Abstract
A 64-year-old woman presented with a 6-month history of right-sided continuous headache, without autonomic symptoms and complete response to indomethacin. Clinical examination and structural brain imaging were normal. A diagnosis of hemicrania continua (HC) was made. We sought to determine the brain structures active during the pain in a patient who met all of the diagnostic criteria for HC with the exception of autonomic symptoms. A brain positron emission tomography study was performed during pain, and completely pain-free after indomethacin administration. Comparing the pain with pain-free states, the region of the dorsal pons was significantly activated. There was no activation in the hypothalamus, as previously reported in HC with autonomic symptoms. Although definitive conclusions can not be drawn from a single observation, the lack of autonomic symptoms along with the absence of hypothalamic activation suggests that the clinical presentation may predict the pattern of brain activation in primary headache syndromes.
Introduction
Hemicrania continua (HC) is a strictly unilateral continuous headache of moderate intensity with superimposed pain exacerbations, which are often accompanied by autonomic symptoms, and complete response to indomethacin (1). This primary headache syndrome shares some clinical features with migraine and the group of trigeminal autonomic cephalalgias (TACs) that include cluster headache, paroxysmal hemicrania and short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT). Neuroimaging studies suggest that the dorsolateral pons and hypothalamic activities are markers of migraine and TACs, respectively (2). A brain positron emission tomography (PET) study in HC patients (3) has previously demonstrated activation of the dorsal rostral pons and hypothalamus in correspondence with clinical features of migraine and TACs.
In this study, we sought to determine the brain structures active during the pain in a patient who met all of the diagnostic criteria for HC with the exception of autonomic symptoms. Based on the clinical features, we hypothesize that HC without autonomic symptoms may involve activation of the dorsal rostral pons, but not of the hypothalamus.
Patient and methods
Patient
A 64-year-old, right-handed woman was referred to our clinic with a 6-month history of daily headache. She described a strictly unilateral headache that was located in the right periorbital and occipital region. It had a boring quality and was associated with nausea, unilateral photophobia ipsilateral to the pain and worsening with movement. The headache was of medium intensity on a daily basis. She rated her daily pain at 7/10 on the verbal rating scale (VRS). The patient also had episodic daytime exacerbations lasting 15 min to 24 h, during which there were no autonomic symptoms, as noted during witnessed episodes (P.I.). She reported some relief with ibuprofen and aspirin, but not with paracetamol, tramadol or oral sumatriptan. General and neurological examinations were normal, except for the presence of tenderness in the right suboccipital region. Her past medical history included hypertension (treated with enalapril 20 mg/day) and kidney stones. There was no personal or family history of headache. The patient had previously been treated with pregabalin (150 mg/day) and amitriptyline (25 mg/day) without benefit. Magnetic resonance imaging (MRI) of the brain and neck was normal.
The response to indomethacin treatment was evaluated using a placebo-controlled intramuscular (i.m.) indomethacin or modified indotest (3). We administered a single dose of i.m. placebo that did not produce any benefit, but after i.m. indomethacin (100 mg), there was a prompt (within 45 min) and absolute response. A diagnosis of HC without autonomic symptoms was made. Chronic treatment with oral indomethacin 75 mg/day brought nearly complete and stable pain relief. Skipping or delaying doses cause reappearance of the headache. After 3 months' treatment, indomethacin was increased up to 125 mg/day because her background headache worsened, but the dose had to be reduced because of gastro-oesophageal reflux. We then performed a greater occipital nerve (GON) blockade with steroids and anaesthetic in the right suboccipital region, as previously reported (4), achieving complete pain relief for about 1 month. GON blockade was repeated on four more occasions with benefit lasting 2–4 weeks. Finally, our patient underwent occipital nerve stimulation (Itrel III, model 7425; Medtronic, Watford, UK). Since implantation the patient has been followed for 18 months. She has been able to reduce indomethacin and finally stop taking this drug without recurrence of pain.
