Abstract
To test the hypothesis that permeability of the blood–brain barrier (BBB) is altered during migraine attack due to enhanced activation of matrix metalloproteinases (MMPs), we investigated MMP-3, MMP-9 and tissue inhibitor of metalloproteases (TIMP)-1 in the external jugular vein during and outside of migraine attacks in 21 patients with migraine without aura. In addition, we measured plasma levels of several other proteins including MMP-7, -8, -10 and TIMP-2. We used Rules-Based Medicine multi-analyte profiling and protein array technologies to study plasma concentration of MMPs. There was no difference in MMP-9 and TIMP-1 levels between ictal and interictal periods. We found significantly decreased plasma levels of MMP-3 in the external jugular (P = 0.002) and cubital (P = 0.008) vein during attacks compared with outside of attacks. We found no correlation of ictal or interictal MMP-3, MMP-9 and TIMP-1 to migraine duration and frequency analysed in 21 patients (P > 0.05). There was no difference between ictal and interictal plasma levels of MMP-7, -8, -10 and TIMP-2 (P > 0.05). Our data suggest that plasma MMP-9 cannot be used as a biomarker of BBB disruption in migraine without aura. Decreased MMP-3 levels are an interesting and unexpected finding warranting further investigation.
Keywords
Introduction
Matrix metalloproteinases (MMPs) are a family of zinc- and calcium-dependent enzymes that are collectively responsible for remodelling of connective tissue (1,2). MMPs are regulated at the transcriptional level by cytokines and growth factors that stimulate the synthesis and secretion of pro-MMPs and also endogenous tissue inhibitors of metalloproteases (TIMPs) (3). The human MMP system is comprised of at least 24 proteases and four TIMPs (3). It has been suggested that diseases involving pathological tissue destruction are associated with aberrant production or activation of MMPs, or a lack of their natural tissue inhibitors, the TIMPs (1,2,4).
Migraine is a neurovascular disorder with a complex pathophysiology (5). The migraine attack consists of different phases that may include aura and headache. There is general agreement that the aura is caused by a cortical spreading depression (CSD), a propagating wave of neuronal and glial depolarization (6–8). A ‘silent’ CSD may occur during migraine attack without aura (9). In animal models of migraine, CSD may trigger nociception (10) and CSD alters blood–brain barrier (BBB) permeability by activating brain MMPs, in particular MMP-9 (11). MMP-9 is capable of degrading proteins found in the extracellular matrix (ECM) (12) and has been implicated in cerebral ischaemia (13) and neural inflammation (14). Furthermore, it has been shown that MMP-3 may contribute to a breakdown of the BBB in relapsing–remitting multiple sclerosis (15).
Two studies have reported elevated ictal and interictal plasma levels of pro-MMP-9 (a latent form) (16) and interictal plasma MMP-9 (17) in the peripheral circulation of migraine sufferers. In the present study we tested these hypotheses: first, that MMPs in the cranial circulation, in particular MMP-9 and MMP-3, are increased during migraine attacks; second, that increased MMPs in the cranial circulation would be detectable in the external jugular blood during migraine attacks. To test these hypotheses we analysed MMP-3, MMP-9 and TIMP-1 in the external jugular vein during and outside of migraine attacks in patients with migraine without aura (MoA). In addition, we measured plasma levels of several other proteins including MMP-7, -8, -10 and TIMP-2, where no significant changes related to migraine were noted (data not shown).
