Abstract
Angiotensin-converting enzyme (ACE) insertion (I)/deletion (D) and methylene-tetrahydrofolate reductase (MTHFR) C677T polymorphisms are linked to endothelial dysfunction and to cerebral white matter lesions. Objectives of this study were to determine if ACE and MTHFR gene polymorphisms are associated with von Willebrand factor (vWF) activity, an endothelial dysfunction marker, and with a distinct headache phenotype. We enrolled 64 women (18–50 years old) with International Classification of Headache Disorders, 2nd edn migraine without aura (MoA) and 61 with aura (MA). Genotypic frequencies: ACE DD 35%, ID 42%, II 23%, and MTHFR TT 17%, CT 40%, CC 43%. Those with ACE DD genotype had higher levels of vWF activity (152%) compared with ID and II genotypes. Levels were highest (179%) with combined ACE DD and MTHFR TT genotypes. ACE DD was associated with higher headache frequency, and MTHFR TT was associated with MA. In migraine, vWF activity may be a marker of endothelial-mediated genetic risk for ischaemic conditions.
Keywords
Introduction
Migraine is a risk factor for stroke, especially in women of reproductive age and those with aura (1, 2). The mechanisms to account for the migraine–stroke association have been elusive (3), but there is mounting evidence that endothelial dysfunction may play a role (4). Endothelial dysfunction is typified by impairment of vascular reactivity and by endothelial activation, which is characterized by a procoagulatory, proinflammatory and proliferative state. Impacted by traditional risk factors and mediated by oxidative stress (5, 6), endothelial dysfunction represents ‘the ultimate risk of the risk factors'. Endothelial dysfunction is associated with, and predicts, an increased rate of cerebro- and cardiovascular ischaemic events (7). There is increasing evidence that migraine may be a non-traditional risk factor for endothelial dysfunction (4, 8–11).
In recent years, genetic factors that increase susceptibility to endothelial dysfunction (12–14) have been linked to both stroke (15) and migraine (16–18), although significant controversy remains. Of particular interest in the migraine–stroke relationship are the angiotensin-converting enzyme (ACE) gene insertion/deletion (ACE I/D) and the methylenetetrahydrofolate reductase (MTHFR) gene C677T polymorphisms. The ACE catalyses the conversion of angiotensin I to angiotensin II, and in individuals carrying the deletion genotype (DD), the circulating level and tissue activity of ACE are higher (19). The ACE DD genotype has been tied to hypercoagulability (12), decreased bradykinin, increased vascular tone and smooth muscle hypertrophy (20, 21). The relationship of ACE DD genotype to ischaemic stroke and cardiovascular disease has been debated (22–25), as has the association with migraine (17, 26, 27). The MTHFR gene regulates a key enzyme in the metabolism of homocysteine (28). The most common mutation of this gene, C677T, is a risk factor for vascular disease (29) and has been linked to stroke (30) and migraine, particularly migraine with aura (MA) (18, 31–33). One recent population-based study, however, noted that women with the MTHFR C677TT genotype were less likely to have MA, although those with aura and this genotype had an increased risk of ischaemic stroke (34). Studies investigating gene–gene interactions have reported that the ACE DD genotype in combination with MTHFR TT genotype increased susceptibility to migraine, particularly MA (35), as well as stroke (36).
One of the most widely accepted and accessible markers of endothelial dysfunction is von Willebrand factor (vWF) (37). Raised levels of vWF are associated with each major cardiovascular risk factor (38, 39). vWF has also been linked to stroke in clinic- and community-based longitudinal studies (40, 41). In particular, vWF has been related to lacunar, non-lacunar and cardioembolic stroke (42, 43). Although there is a paucity of studies of this marker in migraine, two studies have demonstrated that vWF activity and antigen levels are higher in migraineurs than in non-headache controls during the interictal phase (44, 45). Levels of vWF have also been shown to increase during the course of the migraine attack (46), suggesting that migraine may be caused by or a consequence of endothelial dysfunction. Recent work has established that vWF activity is a robust marker of endothelial dysfunction in young women with migraine, being associated with increased platelet aggregation, decreased fibrinolysis, increased inflammation, increased oxidative stress and decreased cerebrovascular reactivity (47).
