Abstract
Migraine is one of the most prevalent neurological disorders with some 30% of patients additionally suffering from focal neurological disturbances: the aura. The underlying mechanism behind the aura is generally considered to be a form of cortical spreading depression (CSD). We used mechanical stimulation to induce hyperaemia associated with CSD in cats and rats, and studied the effect of a glutamate, α-amino-3-hydroxy-5-methyl-4-isoxazole propionate (AMPA) receptor, antagonist, and γ-aminobutyric acid (GABA)A and GABAB receptor agonists, to understand better the pharmacology of CSD. All three were able to inhibit CSD-associated cerebral blood flow changes in the rat and in a proportion of cats studied; non-responders showed altered speed of propagation and time to induction. The data suggest AMPA and GABA receptors may be targets of migraine therapy in inhibiting CSD and thus may alter the frequency of migraine aura.
Introduction
Migraine is a highly disabling (1), common (2) and expensive (3,4) brain disorder (5). About 30% of migraine sufferers (6) experience neurological symptoms, including visual, sensory and motor symptoms, which develop over 5–20 min and generally subside within 1 h (7). The mechanism underlying the cortical blood flow changes believed to result in aura is now widely considered to be a wave of cortical spreading depression (CSD) akin to Leao's spreading depression in the rabbit cortex (8), which spreads across the cortex at a rate of 2–6 mm/min. Thus the pharmacology of CSD is important background to the development of new treatments for migraine aura.
CSD can be triggered by chemical, electrical and mechanical stimulation and results in a transient wave of neuronal and glial depolarization followed by a short-lasting depression or marked decrease in neuronal membrane resistance that spreads across the cortex. It is also associated with cerebral blood flow (CBF) changes and sustained hypoperfusion in the same brain region as the depolarization, as seen in blood flow studies (9–11). The blood flow changes are closely coupled to the underlying neuronal activity seen in CSD and such changes are unlikely to occur in the absence of CSD; however, recent evidence has indicated a degree of divergence in propagation rates (12).
Recent studies have shown migraine preventives, such as topiramate (13,14) and valproate (15), which reduce the frequency and severity of migraine attacks, with and without aura, inhibit the triggering of CSD in experimental animals (9,16). Although the direct mechanism of their preventive action is unknown, some parts of their pharmacology have been identified. Topiramate is believed to modulate voltage-sensitive calcium channels and sodium ion channels, as well as potentiating γ-aminobutyric acid (GABA) inhibition and blockade of excitatory glutamate transmission, including N-methyl-D-aspartate (NMDA) and α-amino-3-hydroxy-5-methyl-4-isoxazole propionate (AMPA)/kainate receptors (17,18). Valproate also acts on sodium ion channels and is involved in GABAergic transmission, particularly in the trigeminovascular system (19–22). It has been shown previously that CSD-associated CBF changes using a similar model to that which we report here are inhibited by Na+ channel blockade (23), and NMDA receptor antagonism (11,24). Therefore, by targeting specific receptors in animal models, using CBF changes caused by mechanical stimulation as a model of CSD, we may understand better some part of how current medicines work and provide directions for the development of new treatments. The work was presented in preliminary form at the 12th Congress of the International Headache Society (Kyoto, Japan, October 2005 (25)).
Methods
Surgical preparation for cats
Four female and four male cats (3.2 ± 0.1 kg) were anaesthetized using α-chloralose (60 mg/kg intraperitoneally) and the cats prepared for physiological monitoring. Halothane (0.5–2% in 40% O2-enriched air) was administered during surgical procedures and discontinued during experimental protocols. The left femoral artery and vein were cannulated for blood pressure recording and infusion of anaesthetic and test compounds, respectively. Supplementary doses of α-chloralose in 2-hydroxy-β-cyclodextrin (26) were given at a rate of 10 mg kg−1 h−1. The cats were intubated after local anaesthesia with lidocaine and placed in a stereotaxic frame. Temperature was maintained throughout using a homeothermic blanket system (CWE Inc., Ardmore, PA, USA). The cats were ventilated with oxygen-enriched air, 30–35 ml/s, 22–26 strokes per minute (Ugo Basile, UK). End-tidal CO2 was monitored (Capstar-100; CWE Inc.) and kept between 2.5 and 3.5% and blood pressure was monitored continually. This allows one to monitor for changes to respiration and blood pressure due to long-term anaesthetic maintenance. Depth of anaesthesia was monitored by testing the sympathetic (papillary and cardiovascular) responses to noxious stimulation and withdrawal reflexes.