Material and methods
Subjects
Blood samples were obtained in 21 patients (17 women, four men, mean age 39 years, range 26–53 years) fulfilling International Headache Society (IHS) criteria 1.1 for MoA (18), during as well as outside of an attack. Patients were enrolled from the out-patient headache clinic at the Danish Headache Centre (Glostrup Hospital, Denmark). Inclusion criteria were MoA fulfilling migraine criteria of IHS 1.1 for at least 1 year; one to six attacks per month for the last 3 months; age between 18 and 65 years; good general health determined by medical history, normal physical examination, neurological examination, and electrocardiogram. Exclusion criteria included more than six migraine attacks or 10 days with migraine per month, use of migraine or headache medication > 12 days/month, > 6 days/month with tension-type headache, migraine prophylaxis within 6 weeks of inclusion, alcohol or drug overuse, regular use of prescribed or over-the-counter medication except oral contraceptive pills and usual acute migraine medication, use of weak analgesics exceeding the equivalent of 18 g paracetamol (acetaminophen) per month. Pregnant or breast-feeding women were excluded from the study. Patients kept a headache diary to ensure the frequency of migraine and tension-type headache. Approvals were obtained from the Scientific Ethical Committee for the County of Copenhagen. Signed informed consent was obtained from each patient. The study was undertaken in accordance with the Helsinki Declaration of 1964, as revised in Edinburgh in 2000.
Clinical study design
After inclusion, all migraine patients were given the number of a pager and instructed to call when they experienced a migraine attack. Patients could call daily between 06.00 and 22.00 h, including weekends and holidays. A physician would personally return their call and as soon as possible come to the patient's home, workplace, or wherever the patient was at the time to take blood samples during the migraine attack. Because of the short distances in the Copenhagen area, all patients could be reached within 40 min, and all blood samples during migraine attack could be taken within 60 min from the patient's initial contact. Patients agreed not to treat their migraine until after the blood samples had been taken; therefore, treatment was delayed by up to 60 min. Blood samples during attacks were taken only if all of the following could be confirmed: the attack fulfilled IHS criteria for MoA and according to the patient was a usual attack; the attack had started < 6 h ago (if the patient awoke with a migraine attack, this time was considered the start time); the migraine was not a recurrent attack (migraine free for at least 72 h); and the patient had not taken 5-HT1B/D agonists within 48 h, ergotamine or similar medication within 72 h, and no form of analgesics had been taken within 24 h. Blood pressure and heart rate were measured before blood sampling. Samples were taken after a minimum of 15 min of rest in the supine position with legs slightly elevated. Samples were taken on the same side as the headache; if the migraine headache was bilateral, samples were taken on the patient's dominant side. Headache characteristics and accompanying symptoms were recorded. Blood samples were taken with a cannula (Venflon or butterfly) in the external jugular vein and the antecubital vein. With a minimum interval of 7 days, the control samples were taken on the same side and in the same way as during an attack, but on a day without migraine or other headache. The patients were contacted by telephone 24 h after the control samples were taken to ensure that an attack had not developed after blood sampling. One patient had new control samples taken because of the development of migraine within 24 h after the initial control samples were taken. All blood samples were taken by two physicians, both with extensive practice in puncturing the external jugular vein and at least 1 year of anaesthesiology training. The blood samples were collected into cooled 10-ml tubes (BD vacutainer™; Becton Dickinson, Plymouth, UK) prepared with 15% ethylenediamine tetraaceticacid (EDTA) (0.12 ml/0.369
Plasma samples were coded, and MMPs and TIMPs were measured blindly with respect to patient identity, attack status, and sampling site. Plasma samples from attack and outside of attack were measured at the same time, and MMP-9 and TIMP-1 samples were measured with two different assays.
Rules-Based Medicine multi-analyte profiling
The samples were thawed at room temperature, vortexed, spun at 13 000
Protein microarray
A rolling cell amplification (RCA) immunoassay was performed by Molecular Staging Inc. (MSI, New Haven, CT, USA) utilizing a protein microarray platform that measured levels of 143 analytes on five separate arrays (19,20). After the incubation and washing of the plasma samples on microarrays, the captured proteins were detected by specific, biotinylated second antibodies, and a universal antibiotin antibody was bound to the secondary antibodies. The antibiotin antibody contained an oligonucleotide DNA primer used for amplification. During the process, a circular DNA hybridizes to the oligonucleotide DNA primer in the presence of DNA polymerase and fluorescent nucleotides to generate a signal. Following RCA, the slides were scanned (L200 scan; TECAN, Durham, NC, USA) using a proprietary software. The fluorescence intensity of microarray spots was analysed and the resulting mean intensity values were measured. Dose–response curves for the biomarkers were determined with increasing intensity indicating increasing analyte concentration.
