Abstract
Late-life onset aura-like symptoms are not rare and can be potentially misdiagnosed as transient ischaemic attacks. The cause is often obscure. Four cases of recurrent aura-like symptoms are presented in whom acute convexity subarachnoid haemorrhage (SAH) contralateral to symptoms was demonstrated. Three experienced subsequent events or groups of events triggered by recurrent SAH. Negative diffusion-weighted imaging, normal electroencephalography and slow symptom march with complete resolution argued against ischaemic and epileptic causes. Aura-like symptoms in the elderly should be investigated with imaging modalities most sensitive for detecting subarachnoid blood, in particular gradient echo magnetic resonance imaging.
Keywords
Introduction
Migraine aura misinterpretation can lead to misdiagnosis of a transient ischaemic attack (TIA) (1, 2). In particular, recurrent brief episodes of sensory disturbance are commonly diagnosed as TIAs but do not presage ischaemic stroke (2).
Late-life onset of migraine-type aura is not rare (3) and can occur without headache or visual symptoms (4, 5). Correct interpretation allows appropriate investigation and treatment. We present four patients whose recurrent migraine aura-like symptoms were triggered by acute convexity subarachnoid haemorrhage (SAH).
Case 1
An 81-year-old hypertensive man, on warfarin for atrial fibrillation, was admitted after four episodes of left leg weakness, progressing proximally over several minutes, with or without left-sided facial dysaesthesia. Episodes lasted between 20 and 120 min. There was no associated headache. His International Normalized Ratio (INR) was 5.9. Magnetic resonance imaging (MRI) demonstrated acute right central sulcus SAH, an older left parietal haemorrhage and white matter disease. Gradient echo (GE) sequences demonstrated convexity siderosis and cortical microbleeds. MR angiography and diffusion-weighted imaging (DWI) were normal. Electroencephalography (EEG) was normal. He was diagnosed with TIAs and his warfarin dose was lowered. Episodes continued and did not lessen with intensification of antithrombotic therapies.
Five years later he re-presented after awakening with a mild left-sided hemiparesis, which had resolved within 1 h. He had returned to sleep, but awoke with left facial numbness and weakness, resolving after 15 min. MRI demonstrated acute right central sulcus SAH, extensive cortical siderosis and cortical microbleeds (Fig. 1A,B). Aspirin was ceased. Over 3 days he then experienced 10 episodes of left-sided sensory disturbance, spreading over 5–15 min, with or without weakness. Valproate was commenced and titrated. Episodes decreased but continued at least daily. An empirical course of oral prednisolone was given hypothesizing a subarachnoid blood-induced inflammatory component. The episodes abruptly ceased. Valproate and prednisolone were weaned without recurrence.

(A) Fluid attenuation inversion recovery MRI demonstrates acute right central sulcus subarachnoid haemorrhage. (B) Gradient echo sequences demonstrate cortical microbleeds (horizontal arrows) and siderosis (vertical arrows).
Case 2
A 73-year-old hypertensive diabetic man was admitted with recurrent stereotyped neurological symptoms. He had previously been warfarinized for recurrent ‘TIAs’. His admission INR was 3.0.
Seven times over the previous 2 days he had experienced left hand paraesthesiae spreading to his neck over 5–20 min, accompanied by clumsiness but no headache. The first episode had resolved after 1 h, subsequent episodes within 30 min. MRI demonstrated acute right central sulcus SAH (Fig. 2A) and white matter disease. Microbleeds were evident on GE sequences (Fig. 2B). EEG and MRI DWI were normal. Valproate was prescribed and anticoagulation reversed. Five episodes then occurred over 3 days, including evolving weakness without sensory symptoms. Two episodes occurred the week following, but none 6 months subsequently.

(A) Fluid attenuation inversion recovery MRI demonstrates acute right precentral sulcus subarachnoid haemorrhage. (B) Gradient echo sequences demonstrate deep and cortical microbleeds.
Case 3
A 72-year-old hypertensive man on aspirin for ischaemic heart disease presented following several stereotypic neurological disturbances. Three months previously he had noted left hand finger tingling, spreading over 5 min to involve arm, face and tongue, associated with dysarthria, resolving within 10 min. There was no headache. An almost identical episode occurred a month later.
Immediately after a further episode of spreading hand tingling 2 months later, an MR head scan was performed, demonstrating acute right superior frontal sulcus SAH (Fig. 3A) and white matter disease. MRI DWI, an EEG and a computed tomography (CT) angiogram were normal. Sodium valproate was commenced and aspirin ceased. One month later an episode of left hand paraesthesia occurred. Repeat MRI with GE sequences demonstrated a new right sulcal SAH, extensive cortical siderosis and cortical microbleeds (Fig. 3B).
