Abstract
We report a case of osteoid osteoma involving the ethmoid sinus, which presented as a unilateral fixed headache with some features suggestive of hemicrania continua.
Introduction
The otolaryngologist is frequently consulted when a headache may be caused by sinonasal pathologies. When a patient presents with unilateral headache, the otolaryngologist will more narrowly look for sinonasal pathologies, especially acute rhinosinusitis, as a potential cause for the patient's complaints. However, occasionally sinonasal pathologies can be manifested solely as headache. On these occasions, the headache is often confused with primary headache such as migraine or cluster headache. Thus, headache attributed to sinonasal pathologies, in the absence of sinonasal symptoms (nasal discharge, nasal obstruction, etc.), can often delay its diagnosis or be misdiagnosed.
Hemicrania continua (HC) is an unusual primary headache disorder probably first described by Medina and Diamond in 1981 (1), and the term ‘hemicrania continua’ was coined by Sjaastad and Spierings in 1984 (2). HC was previously classified as one of the trigeminal autonomic cephalalgias (TACs), but in 2004 the International Headache Society (IHS) moved it to the other primary headache category. HC is an indomethacin-responsive headache disorder characterized by a continuous, unilateral headache without side shift that varies in intensity without disappearing completely (3).
The cause of HC is unknown. In most cases, HC appears to have arisen de novo, without any identifiable trigger (4). However, a number of secondary cases of HC have been described, associated with internal carotid artery dissection, pineal cyst, postpartum, head trauma, pituitary tumour, ipsilateral brainstem lesion, ipsilateral mesenchymal tumour of sphenoidal bone involving the clinoid process at the base of the skull, with HIV infection, and lung adenocarcinoma (4–12).
Here, we report a case of osteoid osteoma involving the ethmoid sinus, which presented as a unilateral fixed headache (side-locked headache) with some features suggestive of HC. We review the literature of this rare but interesting lesion.
Case report
A 35-year-old man was referred to our department for evaluation and treatment of right-sided headache that had persisted for 12 months. The character of headache was atypical, sometimes continuous, deep and pressure-like, but recently it had become more severe and tended to increase at night and frequently disturbed the patient's sleep. There were no precipitating or aggravating factors. There were no symptoms of photophobia, phonophobia, nausea, vomiting or autonomic symptoms (conjunctival injection, lacrimation, nasal congestion, rhinorrhoea, ptosis, miosis) during his baseline headache and nocturnal exacerbations. Although oral analgesics (paracetamol) relieved the headache to some degree, the amount administered had to be increased progressively to relieve the headache.
He had no history of any sinusitis, seasonal allergies or other sinus problems. There was no trauma history or significant past medical history.
The physical examination found neither facial deformity nor eye movement limitation. Sinonasal endoscopic examination did not reveal any mucopus or abnormal secretion in the nasal cavity. Also, there was no deformity or displacement of anatomical structure in the nasal cavity. Neurological examination results were normal and other physical examinations yielded normal findings. The patient underwent a detailed laboratory examination to exclude any other possible causes of headache, but all results were within normal range.
Despite the fact that the patient had no rhinological symptoms, sinus headache induced by acute unilateral right sinusitis or sinonasal pathologies was suspected, and a simple x-ray was obtained. The lateral radiograph of paranasal sinuses revealed a well-defined 2 cm sized round radio-opaque lesion in the ethmoid sinus (Fig. 1). Computed tomographic (CT) scans of the paranasal sinuses revealed a 2.1 × 2.3 cm, well-defined, thin sclerotic margin in the right ethmoid sinus. This lesion was expansile growth, but there was no bony destruction. The right lamina papyracea was remodelled with an outward bulging contour into the right orbit. The midline septum was mildly deviated to the right side (Fig. 2). There was no lesion in other sinonasal cavities. Magnetic resonance imaging (MRI) revealed the same mass with peripheral and mottled enhancement. The right medial rectus muscle was indented by this lesion (Fig. 3).