With the hypothesis that ACE I/D and the MTHFR C677T polymorphisms confer a genetic risk for endothelial dysfunction in young women with migraine, the objectives of this study were twofold: (i) to determine in this population whether ACE I/D, and MTHFR C677T polymorphisms are associated with vWF activity; and (ii) to determine if ACE I/D and MTHFR C677T polymorphisms are associated with a distinct headache phenotype.
Methods
Study population
This study was conducted between February 2006 and October 2006 after approval by the Institutional Review Board. Participants were recruited from advertisements in the ambulatory Headache Center, University campus website, radio and local newspaper. Within the Headache Center, participation was offered to consecutive eligible patients after evaluation by the principal investigator. Each prospective candidate from outside the clinic population was interviewed and examined by the investigator to determine eligibility. The enrolment plan was to include equal numbers of MA and migraine without aura (MoA) patients. Inclusion criteria were: (i) women with MA, MoA as defined by criteria set forth in the International Classification of Headache Disorders (ICHD-II codes: 1.1, 1.2, 1.5) (48); (ii) age 18–50 years; and (iii) headache free for at least 7 days at time of enrolment. Exclusion criteria were: (i) not physically well enough to give blood; (ii) diabetes mellitus, vasculitis, prior stroke/transient ischaemic attack (TIA), pregnancy (self reported), myocardial infarction, systemic lupus erythematosus; (iii) use of anticoagulants; (iv) use of non-steroidal anti-inflammatory drugs (NSAIDs) or other antiplatelet agents in the week prior to testing; and (v) not literate in English. This clinic-based pilot study included only young women, as migraine is more prevalent in this group and the majority of studies demonstrating a link between migraine and stroke are in women, particularly premenopausal women.
Clinical information
Each participant completed a questionnaire regarding age, sex, education, household income, height, weight, age of headache onset, headache-related disability [using Headache Impact Test (HIT)-6] (49), physician-diagnosed medical conditions (including hypertension, diabetes mellitus, smoking, hyperlipidaemia, history of deep venous thrombosis, pulmonary embolism). The principal investigator supplied the following information: ICHD-II headache diagnoses, average monthly days with headache over the previous 3 months, classes of medications (ACE inhibitors, angiotensin II receptor blockers, calcium channel blockers, statins, NSAIDs, antiplatelet agents). If a participant was taking NSAIDs or other antiplatelet agents, and answered yes on the questionnaire, they did not have blood drawn until they had been off the medication for at least 1 week. Participants with frequent headache often had them on multiple consecutive days around the menstrual period, but all participants indicated that they had been headache free for 7 days at the time the blood was drawn.