Statistical analysis
Given that MMP and TIMP variables were not normally distributed, data are presented as median and quartiles. Samples with less than the detection level were excluded from statistical analysis. To test the differences between variables (including baseline variables) we used the Wilcoxon signed rank test.
All analyses were performed with SPSS for Windows 14.0 (SPSS Inc., Chicago, IL, USA). Five percent (P < 0.05) was accepted as the level of significance.
Results
Patient characteristics
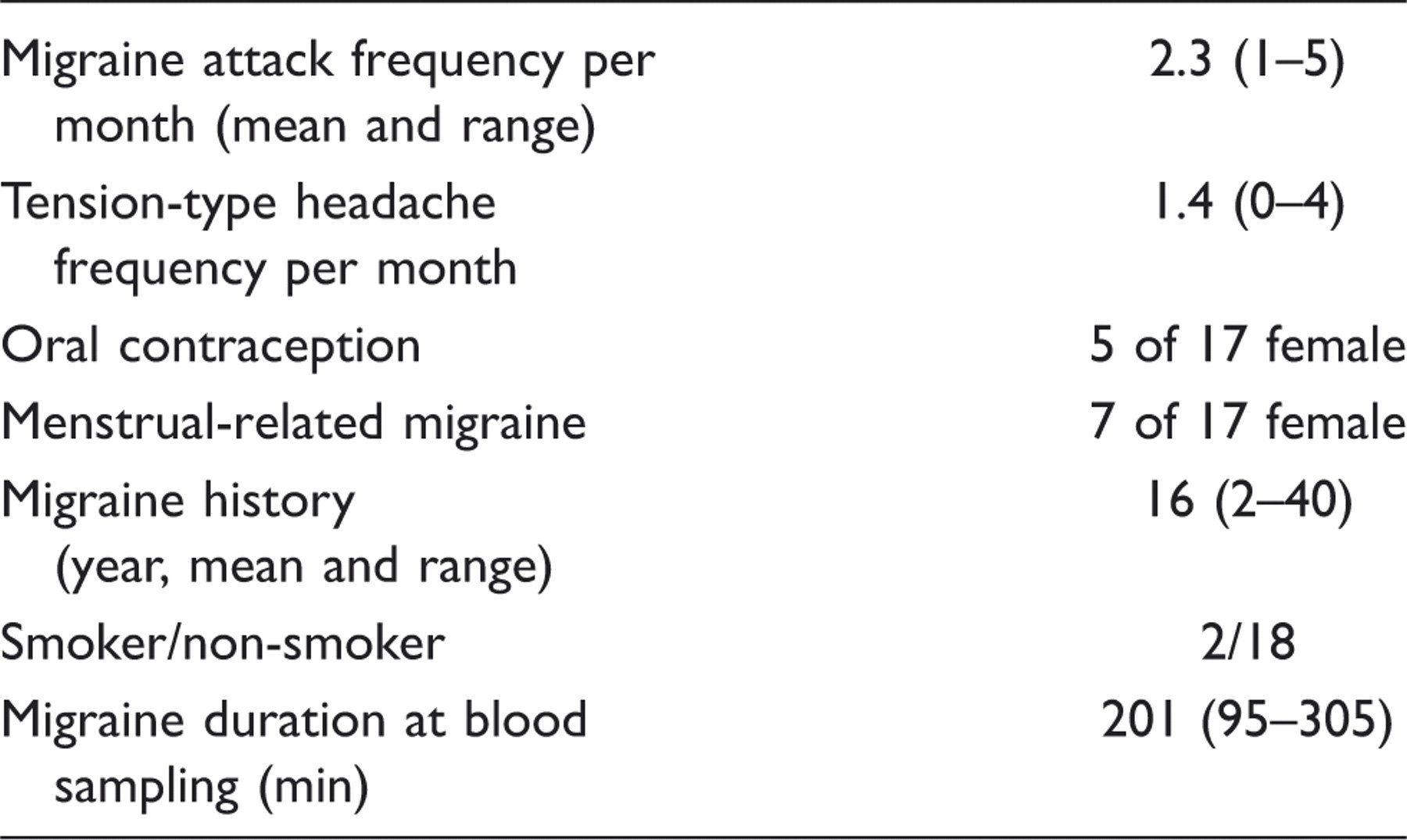
Migraine attack characteristics (n = 21)
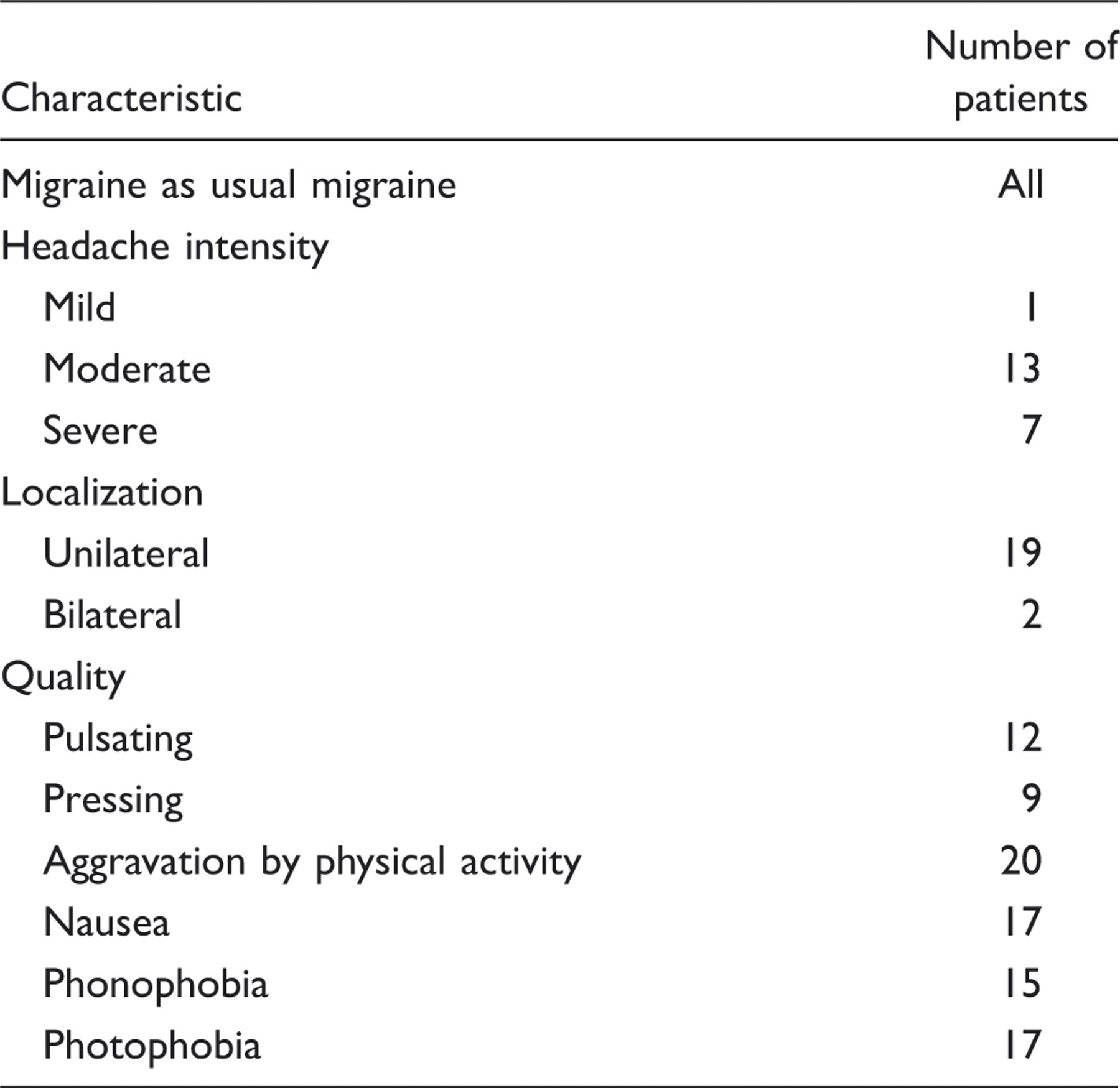
Plasma levels of MMP-3, MMP-9 and TIMP-1
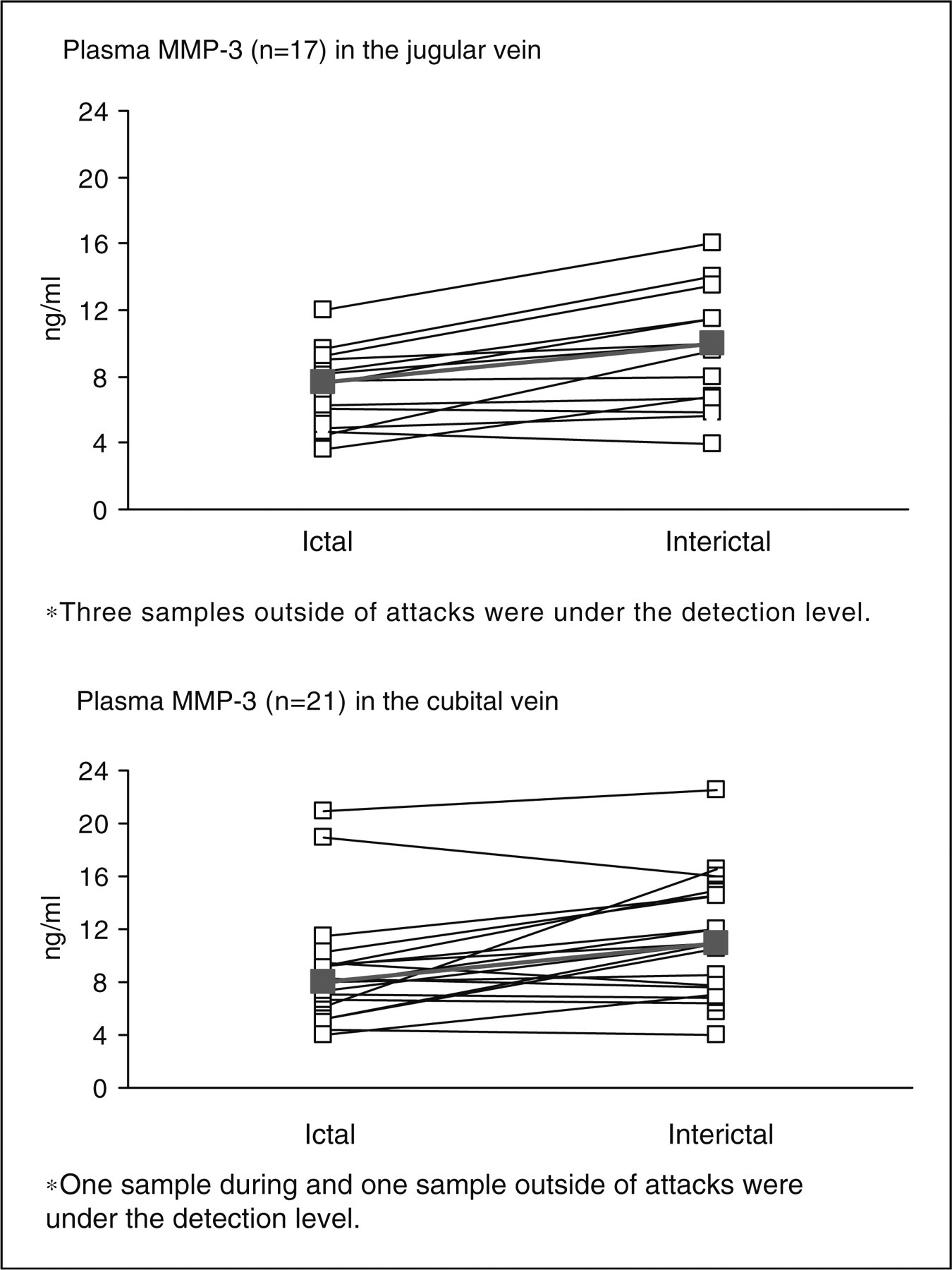
Rules-Based Medicine MAP analysis revealed no difference in MMP-9 and TIMP-1 levels between ictal and interictal periods (Table 3 and Fig. 1). There were significantly decreased plasma levels of MMP-3 in the external jugular (P = 0.002) and cubital (P = 0.008) veins during attacks compared with outside of attacks (Table 3 and Fig. 2). Using protein microarray technology, we found no difference in mean fluorescence intensity of MMP-9 and TIMP-1 between ictal and interictal periods in either jugular or cubital vein (P > 0.05). There was no difference between ictal and interictal plasma levels of MMP-7, -8, -10 and TIMP-2 (P > 0.05) (data not shown).