Brain PET procedure
Informed consent was obtained from the subject and the study was approved by the local Ethics Committee. She was scanned 10 times in a single PET session, and relative cerebral blood flow (rel-CBF) was measured by means of 15O-water with standard laboratory procedures. The scans were performed on an ECAT EXACT HR+ tomograph (Siemens/CTI, Knoxville, TN, USA) using three rotating rod sources of 68Ge/68Ga for transmission scanning. In order to minimize head movement, a softened thermoplastic mask was moulded to the subject's facial contour and fastened to the head holder. Measurements of rel-CBF were made after an intravenous bolus injection of 10–11 mCi of 15O-water in 5–7 ml of saline. Approximately 20 s after the injection (when measured counts exceed a threshold value of 100 kct/s), a 60-s scanning frame started. Data were acquired and reconstructed in full three-dimensional mode. She underwent two blocks of five scans at 15-min intervals, first during the pain and subsequently when she was completely pain-free after indomethacin. A dose of 150 mg i.m. indomethacin was used to provide clear-cut pain relief, because the patient had required an increase from 75 mg to 125 mg/day over the previous 2 months. She was advised to stop taking indomethacin 36 h prior to the scanning session. The patient was scanned before performing any GON blockade.
Brain PET scans collected were mathematically registered to the subject's T1-weighted MRI (Symphony 1.5 T; Siemens) using a mutual information algorithm and rigid body transformations (eSoft; Siemens) (5). Data were analysed using the Statistical Parametric Mapping program (SPM2) (6) in MATLAB (Mathworks Inc., Sherborn, MA, USA). The MRI scan was spatially normalized into MNI space (MNI, Montreal Neurological Institute, Quebec, Canada) using linear transformation, and the transformation parameters were then applied to the co-registered PET scans (SPM2). The resultant images were smoothed using a three-dimensional 12-mm Gaussian filter to suppress noise and minimize effects of normalization errors by increasing the sensitivity of the signal. Differences in global activity within subject were removed by adjusting global means by proportional scaling to a value of 50. Comparisons across the two conditions were made on a voxel-by-voxel basis using the t-statistic. Contrast was defined to assess significant increased metabolism due to hemicrania. An uncorrected threshold of P < 0.001 was chosen for tabular and graphical reporting, and the minimum cluster size was > 20.
Results
Clinical data
Our patient reported her usual headache without autonomic symptoms during the scanning session. She rated her pain at 8/10 on VRS. Indomethacin injection (150 mg) provided complete pain relief after 45 min and the pain-free period lasted approximately 8 h.
PET data
Brain regions significantly activated during headache state compared with the headache-free state after indomethacin administration are shown in Table 1 and Fig. 1. A significant rel-CBF increase was observed in the right insula with some extension to the ipsilateral putamen and inferior frontal cortex (Fig. 2). In addition, significant activation was present in the dorsal aspect of the pons, predominantly lateralized to the right side (Fig. 3). Finally, activation was also found in bilateral temporal cortices, anterior cingulate cortex, bilateral parietal cortices, left frontal cortex, and the left cerebellum (P < 0.001 uncorrected for multiple comparisons).