Laboratory methods
All testing was done when the participant had not used NSAIDs or other antiplatelet agents for at least 1 week, and following an overnight fast. Blood was collected between 08.00 h and 09.00 h in order to decrease diurnal variation, if any, in the measurements. All blood samples were coded and analysis was performed blinded to participant health or laboratory information. Blood was drawn into collection tubes with 3.2% buffered sodium citrate for vWF activity, and the tubes were centrifuged at 20°C at 2500
Genetic analysis
ACE I/D genotyping
Blood was collected in two 4-ml ethylenediamine tetraaceticacid tubes and frozen at −70°C for the ACE I/D genotyping. Prior to testing, genomic DNA was isolated from 200 μl of whole human blood using the QIAamp 96 DNA blood kit (Qiagen, Valencia, CA, USA). The ACE gene polymorphism is located within intron 16 and is identified with presence (insertion, I) or absence (deletion, D) of a 287-bp sequence resulting in one of three possible genotypes—DD, II or ID in an individual. These genotypes were detected by polymerase chain reaction (PCR) followed by visualization of the resultant amplicons on agarose gels (23). Specific sense and antisense oligonucleotide sequences or primers required to amplify the region of ACE intron 16 are as follows: Hace3s, 5′CCCTGCAGGTGTCTGCAGCATGT3′; and Hace3as, 5′GGATGGCTCTCCCCGCCTTGTCTC3′. The primers were custom synthesized using commercial services (Integrated DNA Technologies, Coralville, IA, USA). Primers (1 μM) in a PCR master mixture containing 200 μM deoxynucleotide triphosphates, 1.3 mM magnesium chloride, 50 mM potassium chloride, 10 mM Tris–hydrochloric acid (pH 8.4), 0.1% Triton X-100 and 0.35 U of Taq DNA polymerase were used to set up and amplify 319-bp and 597-bp amplicons corresponding to the D and I alleles of ACE, respectively. The thermocycling conditions included 35 cycles of denaturation at 94°C for 30 s, annealing at 56°C for 45 s and extension at 72°C, followed by a final extension at 72°C for 7 min. The PCR products obtained were subjected to 1.5% agarose gel electrophoresis alongside a size marker DNA ladder. Ethidium bromide intercalated DNA fragments that are size fractionated on the agarose gel were visualized under ultraviolet light. Samples genotypes were recorded as DD if only one band corresponding to 319 bp was observed or as II if only one band corresponding to 597 bp was observed. Samples were recorded as heterozygous ‘ID' if both bands at 319 bp and 597 bp were observed. The D allele in heterozygous samples is known to be preferentially amplified (23). Therefore, the samples with the DD genotype were subjected to a second, independent PCR amplification with primers (hace5a, 5′TGGGACCACAGCGCCCGCCACTAC3′ and 5′TCGCCAGCCCTCCCATGCCCATAA3′) that recognize an insertion specific sequence. All methods were identical as described above except for the PCR annealing temperature of 67°C instead of 56°C. For samples that were heterozygous, a 335-bp amplicon representing the insertion allele was expected, whereas no product was expected for samples that are truly homozygous for DD.
MTHFR C677T genotyping
Genotyping of this polymorphism was performed by direct sequencing method. Genomic sequence of MTHFR around the C677T allele was used to design primers at the Primer3 website ( http://fokker.wi.mit.edu/primer3/input.htm). M13 tags were attached to the primers synthesized. The sense and antisense primers designed were TGTAAAACGACGGCCAGTTGAAGGAGAAGGTGTCTGCGGAA and CAGGAAACAGCTATGACCAGGACGGTGCGGTGAGAGTG, respectively. PCR was performed in a 96-well format and the products were subjected to agarose gel electrophoresis to visualize the quality of the PCR products. On visualizing single, clear bands of expected sizes, the products were purified using Qiagen columns and sequenced using the M13 tags. Multiple bands were excised, gel purified and subjected to sequencing. Sequences (performed by Seqwright, Inc., Houston, TX, USA) were aligned using the DNA analysis software called Sequencher (Gene Codes Corp., Ann Arbor, MI, USA). Alleles were directly visualized on the chromatograms and recorded.
Statistical analysis
Discrete data were analysed using the χ2 test, and continuous data were analysed using unpaired t-test or analysis of variance. Non-parametric statistics were used for comparison of data with deviations from normality or for ordinal variables such as HIT-6 scores. Genotypic frequencies were calculated and Hardy–Weinberg equilibrium analysis was used to ascertain the stability of frequencies within the sample. Information on ACE I/D polymorphism was not available in one, and on the MTHFR C677T polymorphism in two of the study participants. The association of ACE (II, ID, DD) and MTHFR (CC, CT, TT) genotypes with the migraine type (MA, MoA) was examined using logistic regression. The relationship between the genotypes and plasma vWF activity was examined in general linear models of regression. Interaction between the ACE and MTHFR genotypes was tested using the standard method of including a term for ACE × MTHFR analysis in the model. To warrant valid analysis, all regression models were adjusted only for the age of the study participants. The homogeneity of variances in the models was tested, and Dunnett's adjustment for multiple comparisons was done for better statistical power with restricted number of comparisons. Hypotheses were tested at 0.05 level of significance, and for genotypic multiple comparisons a P < 0.025 was used to infer statistical significance. All analyses were performed using SAS version 9.1 (SAS Institute Inc., Cary, NC, USA).
