Abstract
The ability of tonabersat to relieve the symptoms of migraine attacks with or without aura was evaluated in a randomized, double-blind, placebo-controlled, multicentre, parallel-group study. Patients received 20 or 40 mg of tonabersat, or 50 mg of sumatriptan (positive control), or placebo at the onset of a moderate or severe attack. Headache intensity, relief and recurrence were recorded for 24 h after dosing. On the basis of primary or secondary efficacy measures, tonabersat did not provide a clinically or statistically significant advantage over placebo. Tonabersat generally was well tolerated and had no effect on vital signs, electrocardiogram recordings or laboratory values. The lack of efficacy may be a function of the slow absorption of tonabersat. As a consequence of slow absorption, daily administration of tonabersat as prophylaxis for migraine attacks is under investigation in ongoing studies.
Introduction
Despite the advent of the triptans, the potential side-effects, recurrence rate, lack of response in 30–40% of patients, and contraindications associated with their use underscore the need for new drugs in the acute treatment of migraine attacks (1–3). Tonabersat (SB-220453), a novel benzoylamino benzopyran compound, was developed for its ability to inhibit cortical spreading depression (CSD). CSD is a self-propagating wave of neuronal and glial depolarization that advances across the cortical mantle (4). CSD almost certainly causes the migraine aura, but only scarce data support the hypothesis that CSD also plays a role in migraine without aura (5–7). Tonabersat was effective in animal models of migraine—inhibiting neurogenic inflammation, blocking propagation of electrophysiological events, such as CSD, decreasing cortical nitric oxide concentrations during CSD, and inhibiting trigeminal nerve ganglion stimulation-induced carotid vasodilation (8–12). In these studies, tonabersat exerted no cardiovascular effects and was largely devoid of central nervous system adverse events (AEs). Tonabersat also has antiepileptic activity, and some antiepileptic drugs are effective in the prophylactic treatment of migraine (12). The overall broader spectrum of activity of tonabersat, compared with that of triptans, rests on a novel mechanism of action related to an unknown stereo-specific binding site thought to be associated with the neuronal–glial gap junction (13). Triptans show no affinity for that site (14).
For these reasons, tonabersat is a promising, novel approach to migraine treatment. Although the pharmacokinetic characteristics of tonabersat and its preventive effect against CSD indicate suitability as a prophylactic drug, it was decided first to test its efficacy in the acute treatment of migraine attacks (12). This would provide a good first look at the tolerability of tonabersat and the occurrence of any potential harmful side-effects.
This study was conducted to assess the efficacy and safety of tonabersat 20 or 40 mg taken at the onset of acute migraine. A positive control group received sumatriptan 50 mg.
Patients and methods
Patients
The study was conducted from July 1998 to December 1998 and patients were screened at 53 centres in Australia, Belgium, Denmark, France, Germany, Holland, Spain, South Africa, Switzerland, Sweden, the UK and the USA. Patients who met the inclusion and exclusion criteria (Table 1) were randomized to treatment in a 1 : 1 : 1 : 1 ratio using a computer-generated randomization list, and they were allocated in strict sequential order to an investigator-allocated treatment pack. The master randomization list was held by the sponsor (SmithKline Beecham), and individual sealed code envelopes were lodged with the investigator or pharmacist. A total of 688 patients were required to be enrolled in the study. Assuming a 25% dropout rate, this would allow 129 evaluable patients per treatment group, and the study would be projected to have at least 90% power to detect a difference of 20% between groups in the proportion of patients with headache relief 2 h post treatment.
Inclusion and exclusion criteria

Study design
This randomized, double-blind, placebo-controlled, multicentre, parallel-group study included four treatment groups: tonabersat 20 mg, tonabersat 40 mg, sumatriptan 50 mg and placebo. A single oral dose of test drugs or placebo was taken at the onset of a moderate (grade 2) or severe (grade 3) attack (dosing within 6 h after onset of attack). After taking study medication, patients recorded in a study diary details of the onset of their migraine, baseline headache intensity, presence of related symptoms and level of activity prior to treatment; these variables were then recorded again at 0.5, 1, 2 and 4 h post treatment. At 24 h post treatment, patients recorded their current headache pain, level of activity and any recurrence of headache symptoms, and assessed the effectiveness of the study medication. Patients also recorded the time of study medication intake, details of treatment with any other antimigraine medications, details of any adverse symptoms experienced and the time at which they had meaningful relief of their migraine symptoms. Patients attended a follow-up visit within 7 days of receiving treatment.