Regions and coordinates of significant increases in relative cerebral blood flow (P < 0.001, uncorrected) during headache compared with the headache-free state after indomethacin administration
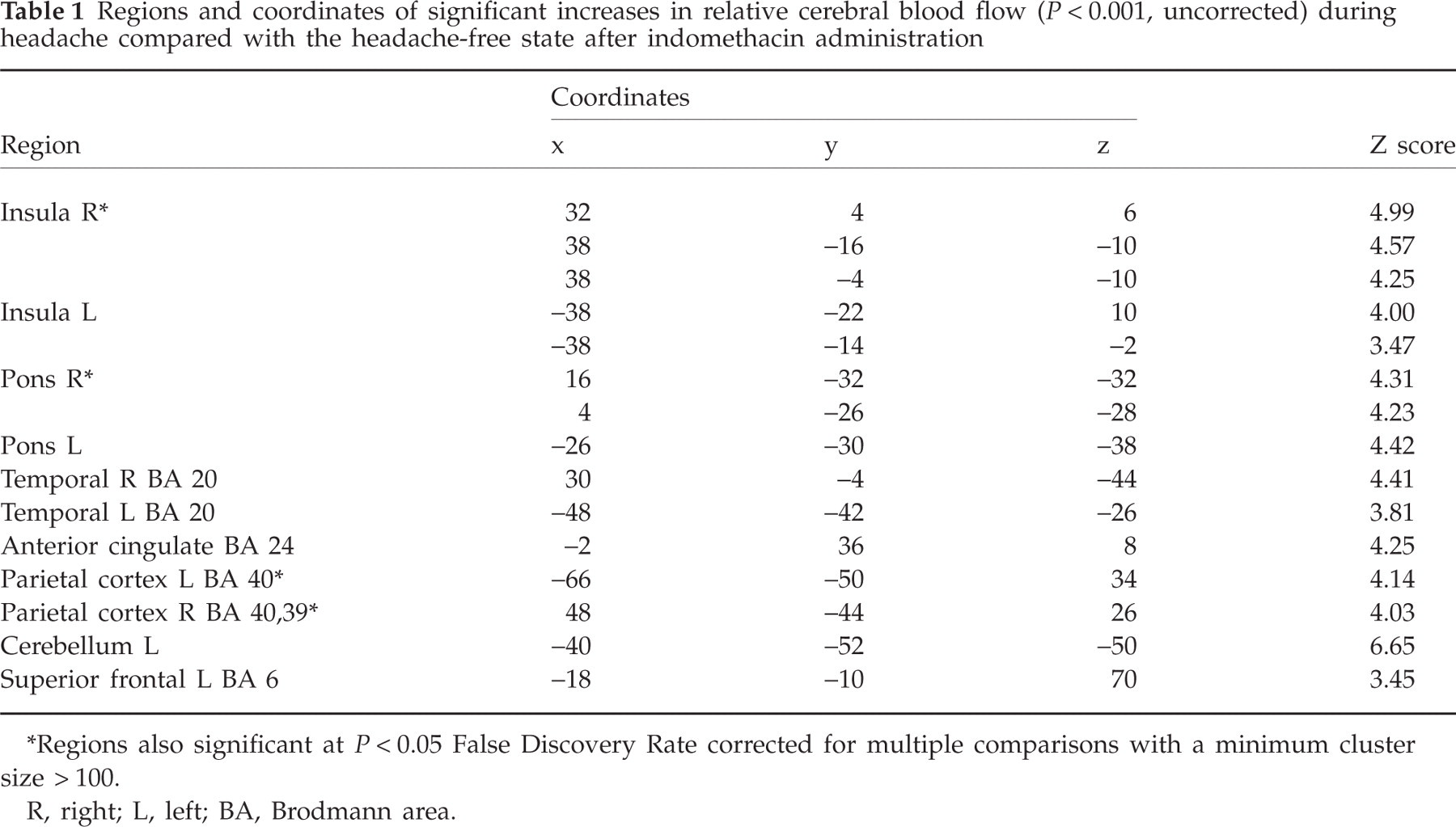
∗Regions also significant at P < 0.05 False Discovery Rate corrected for multiple comparisons with a minimum cluster size > 100.
R, right; L, left; BA, Brodmann area.