Results
All eligible women seen in the clinic during the study period were invited to participate. Only five persons declined, and three people agreed to participate, but were not included as blood could not be obtained. A total of 125 female patients with migraine were enrolled in the study. Thirty-seven of the 125 participants (30%) were from the community (non-clinic based). The mean age was 37 years (
Classification of study population by diagnosis

∗International Classification of Headache Disorders, 2nd edn code for diagnosis of headache.
Characteristics of the study population
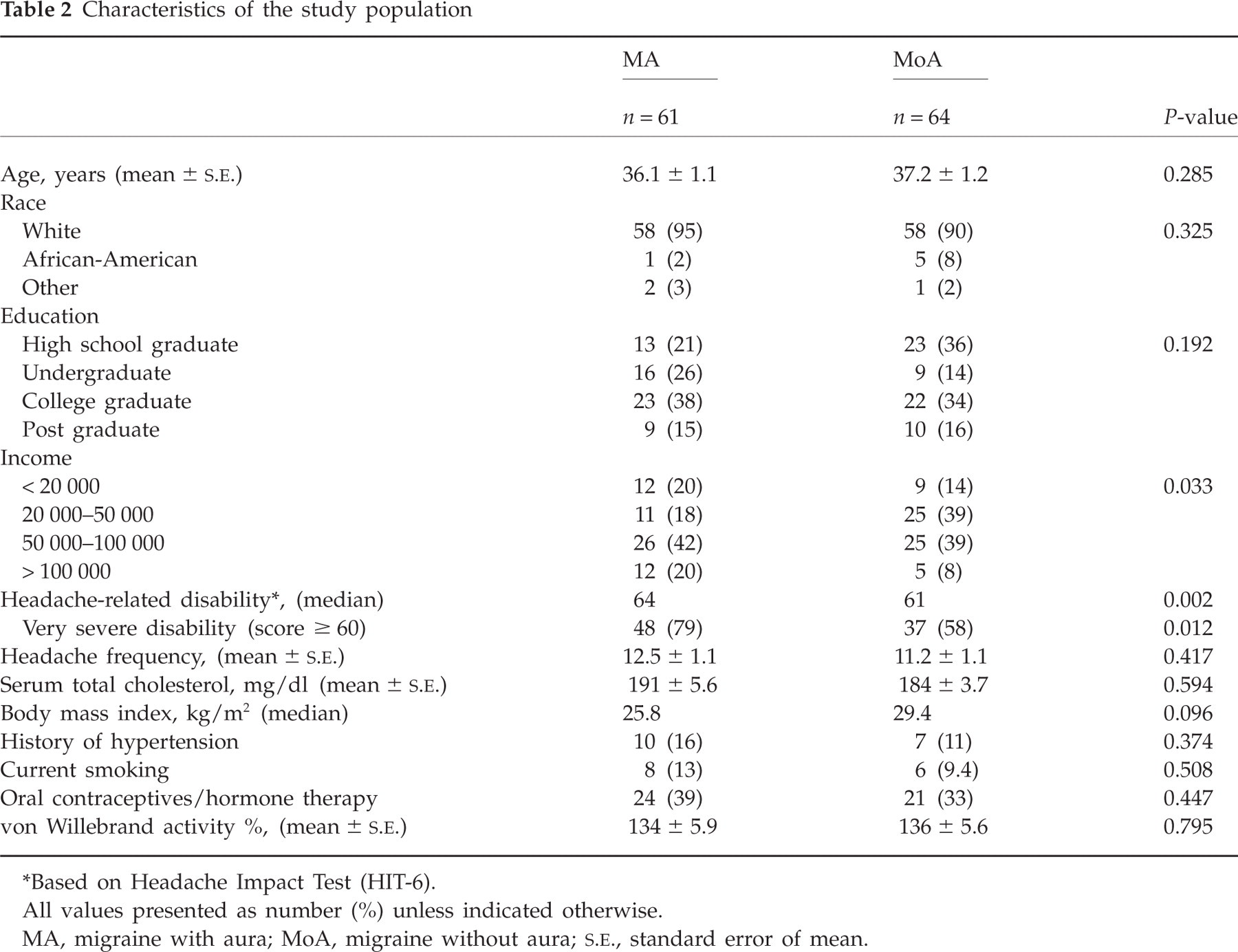
∗Based on Headache Impact Test (HIT-6).
