Abstract
Lidocaine has been used in treatment of patients with refractory headache. Personal observations of neuropsychiatric toxicity in these patients led us to review our cases and the literature systematically for lidocaine side-effects, especially neuropsychiatric symptoms. In our series of 20 patients, side-effects were observed in all, the most frequent being neuropsychiatric (75%) and cardiological (50%). When reviewing published series on intravenous lidocaine use, reports of side-effects range from 0 to 100%, with neuropsychiatric symptoms being reported in 1.8–100%. Thirty-six case reports of lidocaine-induced psychiatric symptoms were also analysed. Psychiatric symptoms of toxicity were similar in most patients, despite their differing ages, pathologies, co-therapies and lidocaine dosages. In conclusion, lidocaine neuropsychiatric toxicity has a well-recognized stereotypical clinical presentation that is probably unrecognized in headache series. As lidocaine represents an emerging alternative therapy in headache, particularly in short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing, clinicians and patients should be aware of the extent of this problem.
Introduction
Lidocaine has been used as a local anaesthetic since 1943 and as an anti-arrhythmic drug since 1950 (1). It has been used systemically in chronic and acute pain states since 1961 (2) and in headache patients since 1984 (3). Lidocaine can be a useful adjunct in some patients with headache, with the most substantial concern for neurologists and headache specialists being that of cardiac side-effects.
Lidocaine is metabolized by the microsomal enzyme system in the liver and excreted in the urine; after intravenous (i.v.) administration lidocaine plasma concentration describes a biphasic curve, with an early rapid fall around 8–17 min and a second slow decrease around 87–108 min (1). Lidocaine exerts its action through sodium channel blockade and subsequent suppression of cellular excitability (4). The effect of lidocaine in headache may be mediated by central neural inhibition at the level of the spinal trigeminal nucleus (5). Although generally regarded as a safe drug, side-effects to lidocaine have long been recognized (6, 7). The most frequent type of adverse reaction described is generally dose dependent and due to central nervous system involvement, and includes perioral numbness, drowsiness, visual disturbances, sweating, weakness, heavy respirations, muscle twitching, euphoria, agitation, difficulty in concentrating and speaking, dysarthria, diplopia, psychotic reactions, shaking and eventually generalized convulsions or coma (8, 9). These events seem to occur in a crescendo sequence but usually resolve quickly after stopping the infusion, as long as hepatic and cardiac function are normal (1, 8, 10).
Prompted by an observation of neuropsychiatric side-effects in patients we treated with i.v. lidocaine for headache, we systematically reviewed our cases. We also performed a literature review on i.v. lidocaine-induced toxicity. Our purpose was to evaluate lidocaine toxicity and its side-effect profile, with special reference to neuropsychiatric symptoms, in order to highlight the problem for the headache community.
Methods
Review of cases
We reviewed charts for all headache in-patients treated with i.v. lidocaine (lignocaine) at the National Hospital of Neurology and Neurosurgery in 2002, 2003 and up to May 2004 as part of a clinical audit of our in-patient care. The headache diagnosis of each patient was made prior to the infusion. The decision to use i.v. lidocaine was made by the clinician in charge of the case usually faced with syndromes intractable to routine medical therapy. Patients were not considered for treatment if they had a previous history of allergic reactions to local anaesthetics, cardiac arrhythmias or if they were pregnant or breast-feeding.
All patients had pretreatment cardiological evaluation, 12-lead electrocardiogram and routine biochemical tests with liver enzyme level estimations. Cardiac monitoring and fluid chart balance were maintained during the course of the infusion. The treatment consisted of a constant infusion of a 0.4% lidocaine solution—2 g lidocaine diluted in 500 ml glucose 5%—that was started at 1 mg/min (11). Dose escalation was determined individually and was typically performed at 1 mg/min increases every 24 h, moving to a maximum dose of 4 mg/min (approximately 3.4 mg kg−1 h−1), if required. At the start and at each dose escalation, pulse rate and blood pressure were measured every 5 min for the first 30 min, every 15 min for the next 4 h and four hourly thereafter. Hourly headache pain charts recording the pain level on a verbal rating scale of 1–10, or tabulation of attacks for short-lasting headaches, was performed throughout treatment. Levels of lidocaine in the blood were not determined.
