Abstract
This study examined the effect of age on placebo response rates in rizatriptan trials in adults. Data from eight rizatriptan adult trials involving patients treating moderate/severe migraine attacks with rizatriptan 5 mg (N = 1819), rizatriptan 10 mg (N = 2046) or placebo (N = 1322) were pooled for post hoc analysis. Logistic regression was used to model 2-h pain relief (reduction to mild or none) and 2-h pain freedom rates by treatment groups. Older patients had lower placebo response rates than younger patients; the estimated odds ratio (older vs. younger) for a 10-year age increase was 0.83 for pain relief [95% confidence interval (CI) 0.75, 0.93] and 0.81 for pain freedom (95% CI 0.68, 0.97). The response proportion vs. age trend was flat for rizatriptan 5 mg and slightly increased for rizatriptan 10 mg. The treatment-by-age interaction was significant for pain relief (P < 0.001) and pain freedom (P = 0.001), suggesting an increasing trend of treatment advantage of rizatriptan over placebo as age increased. Age appeared to be an important predictor of placebo response rate in rizatriptan trials, with older patients being less likely to respond to placebo and more likely to respond to rizatriptan.
Introduction
The efficacy of 5-HT1B/1D receptor agonists (triptans) for the acute treatment of migraine attacks in adult patients ≥ 18 years old is well established (1). However, the efficacy of triptans in patients < 18 years old is less clear. Many of the initial trials of triptans in adolescents failed to demonstrate an advantage over placebo (reviewed in (2, 3)). Various reasons have been proposed for this, including the relatively short duration of migraine attacks, delays in accessing treatment, study design issues, uncertainties about diagnosis, and pharmacokinetic or pharmacodynamic differences in adolescents/children compared with adults (4, 5).
A common observation in adolescent/paediatric migraine studies has been that whereas drug response rates tend to be similar to those seen in adults, placebo response rates tend to be higher, thereby leading to a lack of separation between drug and placebo in adolescents/children. For example, in two studies of rizatriptan 5 mg in adolescents, 2-h pain relief response rates of 56 and 69% were observed for placebo, vs. 66 and 68%, respectively, for rizatriptan 5 mg (6, 7). For comparison, in a meta-analysis of adult studies, a 2-h pain relief response rate of 38% was observed for placebo, with a rizatriptan 5 mg response rate of 64% (8).
Within the adolescent/paediatric group, an inverse relationship with age has been noted with rising placebo response rates with decreasing age (4). It is currently not clear whether a similar age-related effect on placebo response rates exists in adults. If younger adults showed a trend for a higher placebo response than older adults then it would suggest that adolescents/children lie on a continuum. Conversely, a finding of no effect of age on placebo response in adults might indicate a more fundamental divergence between adults and adolescents/children. To investigate this issue, we performed an analysis of placebo data from pooled Phase III adult clinical trials with rizatriptan. We also looked at whether the drug response might vary with age in these trials.
In addition to age in the pooled analysis, we also looked at two other variables with potential relevance for interpreting the lack of treatment differences observed in adolescent/paediatric studies—sex and prior triptan use. A striking observation in adolescent drug trials is the differing proportions of female and male subjects compared with adult clinical trials, which reflects differing real-world prevalence. Patients recruited in adult studies tend to be predominantly female (e.g. 80–90% female in the rizatriptan adult studies) (8), whereas patients recruited in adolescent studies tend to be more equally divided between male and female (e.g. approximately 55% female and 45% male in the rizatriptan adolescent studies) (6, 7). The current literature does not suggest a sex difference in triptan treatment response in adults. However, it is possible that small differences may have been overlooked in analyses based on individual studies. We therefore examined the effects of sex on placebo and drug response in the pooled analysis of adult data. If men showed a higher placebo response or lower drug response than women, then that could be a contributory factor in the lack of separation between drug and placebo in adolescents/children.
