Abstract
Between January 2007 and March 2008, we prospectively studied all patients operated on for intracranial tumours in our Department of Neurosurgery. Preoperatively, all patients were interviewed by a neurologist to collect headache characteristics. Measurements of tumour and oedema volume were made using dedicated software for magnetic resonance imaging studies. Tumour histopathology was established by histological examination postoperatively. If headache improved postoperatively, a diagnosis of ‘headache attributed to intracranial neoplasm’ was made, according to the 2004 International Classification of Headache Disorders (ICHD-II). A multivariate logistic regression model was used to evaluate the association of headache with potential risk factors. We studied 206 subjects. The prevalence of tumour headache was 47.6%. Intracranial tumour headache was non-specific and in most cases could not be classified by current ICHD-II diagnostic criteria for primary headache syndromes. Its prevalence varied depending on volume, location and type of tumour, as well as on the patient's previous headache history.
Keywords
Introduction
The prevalence of headache in patients with intracranial tumours varies in different epidemiological studies. Considering only the studies performed after the advent of modern neurodiagnostic techniques, it ranges between 32.2% and 71% in unselected series (1–5). Likewise, data regarding intracranial tumour headache features and its relationship with tumour characteristics are often discordant. These discrepancies are probably due to differences in patients' samples, study methodology and intracranial tumour headache definition. In fact, although the 2004 International Classification of Headache Disorders (ICHD-II) in subchapter 7.4 (6) clearly states that a diagnosis of ‘headache attributed to an intracranial neoplasm’ can be made only if it resolves after surgical removal or volume reduction of neoplasm, few longitudinal studies have been performed (3,7,8). The main purposes of the present study were to determine the prevalence and clinical features of intracranial tumour headache as defined by ICHD-II (6) among adult patients undergoing surgery and to assess the influence of clinical-demographic variables and tumour histopathology, location and size on the occurrence and on the characteristics of headache.
Patients and methods
Study population and procedures
From January 2007 to March 2008, we examined all patients operated for intracranial tumours at the Department of Neurosurgery (University Hospital of Udine, Italy). Exclusion criteria were age < 16 years, multiple tumours, recurrent tumours, no availability of magnetic resonance imaging (MRI) and severe aphasia or disturbance of consciousness. Informed consent was obtained. The diagnosis and location of the tumours were established by MRI scan and by histological examination postoperatively (for pituitary adenoma, the subgroup was defined both by serum levels of hormones and by