Individual plasma levels of MMP-9 and TIMP-1 in the cranial and peripheral circulation in patients with migraine without aura. There was no difference in MMP-9 and TIMP-1 levels between ictal and interictal periods (P > 0.05). *Samples under the detection level. Thick line shows median values. Individual plasma levels of MMP-3 in the cranial and peripheral circulation in patients with migraine without aura. Plasma MMP-3 levels were significantly decreased during attack compared with outside of attack in the external (P = 0.002) and cubital (P = 0.008) vein. Thick line shows median values. Median (quartiles) plasma levels of MMP-3, MMP-9 and TIMP-1 in the external jugular and cubital veins measured by Rules-Based Medicine Multi-analyte Profiling analysis

Correlation to migraine duration and frequency
We analysed a group of subjects (n = 11) whose blood samples were collected > 3 h after onset (range 3.3–5.1 h). While MMP-3 levels were also decreased during attack, we found no changes between ictal or interictal levels of MMP-9 and TIMP-1 (P > 0.05). No correlation was found of ictal or interictal MMP-3, MMP-9 and TIMP-1 to migraine duration or frequency analysed in 21 patients (P > 0.05).
Discussion
The major result from the present study is that plasma levels of MMP-9 and TIMP-1 were unchanged during MoA attacks. An additional surprising and interesting finding was decreased ictal levels of plasma MMP-3 in the cranial and peripheral circulation during MoA attacks.
Blood–brain barrier in migraine
In spite of considerable interest in the BBB in the aetiology of migraine, few human studies have addressed the underlying physiological changes in migraine. Two pathophysiological mechanisms may cause alteration of BBB in migraine: CSD and activation of trigeminal sensory fibres with leakage of substance P and other messenger molecules. CSD has been implicated in stroke (21), and cerebral ischaemia is associated with increased BBB permeability (22,23). Inflammatory mediators are known modulators of BBB permeability (24), and animal models of inflammatory pain (formalin, Freund's adjuvant and carrageenan injected in hind paw) cause increased permeability of BBB (25–27). Given that CSD (28) and possibly neurogenic inflammation (29) play a key role in migraine, it would be plausible to suggest that the BBB may be opened to the same extent during migraine with or without aura. Neuroimaging is the only tool to examine this. Cutrer et al. (30) used diffusion-weighted magnetic resonance imaging and observed no changes during migraine aura. Furthermore, no gadolinium enhancement was reported in patients with and without migraine aura. However, these data should be interpreted with caution because of possible methodological issues. What if BBB openings vary in size during migraine aura or pain, or it depends on the severity of aura? Thus, two studies have reported gadolinium enhancement over the symptomatic cerebral hemisphere during severe and prolonged attacks of familial hemiplegic migraine (31,32). This indicates that the severity and duration of CSD may influence outcome in imaging studies. Furthermore, it is known that gadolinium-induced enhancement may be dose dependent as shown in multiple sclerosis plaques (33). Collectively, there is no firm evidence of significant BBB disruptions in migraine. Besides, slight changes in permeability of BBB could be missed due to methodological limitations in neuroimaging techniques.