The other variable that we considered in the pooled analysis was the effect of prior triptan use on response. This has been suggested to influence response rates, with triptan-naive patients showing a higher placebo response rate than triptan-experienced patients (9). It might be expected that adolescent/paediatric patients would be less likely than adults to be prior triptan users given the lack of approved labelling for this population. If prior triptan use was associated with a lower placebo response or higher drug response than no prior use, then that could also influence the lack of separation between drug and placebo in adolescents/children.
Methods
Studies included in the analysis
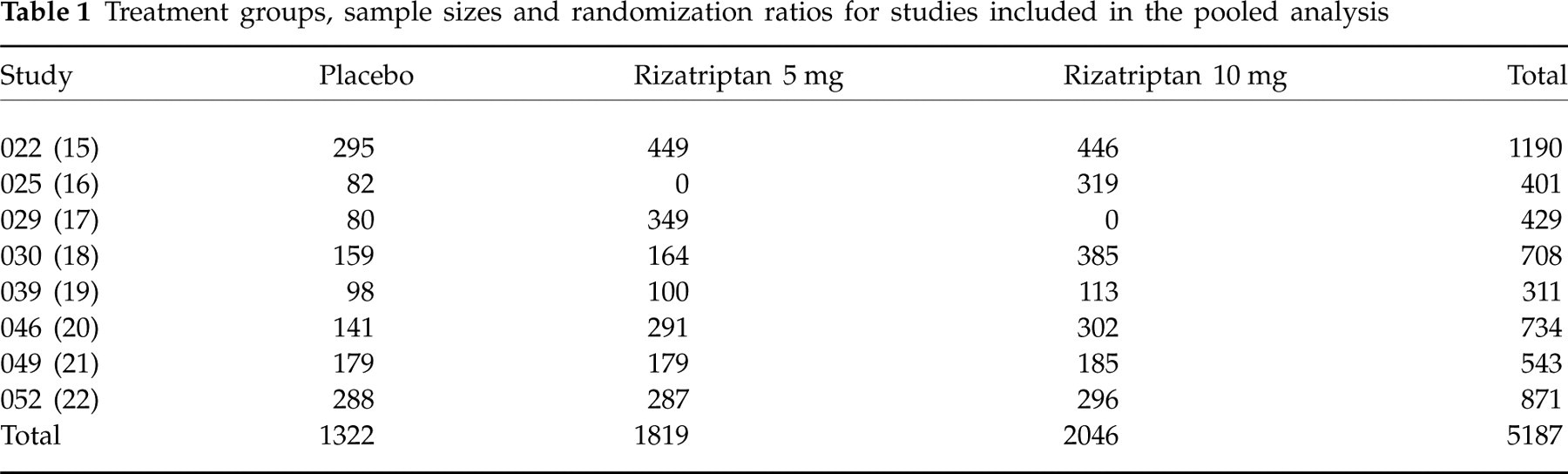
All Phase III randomized, placebo-controlled studies on rizatriptan 10 mg (standard recommended starting dose in most patients) or 5 mg (lower dose) for the treatment of a moderate or severe migraine attack in adults conducted by Merck & Co. Inc. from 1995 to 1998 were included in the analysis (see Table 1).
Treatment groups, sample sizes and randomization ratios for studies included in the pooled analysis
Study design
All studies employed randomized, double-blind, placebo-controlled designs. Five of the studies employed single-attack, parallel-group designs, and three used crossover or multiple attack designs; the present analysis looked at data from the first attack only for each study. All studies were conducted according to good clinical practice guidelines and all were approved by the appropriate Institutional Review Boards.
Patients
Patients were male or female out-patients who had at least a 6-month history of migraine according to the criteria of the International Headache Society (10), were at least 18 years old, and typically experienced one to eight migraine attacks per month. Patients who had previously used a triptan other than rizatriptan could be included in the study. Triptan use within the previous 30 days prior to screening and during the study was recorded. All patients gave written informed consent.
Procedure
Patients were instructed to take test medication when they developed a moderate or severe migraine headache. The patients rated headache severity on a diary card using a four-grade scale (no, mild, moderate, severe headache) at predose and 30-min intervals up to 2 h. Analgesic and antiemetic rescue medications (other than ergotamine or triptans) were allowed from 2 h after dosing.