histological examination). Preoperatively, all patients underwent a thin-slice (1 mm) high-definition MRI for the neuronavigation planning with an Avanto Siemens Scanner (Siemens, Munich, Germany) with a superconductive magnet and field strength of 1.5 T. All examinations included axial, coronal and sagittal T1- and T2-weighted before and after intravenous administration of a gadolinium-based contrast medium. The areas of the tumour and oedema were determined from the axial MRI showing the greatest lesion size and were defined as the product of the greatest length by the greatest width (in cm). After the tumour was encircled and converted into a three-dimensional object, volume assessment was performed with the Medtronic Stealth-Station Treon Surgical Navigation Technology (Medtronic Surgical Navigation Technologies, Louisville, CO, USA). The volume of the surrounding oedema was calculated in a similar fashion. The amount of supratentorial midline shift in millimetres was measured on the axial MRI using the midline structures as landmarks. Increased intracranial pressure (ICP) was defined as the presence of papilloedema at ophthalmoscopic examination or obstructive hydrocephalus on MRI. Preoperatively, all patients underwent a clinical interview and a complete physical and neurological examination, which were performed by one of the authors (L.V.). The interview followed a structured two-part questionnaire. All patients were invited to complete the first part, which included basic demographic data, medical history, medical treatment history, family history of headache, personal history of primary headache (defined as ‘longstanding primary headache’ if the patient had suffered from primary headache for many years, as ‘remote history of primary headache’ if the patient had suffered from primary headache in the past and this headache was no longer present), presence of current headache (if the patient complained of headache in the period just before the diagnosis of brain tumour), and various neurological symptoms related to the tumour. Subjects with a newly appeared headache or with a noticeable change in pre-existing headache pattern were asked additional structured questions about different headache symptoms. The change of pattern was defined as an alteration of headache frequency, localization, severity or quality. Subjects with no history of headache or only with a remote one underwent no further evaluation. We recorded the course, frequency, duration, quality, intensity and location of headache and if there was an increase in intensity, frequency or duration of the pain (‘progressive pattern’ of headache). We also recorded timing of headache, influence on routine activities, interference with sleep, trigger factors, response to symptomatic drugs (painkillers) as well as occurrence of associated symptoms. Patients complaining of headache were interviewed again 3 months after the operation for recording response of headache to surgery. The follow-up included MRI control. If they became free of pain or markedly improved postoperatively, they were diagnosed to have ‘headache attributed to intracranial neoplasm’, according to the ICHD-II 7.4 definition (6). In each case, an attempt was made to classify the tumour-attributed headache in line with the ICHD-II (6), categorizing it as migraine-like, tension-type-like, or cluster headache-like.
