Abstract
Cutaneous allodynia (CA), pain in response to innocuous cutaneous stimuli, is recognized as a sign of central sensitization during migraine episodes. It is either restricted within the pain area on the ipsilateral head, or extends within and outside the head. Moreover, CA can be elicited in response to thermal (heat or cold) and/or mechanical stimuli. This raises the question as to whether cephalic and extracephalic CAs share the same properties. We assessed cephalic and extracephalic CAs in migraine episodic patients using a questionnaire completed at home during migraine attacks. A total of 67 episodic migraine patients (58 women, nine men; 40 ± 13 years old) addressed all questions in the questionnaire. Forty-nine patients (73%) cited one or more allodynic symptoms during or immediately after the migraine attack. Almost all 49 patients reported cephalic CA, whereas 24 (49%) also reported extracephalic CA. Occurrence and extension of CA correlated (P = 0.005) with headache intensity. Modalities of cephalic and extracephalic CA were different (χ2 = 12.03; P = 0.002), extracephalic CA being mostly thermal (75%) whereas cephalic CA was mostly mechanical (92%). This suggests that cephalic and extracephalic CAs involve different mechanisms.
Introduction
Cutaneous allodynia (CA), perception of pain when a non-noxious stimulus is applied to normal skin, is common in migraine. Recently, it has attracted intense interest because it is recognized as a sign of central sensitization during migraine episodes and a risk factor for progression into chronic migraine.
Numerous studies, using quantitative sensory testing (QST) and/or a questionnaire [see (1, 2) for review], have now reported CA in episodic migraine patients, although with very different prevalence. Using a semistructured interview, CA was found in 50–75% of episodic migraine patients (3–5). In studies using the QST, the prevalence of allodynia ranged from 20 to 80% (4,6–9). Other studies, using a questionnaire that is completed by patients at home during migraine attack, found CA in 63–75% of episodic migraine patients. (8,10,11). This variability is, at least, imputable to methodological differences in definition and evaluation of allodynia (12), and the size and type of population under study (1).
However, CA in episodic migraineurs can be restricted to the pain area on the ipsilateral head or occur within and outside the head (3,6,8). Moreover, CA can be elicited in response to thermal (heat or cold) and/or mechanical stimuli (6). This raises the questions as to how cephalic and extracephalic CAs relate to migraine and whether they share the same features. In the present study, using a questionnaire very similar to that of Jakubowski et al. (4) and Ashkenazi et al. (8), we addressed two questions: (i) what is the prevalence of cephalic and extracephalic CA in a cohort of headache out-patients of a Neurology Department in a University Hospital, and is the occurrence of allodynia related to any feature of episodic migraine?; and (ii) what are the clinical features of cephalic and extracephalic CA that develop during the course of migraine attacks?
Methods
The study was approved by the local Ethics Committee (Auvergne). We recruited patients from our out-patient clinic who met the International Classification of Headache Disorders, 2nd edn criteria for episodic migraine. Excluded from the study were patients with traumatic or neuropathic disorders and any sensory symptoms unrelated to migraine. Allodynia was assessed using a questionnaire. The questionnaire and cover letter explaining the aim of study (description of symptoms in relation to migraine pain to better understand the mechanisms underlying this disease) were mailed to all patients on the same day. Patients were instructed (i) to read carefully the questionnaire before the first migraine attack, (ii) to fill it when having an acute migraine attack, (iii) to notice the time at which the questionnaire was filled, and (iv) when completed, to mail it back to the clinic.
The questionnaire included information on demographic features (age, gender), headache history (estimated number years with headache, estimated frequency), characteristics of migraine attack (location, intensity, time of onset and end of attack), and migraine treatment (acute treatment of migraine, preventive treatment of migraine, onset of pain relief). Global headache intensity was scored on a scale from 0 (no pain) to 10 (extremely intense).
The questionnaire assessed symptoms of cephalic CA as follows: do you experience pain or unpleasant sensation on your skin during a migraine attack when you engage in any of the following activities (Yes, No, or Not Applicable): combing your hair; pulling your hair back (e.g. ponytail); shaving your face; wearing eyeglasses; wearing contact lenses; wearing earrings; wearing necklaces; wearing anything tight on your head or neck (hat, scarf); wearing a finger ring; taking a shower (when shower water hits your face); resting your face on the pillow on the side of the headache; being exposed to heat (e.g. cooking; placing heating pads on your face); or being exposed to cold (e.g. breathing through your nose on a cold day; placing ice packs on your face).