Can plasma MMPs be used as biomarkers of altered permeability of BBB?
The anatomical substrate of the BBB is the cerebral microvascular endothelium, astrocytes, pericytes, neurons and ECM (23). Of particular relevance for migraine is the disruption of the ECM that is strongly associated with increased BBB permeability in pathological states (34). Thus, the ECM molecules constitute the basement membrane around the vasculature and play a critical role in maintaining the integrity of the BBB (23).
MMPs regulate ECM degradation during tissue remodelling and can influence the expression of endothelial tight junction proteins (35,36). In an animal model of stroke, MMP-9 expression increased progressively over time after stroke (37). Gursoy-Ozdemir et al. (11) reported that CSD initiates a cascade that disrupts the BBB via an MMP-9-dependent mechanism. The authors showed that MMP-9 levels increased within the cortex ipsilateral to the CSD beginning at 3–6 h, reaching a maximum at 24 h and persisting for at least 48 h (11). However, it is unclear whether increased MMP is specific for the BBB disruption or pathological states such as CSD per se. In an animal model of epileptic seizures, significantly increased intracerebral expression levels of MMP-3 and -9 without any simultaneous BBB disruption or changes in the BBB properties were reported (38). Another question is to what extent intracerebral alterations of MMPs may reach plasma and be detected in blood samples of patients and used as biomarkers? Circulating MMPs are derived from leucocytes, which are also recruited to the brain during pathological and/or inflammatory conditions (38–41). In fact, this recruitment of peripheral leucocytes such as monocytes, macrophages and lymphocytes to the brain parenchyma is highly contributory to the BBB disruptions seen in most neuropathological disorders, as the leucocytic cells secrete tissue-degrading enzymes including MMPs during their invasion of the cerebral vascular endothelium and perivascular space. Hence, when peripheral leucocytes or the bone marrow homeostasis are suppressed, it will lead to deficient brain inflammatory responses due to a lack of recruitment of haematopoietic (peripheral) cells. In such cases the BBB was shown to be intact, indicating that leucocytic invasion of the cerebral endothelium and leucocytic secretion of enzymes (including MMPs) are major contributors to BBB disruption.
These data indicate that plasma MMP fluctuations in migraine patients are likely to be associated with ongoing BBB pathology, although the timing and degree of BBB damage probably affect the outcome. However, the BBB disruption is a local pathology, which mainly works (transports molecules) in a one-way manner (from blood vessel lumen and into brain parenchyma). Accordingly, there are so far no reliable and specific factors (biomarkers of BBB damage) that are released from the central nervous system into the blood vessel lumen and into circulation. Therefore, it is unclear whether fluctuations of brain MMPs may be detected in the plasma of migraineurs.