Statistical analysis
The prevalence of headache was presented as the number and percentage of patients reporting the symptom. The associations of headache with characteristics of the patient and of the tumour were assessed through χ2 tests for categorical variables and through Wilcoxon's rank-sum tests for continuous variables (with results non-normally distributed according to the Kolmogorov–Smirnov test). The association between headache and the size of tumour and oedema was also assessed in analyses stratified by tumour histopathology. All factors with P ≤ 0.15 on univariate analyses were included as independent variables in a multivariate logistic regression model to evaluate the association of headache with each of those factors after adjusting for their potential mutual confounding effect. We did not include in this model factors that showed collinearity. Results are presented as odds ratios (OR) and 95% confidence intervals (CIs). Logistic models were stratified for patient's age (< 65 and ≥ 65 years). Characteristics of the headache are described among patients overall and by tumour histopathology, tumour location (supratentorial, subtentorial, skull base), tumour volume [categorized using tertiles as cut-offs: small (< 16 cm3), medium (16–33 cm3) and large (> 33 cm3)], oedema volume (0 vs. > 0), midline shift (0 vs. > 0) and increased ICP (presence vs. absence). Comparisons between groups of patients were carried out through χ2 tests. P-values < 0.05 were considered significant. The software used for statistical analyses was
Results
Study population
Two hundred and eleven patients were studied during these 18 months, of whom 116 (55.0%) complained of a newly appeared headache or of a changed pattern of a pre-existing headache. Out of these patients, 98 became free of pain or markedly improved postoperatively. They were diagnosed as having ‘headache attributed to intracranial neoplasm’ (subchapter 7.4 of the ICHD-II) (6). In 13 patients no change could be detected following the operation and the headache was considered to be independent of the tumour. Five patients were lost at follow-up and excluded from the study. Therefore, all results reported below refer to a sample of 206 subjects, of whom 98 (47.6%) will be defined as ‘patients with headache’ and the remaining 108 (52.4%) as ‘patients without headache’. The mean (
Intracranial tumour-attributed headache: Univariate analysis
Intrapersonal risk factors for headache development: univariate analysis of 206 patients with intracranial tumours
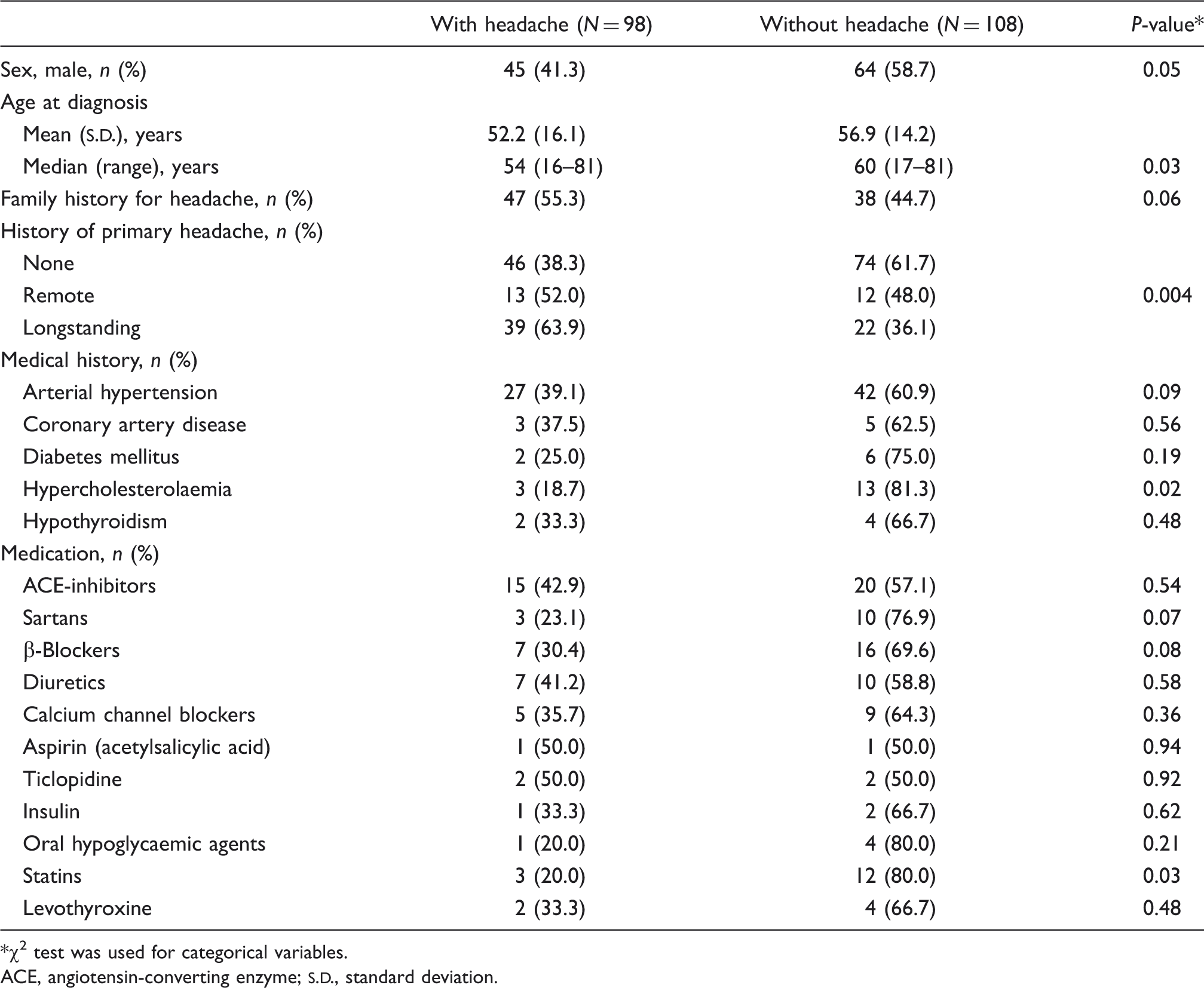
χ2 test was used for categorical variables.