The lateral radiograph of paranasal sinuses shows a well-defined 2 cm sized round radio-opaque lesion in the ethmoid sinus. Computed tomographic scans of the paranasal sinuses (A: axial, B: coronal) show a 2.1 × 2.3 cm sized well-defined and thin sclerotic margin in the right ethmoid sinus. Magnetic resonance imaging shows the same mass with peripheral and mottled enhancement (A: axial T1-weighted image, B: coronal T1WI, C: T2WI, D: gadolinium enhanced-T1 weighted image).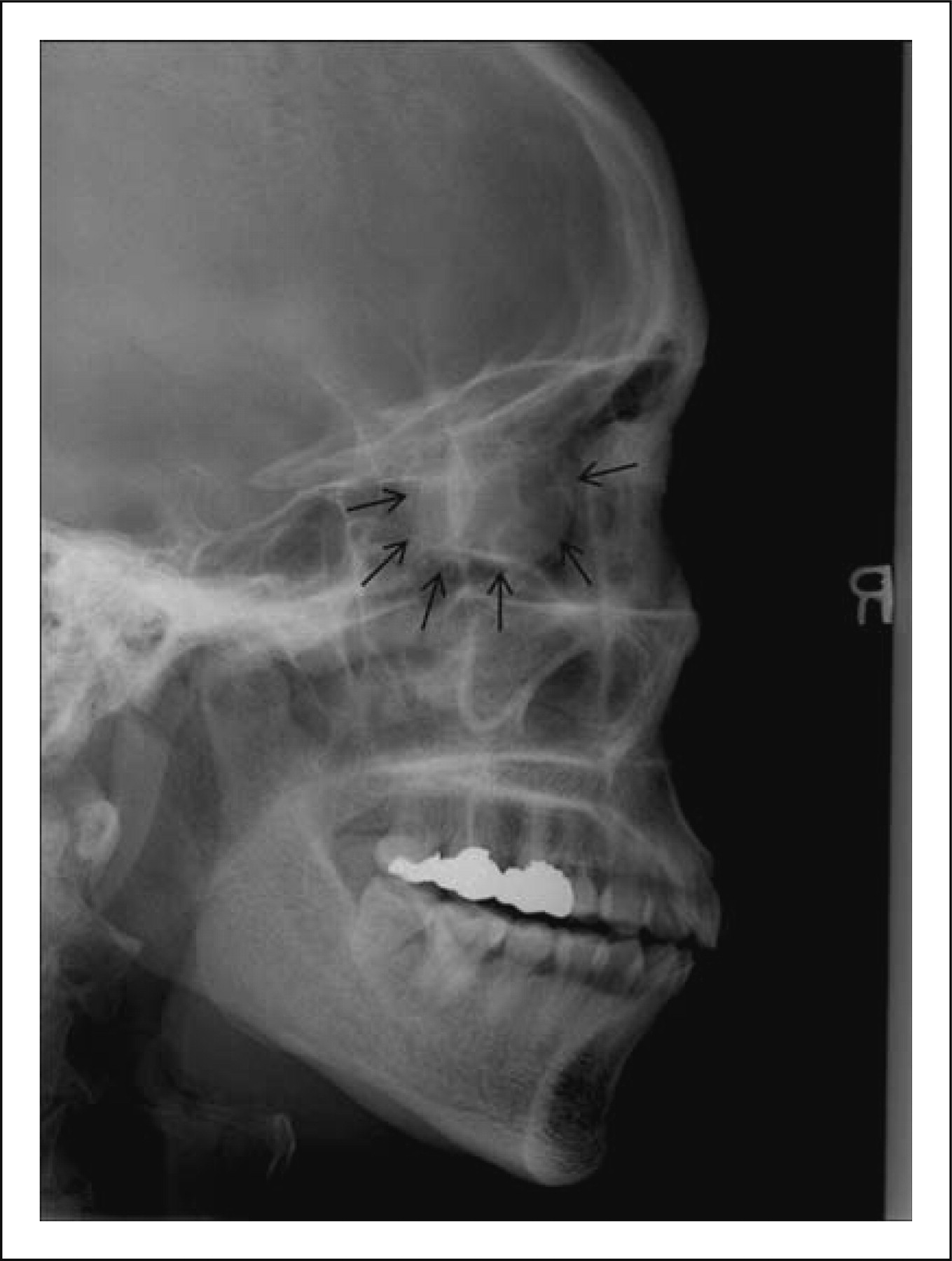


Under general anaesthesia, this mass was surgically removed via intranasal approach with endoscope. During a right uncinectomy, the bony anterior wall of the lesion was detected. During dissection, the lesion was broken into multiple pieces. Although the lesion was well demarcated from surrounding normal tissues, on account of its huge size, its adhesion to the surrounding anatomical structures and profuse bleeding during surgery, complete removal of the lesion was difficult. Finally, using a microdrill and curette, the lesion which was composed of the bony wall and the soft tissue content was removed totally. The lamina papyracea was intact, but severely bowed laterally. Gauzes were lightly packed to prevent postoperative haemorrhage and were removed 48 h later. There were no severe complications such as haemorrhage, ocular disturbance or loss of vision.