Demographic and clinical data were tabulated as were treatment response and adverse events. The later were classified into four major groups including cardiovascular, neuropsychiatric, gastrointestinal and other symptoms. Neuropsychiatric events were further classified into neurological or psychiatric, or both, according to their main characteristics. A full description of the clinical features is included in the Results section.
Statistical analysis was performed using logistic regression to look for a predictor of psychiatric adverse events (
Literature review
We performed a MEDLINE search in order to identify case series regarding the clinical use of i.v. lidocaine in adults and case reports on lidocaine toxicity, with special interest in psychiatric effects. Keywords used included i.v. lidocaine, lignocaine, toxicity, neurological, neuropsychiatric and psychiatric. No time restrictions were applied. Papers related to non-intravenous lidocaine use were excluded. Relevant references were also sought by hand searching headache journals.
Results
Review of cases
Patients
In total 20 patients had i.v. lidocaine in the period reviewed. Twelve patients were female (60%), with an average age for all patients of 44 years (range 19–65 years). Eleven of 20 patients (55%) had short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT)/short-lasting unilateral neuralgiform headache attacks with cranial autonomic symptoms (SUNA) syndromes (13), four had migraine (two chronic migraine and two chronic migraine with medication overuse) and five had other headache types. The latter included chronic post-traumatic headache, glossopharyngeal neuralgia, idiopathic stabbing headache, trigeminal dysaesthesia and pituitary tumour-associated headache (Table 1).
Patient characteristics and lidocaine side-effects

+, yes; −, no; S∗, infusion stopped for side-effects; GBP, gabapentin; CBZ, carbamazepine; TPM, topiramate; LTG, lamotrigine; DPH, phenytoin; VPA, sodium valproate; TAD, tricyclic antidepressants; βB, β-blockers.
Investigations
Patient electrocardiograms demonstrated normal sinus rhythm and no significant conduction abnormalities. Blood tests were within normal ranges with the exception of five patients who presented with borderline elevations of serum potassium (n = 1), alanine transaminase (n = 3) and alkaline phosphatase (n = 1).
Previous treatments for headache
All patients included in the lidocaine protocol were highly refractory to previous treatments. The mean number of preventive treatments that were previously unsuccessful was 10, ranging from three to 20. The most frequent previously used drugs were amitriptyline (18/20), gabapentin (15/20), carbamazepine and topiramate (13/20), sodium valproate (12/20), indomethacin (12/20), lamotrigine and corticosteroids (10/20). Other drugs frequently used included verapamil, pizotifen, propranolol, methysergide and morphine. The range of medicines previously used relates to the fact that many patients had been referred from other clinicians and does not represent simply the range of medicines used by us.
Known comorbid illnesses
All patients had some, usually minor, previous comorbidity. This included hypercholesterolaemia (n = 5), mild asthma (n = 4), rheumatoid arthritis and irritable bowel disease (n = 3 each), high blood pressure, hypothyroidism, peptic ulcer disease and obesity (n = 2 each). Four patients were current mild smokers and four were ex-smokers. There was a previous history of depression in seven patients, one of whom had obsessive symptoms. Two patients had had previous neoplastic diseases—cerebral astrocytoma in one, and breast and cervix carcinomas in another. Other important comorbidities included severe active Crohn's disease, active ischaemic optic neuropathy, benign pituitary adenoma, brainstem lesion of unknown aetiology, left middle cerebral artery territory minor stroke and previous chemical meningitis, reported in one patient each. Three patients had a previous history compatible with cranial nerve pathology, including one patient who had Bell's palsy at age 12 years.
Headache response to i.v. lidocaine
Most patients responded favourably to lidocaine, with 17/20 reporting improvement or complete relief of previous headaches (Table 1). The average number of days of treatment was six, ranging from 2 to 10 days. Rescue treatments for headache and nausea used during the infusion included: chlorpromazine (n = 4), granisetron (n = 7), domperidone (n = 7), naproxen (n = 3) and i.v. aspirin (n = 3).