ACE, angiotensin-converting enzyme;
Tumour-related risk factors for headache development: univariate analysis of 206 patients with intracranial tumours
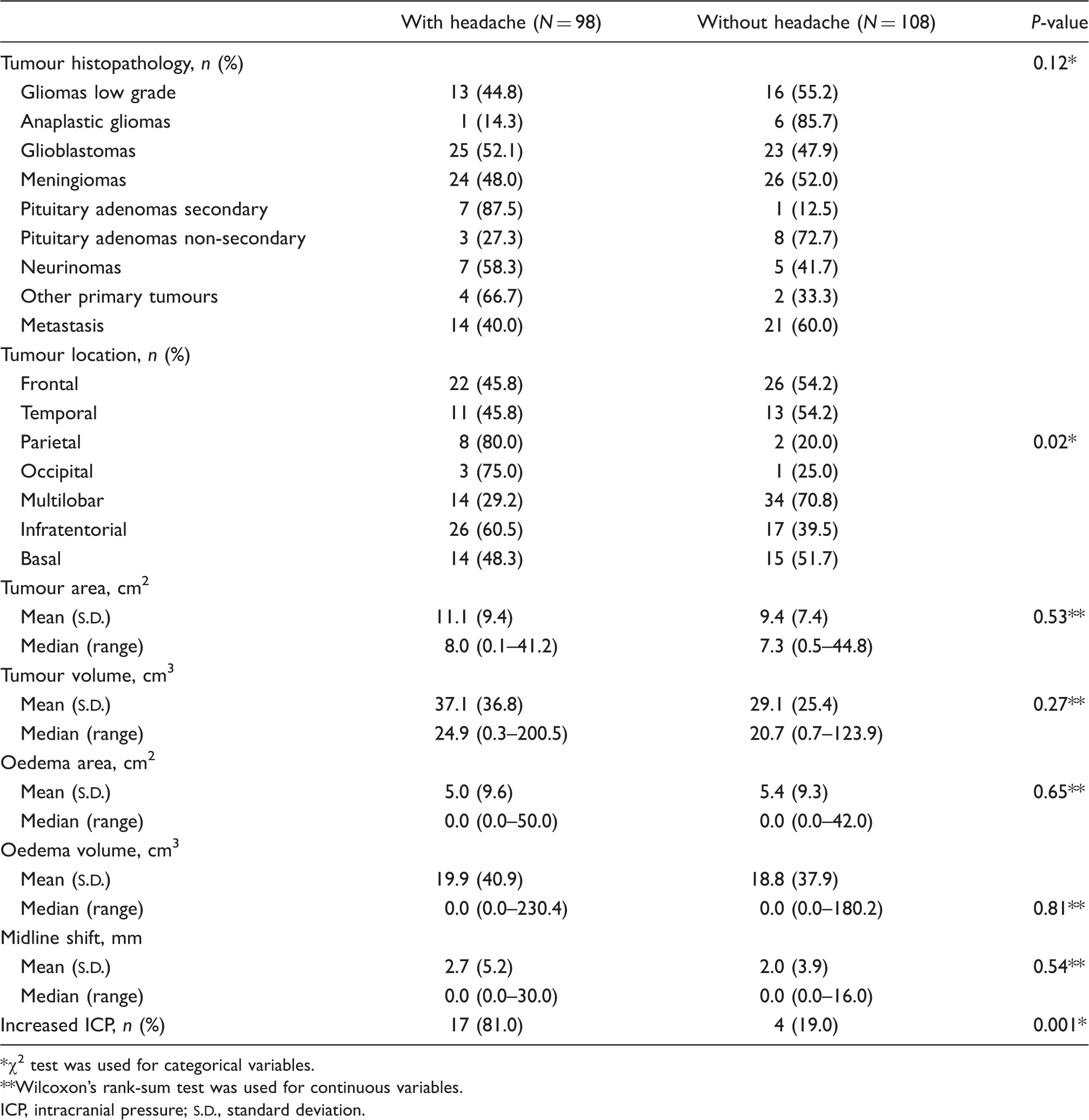
χ2 test was used for categorical variables.
Wilcoxon's rank-sum test was used for continuous variables.