The questionnaire also assessed symptoms of extracephalic CA as follows: do you experience pain or unpleasant sensations outside the face during a migraine attack when you engage in any of the following activities: wearing tight clothes, wearing anything on your arm or wrist (bracelet, watch), being covered with a heavy blanket; taking a shower; being exposed to heat, being exposed to cold. If you experience pain or unpleasant sensation on your skin outside the face, does it take place on the left/right harm, hand, leg, foot or trunk? Patients had to select the best proposition. Patients reporting at least one of these clinical indicators were considered as allodynic.
Results are expressed as mean ± S.E.M. Statistical analysis was performed using Student's t-test, one-way analysis of variance (
Results
Types of medication used for treating acute migraine attacks

NSAID, non-steroidal anti-inflammatory drug.
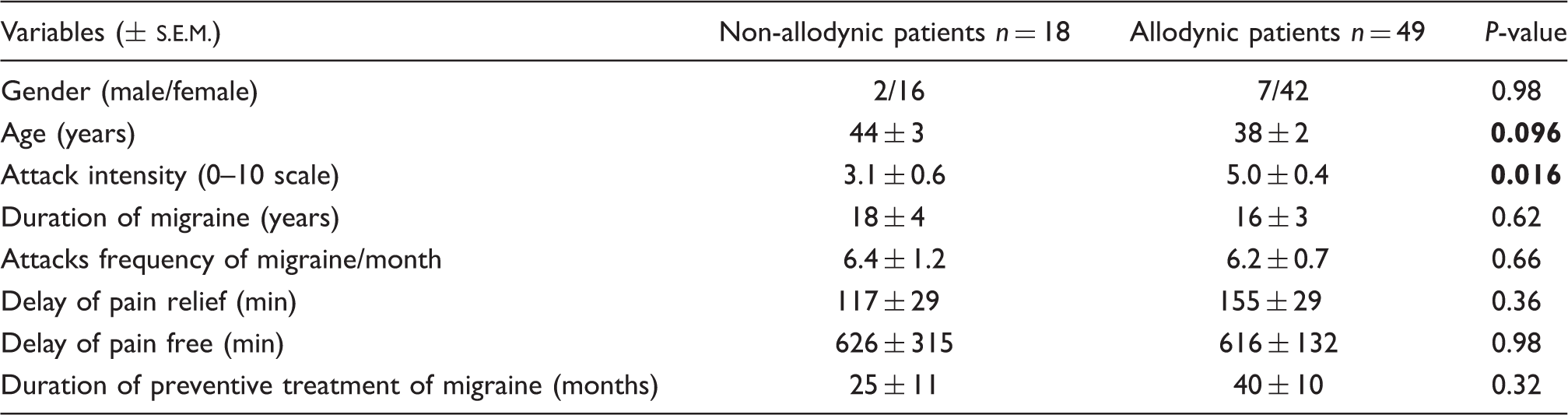
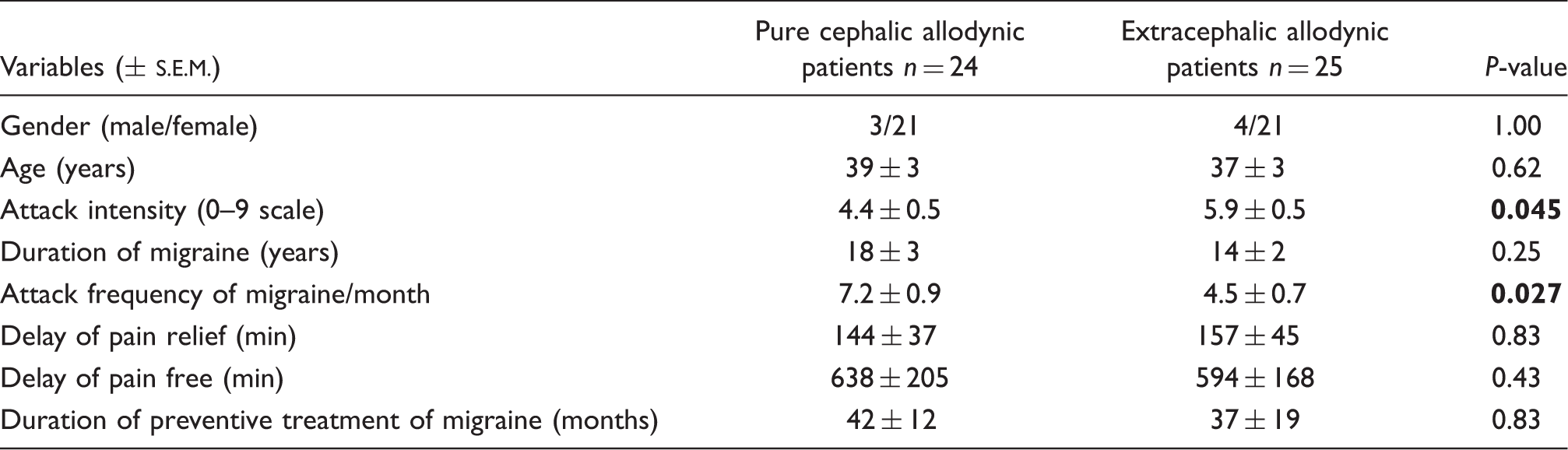
Of the 67 patients, 49 (73%) cited one or more symptom of skin hypersensitivity (allodynia) during or immediately after (within 24 h) the migraine attack (Fig. 1, Table 2). Allodynic symptoms involved cephalic (scalp, face, neck, ears) and/or extracephalic (limbs, trunk) territories (Fig. 1). Of the 49 patients classified as allodynic, 48 (98% of allodynic patients) reported cephalic, 24 (49%) pure cephalic allodynia, 24 (49%) both cephalic and extracephalic and one (2%) pure extracephalic allodynia. There is a correlation (P = 0.005) between the intensity of headache pain and the location/extension of allodynia, with pain intensity progressively increasing for non-allodynic (3.1 ± 0.6), pure cephalic allodynic (4.4 ± 0.5) and extracephalic allodynic (5.9 ± 0.5) patients (Fig. 2). Furthermore, patients with extracephalic allodynia had a lower frequency of attacks than patients with pure cephalic allodynia (P = 0027; Table 3). On the other hand, the presence of allodynia was unrelated to attack frequency, delay for pain relief and pain free, treatment of migraine attack, duration of preventive treatment of migraine, disease duration and age (Table 2).
Percentage of migraine patients with allodynia. Migraine attack intensity in allodynic and non-allodynic patients. Demographic and clinical characteristics of migraine attacks in patient with or without cutaneous allodynia Demographic and clinical characteristics of migraine attacks in patients with pure cephalic allodynia or patients with cephalic and extracephalic allodynia

The modalities of cephalic and extracephalic CA are different (χ2 = 12.03; P = 0.002). Cephalic CA was mostly mechanical (92%). Prominent scalp symptoms were: sensitivity to touch; soreness or tenderness; difficulty brushing, combing, or washing hair; difficulty lying with head resting on the side of allodynia; and circumscribed area of hypersensitivity and tenderness. Mechanical allodynia was either isolated or associated with thermal allodynia in 52% and 40% of the patients, respectively. Pure thermal allodynia was rarely reported (8%) (Fig. 3). Extracephalic allodynia (including trunk or limbs bilaterally in 2/3 of patients) was mostly thermal (75%). Thermal allodynia could be alone (42% of patients) or associated with mechanical allodynia (33% of patients). Pure mechanical allodynia was observed in only 25% of extracephalic allodynic patients.