Medications concurrent with lidocaine infusion
During the course of the lidocaine infusion, patients continued their previous prophylactic headache medication as well as other chronic medication that they might have been on. Headache prophylactics included: gabapentin (n = 5), carbamazepine (n = 3), topiramate (n = 3), lamotrigine (n = 2), phenytoin (n = 1), sodium valproate (n = 1), tricyclic antidepressants (n = 4) and β-blockers (n = 2). Other chronic medication included selective serotonin re-uptake inhibitors (n = 2), benzodiazepines (n = 3) and zolpidem (n = 2), daily aspirin for secondary stroke prophylaxis (n = 2), statins (n = 3), proton pump inhibitors (n = 3), levothyroxine (n = 3) and hormonal treatments (n = 3). Other treatments included methotrexate, vitamin supplements, anti-asthma inhalators and antispasmodics or other drugs altering gut motility.
Patients less likely to benefit from lidocaine infusion were on prophylaxis with tricyclic antidepressants (χ2 4.804, P = 0.028). Phenytoin prophylaxis was also related to lower benefit from lidocaine (χ2 5.965, P = 0.015), although this relates to one patient only, who was also on tricyclic antidepressants (Table 1). Other preventives did not influence the therapeutic response to lidocaine.
Adverse events on lidocaine
All patients suffered some type of adverse event, but none was considered serious or life-threatening. The mean number of adverse events per patient was two, ranging from one to four. In seven out of 20 cases the symptoms led only to transitory infusion suspension, restarting after a short break at the same rate, whereas in four there was a permanent reduction of the infusion rate. Nine of 20 patients had their treatments eventually stopped due to side-effects, seven of which were psychiatric, one neurological and one a cardiac arrhythmia.
Patients on prophylactic headache therapy had no increased incidence of any type of adverse event (cardiovascular χ2 3.810, P = 0.051; psychiatric χ2 0.000, P = 1.000; neurological χ2 0.000, P = 1.000; gastrointestinal χ2 0.848, P = 0.357; and other χ2 0.317, P = 0.573). When analysing each preventive in isolation, two medications were related to the occurrence of cardiovascular events—gabapentin (χ2 6.667, P = 0.01) and tricyclic antidepressants (χ2 5.000, P = 0.025), although again the numbers were small. No other agent influenced any other type of event, not even β-blockers. Analysis was also performed grouping preventives into antiepileptics in general, enzymatic inductor antiepileptics and antiepileptics with action on sodium channels. Still, we found no evidence of influence of these medications on the occurrence of adverse events.
Cardiovascular adverse events
Cardiovascular adverse events were reported in 10 of 20 cases. Nine were mild and included two cases of high blood pressure, two of low blood pressure, four of tachycardia and one of bradycardia. Only three of these patients had their infusion rate reduced or stopped for a period of time (2–15 h). One case had an episode of self-limited atrial fibrillation and the infusion was stopped.
Psychiatric adverse events
Psychiatric symptoms were present in 10/20 (50%) patients, and included dysphoria (n = 2), depressive mood (n = 2), depressive mood with paranoid ideation (n = 2) and agitation with visual and auditory hallucinations (n = 5). One patient with SUNCT with marked paranoid ideation had such an excellent clinical response that she requested the infusion be repeated 1 year later. She had a further almost identical episode of paranoid thinking and behaviour, which was foreshortened as we had suspected it might occur and the infusion was quickly stopped. All but two of these patients had their infusion stopped on account of psychiatric symptoms, whereas the others tolerated the effects and continued for the therapeutic trial.
The occurrence of psychiatric adverse events was not related to any of the variables in the model. The Hosmer and Lemeshow test was not significant (χ2 8 = 9.51, P = 0.33). Wald tests for each of sex (P = 0.36), age (P = 0.36), type of headache (P = 0.18), length of treatment (P = 0.9), previous history of depression or psychiatric illness (P = 0.64) and occurrence of other type of side-effect (P = 0.31) were negative.
Neurological adverse events
Neurological symptoms were frequent (10/20, 50%) and included dizziness, light headiness or drowsiness (n = 5), perioral or limb paraesthesia (n = 4), visual disturbances, blurred vision and transitory double vision (n = 2), confusion, disorientation and cognitive difficulties (n = 1) and frequent visual and sensitive auras (n = 1 each). One patient had the infusion stopped for neurological symptoms, two had permanent reduction of the infusion rates and one had transient infusion suspension for 8 h for blurred vision.
Taken together, neuropsychiatric symptoms occurred in 15 of 20 (75%) patients that we treated.