To prepare the cranium for blood flow monitoring the pericranial musculature was retracted laterally following a midline skin incision. A small unilateral rectangular burr hole was made (20 × 5 mm) over the left parietal cortex, 5 mm from the midline with a low rotational speed, saline-cooled drill, to prevent thermal injury. At the occipital end of the burr hole the dura mater was carefully removed to provide access for a 26-G needle, mounted in a stereotaxic micromanipulator, for mechanical stimulation. More rostrally. two laser Doppler flow probes (Moor Instruments, Axminster, UK) were lowered perpendicular to the brain surface on to the dura mater, 5–10 mm apart, to measure cortical flow, known as probe 1 and probe 2 (Fig. 1a). All experiments were conducted under UK Home Office Animals (Scientific Procedures) Act (1986).
A schematic top view (not to scale) of the experimental set-up in the (A) cat and (B) rat. (A) The two laser Doppler probes on the drilled closed cranial window or (B) the single laser Doppler probe with the recording electrode occipital to this, with the bone and dura removed. The needle stick site is anterior to bregma. MMA, middle meningeal artery.
Surgical preparation of rats
Seventeen male Sprague-Dawley rats (320–500 g) were anaesthetized throughout the experiments with sodium pentobarbitone (Sigma-Aldrich, Poole, UK; 60 mg/kg intraperitoneal and then 18 mg kg−1 h−1 intravenous infusion). Again the left femoral artery and vein were cannulated for blood pressure recording and intravenous infusion of anaesthetic and test compounds, respectively. Temperature was maintained throughout using a homeothermic blanket system (CWE Inc.). The rats were placed in a stereotaxic frame and ventilated with oxygen-enriched air, 1.5–2.5 ml, 80–110 strokes per minute (Small Rodent Ventilator, Model 683; Harvard Apparatus, Edenbridge, UK). End-tidal CO2 was monitored (Capstar-100; CWE Inc.) and kept between 3.5 and 4.0% and blood pressure was monitored continually. The rats were placed in a stereotaxic frame and the parietal bone drilled so the dural blood vessels were clearly visible through the intact skull. At the occipital end of the drilled area a small region of bone and dura was removed to allow insertion of a tungsten recording electrode (WPI, Stevenage, UK; impedance 1.0 MΩ, tip diameter 0.5 µm) to a depth of 500–1000 µm below the surface to monitor cortical single-cell activity. Electrical signals were amplified and passed through filters and a noise eliminator (Humbug; Quest Scientific, North Vancouver, Canada). The signal was fed into a gated window discriminator and an analogue-to-digital converter (Power 1401plus; CED, Cambridge, UK) to a personal computer. Filtered and amplified signal was also fed for audio monitoring and displayed on an oscilloscope. A laser Doppler probe was placed anterior to the recording electrode, perpendicular to the skull surface, to measure CBF changes. Also, just anterior to bregma, a burr hole was created on the bone and dura mater removed so a 26-G needle could be inserted to cause cortical lesioning and induce spreading depression; this method has been used previously to induce reliably CSD-associated cortical blood flow changes as a correlate of migraine aura (see Fig. 1b) (9,10,23,27).
In all experiments CBF was continuously measured using the placed laser Doppler probes (Moor Instruments) and displayed with electrical and physiological monitoring on an online data analysis system (CED spike2 v5 software).
Experimental protocols
In some cats the CBFLDF response to hypercapnia was monitored to assess whether CSD had been accidentally elicited during surgery, since CSD prevents blood flow responses to hypercapnia for up to 8 h in the cat (28). Previous work has shown that α-chloralose has no effect on CSD outcome (29) and that the regional CBF changes can be reliably induced in pentobarbitone-anaesthetized rats (9,23). The animals were left for at least 1 h for physiological monitoring to stabilize, and to allow a refractory period in case spreading depression and blood flow changes had been elicited during surgery. A 26-G needle was briefly inserted into the cortex to a depth of 1–2 mm using a micromanipulator to induce mechanical stimulation, and immediately withdrawn. In the cat there was a 60-min interval and in the rat a 45-min interval between inductions to avoid cortical refractory periods. This has been shown previously to be sufficient time between CSD inductions, and is indicative that depression has taken place (9,11,23,30). Each animal received a control plunge, followed by a plunge after 0.25 ml saline (intravenous) to ensure the reproducibility of the hyperaemic response (HR), as not all mechanical stimulations of the cortex induce a CSD.