Sensory modality of allodynia.
Discussion
We assessed the presence, localization and features of CA in patients with episodic migraine using a questionnaire. CA was observed in 73% of patients. The two main findings of this study are that (i) the location of CA—cephalic and cephalic + extracephalic—is correlated with the intensity of headache pain, and (ii) cephalic allodynia is mostly mechanical, whereas extracephalic allodynia is mostly thermal.
We used a questionnaire. The reliability of the questionnaire in identifying allodynia has recently been validated by comparing its outcome with that of QST (4, 8). Thus, the questionnaire appears to be highly reliable in detecting allodynic patients.
The prevalence of allodynia here is similar to that previously observed in migraineurs seeking secondary and tertiary care (4, 8), but slightly higher than that reported in a population-based study (11). Interestingly, however, these are two different types of population with the patients from subspecialty headache clinics (i.e. highly selected populations probably with more severe migraines) showing the highest prevalence of CA.
Our results show that the presence of CA is not related to the frequency of migraine attack, disease duration or age but with the intensity of headache pain. This last result is in agreement with previous work (7–9,11). We have shown here, in addition, that the location—and probably the extension from cephalic to cephalic and extracephalic—of CA is correlated with pain intensity.
The triptans, a family of 5HT1B/1D receptor agonists, are commonly prescribed migraine drugs. For many migraine patients, however, triptan therapy appears to provide complete pain relief in some attacks but not in others. Burstein et al. (13) have shown that it is the presence of allodynia that is detrimental to successful triptan therapy. However, this conclusion has recently been challenged (14–16). Moreover, others have shown that headache intensity at treatment intake, rather than allodynia, significantly influences most outcome measures in attacks treated with triptans (9, 17). Our result, that the presence of CA is related to the intensity of headache pain, can account for these apparently contradictory results. On the one hand, patients who develop allodynia during the course of an attack might thus have the highest headache intensity at treatment intake, and they should be rendered pain free when triptan therapy is administered before rather than after the establishment of cutaneous allodynia and high-intensity pain. On the other hand, those who never develop allodynia and have low headache intensity are likely to be rendered pain free at any time after the onset of pain.
