Abstract
We report on a female patient who developed post-dural puncture headache (PDPH) after epidural analgesia for delivery. Treatment with epidural blood patch led to complete headache remission and the patient was discharged. Two days later the patient was readmitted with hemihypaesthesia and mild hemiparesis of the right side. Magnetic resonance imaging showed a small left parietal cortical haemorrhage probably following cerebral venous thrombosis (CVT). Coagulation screening detected heterozygous Factor V mutation. Headache is a common symptom of PDPH and CVT. Review of the literature revealed five patients in puerperal state, who developed CVT in close temporal relationship after blood patch treatment for PDPH. Change of headache character with loss of postural influence was reported frequently before diagnosis of CVT was confirmed. These findings may indicate a causal relationship.
Case report
A 30-year-old female was admitted to the hospital with headache and neck pain which had developed 2 h after delivery with uneventful epidural analgesia 6 days before. Oral medication with paracetamol was not effective and the patient reported a postural fronto-occipital headache (severity of 8 out of 10) with relief by lying down. She also complained of tinnitus as an accompanying symptom. There was no history of a primary headache. Furthermore, she never had deep vein thrombosis, pulmonary embolism or spontaneous abortion. She did not smoke. Neurological examination showed mild meningeal signs but no focal deficit. She was afebrile and all vital signs were normal. Magnetic resonance imaging (MRI) with venous time of flight angiography (MRV) and gadolinium contrast was performed to exclude cerebral venous thrombosis (CVT). Pachymeningeal enhancement could be visualized and a diagnosis of post-dural puncture headache (PDPH) was made. Autologous epidural blood patch (EBP) (20 ml) led to complete resolution of headache within 4 h. The opening pressure was not obtained when EPB was performed. The patient was subsequently discharged.
Two days later the patient was admitted again complaining about right hemihypaesthesia and mild hemiparesis. MRI was requested. The following MRI sequences were performed: fluid attenuated inversion recovery, diffusion weight imaging, T2∗ and MRV. A small left parietal cortical haemorrhage was detected, but no direct sign of CVT (Fig. 1). Heterozygous Factor V Leiden mutation could be diagnosed in coagulation screen. Protein C and S, antithrombin III, lupus anticoagulant and antiphospholipid antibodies were within normal limits. Anticoagulation therapy with enoxaparin (40 mg/day) was started and the patient was discharged 4 days later after all neurological symptoms had disappeared.