In the cat GYKI52466 (4 mg/kg), an AMPA receptor antagonist, muscimol (4 mg/kg), the GABAA receptor agonist and (R)-baclofen (4 mg/kg), the GABAB receptor agonist, were all given 5 min prior to mechanical stimulation and was repeated 60 min later if the HR was inhibited. If the HR did occur, a repeated dose was administered and needle plunge repeated. Each cat received up to two drug doses.
A second series of experiments took place in the rat to measure the effects of the same compounds GYKI52466 (2 mg/kg), muscimol (1 mg/kg) and (R)-baclofen (2 mg/kg) on the electrical spiking that occurs during CSD as well as blood flow changes. Each drug was again given 5 min prior to the cortical mechanical stimulation and was repeated after 45 min if the spreading depression was inhibited, or did not occur.
It has been shown previously that repeated blood flow changes can be induced in both cats and rats using the needle prick as a method of mechanical stimulation, without subsequent changes to latter blood flow responses, implying there is no additional damage caused to the brain by repeated inductions at the same site (9,27).
Data analysis
CBF changes were observed and recorded online and evidence of CSD observed as a spike of electrical activity caused by depolarization, followed by electrical depression (Fig. 2). CSD and the CBF changes that take place are considered an all-or-nothing event, therefore any CBF changes were deemed to be evidence that spreading depression had taken place. The change in electrical activity from single cell recording was compared with baseline level of cell firing (Hz) for the minute prior to mechanical stimulation. Speed of propagation (SP) was derived by the difference in the latencies to the beginning of the hyperaemic phase of the probes and the response across the cohort was compared using ANOVA for repeated measures with Bonferroni correction applied, followed by Student's paired t-test if any significance was found.
Example tracing of a single laser Doppler probe and single cell cortical electrical activity occurring during mechanical stimulation to induce spreading depression in the rat. Cortical mechanical stimulation was followed by a hyperaemic response and a cortical spike and subsequent electrical depression that spreads across the cortex. Intravenous injection of GYKI52466 (2 mg/kg) inhibits the spreading depression and cortical spiking; after 45 min the spreading depression response returns.
It is important to note that previous studies have shown the time relation of neuronal discharges/spiking and CSD are not always coincident (31,32), indicating that the spread is probably not due to the discharge, but might be another result of the spreading depression and glial depolarization itself (32). To add to this, a recent study has shown that there may be dissociation between the vascular and neuronal/metabolic changes that take place during spreading depression, with possibly independent mechanisms of action (12). This has particular relevance as regards the study of aura in migraine, where the aura response is believe to be a response to the cortical blood flow that is observed (33,34), but it is the neuronal and metabolic changes that take place during CSD that underlie these vascular changes. The Brennan study implies that cortical blood flow changes may indeed be a direct response of spreading depression under a separate mechanism. Therefore studies on the vascular changes, particularly in relation to migraine aura, are very relevant to the physiology of this syndrome.
Drugs
The infusion of anaesthetic and test compounds was via the same catheter; however, the line was always flushed with saline first, several minutes before administering the different compounds. Muscimol (5-aminoethyl-3-hydroxyisoxazole), (R)-baclofen [(R-40amino-3-(4-chlorophenyl) butanoic acid] and GYKI52466 {4-[8-methyl-9H-1,3-dioxolo(4,5-L)(2,3)benzodiazepine-5-yl]-benzamine} (all Sigma Aldrich) were all dissolved in 0.9% NaCl. α-Chloralose (Sigma-Aldrich) was dissolved in either water for injection or 2-hydroxy-β-cyclodextrin (Sigma-Aldrich) (26). Lidocaine hydrochloride (Intubeaze; Arnolds, Shrewsbury, UK) and halothane (May and Poulenc, Dagenham, UK) were also used.