Histopathological examination revealed that the lesion showed well-demarcated central nidus with surrounding bone trabeculae. The central nidus was composed of calcified osteoid and vascular connective tissue stroma and osteoblastic rim (Fig. 4).
The tumour shows a well-demarcated central nidus with surrounding bone trabeculae (T) (A, H&E, ×100). The central nidus is composed of calcified osteoid (O) and vascular connective tissue stroma (S). High-power view of central nidus (B, H&E, ×400) (O, calcified osteoid; S, vascular connective tissue stroma; arrow, osteoblast).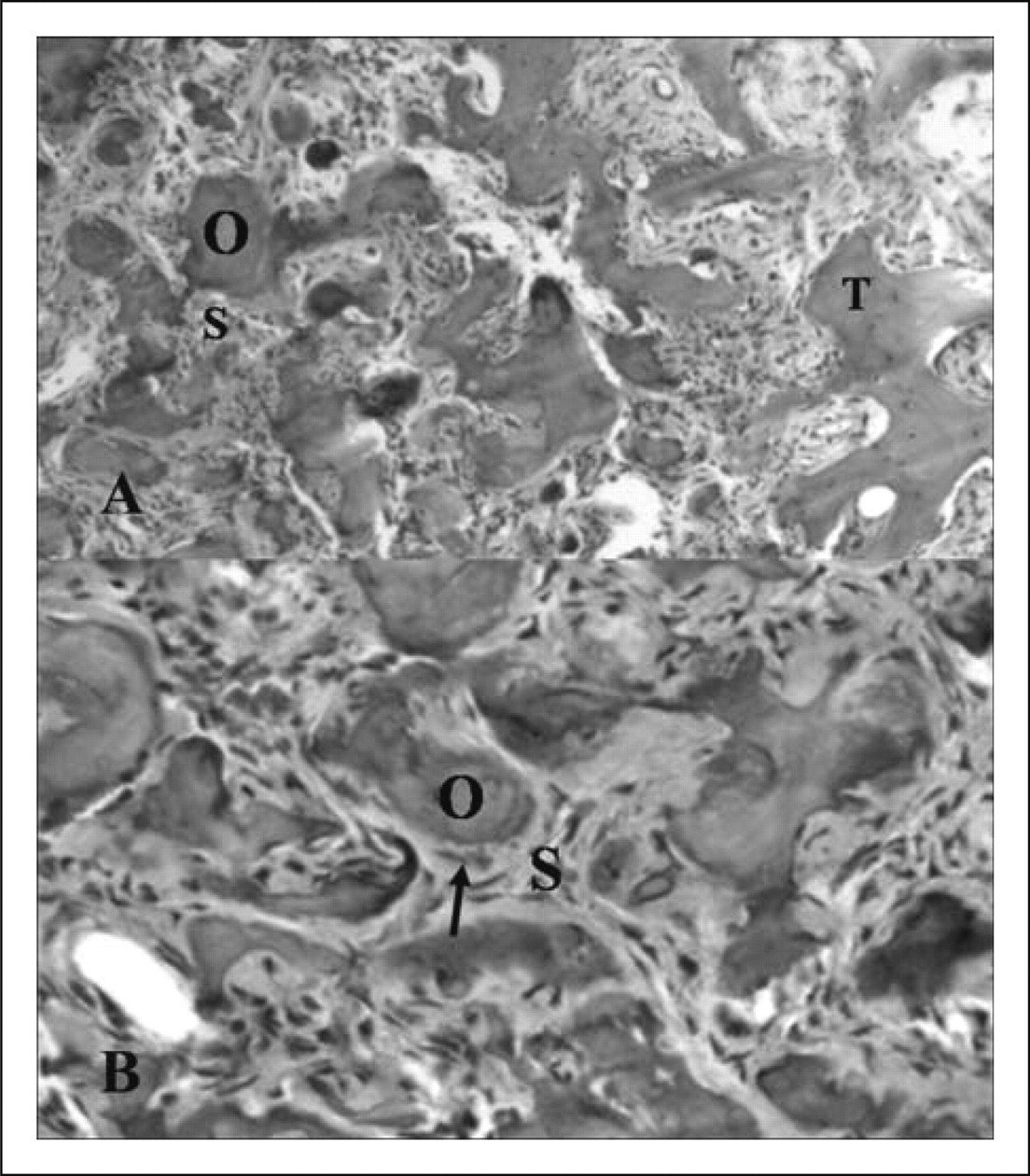
On the basis of clinical characteristics, and radiographic and histopathological findings, the diagnosis of ‘osteoid osteoma of ethmoid sinus’ was confirmed.
The patient had an excellent postoperative recovery without postoperative bleeding or other complications, and was discharged 3 days later. The postoperative CT scan performed 12 months later showed complete removal of the tumour. The patient reported prompt relief of his headache and at 15 months' follow-up postoperatively he was free of headache clinically with no evidence of tumour recurrence.
Discussion
What is osteoid osteoma?
Osteoid osteoma is a solitary, benign and osteoblastic tumour that is most commonly located in the cortex of long bones of the lower extremities and vertebrae (13,14). The incidence of osteoid osteoma is around 1–3% of all primary bone tumours and 11–12% of benign bone tumours (15,16). Cranial osteoid osteoma, especially in the ethmoid sinus, is an extremely rare finding (17). To the best of our knowledge, this report is the fifth case of ethmoidal osteoid osteoma in English literature (17–20). Osteoid osteoma consists of a central core of vascular osteoid tissue (nidus) and a peripheral zone of sclerotic bone (15). Osteoid osteoma occurs mainly in the second and third decades (13). It occurs predominantly in men, with a 2–3:1 male : female ratio (13,15). The pathogenesis of osteoid osteoma remains controversial (13). Unusual healing and reparative process after trauma is