All values presented as number (%) unless indicated otherwise.
MA, migraine with aura; MoA, migraine without aura;
Table 3 illustrates the distribution of ACE I/D and the MTHFR C677T polymorphisms in the study population and the association of genotypic frequencies with migraine type, headache frequency and level of vWF activity. In this study, the genotypic frequencies of ACE I/D (χ2 = 2.8, P = 0.09) and MTHFR C677T (χ2 = 2.6, P = 0.11) polymorphisms did not differ significantly from those predicted by Hardy–Weinberg equilibrium. Homozygous DD genotype of ACE I/D polymorphism was observed in 35% and the ID genotype in 42% of the study population. The individual frequencies of D and I alleles were 0.56 and 0.44, respectively. Most of the study population (53%) had the CC genotype, and the high-risk TT genotype was observed in 17% of all migraineurs. Frequencies of T and C alleles were 0.37 and 0.63, respectively. Genotypic frequencies of ACE I/D polymorphism did not differ by migraine type, but the frequency of the TT and CT genotypes of MTHFR C677T polymorphism were significantly higher in the MA group (χ2 = 8.72, P = 0.013). Odds ratios indicate that the relationship between MTHFR genotypes and aura increased in strength with an increase in the number of T alleles. Persons with TT genotype were nearly four times more likely to have aura compared with those with CC genotype. A trend was noted for higher headache frequency in persons with ACE DD and ID genotypes, but no such relationship was observed with the MTHFR genotypes. The average monthly headache frequency was highest in persons with DD genotype. There were no differences in the other headache characteristics such as headache-related disability, age at onset of headaches or duration since onset between the ACE or MTHFR genotypes.
Association of the ACE and MTHFR genotypes with migraine aura, frequency and vWF activity

∗Values do not add to the total due to missing information (see Methods).
†Odds ratios (unadjusted) for MA vs. MoA.
‡ P < 0.05 vs. CC.
§ P < 0.05 vs. II, from Dunnett's adjustment for multiple comparisons.
¶ P < 0.025 vs. ID and P < 0.005 vs. II, from Dunnett's adjustment for multiple comparisons.
MA, migraine with aura; MoA, migraine without aura; OR, odds ratio; CI, confidence intervals; vWF, von Willebrand factor; ACE, angiotensin-converting enzyme; MTHFR, methylenetetrahydrofolate reductase.
Levels of vWF activity were significantly different between ACE DD, ID and II genotypes in age-adjusted regression analysis (F = 4.88, P = 0.003). Migraineurs with the DD genotype had higher levels of vWF activity compared with ID or II genotypes. Similarly, migraineurs with MTHFR TT genotype had higher levels of vWF activity, but the difference was not significant. Using the standard method for examining interaction, no evidence was observed for a gene × gene relationship. Analysis indicated that the study sample (n = 125) lacked sufficient power to detect a difference in vWF activity between the MTHFR genotypes, or evaluate the interaction between the genotypes. However, data in support of a combined genetic risk were found by examining the distribution of vWF activity based on the combination of high risk, homozygous mutations of the ACE and MTHFR genes. Four groups generated based on the homozygous genotypes (DD and TT, DD and no TT, TT and no DD, and no DD and no TT) recorded different levels of vWF activity. Migraineurs homozygous for both the gene mutations (DD and TT) had the highest level of vWF activity (F = 2.53, P = 0.043) (Fig. 1).