Headache parameters
The primary efficacy measure was headache relief, defined as the change in severity from grade 2 or 3 at pretreatment to grade 0 (none) or 1 (mild) at 2 h post treatment. Intensity was rated on the International Headache Society scale. Pain was scored in terms of its functional consequence on a verbal four-point scale: 0, no pain; 1, mild pain, does not interfere with usual activities; 2, moderate pain, inhibits but does not wholly prevent usual activities; 3, severe pain, prevents all activities. Secondary efficacy measures were headache relief at 1 and 4 h post treatment; abolition of headache at 1, 2 and 4 h post treatment; need for additional migraine medication within 60 min of the 2-h post-treatment assessment; and self-defined meaningful relief 2 h post treatment. Other efficacy measures included headache relief and abolition of headache at 0.5 h post treatment; headache recurrence between 2 and 24 h post treatment in the responder subpopulation; the effect of treatment on associated symptoms (nausea, vomiting, phonophobia, photophobia); and the use of additional migraine medication up to 24 h post treatment. Patients also provided an overall evaluation of study medication, compared study medication with their usual medication and assessed functional impairment. All assessments were done for the intent-to-treat (ITT) population.
Safety and tolerability measures
Routine laboratory tests, electrocardiogram (ECG) recordings and vital signs were performed at the screening and follow-up visits. AEs within 6 days of dosing were noted at follow-up. The investigators judged the relationship of AEs to the study drugs as not related, unlikely, suspected (reasonable possibility) or probable. Treatment-related AEs were those judged as suspected or probable.
Statistical analysis
Primary inference of the efficacy of tonabersat used the ITT population and the observed case dataset at 2 h post treatment. Patients were excluded from analysis if they took study medication when they did not have a headache or when their headache was mild in severity, ergotamine or derivatives (e.g. dihydroergotamine) or a triptan within 24 h prior to taking study medication, any analgesic within 6 h prior to taking study medication, any additional migraine medication within 2 h after taking study medication or a triptan or ergotamine or derivatives as additional migraine medication within 24 h after taking study medication. Logistic regression compared responder rates for both tonabersat doses with those for placebo. The modified Bonferroni method for multiple comparisons determined whether these comparisons were statistically significant. The study was not designed to compare statistically the efficacy of sumatriptan with tonabersat or placebo.
Results
Patient demographics and baseline characteristics
A total of 693 patients suffering from migraine headache with or without aura were screened. Of these patients, 667 were assigned to treatment and 541 took study medication (Fig. 1). Among the 126 patients who did not take study medication, reasons included being lost to follow-up after randomization, screening laboratory results that did not meet the inclusion/exclusion criteria, or study medication that was returned to the clinic at the follow-up visit. In the ITT population, 85% of the participants were women and > 95% were white (Table 2). The mean age in each treatment group ranged between 38.8 and 40.4 years. There were no statistically significant differences among groups for any demographic characteristic. Migraine attacks began at a mean age of 20 years, and the disorder had a mean duration of 20 years. Overall, 73.6% of patients had migraine without aura, 12.4% had migraine with aura, and 14% had migraine with and without aura. A monthly mean of 2.9 attacks with or without aura had occurred in the previous 6 months. Previous acute migraine medication included triptans (47%) or other analgesics (61%), or both. The vast majority of patients used medication when their migraine was still mild or moderate to severe, and 24% were very satisfied with their usual treatment.
Demographic characteristics for the ITT population

Patient disposition.
Between 56 and 69% of each group had had migraine for 2–6 h before receiving study medication. Intensity at treatment was moderate for 60–65% of patients and severe for 35–40%. Almost all patients experienced at least one other symptom; photophobia (71–78%) and phonophobia (65–74%) were most common. Before ingestion of study medication, functional ability in the majority of patients was either mildly (39–53%) or severely (26–34%) impaired by the migraine. Overall, 87% of patients took concomitant migraine medication—paracetamol (39%), salicylic acid (16%), caffeine (20%), codeine (15%), sumatriptan (28%), zolmitriptan (9%) and ibuprofen (9%). This includes medications that the patient would normally take to treat a migraine attack. Treatment with these medications was therefore ongoing during the study period; however, they were not supposed to be used to treat the evaluable attack.