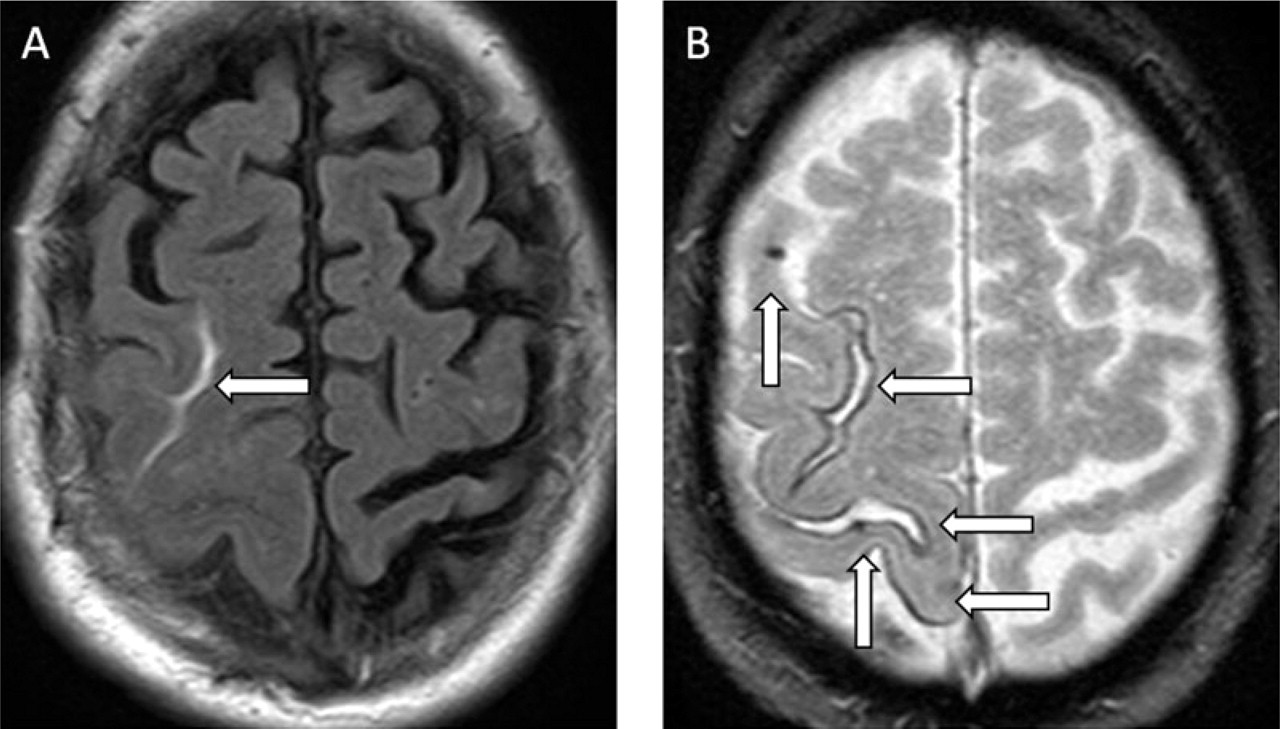
(A) Fluid attenuation inversion recovery MRI demonstrates acute right superior frontal sulcus subarachnoid haemorrhage. (B) Gradient echo sequences demonstrate cortical microbleeds (vertical arrows) and siderosis (horizontal arrows).
Case 4
An 82-year-old hypertensive man was admitted with left facial droop and arm numbness. Over the past 2 years he had experienced multiple episodes of left hand paraesthesia spreading to his shoulder over 5–10 min, with subsequent numbness resolving within 1 h. One episode was associated with leg clumsiness. Recurrent TIAs had been diagnosed and treated with escalating antiplatelet therapy.
Two days prior to admission he had experienced paraesthesia of his left face, spreading to his hand over 5 min, associated with dysarthria, drooling and left hand clumsiness, resolving in 2 h. An identical episode occurred 2 days later but lasted 6 h. MRI demonstrated acute right convexity SAH (Fig. 4A) and diffuse white matter disease, as well as two right frontal cortical haemorrhages of differing ages. GE imaging demonstrated cortical siderosis (Fig. 4B). DWI was normal. Ten days later the patient experienced six brief episodes in 2 days of left facial droop and dysarthria. Repeat CT scanning demonstrated new right central sulcus SAH. Antiplatelet therapy was ceased and symptoms have not recurred.