considered to be a contributing factor, but the correlation between injury and the onset of osteoid osteoma remains unclear (21). Also, some believe it to be a neoplasm (13). However, it has limited growth potential and typically measures < 1.5 cm in diameter (22). Because of the age distribution of affected patients and self-limited growth, it was thought that the lesion may heal spontaneously. Spontaneous thrombosis of the blood vessels within the lesion may explain the self-limiting course of the disease (23). However, the reported numbers of untreated osteoid osteomas suggest that symptoms may occasionally last as long as 20 years, and surgical removal of the tumour is the treatment of choice (22).
Although clinical symptoms depend on the location of the lesion, the most common and often the only symptom is sharply localized pain, typically described as a mild intermittent vague pain in the beginning of increasing severity and duration, with nocturnal paroxysm (19,24). Despite its small size, pain and discomfort at the site are presented significantly for prolonged periods (13). This pain is relieved dramatically by oral salicylates or non-steroidal anti-inflammatory drugs (NSAIDs) (25). This pain pattern and characteristic pain relief by drugs are highly indicative of osteoid osteoma (26). In our case, osteoid osteoma was seen to originate from the ethmoid sinus and atypical headache was the only presented symptom. This headache was relieved by NSAIDs to some degree. However, this relief can only be transient and an increased amount of salicylates may be needed to control the pain.
There are two most important points to explain the pathophysiology of pain in osteoid osteomas. First, the free nerve fibres, presumed to be components of the autonomic nervous system, are identified in the nidus and the reactive zone. These fibres are larger and more abundant in the reactive zone than in the nidus. Because most of the nerve fibres are found in close proximity to blood vessels, vascular pressure stimulating afferent nerve fibres might cause visceral pain (27,28).