Results
In all cats the respiratory parameters were maintained at physiological levels throughout the experiments: pH 7.44 ± 0.01, pCO2 2.8 ± 0.2 kPa and O2 32.7 ± 2.2 kPa. To avoid hypovolaemia in the rat experiments, arterial samples were not taken, but blood pressure and end-expiratory CO2 levels were observed and physiological throughout the studies.
Summary of raw data for the GABA receptor or AMPA receptor mediators that highlights whether cerebral blood flow changes or neuronal discharge occurred
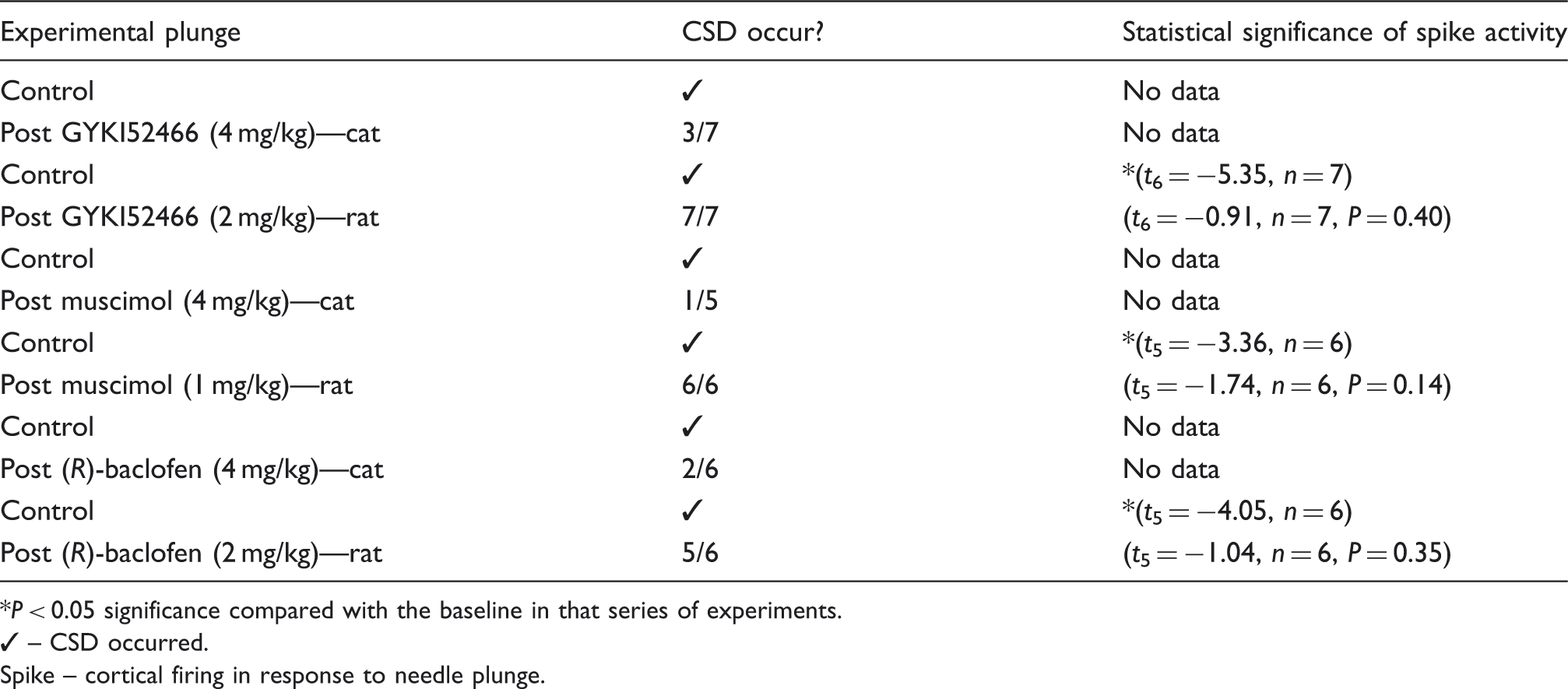
P < 0.05 significance compared with the baseline in that series of experiments.
– CSD occurred.
Spike – cortical firing in response to needle plunge.
Cat work
There were no sex differences between responders and non-responders.