Gastrointestinal adverse events
The principal gastrointestinal adverse event registered was nausea (n = 6), and two patients complained of constipation. Two patients had transient infusion suspension for nausea.
Other adverse events
Other symptoms reported included feeling hot in the body (n = 3) and tiredness (n = 1), and another patient had a minor local infusion site reaction due to extravasation and subcutaneous infiltration of lidocaine. Two patients who complained of feeling hot had the infusion suspended for 30 min and then re-started at the same rate, uneventfully.
Review of literature
Results from the literature review on unwanted effects of i.v. lidocaine treatment in adults are presented in Table 2, including only case series (9, 10, 14–63). It includes 50 series and around 6000 patients treated for different reasons: surgical patients (for anaesthesia), pain and headache patients (for analgesia), cardiac patients (for ventricular arrhythmias), epileptic patients (for seizure control) and also healthy volunteers in experimental conditions. The side-effects rate varies in the different series, ranging from 0 (27, 44, 45, 49, 50, 60) to 100% (16, 18, 21). Major or life-threatening side-effects are infrequent, most series reporting rates between 0.3% and 33.3% for seizures, coma, severe heart block and cardiovascular arrest (16, 28, 37). Psychiatric side-effects are reported in 1.8–100% (17, 27).
Case series involving i.v. lidocaine treatment

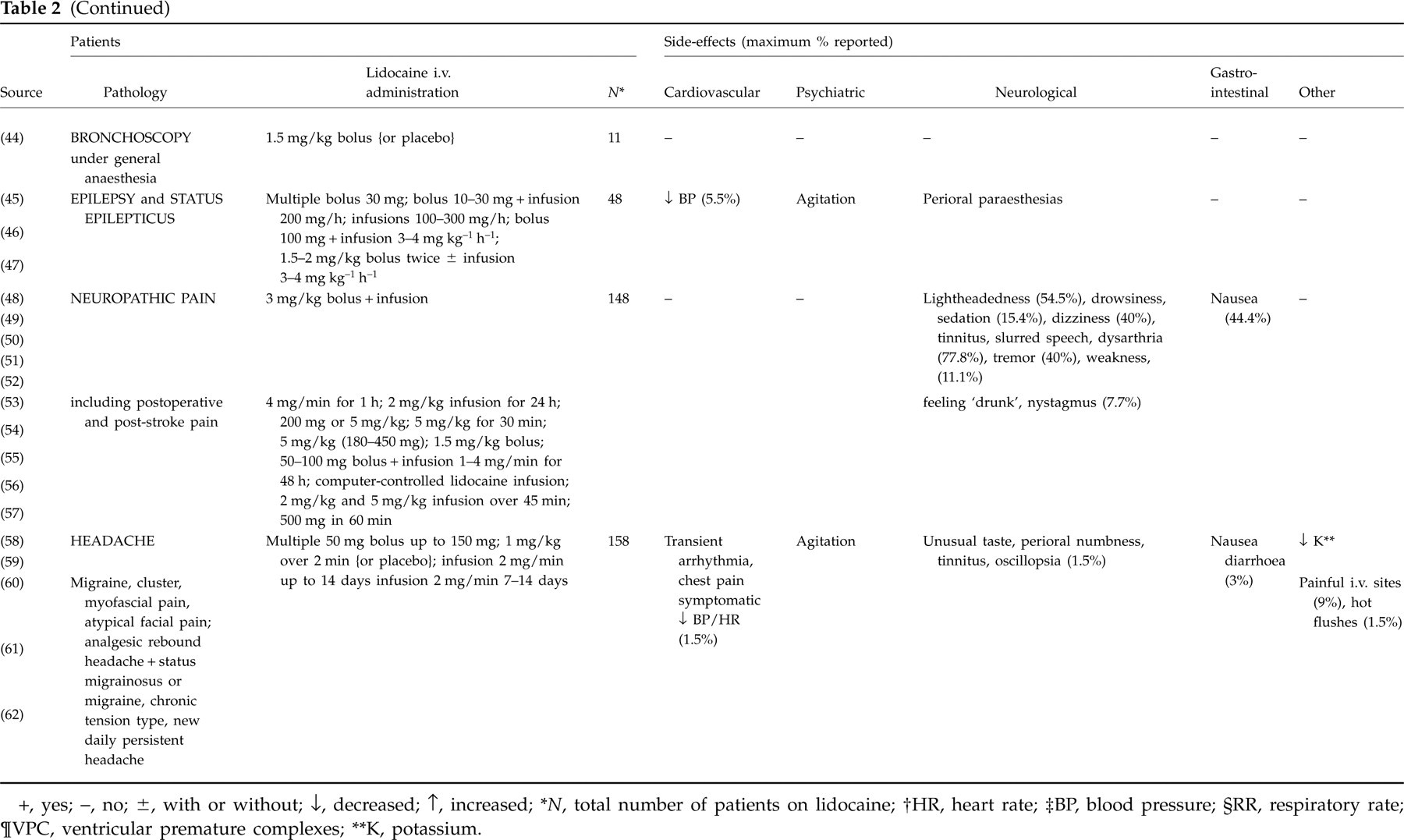
+, yes; −, no; ±, with or without; ↓, decreased; ↑, increased;
∗ N, total number of patients on lidocaine;
†HR, heart rate;
‡BP, blood pressure;
§RR, respiratory rate;
¶VPC, ventricular premature complexes;
∗∗K, potassium.