Statistical analysis
The analysis was performed on a post hoc basis. The end-points analysed were: (i) 2-h pain relief, defined as the percentage of patients with a reduction of headache severity from moderate or severe at baseline to mild or none at 2 h; and (ii) 2-h pain freedom, defined as the percentage of patients with a reduction of headache severity from moderate or severe at baseline to none at 2 h. These were primary or secondary end-points in each of the studies. The statistical analysis was based on attack 1 data only, and therefore the data can be regarded as originating from parallel group studies. The analysis included all patients who took study treatment and provided a baseline efficacy evaluation and data for at least one time point within 2 h after dosing. A last observation carried forward approach was used for missing data, with the proviso that no imputations were made using baseline data. Attacks that were mild or had missing baseline severity data were excluded.
The age data were analysed using logistic regression models for response rates by treatment group, with baseline headache severity (moderate or severe) and age (continuous variable) as covariates. Baseline headache severity was included as a covariate because it is a predictor of response, in that moderate headaches are associated with higher response rates than severe headaches. The homogeneity of the studies (‘poolability’) was tested by adding terms for study and study-by-age interaction in the logistic models. An age covariate, with a unit of decade, was included in the models to estimate the odds ratios (ORs) and corresponding 95% confidence intervals (CIs) of the response rates for an average of a 10-year age increase. Age-by-treatment interactions were also assessed separately by adding treatment group and interaction in the models. Response curves were estimated using a local regression smoothing model (Loess) with age as the predictor.
Sex and prior triptan use data were analysed as categorical variables. The analyses were limited to the 10-mg dose and placebo. The data were analysed with logistic regression models using treatment group, baseline headache severity, sex (or prior triptan use), and treatment-by-sex (or prior triptan use) interaction. The percentage of patients in each treatment group who achieved each end-point was calculated, along with 95% CIs. In addition, the therapeutic gains (difference between active treatment and placebo) along with the associated standard error were calculated.
Results
Patient characteristics and sample sizes