Efficacy of tonabersat
Primary efficacy results
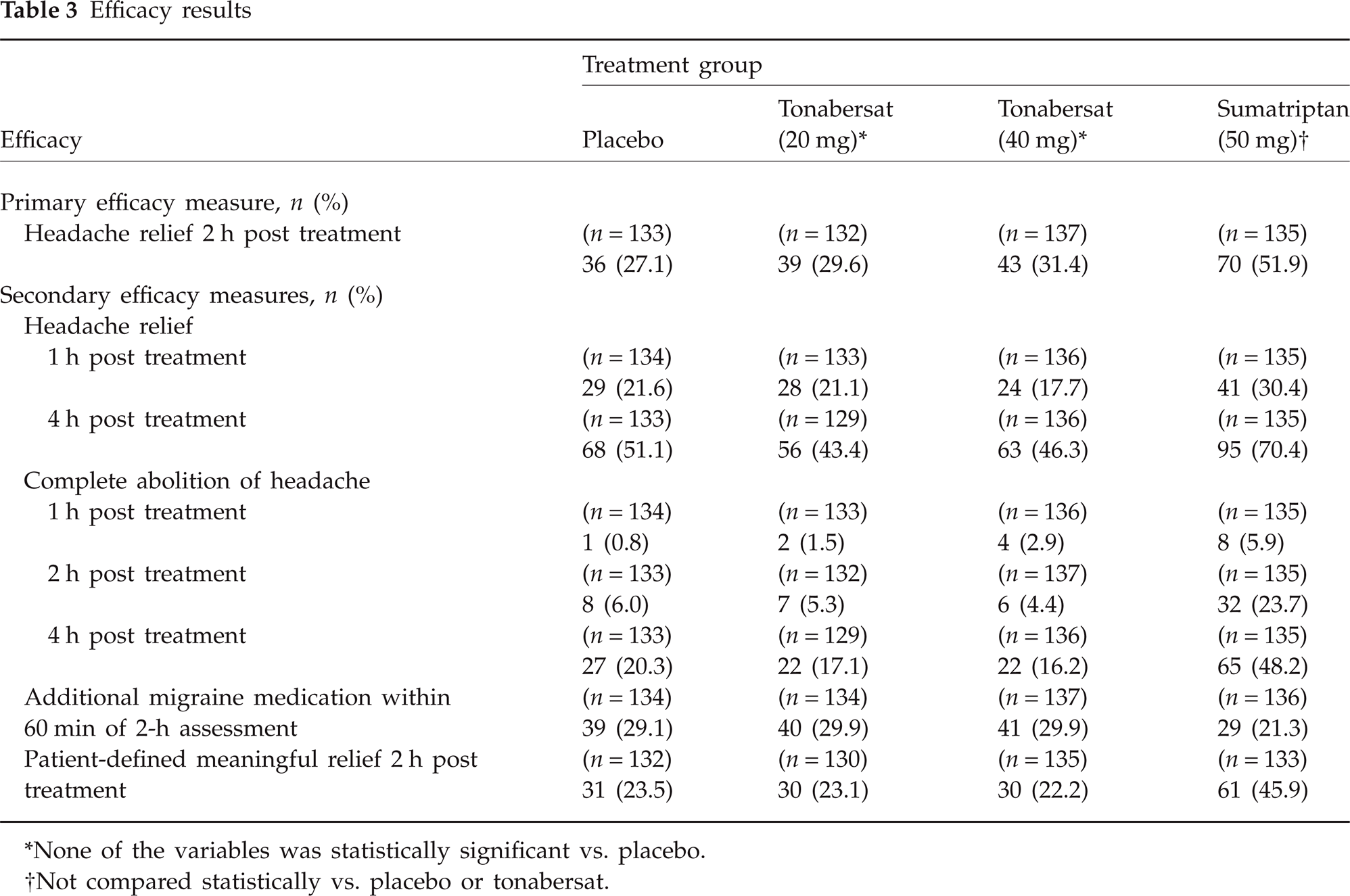
Tonabersat 20 or 40 mg did not provide a clinically or statistically significant advantage over placebo in the relief of acute migraine, according to the primary efficacy measure. Two hours after ingestion of tonabersat, 29.6% of patients taking 20 mg and 31.4% taking 40 mg experienced relief, compared with 51.9% of patients treated with sumatriptan 50 mg and 27.1% given placebo (Table 3).
Efficacy results
∗None of the variables was statistically significant vs. placebo.
†Not compared statistically vs. placebo or tonabersat.
Secondary efficacy results
Tonabersat had no significant advantage over placebo in acute migraine relief either 1 or 4 h post treatment. At 1 h, 21.1% of the tonabersat 20-mg group and 17.7% of the 40-mg group reported relief, compared with 30.4% of the sumatriptan group and 21.6% of the placebo group. Respective results at 4 h were 43.4 and 46.3% with tonabersat, 70.4% with sumatriptan and 51.1% with placebo (Table 3).