GYKI52466 was observed to prevent the HR induced by cortical mechanical stimulation in three out of seven trials, and in those animals where a response occurred GYKI52466 significantly affected the time to onset of hyperaemia (F 1,9 = 5.62, P < 0.05), but there was no significance in the SP across the cohort (F 2,12 = 1.58, P = 0.25). Muscimol was able to inhibit the HR in one out of five trials and it did not significantly effect the time to start the HR (F 1,6 = 1.53, P = 0.26), but the SP was significantly affected over time (F 1,6 = 16.49, P < 0.05). In the case of (R)-baclofen, two out of six responses were observed to have inhibited hyperaemia and it did not significantly affect either the onset of HR (F 1,7 = 3.87, P = 0.09) or the SP (F 1,7 = 0.62, P = 0.46).
Rat work
When CSD was induced in rat, cortical electrical changes were made as previously described (9,23). GYKI52466 (2 mg/kg) inhibited CBF in 7/7 trials. When cellular recording was monitored, the spike of electrical activity that is time-matched to the hyperaemia (see Fig. 2) was also inhibited by GYKI52466 (F 3,18 = 13.16, P < 0.05, n = 7) and specifically when compared with control (t 6 = 5.02, P < 0.05). Vascular changes and neuronal discharge returned when mechanical stimulation was repeated after 45 min. Muscimol (1 mg/kg) also inhibited CBF changes in 6/6 trials and the cortical electrical firing (F 3,15 = 10.96, P < 0.05, n = 6) across the cohort and specifically when compared with the control spreading depression (t 5 = 3.33, P < 0.05). (R)-Baclofen (2 mg/kg) inhibited CBF changes in 5/6 trials and cortical electrical spiking (F 3,12 = 6.12, P < 0.05, n = 6) across the cohort. Specifically, when the responses after (R)-baclofen were compared with the control, CSD electrical activity (t 5 = 2.65, P < 0.05, n = 6) was significantly inhibited.
Blood pressure changes
Summary of mean arterial blood pressure changes with drug intervention

P < 0.05 significant change from baseline.
↓↑, direction of blood pressure change.
Discussion
The data demonstrate that an AMPA receptor antagonist was able to inhibit the vascular changes and neuronal discharge occurring during cortical mechanical stimulation and the time to onset of the hyperaemic response, although not the speed of propagation. This suggests involvement in the initiation but not propagation of the blood flow changes. Both GABAA and GABAB receptor agonists inhibited the wave of hyperaemia and neuronal firing in the rat model, but much less so in the cat. The GABAA agonist also inhibited the speed of propagation, indicating that it may affect the propagation of the blood flow changes.
Among the considerations, the data do highlight a disparity between cat and rat models, similar to that found with other studies (9,23), which may be explained by the developmental differences between the two species (35); however, a possible influence of the different anaesthetic agents used cannot be ruled out. It has been shown that different anaesthetic regimes can alter CSD susceptibility; however, this is more common for the inhalation anaesthetics such as halothane (36). Pentobarbital anaesthesia was shown to result in higher CSD frequencies when compared with α-chloralose in the KCl induction method (29) and, as such, is unlikely to explain the reduced levels of CSD observed in the pentobarbital anaesthetized rat when compared with the α-chloralose-anaesthetized cat in this study. While sustained hypoperfusion is a prominent and long-lasting phenomenon following CSD, there is evidence for cortical laminar differences (37), with pial vessels demonstrating a biphasic transient hypoperfusion followed by a more sustained hyperaemic response (38). As such, laser Doppler flowmetry is more biased towards superficial layers, especially in larger mammals, introducing a likely species variation, and the overall oligaemic response may not be seen, as is the case with laser speckle imaging in the cat (39).
Glutamate receptor agonists that activate quisqualate, kainate and NMDA receptors are all able to induce CBF changes (40) and an NMDA receptor antagonist was able to inhibit the hyperaemia induced by cortical mechanical stimulation (30). In the data presented we show that an AMPA receptor antagonist was able to inhibit the hyperaemia in nearly half of cats tested, and the hyperaemia and neuronal discharge in all rats. The time to hyperaemic response was also significantly altered, although the rate of propagation was not. This would seem to indicate some component of the activity of topiramate at the AMPA receptor may help to inhibit spreading depression in animal models and reduce the occurrence of aura in patients and therefore may be involved in the mechanism of aura.