Sample sizes for each study are summarized in Table 1. A total of 5187 patients were included in the analysis: 2046 in the rizatriptan 10 mg group, 1819 in the rizatriptan 5 mg group, and 1322 in the placebo group. Patient characteristics are summarized in Table 2. Most patients were women (86%) and the mean age was 41 years. The distribution of characteristics was similar across treatment groups.
Patient characteristics in the pooled studies
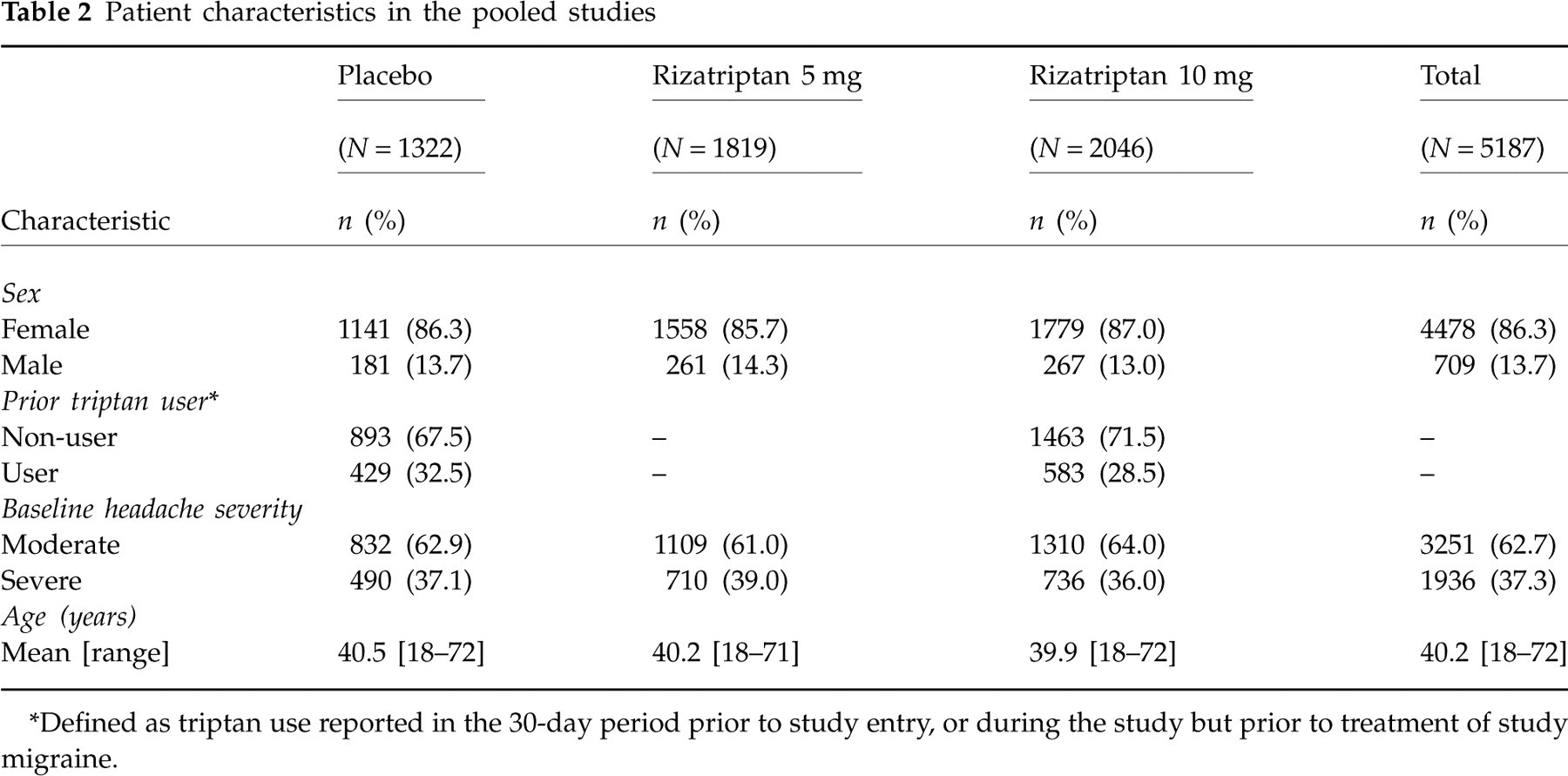
∗Defined as triptan use reported in the 30-day period prior to study entry, or during the study but prior to treatment of study migraine.
Age
There were no significant study or study-by-age interactions for any treatments for either pain relief or pain-free end-points (P-values > 0.10), indicating that the studies were homogeneous with respect to the treatment effect and had a consistent pattern of age effects. The final models did not include the terms for the study covariate or the study-by-age interaction.
Figure 1 shows the estimated pain relief and pain freedom response rates in each treatment group by age. In the placebo group, older patients had lower pain relief and pain freedom response rates than younger patients; the estimated OR for a 10-year age increase (older vs. younger) was 0.83 for pain relief (95% CI 0.75, 0.93) and 0.81 for pain freedom (95% CI 0.68, 0.97). Similar analyses showed that the response proportion vs. age trend was flat for rizatriptan 5 mg dose (estimated OR for a 10-year age increase: pain relief 0.94, 95% CI 0.85, 1.04; pain freedom 1.05, 95% CI 0.95, 1.17), and slightly increasing for the rizatriptan 10 mg group (estimated OR for a 10-year age increase: pain relief 1.14, 95% CI 1.03, 1.25; pain freedom 1.16, 95% CI 1.07, 1.27). In the combined population of 5187 patients, the treatment-by-age interaction was significant (P < 0.001 for pain relief, P = 0.001 for pain freedom), suggesting an increasing treatment advantage of rizatriptan over placebo as age increases. Visual inspection of the data in Fig. 1 suggested that the rizatriptan vs. placebo treatment differences were particularly affected by age up to 50 years and that the treatment difference were not as affected by age in those > 50 years old. This observation was supported by an analysis looking at patients < 50 years (N = 4235) and ≥ 50 years (N = 952) separately. The treatment-by-age interactions were significant for pain relief (P < 0.001) and pain freedom (P = 0.016) in the < 50 years group, suggesting that treatment differences were affected by age in the younger group. In the ≥ 50 years group, no significant treatment-by-age interactions were observed (P-values > 0.9 for both pain relief and pain freedom), suggesting that the treatment differences were not as affected by age in the more elderly group.