Table 3 describes case reports on psychiatric effects of lidocaine, independently of route of administration (6, 8, 64–71, 73–78). It includes 17 reports involving 36 patients, mostly male (69%) adults with cardiac disorders (78%). Most of these toxicity reports involve i.v. administration of lidocaine (8, 65–69, 73–75), but some are due to subcutaneous (6, 64, 76–78) or oral (accidental) (71) administration. Twenty patients (56%) had concurrent conditions or administration of other drugs that could influence lidocaine toxicity. The psychiatric symptoms include excitement, euphoria and agitation, sometimes evolving to doom anxiety, delirium and frank psychosis with hallucinations.
Case reports involving neuropsychiatric side-effects with lidocaine treatment

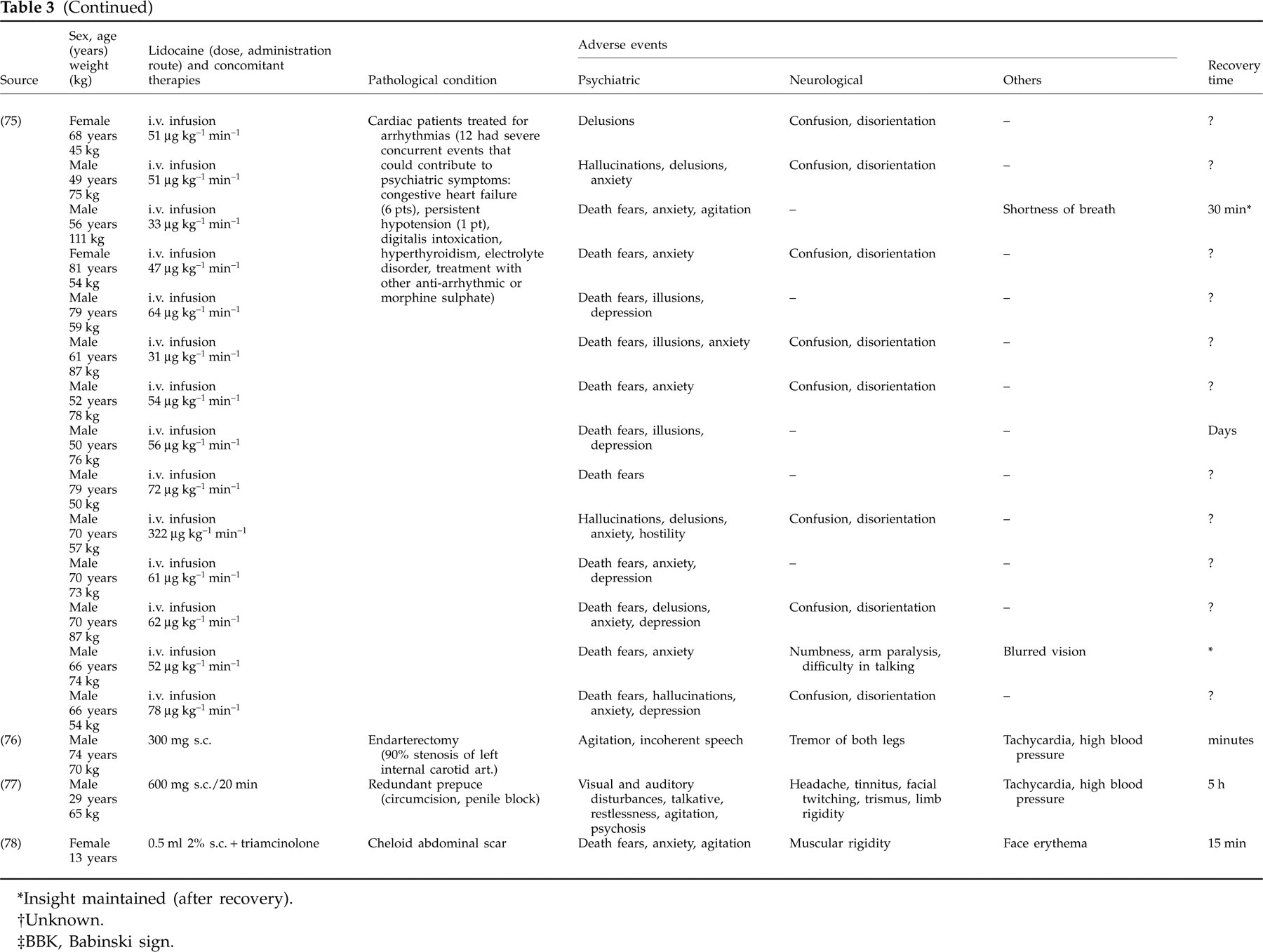
∗Insight maintained (after recovery).
†Unknown.
‡BBK, Babinski sign.
Discussion
Use of i.v. lidocaine to treat intractable headache produced psychiatric side-effects in 50% of our patients. Although recognized in the literature, the incidence seems high and not widely discussed in the neurological or headache community, in contrast to neurological side-effects that were as common and more widely recognized. Our series and the literature at large suggest that patients being considered for i.v. lidocaine should be warned that psychiatric side-effects are possible, and may be disturbing for both patient and carers.
Neurological side-effects with lidocaine have been recognized for many years, with the initial description of generalized tonic-clonic seizures in two surgical patients in a series of 853, an incidence of 0.2% (79). Apart from nausea in two patients, there were no other major unwanted events (79). Several reports have been published over the