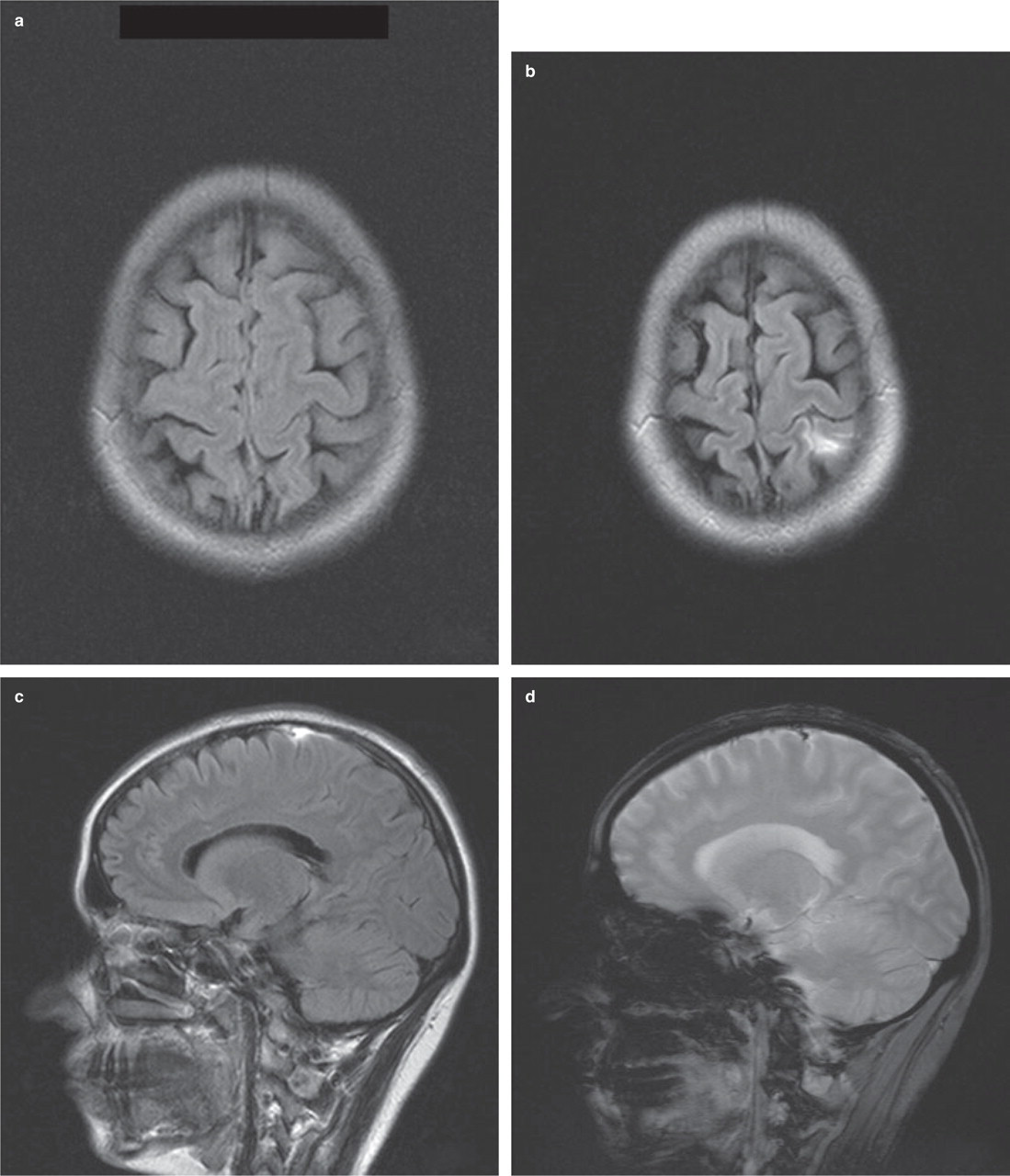
(a) Inconspicuous initial fluid attenuated inversion recovery (FLAIR) imaging. (b,c) FLAIR imaging shows a small left parietal cortical haemorrhage. (d) T2∗ shows equivalent result suggestive of a bleeding following cortical vein thrombosis.
Discussion
Headache following delivery is a common problem. Thirty-nine per cent of women develop headache within 1 week postpartum (1). Migraine tends to relapse after childbirth (2), but in our patient there was no history of previous migraine. In some patients a first migraine attack occurs in the postpartum period (3). The incidence of accidental dural puncture in obstetric patients receiving epidural analgesia ranges between 2% and 6% (4). In 70–85% of these patients headache develops (5, 6).
According to the International Headache Society (IHS), PDPH worsens on sitting or standing and improves after lying (7). Accompanying symptoms are neck stiffness, tinnitus, hypacusia, photophobia and nausea, of which at least one must be present for diagnosis (see Table 1). Thus PDPH was consistent with IHS diagnostic criteria in our patient at this point. Nevertheless, MRI was performed to exclude CVT, as the puerperal state is an important risk factor for CVT, which may present with headache without accompanying neurological deficit. This MRI showed pachymeningeal enhancement, which is frequently found in patients with PDPH (8). Further signs of PDPH such as descending tonsils or subdural fluid collection were not present. Subsequently EBP was performed with excellent response.
Diagnostic criteria for postdural puncture headache according to International Headache Society (ICHD-II) classification

The cortical haemorrhage found in MRI was suggestive of a bleeding following CVT (Fig. 1). MRV could not detect direct signs of thrombosis. However, MRV has a rather low sensitivity (37%) in detecting isolated cortical venous thrombosis (9). T2∗ susceptibility-weighted images have recently been shown to have the highest sensitivity (97%) in imaging cortical vein thrombosis (9). Altogether, MRI could not visualize cortical vein thrombosis. Given the patient's risk factors and the configuration of the small haemorrhage, CVT with spontaneous recanalization is in our opinion the most likely underlying explanation. Nevertheless, other pathologies (e.g. venous angioma, tumour bleeding) have to be taken into account. However, no such pathology could be found either in the first MRI scan (performed with gadolinium contrast) or in the second scan.