Estimated pain relief and pain-free rates at 2 h by age with 95% pointwise confidence intervals in pooled data from eight Phase III studies. Response curves were estimated using a local regression smoothing model (Loess) with age as the predictor. Sample sizes were 1322 for placebo, 1819 for rizatriptan 5 mg, and 2046 for rizatriptan 10 mg.
Sex
The percentage of patients with pain relief and pain freedom by sex for rizatriptan 10 mg and placebo, as well as therapeutic gains (rizatriptan 10 mg minus placebo), are shown in Table 3. There were no significant treatment-by-sex interactions for pain relief (P = 0.604) or pain freedom (P = 0.659).
Percentage (95% confidence interval) of patients with pain relief and pain freedom at 2 h, and percentage therapeutic gain (standard error), by sex and prior triptan use in the pooled studies

∗Rizatriptan 10 mg minus placebo.
†Sample sizes were: female, placebo 1141, rizatriptan 1779; male, placebo 181, rizatriptan 267.
‡Sample sizes were: non-user, placebo 893, rizatriptan 1463; user, placebo 429, rizatriptan 583.
Prior triptan use
The percentage of patients with pain freedom and pain relief by prior triptan use for rizatriptan 10 mg and placebo, as well as therapeutic gains (rizatriptan 10 mg minus placebo), are shown in Table 3. There were significant treatment-by-prior triptan use interactions for pain relief (P = 0.003) and pain freedom (P = 0.024). As shown in Table 3, therapeutic gains were greater in patients with prior triptan use; this reflects both a lower placebo response rate and a higher active treatment response rate in these patients.
Patients with prior triptan use tended to be slightly older than those without prior triptan use [mean (
Discussion
The results of this pooled analysis of > 5000 adult patients with migraine suggest that age was an important predictor of placebo response, with younger adult patients being more likely to respond to placebo. This finding suggests that the high placebo response rate observed in adolescents and children represents one end of a continuum rather than there being a fundamental difference between the two populations. An effect of age on drug response was also observed, with treatment efficacy tending to increase with age for the 10-mg dose of rizatriptan. However, for the lower rizatriptan 5-mg dose, which was the dose investigated in the adolescent studies, there was no clear evidence of such an effect. Consequently, the relevance of a possible age-related difference in drug response in adolescents is less clear; as noted in the Introduction, response rates for rizatriptan 5 mg in adolescents tended to be similar to those seen in adults.
The adolescent migraine population differs from the adult migraine population in being comprised of approximately equal proportions of males and females, whereas in adults, women are much more common than men. The present analysis did not suggest that the patient's sex influenced placebo or drug response, so it seems unlikely that sex differences per se would be a confounding factor in interpreting the effect of age on placebo response. However, migraine has a hormonal component, and it is possible that oral contraceptive or hormone therapy use in younger or older women could be a confounding variable in interpreting age effects.
We found that use of triptans by subjects within 30 days of a study was associated with lower placebo response rates and higher drug response rates compared with non-use. This may be due to selection bias, since such prior triptan users may be experienced with the triptan side-effects and consequently better able to discriminate between drug and placebo, and/or the likelihood that active triptan users are triptan responders. Since adolescents in the rizatriptan studies were unlikely to have been prior triptan users (due to the lack of Food and Drug Administration approval for use in this age group), this may have had an influence on the results. Indeed, approximately 15% of patients in the prior rizatriptan adolescent studies were previous triptan users, vs. approximately 30% in the present adult analysis. In the present analyses, the effects of age and prior triptan use were somewhat confounded in that prior triptan users tended to be slightly older than non-triptan users, as might be expected since older patients would presumably have had a greater window of opportunity to use a triptan. However, the finding that treatment-by-age and treatment-by-prior triptan use interactions remained significant when age and prior triptan use were assessed in the same statistical model suggests that the effects of age are not entirely attributable to prior triptan use or vice versa. This does not exclude the possibility that there may be some common element contributing to the effects of age and prior triptan use on placebo or drug response.