years describing mechanisms that might induce systemic toxicity when using lidocaine for local, subcutaneous, topic or infiltrative, anaesthesia (6, 7, 64, 77, 78, 80). These might be due to increased systemic absorption of the drug, either by failure to add adrenaline to the local solution (6, 79), excessive dosage (79), highly vascularized injection site (77, 80) or accidental i.v. or oral administration (70, 80). Absorption through mucous membranes diminishes the hepatic metabolism of lidocaine by avoiding the first-pass effect (81). Other causes of decreased drug metabolism are hepatic insufficiency, congestive heart failure (1, 8, 10, 38, 39) or concomitant use of other drugs that might either involve the P450 cytochrome system, such as cimetidine, alter haemodynamic status, such as propranolol, or have the same profile of side-effects, such as procainamide (1, 10, 32, 67, 70, 73). More than half of reviewed case reports with psychiatric manifestations had concurrent disorders or medications that could theoretically facilitate toxicity. Interestingly, in our headache series 13 patients had concomitant prophylactic therapy with antiepileptics (n = 11), β-blockers (n = 2) and tricyclic antidepressants (n = 4). However, none of these therapies influenced the occurrence of neuropsychiatric adverse events in our series.
The purpose of our review was to document the effects of the use of lidocaine not as a local anaesthetic, but as a systemic, i.v. treatment. Generally, toxic reactions to either local (subcutaneous) or i.v. administration probably differ only in the time-frame they occur (80), although there is evidence supporting the existence of local direct neurotoxicity of intrathecal administration of lidocaine for spinal anaesthesia (82, 83).