GABAA and GABAB receptors, although apparently not directly involved in the initiation and propagation of CSD, may have a role to play in maintaining the state of excitation of the neurons, and if this is interrupted may contribute to inhibiting the changes that take place. We found that a GABAA receptor agonist was able to inhibit CSD-associated CBF changes in one of five cats and all rats, and a GABAB receptor agonist in two of six cats and five of six rats in keeping with the proposed use of baclofen in headache. It must be said that whereas the clinical reports are promising in migraine (41) and cluster headache (42), clinical experience suggests both randomized controlled trials are required and indeed that a more complex interplay is involved. Furthermore, the GABAA agonist was able to reduce significantly the speed of propagation, although Kaube and Goadsby (30) did find that this may be a time effect. Again, there is disparity between the cat and rat data which is interesting in terms of what this may imply for humans and may be explained by receptor differences both at the cortical and sub-cortical levels (43,44). Certainly the rat data imply that GABAergic activation by topiramate and valproate may contribute to their ability to inhibit CSD and reduce migraine aura. It would be premature to conclude that GABA receptor activation plays a crucial role in the CBF changes given the number of non-responders; however, it is clear that manipulating the state of excitability of the cells with GABA changes the likelihood of blood flow changes occurring. It is reported that the role of glutamate may be important in creating the wave seen in spread of blood flow changes (45); perhaps GABA can contribute to this by stabilizing the excitable state of the neurons. It must also be noted that the interventions reported are single dosing regimes in each animal and, as such, a lack of effect does not rule out a possible action with longer chronic administration. In this regard, it has previously been shown that following chemical activation only repeated dosing was able to inhibit the number of CSDs initiated (16). Although it is convenient to think mechanistically about preventive and acute administration/treatments being different in principle, this is not as clear as it is painted. Some medicine classically considered preventives, such as flunarizine (46), have been shown to be affected in randomized controlled trials in acute therapy (47), whereas the time-honoured preventive methysergide (48) was the pharmacological inspiration for the most successful acute treatments yet developed—the triptans (49).
The differences between the cat and rat results are potentially challenging. The fact that some animals responded implies modulating GABA transmission has some effect. Why is there a variation in the species here? The greatest difference between rat and cat brains is there level of development. Cats are gyrencephalic with a more complex neural network. Rats are lissencephalic, therefore the cerebral cortex is smooth and less complex. This may contribute to the responsiveness of the two brains, as was also seen with topiramate (9); however, the use of different anaesthetic agents must also be highlighted, as previously discussed. Another known difference between the species is that CSD inhibits cerebrovascular responses to hypercapnia for 8 h in the cat (28), but for only 1 h in rats (50,51), indicating a clear difference in their physiological responses with regard to CSD. It would seem that the variability of response in the cat better models the clinical condition, where not all patients respond identically to migraine preventives, while the seemingly all-or-nothing response in the rat is ideal for understanding the mechanism of the vascular changes and predicting what pharmacology may be involved in CSD.
The cat data may also be indicative of the very specific pharmacology of the agonists and antagonists used. We used a very specific and potent AMPA receptor antagonist and GABA receptor agonists to dissect the receptor populations involved. We found virtually 100% response in rat, and a partial response in cat, with their more highly developed cortex. Some migraine preventives have actions at three or more different sites, and therefore the cumulative effect of this action at the different receptor systems, particularly in cats, as was found with topiramate (9) and in clinical studies, is likely to be a cumulative affect on these different pharmacologies. Perhaps these data partly explain the apparent conundrum that pharmacologically dirtier medicines, such as topiramate (52) and valproate (53), work in migraine, whereas more specific anticonvulsants, such as oxcarbazepine (54), do not.
In summary, AMPA as well as NMDA receptors seem to influence the initiation and, more predominantly, the propagation of the spreading oligaemia and are likely to be among the targets in reducing frequency of migraine aura of topiramate. GABAA and GABAB receptors, although perhaps not playing a major role in the initiation and propagation, may play a role in increasing the likelihood of vascular changes and therefore of aura to occur. As such these receptors may also contribute to the preventive action of both topiramate and valproate. It does seem clear that the cumulative impact of topiramate and valproate in many pharmacological systems contributes to their efficacy as therapeutics.
Footnotes
Acknowledgements
The authors thank Kevin Shields and Paul Hammond of the Headache Group for both assistance and technical support during these experiments.