Present findings
The present study focused upon a carefully characterized and monitored population, and had shown that plasma levels of MMP-9 are unchanged during attacks compared with outside of attacks. All samples were assessed blindly in respect of ictal and interictal periods. These data are in contrast to two previous studies showing elevated plasma MMP-9 in the first 3–6 h from onset of migraine attack (16) and the highest plasma MMP-9 levels in subjects from whom blood samples were taken 2–4 days after their latest attack (42). Leira and colleagues (16) also found increased interictal plasma levels of MMP-9 in the peripheral circulation in patients with (n = 14) and without aura (n = 20) compared with controls. Given that up-regulation of MMP-9 is a hallmark indicating nociception in trigeminal sensory afferents (11), it is difficult to interpret these data because one would expect no difference between patients outside of attacks and controls. Furthermore, Leira and colleagues (16) did not register the time from migraine attack to pain-free period, or the time from the last migraine attack to the current attack. Furthermore, it is unclear whether ictal levels in patients with aura were examined during attacks with or without aura (16). In the present study the patients had been migraine free for at least 72 h, and ictal samples were collected between 1.6 and 5.1 h after onset of migraine pain. We did not collect samples in healthy controls, and it is therefore unknown whether baseline levels in patients differ from those without migraine. It has been suggested that CSD initiates a cascade that disrupts the BBB via an MMP-9-dependent mechanism. This would lead to release of inflammatory mediators and subsequent sensitization of meningeal nociceptors and head pain. In the present study all patients reported moderate to severe pain. If one assumes that all of them experienced a ‘silent’ CSD, we would expect increased MMP-9 levels during ongoing nociception. We found no changes in plasma MMP-9 even in the group of subjects whose blood samples were collected > 3 h after onset (range 3.3–5.1 h). This is in contrast to Leira et al. (16), who reported up-regulation of pro-MMP-9 in the first 3–6 h from onset of migraine attack compared with outside of attacks. We have no satisfactory explanation for this discrepancy. In the present study plasma samples were measured with two different assays. Quantitative measurements were made by Rules-Based Medicine, a US Clinical Laboratory Improvement Amendments-certified biomarker testing laboratory delivering reproducible, quantitative, multiplexed immunoassay data. For these measurements immunobead technology was used, which, unlike microarray or enzyme-linked immunosorbent assay, performs assays in a fluidic phase. Fluid phase assays allow greater availability for antibody binding due to the three-dimensional nature of the microparticle.
MMP-3 levels have not previously been examined in patients with migraine. In the present study, we found decreased levels of MMP-3 in both cranial and peripheral circulation during attack. Could decreased plasma concentrations of MMP-3 indicate increased ECM degradation and thereby possibly increased permeability of BBB in migraine? It has been reported that MMP-3 knockout (KO) mice have less disruption of the BBB after lipopolysaccharide-induced opening of the BBB than WT mice (43). Interestingly, MMP-9 mRNA levels were increased to a similar level in both the MMP-3 KO and WT. The authors suggested that MMP-3 may attack the basal lamina and tight junction proteins, opening the BBB and facilitating neutrophil influx (43). Interestingly, MMP activation is often the result of a complex proteinase cascade, and some of the activated MMPs can activate other pro-MMPs, e.g. MMP-3 can activate pro-MMP-9 (44). It was recently demonstrated that serum MMP-3 concentration is significantly lower in the acute phase of myocardial infarction than during recovery (45). In contrast MMP-3 brain tissue levels could not be detected between 6 and 24 h after focal stroke in the rat (37). However, the authors reported increased MMP-9 expression in endothelial cells and infiltrating neutrophils. It is possible that this study was unable to identify transient expression of MMP-3 in the early phase of the time course (37). Based on these data, it would be plausible to suggest that MMP-3 decreases during the early phase of acute migraine attacks. However, these data should be interpreted with caution and should be replicated before any firm conclusions are drawn.
Conclusions
MMP-9 and TIMP-1 levels in the external jugular vein are unchanged during migraine attacks without aura 1.6–5.1 h after onset of migraine pain. However, it is possible that delayed up-regulation of MMP-9 might occur, but this would hardly be relevant for migraine pain. Our data suggest that plasma MMP-9 cannot be used as a biomarker of BBB disruption in MoA. Decreased MMP-3 levels are an interesting and unexpected finding warranting further investigation.