The case series reviewed are quite heterogeneous in patient selection, pathologies involved and mode of lidocaine administration, and discrepancies in side-effect reports would therefore be expected. Another fact contributing to this variability was each author's notion of relevance of side-effects. For example, mild sedation, lightheadedness, dizziness, tinnitus, paraesthesias and twitching, nausea or minor heart rate or blood pressure changes are often viewed as normal effects of lidocaine and are not considered unwanted or secondary (16, 27, 35, 37, 43, 46). Patients themselves often do not report these symptoms spontaneously, even when involved in clinical trails (37, 38, 50). Our review of the literature suggests that most authors actively seek out neurological and cardiovascular effects, whereas psychiatric are often not mentioned (15, 28, 38, 41, 43, 46, 50).
Central nervous system toxicity is 0.3–100% in the published series (14, 16, 17, 21). The clinical pattern of this type of symptomatology seems to be stereotyped and progressive, but sometimes psychotic reactions are described either with minimal or without neurological manifestations (67, 68, 70–73). This is also the case in our series, in which four patients (20%) had marked psychiatric symptoms that precluded further utilization of lidocaine but had no neurological symptoms. In our series the occurrence of neuropsychiatric adverse events was 75%, which is high when compared with the low occurrence, 1.5% (59) to 29% (62), or total absence (60) of this type of side-effect in other headache series. Data from series with cardiac or other pathologies are unhelpful in this regard because of the encompassing range of 0.3–100% of patients with the effect. Of note, SUNCT/SUNA patients seem to have a higher rate of psychotic side-effects than other headache patients (64% vs. 33%), although the numbers are too small to draw any definite conclusion. This difference could not be accounted for in the previous history of depression or psychiatric illness, absence or presence of beneficial therapeutic effect on headache, presence or absence of other side-effects, infusion doses or duration, or age. It is a clinical point to bear in mind when considering SUNCT/SUNA patients for such a treatment.
The toxic i.v. dose of lidocaine has been described as > 4–8 mg/kg of body weight (6, 64). Two studies (10, 39) determined signs of toxicity when blood levels were > 5.9 μg/ml and 5.0 μg/ml, respectively, but only studied the blood levels of toxic patients. Other authors have reported on the importance of rate of infusion in determining toxicity (19, 28). Drayer and colleagues (32) evaluated serum levels of lidocaine and its metabolites monoethylglycinexylidide (MEGX) and glycinexylidide (GX). They reported six of 27 patients without toxicity symptoms with lidocaine levels > 8.0 μg/ml, whereas five of six patients with toxic symptoms had levels < 8.0 μg/ml. The difference observed between toxic and non-toxic patients in this study was of higher MEGX levels in toxic patients. Korbon et al. (20) have suggested that there might be an individual susceptibility for lidocaine toxicity, demonstrating a significant decrease in experimental pain and tolerance in toxic vs. asymptomatic individuals. It is not then clear whether side-effects can been attributed to lidocaine itself or to its metabolites, MEGX and GX (1, 32).
Animal electroencephalographic studies (84) have demonstrated rhythmic amygdaloid electric activity after the administration of sub-convulsant doses of lidocaine, sometimes spreading to the fronto-orbital cortex, nucleus medialis dorsalis, mesencephalic reticular formation, globus pallidus, putamen and hippocampus. These discharges are accompanied by vocalizations, respiratory changes and a decrease in motor activity and in the response to external stimuli. Cortical slow high-voltage activity would follow and outlast the amygdaloid changes and could be related to staring and stupor. Convulsant doses would produce focal discharge from the amygdala and seizures, sometimes preceded by behavioural alterations (84). These observations lead to the hypothesis that the neuropsychiatric effects of lidocaine in humans were related to limbic hyperactivity with manifestations similar to temporolimbic epilepsy, such as hallucinations, emotional and behavioural changes (85). This hyperactivity could be related to the development of lidocaine-induced pharmacological kindling of the limbic system (86). A similar syndrome occurs as an acute non-allergic reaction to procaine penicillin, the Hoigné's syndrome (86).
In conclusion, we have reported a series of patients treated with i.v. lidocaine for primary headache indications who developed neuropsychiatric symptoms. Half of the cohort had psychiatric symptoms, some of which were highly disturbing, and in one case a re-exposure established causality. Our review of literature indicates that such problems have been recognized, although not in the headache and pain literature. Because these symptoms do seem relatively common, and may be disturbing, patients and physicians should be aware that they might occur prior to commencing treatment with i.v. lidocaine. We recommend routinely counselling patients and relatives of these symptoms prior to treatment.
