate by a wide variety of mechanisms [29]. The abnormal production of growth factors and cytokines by the tumor cells and stroma including resident macrophages causes expansion of MDSCs by inhibiting normal myeloid differentiation [30]. The expansion of MDSCs has been found to extend beyond the tumor to the spleen of tumor-bearing mice as well as bone marrow cells cultured with tumor cells [31]. Within the cell groups comprising immature MDSCs, a small group phenotypically similar to monocytes (M-MDSCs) can differentiate into macrophages, dendritic cells, and osteoclasts that primarily have immunosuppressive functions both at the tumor site and periphery [31, 32]. Similarly, macrophages induced with growth factors and other innate inflammatory mediators as well as tumor-associated macrophages (TAM) have been found to overexpress Siglec-15 suggesting that a subset of the M-MDSC cells are Siglec-15+ [9]. Taken together the increased cytotoxic activity of macrophages and then the allograft immune response resulting in the expansion of Siglec-15+, M-MDSCs in the spleen, lymph node, and bone marrow of athymic tumor-bearing mice may be expected to account for the increased [89Zr]Zr-DFO-NC318 uptakes observed in these lymphoid tissues.
The in vivo [89Zr]Zr-DFO-NC318 uptakes of the 624-MEL+S15, LOX-IMIV, HCC-827, and 624-MEL WT tumors exhibited similar rank order compared to the in vitro (
These promising preclinical results would suggest that the [89Zr]Zr-DFO-NC318 would be appropriate for Siglec-15 immuno-PET imaging in human subjects to establish the predictive value of Siglec-15 expression as a reliable biomarker for patient selection in clinical trials, monitoring therapeutic responses and evaluating the efficacy of this new class of immunotherapeutics. Siglec-15 expression in humans is generally absent from normal tissue and confined to myeloid cells and osteoclasts in lymphoid tissues [9, 34]. In normal human spleen and lymph nodes, Siglec-15 expression was found on a small number of dendritic cells and macrophages suggesting that Siglec-15 expression occurs on a subset of dendritic cells or macrophages [35]. In contrast, Siglec-15 expression levels are upregulated in tumors and tumor infiltrating macrophages which may also extend to the spleen, lymph nodes, and other lymphoid tissues. Therefore, in PET images, lesions of cancer patients in which Siglec-15 has been upregulated should be discernible from normal tissue. In addition with whole body PET imaging, changes in Siglec-15 expression levels in the spleen, lymph nodes, and other lymphoid tissues can be assessed to determine the role Siglec-15 plays in cancer progression and M-MDSC biology [31]. Additionally, since NC318 is currently in clinical trials, [89Zr]Zr-DFO-NC318 could be used as a companion diagnostic imaging agent as well as assist in establishing dosing and tracking in vivo the tissue distribution of NC318 to gain a better understanding of off-target side effects. Since NC318 has been found to be safe in on-going clinical trials, clinical translation of [89Zr]Zr-DFO-NC318 can be completed relatively more easily for “proof of concept” PET imaging studies. Providing these studies corroborate the value of Siglec-15 as a predictive biomarker further development of small molecules or Ab fragments targeting Siglec-15 labeled with shorter lived radionuclides (i.e., fluorine-18) would be justified. The faster pharmacokinetics of these imaging agents would allow for same day imaging rather than waiting several days to acquire images which is preferable with whole mAb-based imaging agents.
In conclusion, [89Zr]Zr-DFO-NC318 would make available real-time PET images that represent Siglec-15 tumor expression levels not only for the primary tumor but metastatic lesions as well. In patients with metastatic disease, [89Zr]Zr-DFO-NC318 imaging would make possible the measurement of all Siglec-15+ lesions thereby providing a potentially better tool for patient selection than a biopsy of the primary tumor. Such imaging could also potentially serve as a biomarker to monitor responses in patients undergoing Siglec-15-targeted therapies. Validated PET imaging agents that could identify and quantify tumor Siglec-15 expression levels would be beneficial not only for clinical diagnostic and prognostic applications but also for the drug development process as well.
Footnotes
Data Availability
The data used to support the findings of this study are included within the article and the supplementary information files.
Disclosure
The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services nor does mention of trade names, commercial products, or organization imply endorsement by the U.S. Government.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Acknowledgments
The authors would like to thank Dr. Linda Liu and Dr. Han Myint at NextCure for providing materials (NC318 and 1F7 mAbs) and expertise. This project has been funded in whole or in part with federal funds from the National Cancer Institute, National Institutes of Health, under contract No. 75N91019D00024.