Second, increased prostaglandin synthesis, particularly of PGE2 with as much as a 1000-fold increase within the nidus, suggests an important pathophysiological role as a mediator of pain in osteoid osteomas. This increase in prostaglandins mediates inflammation and vasodilation, which stimulate pain (29).
Vasodilation mediated by PGE2 may cause expansion of the osteoid osteoma and compression of the abundant nerve endings in the tumour. Since PGE2 also induces hyperalgesia, the 1000-fold increase of PGE2 synthesis probably causes the intense local pain produced by osteoid osteomas. This hypothesis is reinforced by the significant analgesic actions of NSAIDs in this condition (29).
The blood flow in osteoid osteomas at night is 1.5 times greater than during the day. This increase in pressure within the nidus could be responsible for increased night pain (30).
Diagnosis depends on clinical history and imaging techniques. Because of its characteristic radiographic findings and clinical symptoms, diagnosis of cortical osteoid osteoma is not difficult. Osteoid osteoma can be identified on plain radiographs. The characteristic radiographic findings include sharp, round-to-ovoid contours, and dense sclerotic bony margin with a nidus demonstrated in 85% of cases (31). They are indicative of the diagnosis. In our case, no characteristic radiographic findings could be visualized, probably due to the large size and unusual location. Other imaging studies may be needed to make a correct diagnosis, for two reasons. First, the characteristic radiographic finding may not become evident until several months after the first clinical symptoms (32). Second, the lesion may be in an atypical location and lack the associated reactive sclerosis (13). CT and isotope bone scan are reliable tools for correct diagnosis.
Technetium-99 bone scan is mainly used as a isotope bone scan and the classic scintigraphic appearance of a double density is very specific and indicative of the diagnosis. Also, it is used as a guide for CT study (33). CT is the best imaging modality for diagnosis of osteoid osteoma, because it is usually able to demonstrate a small radiolucent cortical nidus with a dense sclerotic ring, which in 20–30% of cases has central calcifications (34).
MRI in the diagnosis of osteoid osteoma is not essential. MRIs findings are ambiguous and not diagnostic, because they depend on the amount of calcification within the nidus, the size of the fibrovascular zone, reactive sclerosis and the amount of oedema in the bone (35). However, MRI is more sensitive than CT in detecting soft tissue and bone marrow changes adjacent to the nidus. Thus, MRI should be interpreted with reference to plain radiographs and CT (36). In our case, MRI was not useful because there were small amounts of soft tissue and bone marrow in the site of origin.
The histological hallmark of osteoid osteoma is the nidus, which is a small, well-circumscribed lesion composed of irregular trabeculae of osteoid tissue or mineralized bone lined by a prominent osteoblastic rim within a highly vascularized connective tissue stroma. The nidus is often sharply demarcated from the surrounding variably thick layer of dense bone (15). It does not invade the adjacent tissue or undergo malignant transformation (20).
The most important differential diagnosis is osteoblastoma, because both lesions appear very similar histologically and radiographically. Differentiation between them can be on the basis of size, clinical presentation and distribution. First, a lesion < 1.5 cm is considered as osteoid osteoma and a lesion > 1.5 cm as osteoblastoma. Second, osteoid osteoma is usually accompanied by nocturnal pain, promptly relieved by salicylates; osteoblastomas have variable, inconsistent pain and more potentially aggressive behaviour. Third, osteoid osteoma arises predominantly in the long bones of the lower extremities and vertebrae; osteoblastoma in the axial skeleton, spinal lesions (15,24). In our case, although the lesion was > 1.5 cm, like osteoblastoma, and with atypical clinical findings, osteoid osteoma was confirmed due to typical histopathological findings.
If the lesions are asymptomatic and small, there is a definite role for conservative treatment because of a tendency to regress spontaneously. Indication for surgery depends on symptom severity and the patient's pain tolerance. Generally, complete surgical nidus removal is sufficient and essential for immediate, lasting pain relief. Accurate localization of the nidus by CT is crucial for predictable success of surgical intervention (13,37). Recurrence is likely due to incomplete excision of the nidus, with a recurrence rate of about 10%. Such recurrences have been reported after curettage or drilling (38).