ICP, intracranial pressure;
Intracranial tumour-attributed headache: Multivariate logistic regression analysis
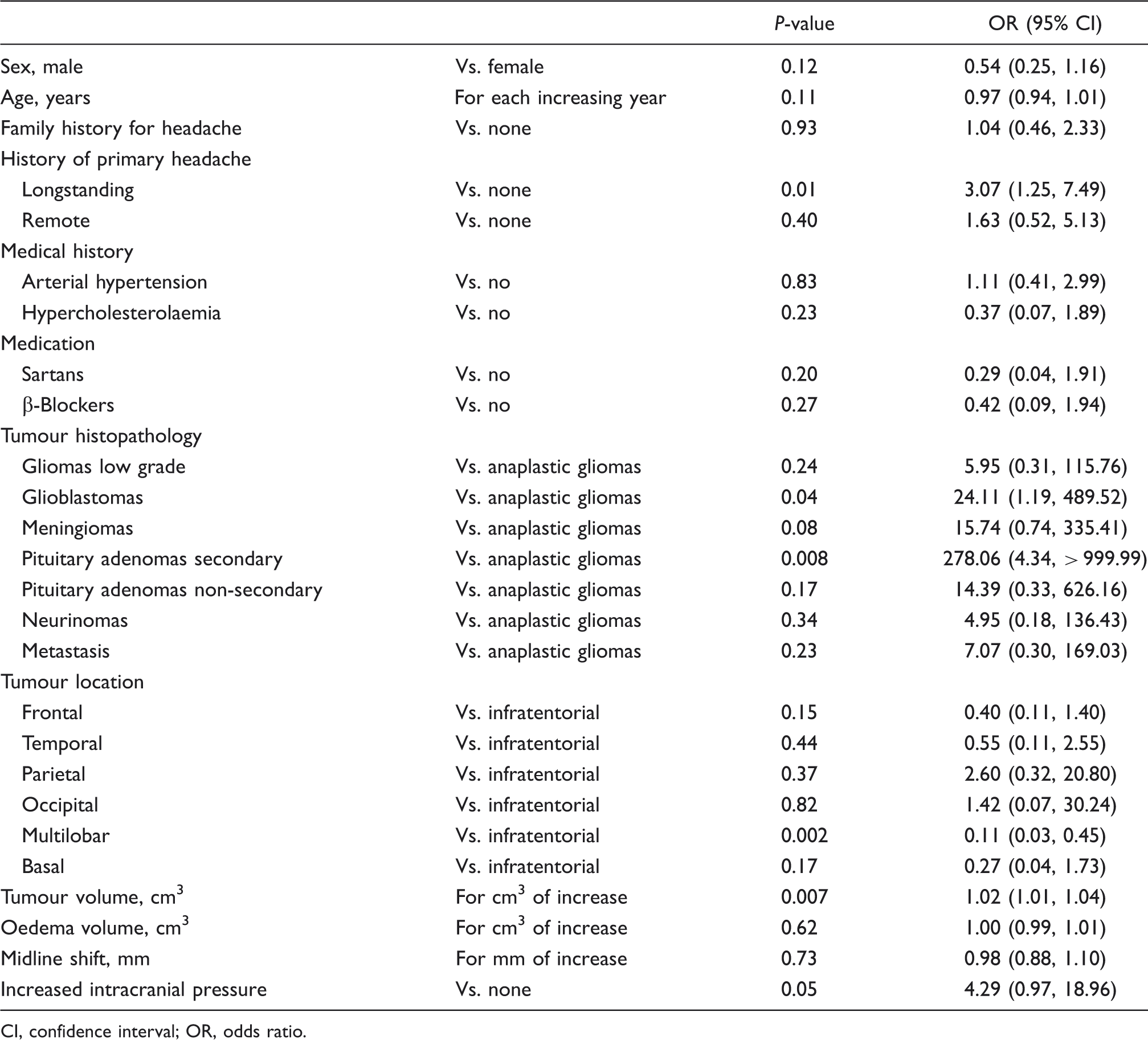
We performed a multivariate logistic regression analysis including in the model all variables with a P-value < 0.15 (Table 3), except statins, which were collinear with hypercholesterolaemia. These variables were sex, age, positive family history of headache, individual pre-existing headache, hypercholesterolaemia, arterial hypertension, intake of sartans, intake of β-blockers, tumour histopathology and location, and presence of increased ICP. In the multivariate analysis we included also the volume of the tumour, the volume of the surrounding oedema and the midline shift, which correlated significantly with the occurrence of the headache in a subgroup of patients (those with GBM). A personal history of remote primary headache (no longer present) did not significantly correlate with the occurrence of intracranial tumour headache, while a longstanding primary headache history was an independent risk factor (OR 3.07, 95% CI 1.25, 7.49; P = 0.01) (Figure 1). The probability of developing intracranial tumour headache was higher in patients affected by secreting adenomas (three producing prolactin, three growth hormone and one adrenocorticotropic hormone; OR 278.06, 95% CI 4.34, 999.99; P < 0.01) and GBM (OR 24.11, 95% CI 1.19, 489.52; P = 0.04) compared with those affected by anaplastic gliomas. In the group of patients with supratentorial multilobar tumours, the prevalence of headache was significantly lower than in those with infratentorial tumours (OR 0.11; P < 0.01). The presence of increased ICP was confirmed to have a marginally significant association with the occurrence of intracranial tumour headache (OR 4.29, 95% CI 0.97, 18.96; P = 0.05). Under the same histopathological diagnosis, for each cm3 of increase in tumour volume there was a significant increase in the risk of developing headache (OR 1.02, 95% CI 1.01, 1.04; P < 0.01). If the model was stratified for patient's age (< 65 and ≥ 65 years), an increasing risk of developing headache for each cm3 of increase in tumour volume was confirmed in the younger age group (OR 1.017, 95% CI 1.004, 1.030; P = 0.01) but not in the older one (OR 1.004, 95% CI 0.981, 1.027; P = 0.75). In younger patients both the presence of increased ICP (OR 10.173, 95% CI 1.962, 52.746; P < 0.01) and the increasing midline shift (OR 1.146, 95% CI 1.023, 1.283; P = 0.02) increased the probability of headache, whereas this did not happen in patients ≥ 65 years old. Finally, intracranial tumour headache was more common in the younger age group (51.4% vs. 39.4%), even if this difference was not significant.
Stratification of all patients with intracranial tumour (n = 206) by presence or absence of a history of primary headache. For patients with a history of primary headache, an additional subdivision was made according to the presence of current primary headache (longstanding primary headache) or absence of current primary headache (remote history of primary headache). We categorized patients with longstanding primary headache into those who presented with a relevant alteration of the pre-existing headache pattern (defined as change in frequency, localization, severity or quality) and those who reported a new type of headache (i.e. the patients recognized a difference from their usual headache symptomatology). The headache was attributed to intracranial neoplasm if there was marked postoperative improvement (or a return to previous pattern as in longstanding primary headache). The respective number of patients is shown in boxes. PO, postoperative. Risk factors for headache development: multivariate logistic regression analysis of 206 patients with intracranial tumours CI, confidence interval; OR, odds ratio.
Headache characteristics
Characteristics of headache attributed to intracranial tumour

ICHD-II, International Classification of Headache Disorders, second edition; NAS, nominal analogue scale.
Localizing value of the headache
A great localizing value of the headache (i.e. the headache overlies the projection of the tumour to the nearest skull surface) was found in 13 cases. Of the 29 patients with strictly unilateral side-locked headache, the location of the tumour was on the ipsilateral side in 24 cases, on the opposite site in two and in midline in three cases. Of the 60 patients with strictly bilateral headache, 53.3% had hemispheric tumours and only 25% had midline or bilateral tumours.