Our patient had several risk factors for CVT. First, puerperal state is a well-established risk factor (10). Lanska and Kryscio have estimated a risk of 11.6 cases of intracranial venous thrombosis per 100 000 deliveries (11). Second, heterozygous Factor V Leiden mutation, as confirmed in our patient, has been shown to increase the risk of CVT in a meta-analysis of 17 studies (12) with an odds ratio of 3.38 (95% confidence interval 2.27, 5.05; P < 0.001). Third, there have been several reports of CVT following dural puncture. In a retrospective study of 66 patients with CVT, Wilder-Smith et al. (13) found preceding dural puncture the fourth most frequent risk factor, which was present in 8% of cases. The authors also described five case reports of CVT following dural puncture. It was hypothesized that traumatic injury of the fragile venous endothelial lining due to stretching of the cerebral vessels by a negative spinal-cranial pressure gradient and stasis of blood flow via venous vasodilation contributes to thrombosis formation after dural puncture. This hypothesis is also supported by a study showing that dural puncture led to a 47% decrease of mean blood flow velocity in the straight sinus measured by transcranial Doppler ultrasound (14).
Review of the literature has revealed five cases (see Table 2) in whom diagnosis of CVT was made after EBP was performed for treatment of PDPH following epidural analgesia for delivery. The IHS criteria for PDPH (see Table 1) were positive for three of five patients (B, C, D). In one patient with presumed PDPH there was no postural component mentioned (A) and in one patient (E) there was neither a clear history of dural puncture in epidural analgesia nor accompanying signs (nausea, photophobia, neck stiffness, hypacusia, tinnitus). Blood patch treatment led to significant improvement of headache in those cases (B, C, D) who met the IHS criteria for PDPH, but headache relapsed within 2–48 h. Three patients (C, D, E) reported a change of headache characteristic with less or no further postural component after EPB. Blood patch was performed twice in patients B and D as well as three times in patient C. Due to headache persistence (C, E) and/or occurrence of neurological symptoms (A, B, D), cerebral imaging was performed (cerebral arteriogram in patient A; MRI in patients B, C, D, E), which confirmed cerebral sinus thrombosis (A, B, C, E) or cortical vein thrombosis (D). In our patient blood patch led to complete and constant relief of headache, but she developed neurological symptoms 2 days after treatment.
Overview of patients with development of cerebral venous thrombosis (CVT) after epidural blood patch treatment for post-dural puncture headache

Headache is the most frequent symptom (80–90%) of CVT and may mimic PDPH. It is therefore questionable whether headache in these cases, in spite of being consistent with IHS diagnostic criteria majoritarian, can be attributed to PDPH, as it may have been a feature of CVT or a mix of both conditions. The response to blood patch treatment (B, C, D and our patient) points to the initial presence of PDPH followed by CVT, which caused a different, less postural, headache in patients C and D. In patients A and E, blood patch was not at all effective, and IHS criteria for post lumbar puncture were not met. This makes CVT as the initial cause of headache more likely. Interestingly, patient E also reported a change of headache character after EBP with loss of postural component.
A pathophysiological link between blood patch and CVT is difficult to establish. Blood injected in the epidural space is hypothesized to clot and occlude cerebrospinal fluid leakage. MRI studies have shown a spread of blood over 3.5 intervertebral spaces above and one intervertebral space below the level of injection after performance of a 20-ml blood patch (15). Animal studies have demonstrated that 7 days after administration there is widespread fibroblastic activity and collagen formation (16, 17). However, none of these findings can explain the development of CVT.
Whether EBP is a risk factor for CVT is not clear. Yet the close temporal relationship between the two in some cases may suggest a causal relationship. However, all patients were in a puerperal state and thus at risk of developing CVT. Screening for CVT of high-risk patients after successful EBP is not indicated, but headache persistence or relapse with different characteristics after EBP treatment in puerperal patients indicates further diagnostic work-up to exclude CVT.