The present analysis has a number of limitations typical of post hoc analyses. First, there were imbalances with regard to the number of patients in each group for each of the analyses (i.e. fewer patients > 50 vs. < 50 years old, fewer men, fewer patients with prior triptan use). In the instances where significant differences were observed (age, prior triptan use) this may be less of a concern, but the lack of sex difference should be interpreted with caution. Another caveat is that the reporting window for prior triptan use extended back only 30 days prior to study screening. It is not known whether patients took triptans before this. Finally, it is possible that our findings were influenced by other covariates that we did not consider, e.g. body weight, duration of disease history or duration of migraine attack. Migraine duration tends to be shorter in adolescents and children compared with the overall adult population, but within the adult population it is unknown whether there is an age-related increase in duration. Since the primary end-point of most migraine trials is efficacy at 2 h, spontaneous improvement of migraine pain in younger patients may give an impression of increased placebo effect.
For all these reasons, the present data should be regarded as suggestive rather than conclusive. If the finding that increasing age is associated with reduced placebo response is valid, then it may have some practical implications. For example, in performing a proof-of-concept trial for a novel compound, it might be possible to increase the signal-to-noise ratio by including only patients older than some age cut-off of, for example, 30 or 40 years. Also, future studies might consider stratification by age and prior triptan use.
Ideally, it would be desirable to understand more fully the reasons for the effects of age and prior triptan use on placebo and drug response. For example, it might be speculated that a common factor contributing to the effects of both age and prior triptan use is patients' awareness or experience/expectations about how their migraines normally progress and the nature of the effects of treatment on the symptoms. Placebo rates can be influenced strongly by subjects' expectations and their understanding of the trial (11). Therefore, efforts to increase patient experience or education about their migraine may be beneficial in improving separation between drug and placebo. Children, in particular, may need to be educated that it is acceptable to report that a treatment did not work (4).
To some extent, this speculation may be supported by a recent child/adolescent study that successfully demonstrated a treatment advantage over placebo for rizatriptan (12). The study included only adolescents who had previously had an unsatisfactory response to non-steroidal anti-inflammatory drugs, and who experienced two or more migraine attacks per month lasting ≥ 4 h. Thus, patients were experienced both in having migraine attacks and in understanding that a drug may not be effective. (Factors such as weight-adjusted dosing and controlling the duration of attack may have also contributed to the success of this study.) Other recent adolescent/paediatric studies which have had success in separating drug efficacy from placebo have used placebo-challenge or crossover designs, which may also have had the effect of improving patient awareness or expectations of differences between drug and placebo (13, 14). Whether there is a difference in expectations about treatment effects in clinical trials between younger and older patients that contributed to our observation requires further exploration.
Competing interests
This study was funded by Merck Research Laboratories. P.W. and R.E.S. have received research funding from Merck. T.W.H., X.F., C.R.L. and A.R. are employees of Merck and have an equity interest in Merck. Merck employees were involved in the design and conduct of the analysis, interpretation of the data; and preparation, review and approval of the manuscript. The authors are responsible for the work described in this paper. All authors were involved in at least one of the following: conception, design, acquisition, analysis, statistical analysis, interpretation of data, drafting the manuscript and/or revising the manuscript for important intellectual content. All authors provided final approval of the version to be published.
Footnotes
Acknowledgements
This analysis was funded by Merck Research Laboratories. These data were previously presented at the 49th annual meeting of the American Headache Society, Chicago, IL, USA, 7–11 June 2007.