Headache characteristics and tumour pathology, location and size
Patients with secreting adenomas or GBM more often presented with progressive headache in comparison with those with meningiomas or low-grade gliomas (60.0, 40.0, 29.2 and 0.0%, respectively; P = 0.03). Phenotypes of intracranial tumour headache, classified according to the ICHD-II criteria (6) for primary headache, demonstrated a non-uniform distribution in various groups (P < 0.01). Meningioma-related headaches more often satisfied the criteria for tension-type headache (45.8%), whereas headaches related to GBM or to metastasis were more often non-classifiable (68.0% and 85.7%, respectively). The response to analgesics seemed to be more favourable in patients with meningioma-related headaches (complete relief in 45.8% of cases) in comparison with those with GBM- or metastasis-related headaches (complete relief in 10.0 and 7.1% of cases, respectively), but this difference was not significant (P = 0.10).
Headache was more frequently accompanied by vomiting in the group of patients affected by tumours in the posterior fossa (42.3%) than in the supratentorial (19%) and skull base (7.1%) groups (P = 0.02). The location of the headache was not uniformly represented among the three groups (P = 0.04); in fact, occipital headache was more frequently associated with infratentorial tumours (38.5%), whereas frontal headache was more often seen with supratentorial (34.5%) and skull base (57.1%).
Headache attributed to intracranial tumours of large dimension was more often accompanied by vomiting (35.0% vs. 24.1% and 6.9% of medium and small dimension tumours, respectively; P = 0.02) and presented more often an early morning or nocturnal occurrence (40.0% vs. 13.8% and 27.6% of medium and small dimension tumours, respectively; P = 0.04). The presence/absence of brain oedema and of midline shift showed no influence on headache features.
Headache characteristics in patients with increased ICP
Increased ICP was more often associated with an early morning or nocturnal occurrence of headache (P < 0.001). The headache was more frequently accompanied by vomiting (P < 0.001) and showed a poorer response to common analgesics (P = 0.04). Quality, intensity and location of pain were not distinctive in patients with evidence of increased ICP.
Discussion
In this study, 98 out of 206 neurosurgical patients were diagnosed to have ‘headache attributed to intracranial neoplasm’ (6), because the improvement in headache following surgery reasonably points to a cause–effect relationship between the tumour and the headache. The prevalence of 47.6% in our series was comparable with 49.8% reported by Pfund and colleagues (3), who used a similar study methodology. To our knowledge, only two other longitudinal studies have been performed (7,8), but as they referred only to pituitary adenomas, we can make no comparison. Since this study was based in a neurosurgical centre, our population is likely to have contained larger numbers of symptomatic patients compared with a non-surgical centre. This might have overestimated the occurrence of headache in patients with intracranial tumours. However, the higher prevalence of headache reported in other studies argues against this (4,5). Furthermore, thanks to the prospective evaluation of patients, we avoided running the risk of attributing to the tumour a headache of ‘primary’ origin. Nevertheless, we cannot rule out the possibility that surgery may also have provided some placebo effect. Moreover, it is possible that some of the 13 patients who still complained of headache 3 months after surgery actually suffered from ‘chronic post-craniotomy headache’, and this fact could have caused the lack of diagnosis of ‘brain tumour headache’. In accordance with Forsyth and Posner (2), the ‘classic’ brain tumour headache has been found infrequently in our adult patients with intracranial tumours. The intracranial tumour headache profile in our patients was that of a pain located bilaterally over the frontal region, pressing/tightening in quality and of moderate intensity, rarely associated with nausea or vomiting and with a favourable response to common analgesics. The headache was usually intermittent, lasted for few hours and presented no typical daily distribution. In most patients, intracranial tumour headaches did not meet ICHD-II diagnostic criteria for primary headache syndromes (6), although migraine and tension-type presentations were observed. As regards patients whose intracranial tumour headache could resemble primary migraine, it usually represented a change of a pre-existing migraine pattern and was nearly always associated with at least one atypical feature. Conversely, as regards intracranial tumour headaches with tension-type pattern, headaches were more frequently of new onset and the presence of atypical features was less common. In every instance, at the time of diagnosis almost all headache patients presented other neurological symptoms or signs. This is congruent with findings of previous works (1,2,4).