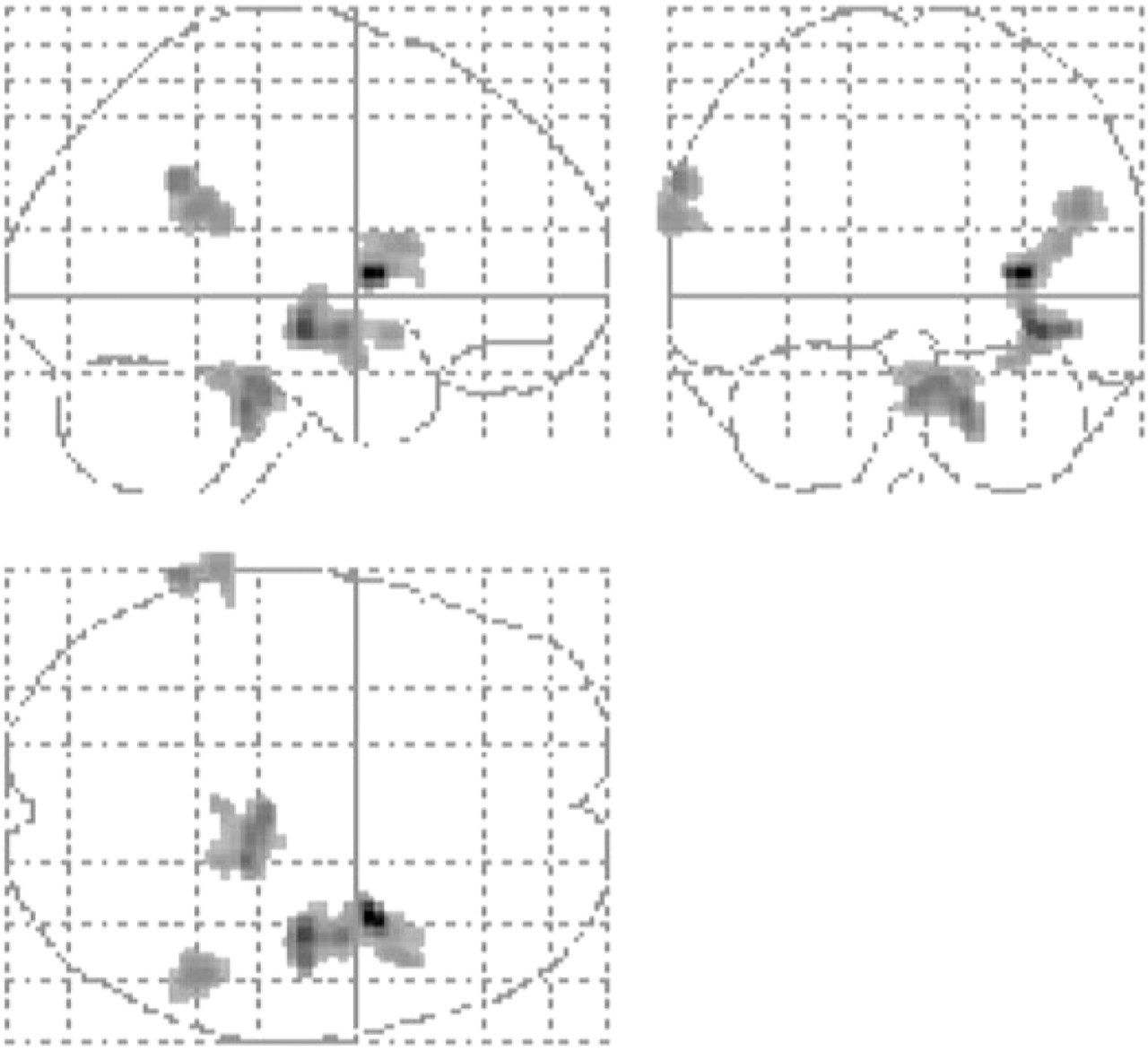
Glass brain figure for statistical parametric maps showing significant increased relative cerebral blood flow during headache attack (P < 0.05 False Discovery Rate corrected for multiple comparisons; cluster size > 100).

Statistical parametric maps showing significant increased relative cerebral blood flow during headache attack, in the right insula with extension to the putamen, presented over the patient T1-weighted magnetic resonance image (P < 0.001, uncorrected).

Statistical parametric maps showing significantly increased relative cerebral blood flow during headache attack in the dorsal aspect of the pons, lateralized to the right side, presented over the patient T1-weighted magnetic resonance image (P < 0.001, uncorrected).