The proportion of patients reporting complete abolition of headache was similar with placebo and tonabersat but was considerably higher with sumatriptan at 1, 2 and 4 h. Approximately 29% of the tonabersat and placebo groups required additional migraine medication within 60 min of the 2-h post-treatment assessment, compared with 21% of the sumatriptan group. About 23% of patients experienced meaningful relief at 2 h with tonabersat and placebo, compared with approximately 46% of sumatriptan-treated patients. Overall, none of the treatment comparisons was statistically or clinically significant for any of the secondary efficacy parameters (Table 3).
Several additional efficacy variables were tabulated: (i) headache recurrence between 2 and 24 h post treatment; (ii) time to headache recurrence for responders to treatment at 2 h; (iii) headache relief 30 min post treatment; (iv) complete abolition of headache 30 min post treatment; (v) maintenance of headache relief between 2 and 24 h; (vi) use of additional migraine medication between 0 and 24 h; (vii) nausea, vomiting, photophobia or phonophobia in the 4 h post treatment; (viii) patient assessment of functional impairment at 30 min, and 1, 2, 4 and 24 h; and (ix) patient evaluation of study medication. Neither the 20-mg nor the 40-mg dose of tonabersat demonstrated statistically significant efficacy on any of these measures.
Safety
In the 6 days post treatment, 32% and 41% of patients receiving tonabersat 20 and 40 mg, respectively, reported AEs, compared with 32% of patients taking sumatriptan and 25% of patients given placebo. AEs occurring in at least 5% of any group were dizziness, nausea, vertigo and vomiting. Twenty-one percent and 32% of patients receiving tonabersat 20 and 40 mg, respectively, 20% of patients in the sumatriptan group and 14% of patients given placebo reported AEs that were suspected to be or probably related to study medication. Treatment-related AEs occurring in at least 3% of any group were dizziness, paraesthesia, vertigo, nausea and somnolence (Table 4). Although at least 14% of each group had treatment-related AEs overall, no single AE was reported by > 9.5% of patients in any one group. AEs generally appeared early after treatment, most within the first 2 h. Overall, severe AEs were limited to ≤ 11% of patients in any treatment group in the 6-day follow-up. Of the reports of dizziness and vertigo, ≤ 12% were severe, as were fewer than half of the reports of nausea and vomiting. In the ITT population, two serious AEs occurred post treatment, both considered by the investigator to be unrelated to study medication. One patient was hospitalized for longstanding depression, and another had excision of a malignant melanoma. No patients withdrew from the study because of AEs. No deaths were reported during the study or within 30 days of ingestion of study medication.
Adverse events (AEs) suspected or considered to be probably related to treatment∗

∗Occurring up to 6 days after treatment in ≥ 3% of patients.
Blood pressure and pulse rate results raised no safety issues; only four values, one in each treatment group, were of potential clinical concern at the follow-up visit. One patient, with previous ECG abnormalities, had an abnormal tracing at follow-up. Of the 541 enrolled patients, only 11 had clinical laboratory values of potential concern. Laboratory results did not differ in the active-treatment and placebo groups.
Discussion
CSD may have a role in the initiation of the migraine attack, as well as development of sensitization (15). The calcitonin gene-related peptide (CGRP) is a neuropeptide modulator within the pain control systems of the brain (16–19), which is released from peripheral trigeminovascular afferents, as well as brain neurons (20). Release of CGRP outside the blood–brain barrier (BBB) leads to vasodilation and plasma leakage within the dura mater (21, 22). Within the brain (inside the BBB), CGRP causes sensitization of the trigeminal system and facilitation of normally filtered external (afferent) sensory impulses (16–18, 20, 23).
Migraine is associated with neurological aura in about one-third of sufferers. The aura of migraine is typically visual or sensory, and is accompanied by an initial frontal hyperaemic phase followed by a spreading oligaemia (24–26). The observation of a hyperaemic phase reinforces the validity of comparisons of migraine aura with CSD (27). The slow movement of a CSD wave (2–4 mm/min) along the cortex is associated with the release of potassium, arachidonic acid, nitric oxide and proteases (28). As a consequence, CSD alters the permeability of the BBB and activates matrix metalloproteinases (MMPs), in particular, MMP-9 (29). A recent positron emission tomography study showed that posterior cerebral hypoperfusion, which accompanies migraine attacks with aura, could also be present in migraine attacks without aura (6). In addition, the headache begins even while the oligaemia is still present (30). The diminution of cerebral blood flow during CSD does not generally fall below the ischaemic threshold at the macroscopic level (28). Overall, these study results suggest that the aura is not caused by vasoconstriction and the headache is not due to reactive vasodilation as Wolff had once considered (31).