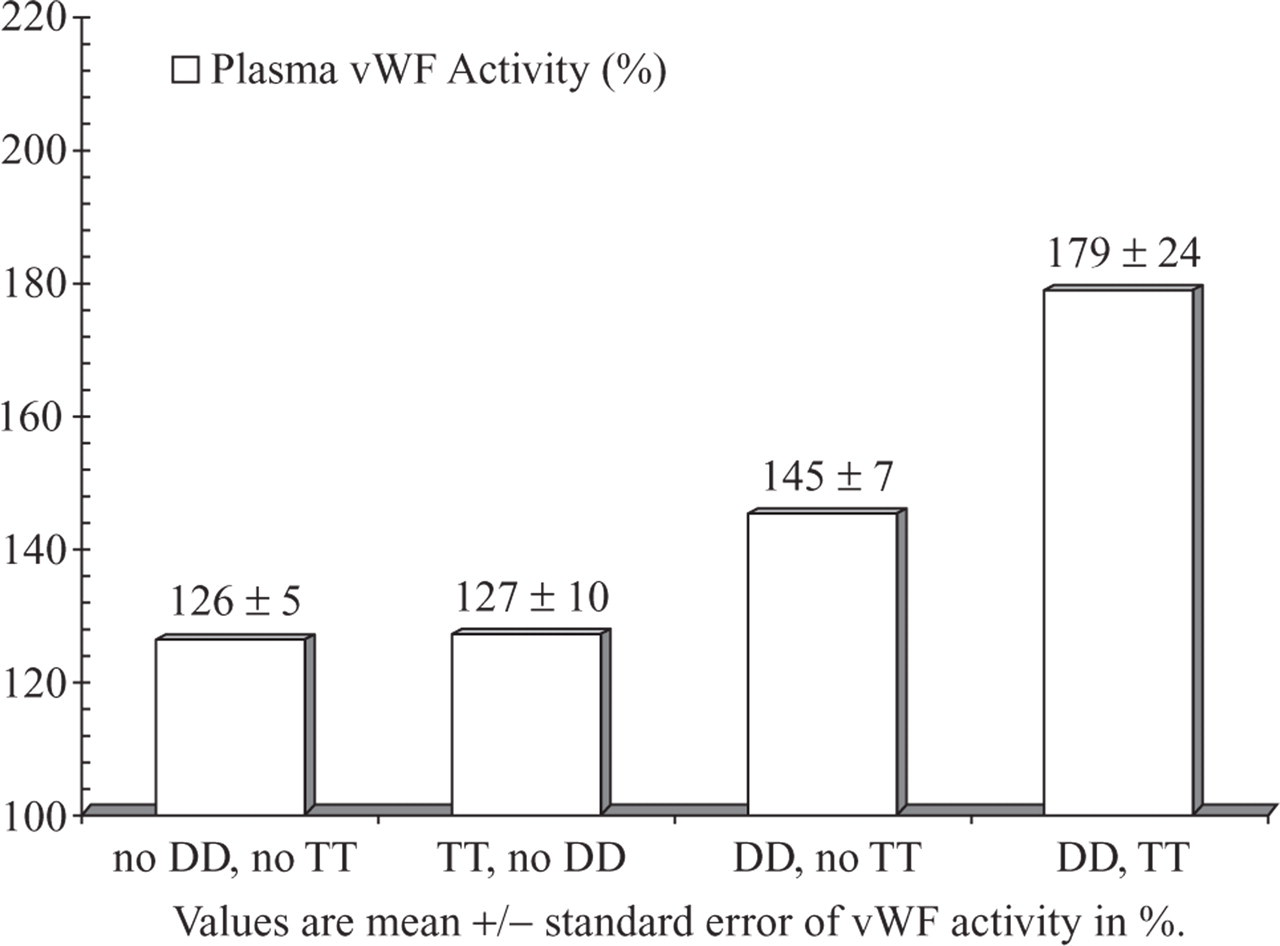
Combined effect of angiotensin-converting enzyme (ACE) DD and methylenetetrahydrofolate reductase (MTHFR) TT genotypes on plasma von Willebrand factor (vWF) activity.