Discussion
This study has confirmed the activation of the dorsal pons ipsilateral to the pain in HC without autonomic symptoms and provided strong evidence for the involvement of the brainstem in the pathophysiology of HC. This finding is consistent with a previous report of one patient who developed HC ipsilateral to a brainstem lesion (7). Even more striking is the fact that in our patient no hypothalamic activation was found during the headache. Interestingly, the common clinical features of TACs, in which hypothalamic activation has been reported, are prominent cranial autonomic symptoms. Although definitive conclusions can not be drawn from a single observation, the lack of autonomic symptoms in our patient, along with the absence of hypothalamic activation, suggests that the clinical presentation may predict the pattern of brain activation in primary headache syndromes.
PET studies in spontaneous or provoked attacks of migraine have shown brainstem activation (8–10), specifically localized in the dorsolateral pons usually ipsilateral to the pain (11). A significant increase of cerebral metabolism in the brainstem has been also reported in chronic migraine patients (12, 13). A PET study in cluster headache patients (14) showed activation in the hypothalamic region during the attacks. Subsequent publications have demonstrated hypothalamic activation during pain attacks in paroxysmal hemicrania (15) and SUNCT syndrome (16). In 2004, Matharu and coworkers (3) carried out PET studies on seven patients with HC (all of them with autonomic symptoms), observing significant activation of the ipsilateral dorsal rostral pons, contralateral posterior hypothalamus and ventral midbrain during HC. Their study demonstrated that the neuroimaging markers of migraine (brainstem) and TACs (hypothalamus) appear in HC, in correspondence with clinical features of both headache forms. However, the specificity of neuroimaging markers of migraine and TACs has been questioned on the basis that it has been reported in other primary headache syndromes. Recently, Denuelle et al. (17) observed brainstem but also hypothalamic activation in seven patients with migraine without aura attacks, all of them apparently without autonomic symptoms. In the case presented here, we observed brainstem activation in the same region previously reported in migraine patients.
In our patient, the diagnosis of HC without autonomic symptoms is supported by the clinical phenotype and exquisite response to indomethacin, and chronic migraine should be considered in the differential diagnosis. The pain was persistent and strictly unilateral, centred in the periorbital region, and there had been no previous episodic headache attacks. Migraine, although often more severe on one side, is frequently bilateral or with side-alternating attacks (18), and chronic migraine usually evolves from the episodic form (19). Also, unilateral photophobia ipsilateral to the pain, as occurs in this case, is much more common in HC patients than in migraine sufferers (20). The presence of at least one autonomic symptom during pain exacerbations is mandatory for HC diagnosis according to The International Headache Classification (1). However, autonomic features in HC are not as prominent as in TACs, and may be absent. In the largest series of HC, autonomic symptoms were not present in 26–37% of patients (21). Neck tenderness ipsilateral to the pain, as occurred in our patient, is also common in HC (21), and occipital nerve stimulation appears to offer an effective treatment option, particularly when indomethacin is not tolerated (22, 23). Finally, complete response to indomethacin confirmed our diagnosis of HC (1, 3). The effect of indomethacin is rapid, reproducible, and can be easily differentiated from placebo (3).
The main limitation of this study is that PET was performed on one patient only, but it is important to emphasize that is extremely difficult to recruit patients with HC without autonomic symptoms. HC is an uncommon condition—in fact there are no more than a few hundred cases in the literature—and HC without autonomic symptoms is even rarer. Consequently, this patient offered a unique opportunity to study some fundamental aspects of HC pathophysiology. Also, it could be argued that data based on a single patient can not be taken as proof of missing activation in the hypothalamus. In our study this limitation has been overcome by increasing the number of scans to five for each condition and using a permissive threshold to analyse the PET data. Even after this analysis we did not observe any activation trend in the hypothalamic region.
In conclusion, we observed activation of the brainstem but not of the hypothalamus during pain attacks in HC without autonomic symptoms. Further neuroimaging studies comparing patients who suffer idiopathic headache syndromes with and without autonomic features are needed to extend our results, and may also be helpful to seek the anatomical or functional basis for the variation in symptoms.
Footnotes
Acknowledgements
This study was supported by an unrestricted grant from Almirall-Prodesfarma Pharmaceuticals, Barcelona, Spain. We thank Dr María Rosario Luquin for her critical review of this manuscript.