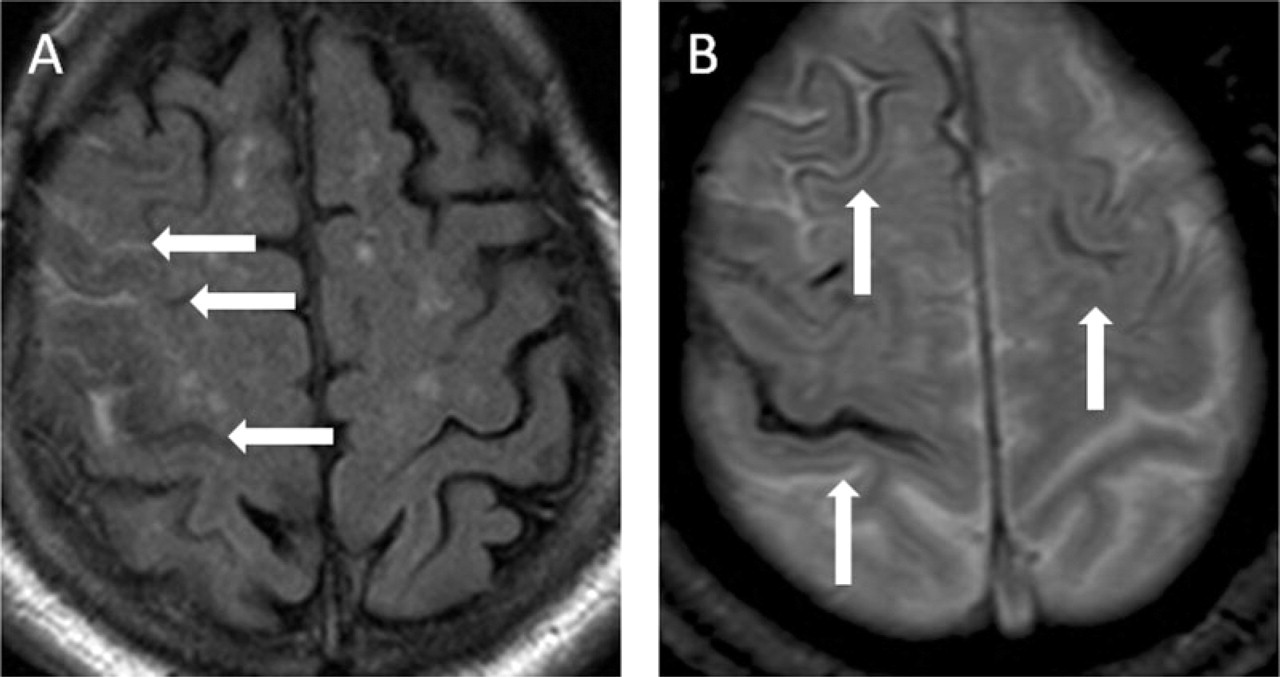
(A) Fluid attenuation inversion recovery MRI demonstrates right sulcal subarachnoid haemorrhage and two small cortical haemorrhages (not shown). (B) Gradient echo sequences demonstrate cortical siderosis, especially in the right central sulcus.
Discussion
We present four cases in whom late-life onset of aura-like symptoms was precipitated by acute contralateral SAH in or adjacent to the central sulcus. After variable symptom-free intervals, three had recurrent events or groups of events associated with new acute SAH. The underlying cause of the SAH was not established, but all four patients demonstrated periventricular white matter disease and at least two cortical haemorrhages, suggestive of a diffuse microvasculopathy. This may have been age- and hypertension-related (6), although the exclusively cortical haemorrhage location in three patients is suggestive of cerebral amyloid angiopathy (CAA) (7).
Fisher's landmark studies in late-life onset aura-like symptoms noted the frequent absence of headache, the tendency of episodes to occur in a ‘flurry’, and a generally benign prognosis (4, 5). Our cases were concordant with Fisher's observations. Few of his patients were investigated with modern imaging techniques.
Convexity SAH has been reported to occur in trauma, cortical vein thrombosis (8), moyamoya disease (9), postpartum and drug-related acute hypertension (10–12), primary CNS angiitis (13) and CAA (14). Patel et al. (12) have recently reported that in their case series of convexity SAH, three idiopathic central sulcus SAH patients experienced recurrent stereotyped neurological symptoms. It has long been recognized that patients with CAA may experience similar events (15), but it has not been widely appreciated that convexity contralateral SAH is a common accompaniment (14, 16, 17).
It has been suggested that epilepsy may underlie these transient symptoms in CAA patients (15). However, the reported spread of sensory symptoms in our cases and others (whether associated CAA is proven or not) is slow, spreading over 2–20 min. Furthermore, some cases have demonstrated weakness without positive neurological phenomena, which would be an unusual manifestation of epilepsy (e.g. cases 1, 3 and 4 above, Karabatsou et al. case 1 (14) and Roch et al. case 4 (17)). No cases in the literature have reported motor or generalized seizure activity or EEG evidence of epileptic activity during an event. An ischaemic aetiology has also been suggested (14), but DWI is invariably normal.
The clinical features of our cases and others are more in keeping with migrainous aura. Spreading cortical spreading depression (SCD), a putative mechanism for migraine aura (18), can be triggered by subarachnoid blood products (19) and has been demonstrated in human diffuse SAH (20). SCD initiated in or adjacent to the central sulcus would produce symptoms identical to those of reported cases. The apparent response to anticonvulsant medications (15, 17) could be explained by suppression of SCD rather than epilepsy (21).
All our patients were on antithrombotic therapy, often intensified by other practitioners after misdiagnosis with cerebral ischaemia. It is unclear whether antithrombotic therapy increases the propensity to convexity SAH; however, cessation seems prudent.
Late-onset aura-like symptoms can be triggered by convexity SAH. Links between this condition and CAA warrant further elucidation. Awareness of this entity will prevent unnecessary investigations and dangerous initiation or escalation of antithrombotic therapies. Anti-migraine therapies can anecdotally cause remission, although event groups are self-limiting. MRI, including gradient echo sequences, should be performed in all cases of late-onset aura-like symptoms.