Was this patient's fixed unilateral headache (or side-locked headache) consistent with hemicrania continua?
In summary, our patient's headache:
Was fixed unilateral (or side-locked)
Was continuous, deep and pressure-like, of fluctuating quality
Disturbed the patient's sleep by nocturnal paroxysm
Had no ipsilateral autonomic features during nocturnal exacerbations.
According to the criteria for the diagnosis of HC by the IHS classification 2004, HC presents four characteristics: (i) strictly unilateral, moderate intensity, continuous and fluctuating pain; (ii) at least one ipsilateral autonomic feature during exacerbations; (iii) complete response to indomethacin; and (iv) not attributed to another disorder (3).
We discussed whether our patient was consistent with HC according to the above-mentioned four characteristics.
First, although the revised 2004 IHS diagnostic criteria provide no characterization for quality of pain, we think the quality of headache is very similar between the two diseases.
HC is a headache disorder characterized by a continuous, unilateral headache without side shift that varies in intensity without disappearing completely. The baseline headache is typically described as dull, aching, or pressure-like and not associated without autonomic symptoms. Superimposed on the continuous but fluctuating pain are occasional attacks of more severe pain. Exacerbations are accompanied by functional disablement and increase at night in one-third of cases (39,40).
In cases of osteoid osteoma, the pain depends on the location of the lesion. In our case, because osteoid osteoma was seen to originate from the ethmoid sinus, fixed unilateral headache (or side-locked headache) was the only presenting symptom. The quality of the headache with its nocturnal paroxysm and deterioration of life quality during the exacerbations are features seen in HC.
Second, at least one of the autonomic features during the exacerbations is one of the essential features of the IHS diagnostic criteria. However, ipsilateral autonomic features during nocturnal exacerbations are absent in our case. Nevertheless, we think HC cannot be ruled out, because ipsilateral autonomic features during exacerbations are absent in up to 30% of HC cases (40). Also, symptomatic (secondary) HC tends to be characterized by absent or inconspicuous autonomic features in comparison with primary HC (41,42).
Third, response to indomethacin is an essential feature in IHS diagnostic criteria. However, our case was unfortunately not given a trial of indomethacin, so we do not know for certain about indomethacin responsiveness in our case. Nevertheless, we think HC cannot be ruled out because some cases of HC are not responsive to indomethacin and an absolute response to indomethacin is not specific for the diagnosis of HC (43). In any case, many authors have suggested the term HC ‘possible’ or indomethacin-resistant HC for patients who fulfil criteria except the indomethacin response (44–47). Also, the effective dose and response of indomethacin are variable in symptomatic HC (48). The need for high doses of indomethacin or wearing off of its effect are also regarded as signs of associated pathology (symptomatic HC) (7). In our case, as stated above, because the osteoid osteoma was located in the ethmoid sinus in contrast to typical osteoid osteoma, our patient's pain was the atypical, fixed unilateral headache as the only presented symptom. This headache was relieved to some degree by NSAIDs, but this relief was only transient and an increased amount of NSAIDs was needed to control the headache. So, if the patient were given a trial of indomethacin, the headache might be relieved to some degree as expected, but would require a high dose of the drug.
Fourth, as stated above, many cases of HC with atypical features and in association with other pathologies (symptomatic HC) have been reported. There is no clinical characteristic that differentiates between primary and symptomatic HC. Also, to clarify the causal relationship between the associated pathology and the headache, or the possibility of a coincidental association, Trucco and colleagues recently proposed specific criteria, including close temporal relationship between the associated disease and the onset of pain, side concordance between the unilateral pain and the lesion, surgical remission or prompt remission after aetiological medical therapy and prolonged post-treatment follow-up (49). Our case met the criteria of side concordance, remission of fixed unilateral headache after surgical therapy of the primary pathology and prolonged post-treatment follow-up. So, our case demonstrates that a clinical presentation compatible with HC may be causally related to ethmoid sinus pathology.
Conclusion
Our patient was symptomatic with a strict unilateral headache syndrome with features suggestive of HC, raising the possibility of a new possible cause for symptomatic HC, thereby reinforcing the indication for investigations in any case of HC or TAC with atypical features.