All things considered, migraine can be viewed as a failure of normal sensory processing (32). The initiating events of migraine appear to occur through peripheral sensitization of dural and meningeal nociceptors, which send increasingly intense stimuli to the spinal cord. Central sensitization in response to these stimuli is associated with abnormal neuronal excitability in the trigeminal nucleus caudalis, and amplification of pain (33). Central sensitization, in turn, may be responsible for cutaneous allodynia and hyperalgesia, which are common in migraine (32). The presence of neuropathic-like pain components of migraine pathophysiology may suggest mechanism-based treatment strategies that can improve the acute and preventive management of migraine attacks and also provide opportunity for the discovery of novel therapeutic targets (34).
In patients suffering from migraine attacks, with or without aura, tonabersat 20 and 40 mg showed no statistically significant advantage over placebo in headache relief determined 2 h after treatment (primary end-point). In addition, neither dose of tonabersat, compared with placebo, had an effect on any secondary outcome variables (e.g. complete abolition of headache at 2 h and headache relief at 1 and 4 h). By contrast, sumatriptan, though not compared statistically with placebo or tonabersat, produced significant relief and abolition of headache within the range expected from published literature on triptans (1–3).
The efficacy of tonabersat in the acute treatment of migraine attacks has also been assessed in two other large clinical studies (35). In one international dose-ranging trial, significantly more patients who received tonabersat 40 mg than those who received placebo experienced headache relief at 2 or 4 h, or complete abolition of headache at 4 h. A North American dose-ranging study, however, showed no statistically significant benefits of tonabersat compared with placebo, perhaps as a result of previous use of triptans. In the study reported here, sumatriptan-naive patients who took tonabersat 40 mg [23 of 54 patients (42.6%)] reported similar response rates at 2 h to those who took sumatriptan [25 of 57 patients (43.9%)]. The effects of previous triptan use on the efficacy of tonabersat may require further study.
In pharmacokinetic analyses, the time to maximum absorption (T max) of tonabersat occurred 1–2 h postdose in healthy subjects, although substantial delays ensued when tonabersat was taken within 6 h of the onset of migraine. Therefore, tonabersat is absorbed relatively slowly, and the 2-h acute efficacy time point in this study is earlier than the time to maximum plasma concentration. Assuming that higher plasma tonabersat concentrations would be efficacious, slow absorption could result in an insufficient therapeutic effect on acute migraine when dosing is initiated several hours after symptoms begin.
Slow absorption, however, would not be expected to limit prophylactic use. One Phase 2a prevention study demonstrated a small reduction in the number of migraine days and migraine attacks in tonabersat-treated patients, who, compared with a placebo group, required less rescue medication, reported less severe headaches and had an overall higher responder rate (50% reduction in migraine attacks) (36). Two ongoing prophylaxis studies are examining once-daily administration to determine whether tonabersat can decrease the number of migraine attacks.
The present study has demonstrated that tonabersat was well tolerated and raised no major safety concerns. The incidence of treatment-related AEs generally was numerically higher and dose dependent in patients receiving tonabersat; however, no single AE was reported by > 9.5% of patients in any one group. Very few cardiovascular AEs were reported in any of the groups, and no abnormal ECG results at follow-up were considered significant to study medication. An earlier study has shown that tonabersat does not constrict the coronary artery, saphenous vein and cardiac trabeculae and is not expected to cause adverse cardiac side-effects (37). In general, very few patients had laboratory or vital sign values of potential clinical concern, and there were no differences in this regard between active treatments and placebo.
In this double-blind, placebo-controlled study, tonabersat 20 or 40 mg showed no difference compared with placebo in the acute treatment of migraine attacks; however, based on pharmacokinetic data, tonabersat is being studied for use as a prophylactic agent. Tonabersat generally was well tolerated and had no effect on vital signs, ECGs or laboratory values.
Study investigators
Footnotes
Competing interests
J.O. has within the last 2 years received grants/research support from and/or has been a consultant/scientific advisor for, and/or has been on the speakers bureau of Merck and Company, Pfizer Inc., Minster Pharmaceuticals, RoxRo Pharma, MAP Pharmaceuticals, Neur Axon, and Lundbeck; C.G.H.D. and A.W.H. declare no conflicts of interest.
Acknowledgements
R. Blumenthal, PhD (RBH Associates, Inc., Elmhurst, NY, USA), and P. Kontur, PhD and M. Kersting, PhD (JL Shapiro Associates Inc., Edison, NJ, USA) assisted with manuscript preparation with financial support from Minster Pharmaceuticals.