Discussion
In this study of premenopausal women with migraine, our findings may have implications for migraine pathogenesis and the migraine–stroke association. A novel discovery in a neurovascular condition, elevated vWF activity was found to be associated with the ACE DD genotype, and highest when combined with MTHFR TT genotype. Supportive of previous studies, we have demonstrated that higher headache frequency was associated with ACE DD genotype, whereas diagnosis of aura was linked to the MTHFR TT genotype. The implications of these findings are best considered in the context of the related literature.
vWF activity is related to endothelial dysfunction and is a marker of stroke risk. The two genetic polymorphisms we studied have both been linked to endothelial dysfunction (12–14, 50). Our finding of increased vWF activity in the ACE DD subgroup, particularly when combined with MTHFR TT genotype, supports the conjecture that increased risk of vascular events in migraine may be related to a genetic predisposition to endothelial damage (51).
Our study was not designed to address whether these polymorphisms predicted migraine risk. Many (16, 17, 31–33), but not all (27, 34, 52, 53) earlier studies have associated ACE DD and MTHFR TT with migraine, predominantly MA. A positive association may implicate genetically mediated vascular dysfunction as a cause of migraine or at least a determinant of the clinical phenotype. We found that headaches were more frequent in persons with DD genotype. This supports an earlier study in MoA (26) and implicates a prominent role of the vasculature in determining frequency. Experimental studies have demonstrated that there is a mechanism, intrinsic to the vasculature, for propagation of vasodilation (54), although this has not been tied to any specific genotype. The finding that aura is positively linked to MTHFR TT has also been supported by earlier studies (18, 31–33), although not uniformly (34, 53). A potential mechanism is homocysteine-mediated platelet activation and hypercoagulability leading to ischaemia-induced cortical spreading depression (55, 56). Although speculative, aura might, in this context, be considered a TIA equivalent and, as such, a predictor of ischaemic stroke (33).
Limitations of our study include the small sample size and the fact that certain associations may have been missed due to lack of statistical power. Study participants came from both the clinic and the general population, and the generalizability of the findings needs confirmation in larger samples. We restricted this study to premenopausal women, but there are prospective data suggesting that the risk of stroke with migraine is increased in men as well as older women (51, 57). By excluding women with a history of cardiovascular disease, certain relationships with endothelial dysfunction markers may have been missed, especially if cardiovascular disease is related to migraine in the same causal pathway. Despite the fact that consecutive clinic patients were invited to participate during the recruitment period, and the study was advertised to the general population, selection bias is possible. Subjects in the study were taken off antiplatelet agents, including NSAIDs, but other medications were not restricted and medication use may influence headache phenotype. Because we did not gather specific information on frequency of attacks of aura, we cannot ascertain the relationships of aura frequency and genotype or vWF activity.
In young female migraineurs, the ACE DD genotype is associated with elevated plasma vWF activity, a robust marker for endothelial dysfunction and stroke risk. This association is strengthened in the presence of the MTHFR TT genotype. In migraine, vWF activity appears to be a marker of endothelial-mediated genetic risk for ischaemic conditions.
Footnotes
Acknowledgements
This study was funded by a research grant from GlaxoSmithKline and by a Translational Research Stimulation Award from the University of Toledo College of Medicine.