Very few studies have actually assessed the anatomical location of CA in migraine. Burstein et al. (6) initially found allodynia in extracephalic areas in 73% of allodynic patients. Extracephalic CA has since been reported in patients with episodic (26.5% of patients (3); 49% in the present study) as well as chronic migraine [66% of patients (18)]. The nature of such a symptom is reminiscent of the fibromyalgia syndrome, characterized by diffuse pain. Fibromyalgia is observed in 22–36% of migraine patients (10, 19). It has been hypothesized that episodic migraine and fibromyalgia belong to the same disorder (19, 20) and that a common mechanism underlies the widespread hypersensitivity in fibromyalgia and migraine patients.
Cephalic and extracephalic allodynia exhibit different features, with the former being mostly mechanical (92% of patients) and the latter thermal (75% of patients). That cephalic allodynia is mostly mechanical is consistent with previous studies using either QST (4,6,7) or questionnaire (4, 8). Also, Burstein et al. (6), using QST, found that extracephalic CA is mostly thermal (∼60% of their patients). Interestingly, it has been reported that migraine patients have a lower heat pain threshold within 24 h before their next migraine attack, compared with paired interictal recording, for the forehead, neck and hand (21). Similarly, widespread thermal hypersensitivity has also been reported in fibromyalgia (22).
Thus, patients with episodic migraine can exhibit both cephalic and extracephalic CA, but the occurrence of cephalic and extracephalic CA is correlated with headache intensity. The modalities of these two CAs are different. This suggests that cephalic and extracephalic CA involve different mechanisms recruited by different levels of pain experience.
That in patients suffering from chronic painful neuropathies and in normal subjects with acute experimental chemogenic pain the severity of mechanical allodynia correlates with the intensity of background pain (23) suggests that the greater the pain experience is the greater the consecutive changes in nociceptive pathways are. A short barrage of nociceptor input, very much like that occurring during a migraine attack, is known to induce central sensitization, a form of use-dependent synaptic plasticity that outlasts afferent activation (24). One manifestation of such central sensitization is mechanical allodynia (6). Thus, use-dependent central sensitization, which probably follows the persistent excitation of unmyelinated primary afferents during the migraine attack, may account for cephalic mechanical allodynia. On the other hand, the mechanisms underlying extracephalic CA remain poorly understood. Extracephalic CA has been suggested to result from the hyperexcitability of cortical and subcortical brain regions (6). A recent animal study has shown that chemical inflammation of the dura elicits cephalic CA that extends beyond the trigeminal dermatome (25). Such extracephalic hypersensitivity requires the rostral ventromedial medulla (RVM), the chemical inflammation of the dura activating RVM pain-facilitating ‘on’ cells. In accordance with these findings, functional imaging studies (26, 27) performed on migraineurs show dysfunction of brainstem structures [i.e. the periaqueductal grey matter (PAG) and the nucleus cuneiformis (NCF)], that are prominent components of pain modulatory pathways (23). The RVM receives inputs from the PAG and NCF, and exerts bi-directional control over nociception under different physiological and pathophysiological conditions (28). Thus, we propose that the pronociceptive role of the descending modulatory circuit is critical for the development of extracephalic CA associated with headache. This is consistent with the evidence that thermal allodynia is mediated by C and A δ nociceptive fibres and descending control that originates from the PAG is highly selective for C and A δ nociceptive input (29). Amongst the many neurotransmitters in the brain, the serotoninergic system from the RVM has been implicated in migraine and fibromyalgia pathophysiology (30). Interestingly, one animal study has found that mice deficient in the serotonin transporter develop mechanical allodynia, but not thermal hyperalgesia after the nerve injury (31). Dysregulation of the serotonin transporter has also been reported in migraine (32) and fibromyalgia (33) patients. Altogether, these observations lead to the hypothesis that alteration of the serotonin transporter is one of the factors determining the development of thermal hypersensitivity in chronic pain states.
In summary, the present study has confirmed that cephalic and extracephalic CA are common in episodic migraine. It provides evidence for their occurrence being correlated with headache intensity and their modality being different. This suggests that cephalic and extracephalic CA involve different mechanisms, at different levels of the brain. Thus, assessing the localization and modality of CA might be pivotal to our understanding of migraine pathophysiology and the treatment of patients.
Footnotes
Acknowledgements
We thank Pr A. Artola for helpful comments on this manuscript and A. M. Gaydier for secretarial assistance. This work was supported by funding from Inserm, MENESR, CHU Clermont-Ferrand and Université Clermont1 (France).