Univariate analysis of clinical and demographic variables indicated that patients with a positive personal history of headache or with a younger age at diagnosis have a higher risk to develop a headache secondary to an intracranial tumour. Nevertheless, multivariate logistic regression analysis demonstrated that a longstanding headache history was the only significant independent intrapersonal risk factor. This finding confirms earlier reported data and suggests that genetic factors are important in predicting whether a patient who has an intracranial tumour will develop headache as part of its natural history (2,4). In fact, since the neuronal pathways that lead to headache pain are probably common between the primary and secondary forms of headache, we can hypothesize that in predisposed individuals the tumour may trigger headache attacks, or, alternatively, may determine alterations in the internal milieu resulting in a lowering of the threshold for headache onset. Univariate analysis of clinical variables indicated also that patients affected by hypercholesterolaemia and those undergoing treatment with statins seemed to be protected in respect of the development of headache. Since nearly all hypercholesterolaemic patients were on statins, it was difficult to evaluate the meaning of these findings. A recent open-label study supported the potential efficacy of statins for migraine prevention (9). As suggested by the authors, the pleiotropic effects of statins could influence the neurogenic inflammation involved in the pathophysiology of migraine. These same properties may explain the ‘protective effects’ of statins in respect of brain tumour headache development. However, since many variables were tested for an association with headache, we cannot exclude that chance is the explanation for some of the results. As regards tumour-associated risk factors, we found a trend towards an increased prevalence of tumour-related headache in association with increased ICP, consistent with previous studies (2,5). As documented in other studies, headache seems more common in infratentorial tumours (3,5), and this is probably related to the small space of posterior fossa and the obstruction of cerebrospinal fluid pathways (10). Moreover, even if the percentages of histopathological diagnoses did not differ significantly between the two groups of patients (with and without headache), multivariate logistic regression analysis revealed that GBM and secreting adenomas are associated with a higher risk of developing headache if compared with anaplastic gliomas. In the case of GBM, it can be hypothesized that their rapid growth does not permit the pain-sensitive structures to adapt themselves to the traction created by the growth. A mass effect, on the other hand, is less likely in the case of a secreting pituitary adenoma (usually a microadenoma), which probably involves neuroendocrine mechanisms as suggested by Levy and colleagues (11). On multivariate logistic regression analysis, each cm3 of increase in tumour volume was associated with an increased risk of developing headache (even if, considering the findings of the univariate analysis, we cannot rule out the possibility that this may have been valid for GBM only). Forsyth and Posner reported a link between tumour area and the presence of headache (2), but their findings have not been confirmed in later studies (3,4). In our opinion, this discordance can in part be attributable to differences in the methods used to measure tumour dimensions. However, we consider that the most plausible indicator of the mass effect exerted by a tumour is its volume and not its area. To measure volume we used an advanced computerized calculation system. Stratifying the logistic model for patient's age, we found a trend towards a reduced prevalence of headache in older patients (even if this was not significant). Lowry and colleagues (12) reported that headache was significantly more common as the main presenting symptom of a brain tumour in younger age groups compared with older ones. According to them, this may be related in part to varying degrees of general brain atrophy, which leaves the subarachnoid space and ventricles larger, allowing for more expansion of space-occupying lesions (12). In support of this is our finding that headache occurrence is significantly associated with variables such as tumour volume, midline shift and increased ICP in patients < 65 years old, but not in those ≥ 65 years.
In summary, headache attributed to intracranial tumours is common. Its prevalence varies depending on location, type and volume of the tumour, as well as on the patient's previous headache history. The majority of patients presented headache that could not be classified by ICHD-II diagnostic criteria for primary headache syndromes, although migraine and tension-type presentations were observed, thus making early diagnosis problematic. According to this, a tension-type headache pattern is more likely to deceive clinicians, being less often associated with atypical features.
Footnotes
Competing interests
None to report.
