Abstract
Intolerance to smell is often reported by migraine patients. This study evaluates osmophobia in connection with the diagnosis of migraine and episodic tension-type headache (ETTH). The characteristics of this symptom are also investigated. We recruited from our Headache Centre 1005 patients (772 female, 233 male; age 37 ± 11 years), of whom 677 were migraine without aura (MoA), 130 migraine with aura (MA) and 198 TTH. Patients with two or more forms of primary headache were excluded. Among migraine patients, 43.9% with MoA and 38.5% with MA reported osmophobia during the attacks; none of the 198 TTH patients suffered this symptom. Most frequently offending odours were scents (63.9%), food (55.2%) and cigarette smoke (54.8%). Osmophobia appears structurally integrated into the migraine history of the patient. It seems to be a peculiar symptom favouring the diagnosis of migraine (MoA and MA) in the differential diagnosis with ETTH.
Introduction
In recent years there has been an increase in scientific interest in olfaction, with alterations found in the initial phase of degenerative neurological conditions, such as Alzheimer's disease and Parkinson's disease, and recognition of the importance of early and differential diagnosis between the idiopathic and non-idiopathic degenerative forms (1, 2).
The diagnosis of primary headaches is based on the application of clinical-anamnestic criteria, whereas laboratory or instrumental examinations are used only to exclude a secondary cause. No criterion has been identified as sufficiently sensitive and specific to be considered pathognomonic. It is a common experience that migraineurs frequently report the occurrence of osmophobia. It can be defined as an unbearable perception, during a migraine attack, of odours that are non-aversive or even pleasant outside the attack. In the Appendix of the International Classification of Headache Disorders (ICHD)-II (3), osmophobia has been proposed as an additional criterion for migraine diagnosis among the accompanying symptoms. Moreover, it has been demonstrated that migraine patients in the interictal period present an altered threshold to an olfactory stimulus (4–8): this is in keeping with the concept of cortical hyperexcitability in migraineurs with reference also to the olfactory processes, as already reported for the visual and auditory systems (8, 9). However, the relationship between osmophobia and migraine attack has not been thoroughly studied, even though as early as the second century AD, Aretaeus of Cappadocia asserted that the sense of olfaction was altered during a headache attack (‘to the sick one are equally intolerable scents and unpleasant odours’ (10)), and in the 19th century Gowers observed that an olfactory stimulus could trigger migraine attacks (11). Only two studies have specifically investigated the occurrence of osmophobia in adults during migraine attack. The first, prior to the formulation of International Headache Society (IHS) criteria (12), reported it in 40% of 50 migraine patients studied (13); the second, more recent study of a large sample of patients found the presence of osmophobia in 25% of migraineurs (14). In an epidemiological study of a Latin American population, osmophobia in migraineurs was referred to as ‘almost always’ present in 47.7% of subjects and ‘only rarely’ in 7.1% (15). In a young population, in a recent study, osmophobia had a prevalence of 20% (16). In a previous preliminary report (17) we have stressed the importance of osmophobia for the diagnosis of migraine.
This paper presents the final results of a clinical study on patients suffering from migraine and episodic tension-type headache (ETTH) and also reports the characteristics of osmophobia in migraine patients.
Materials and methods
A clinical study was conducted on a consecutive series of patients referred to our Headache Centre from January 2003 to July 2006. The study, which began before publication of ICHD-II, used the 1988 IHS diagnostic criteria; nevertheless, when ICHD-II was applied to the study, the diagnoses of our population were not modified. The patients studied suffered either from migraine, divided into those without (MoA) and with aura (MA), or from ETTH. The diagnosis was made on the basis of the patient's medical history, carried out with a semistructured questionnaire; a general and neurological examination; and the exclusion, when needed, of secondary causes by means of laboratory and/or instrumental examinations. Subjects presenting with more than one form of headache were excluded.
All patients were orally administered a semistructured questionnaire to evaluate the eventual presence of osmophobia, as defined in the Introduction, during headache attack.
In the first part of about two-thirds of patients who proved to be osmophobic, the following aspects were assessed more closely: the time of appearance in the headache history (present from the beginning; within 5 years; after 5 years); the frequency of osmophobia recurrence (n/10 attacks); the time of its appearance in relation to the pain phase, which was divided by convention into three time periods: onset before the pain phase (osmophobia appeared within 30 min before the headache), onset at the start (within 10 min after the start of the pain phase), onset during the pain phase (after 10 min from the start of the pain phase); duration of osmophobia during the course of headache attacks; number and type of odours, freely indicated by the patients, divided by convention into four categories (scents, foods, cigarette smoke, other) in accordance with Blau and Solomon (13). The patient was given the further possibility of specifying the frequency with which the olfactory stimulus triggered an attack, and the odour capable of provoking the crisis.
Statistical analysis was performed using χ2 test and analysis of variance (
Results
The study population (Table 1) consisted of 1005 patients ranging in age between 14 and 71 years (mean 37.2 ± 11.4 years), of whom 772 were female (76.8%, mean age 37.6 ± 11.4 years) and 233 male (23.2%, mean age 36.0 ± 11.4 years). Of the 1005 patients, 807 were migraineurs (80.3%) with a mean age of 37.4 ± 11.3 years; in particular, 677 had a diagnosis of MoA (67.4%, mean age 37.6 ± 11.3 years) and 130 diagnosis of MA (12.9%, mean age 36.4 ± 11.1 years); 198 suffered from ETTH (19.7%, mean age 36.4 ± 11.7 years).
Patient characteristics according to diagnosis

MoA, Migraine without aura; MA, migraine with aura; ETTH, episodic tension-type headache.
The 807 migraine patients were divided on the basis of gender (Table 1) into 78.7% (635/807) female and 21.3% (172/807) male. Of the 677 patients affected by MoA, 79.8% (540/677) were female and 20.2% (137/677) male; of the 130 diagnosed with MA, 73.1% (95/130) were female and 26.9% (35/130) male; of the 198 patients diagnosed with ETTH, 69.2% (137/198) were female and 30.8% (61/198) male.
In our study (Table 2), 43.0% (347/807) of migraine patients reported osmophobia during an attack. Of the 677 patients affected by MoA, 43.9% (297/677) reported osmophobia, whereas of the 130 with MA, 38.5% (50/130) complained of the same disturbance. Of the 198 patients diagnosed with ETTH, none reported the presence of osmophobia during an attack (P < 0.00001). Therefore, the symptom osmophobia in migraine patients gave a sensitivity of 43% and a specificity of 100%.
Patients who refer osmophobia during attacks

MoA, Migraine without aura; MA, migraine with aura; ETTH, episodic tension-type headache.
On the basis of gender, 45.0% (286/635) of women with migraine (MoA + MA) complained of osmophobia, compared with 35.5% (61/172) of men (P = 0.02). In women, in particular, it occurred in 45.5% (246/540) of those suffering from MoA and in 42.1% (40/95) of those with MA, whereas in men it occurred in 37.2% (51/137) of those suffering from MoA (P = 0.07) and in 28.6% (10/35) of those with MA (P = 0.16).
Migraine patients (MoA + MA) who reported osmophobia had a mean age of 37.5 ± 11.1 years; those who did not report osmophobia had a mean age of 37.3 ± 11.4 years. The statistical difference was not significant.
Osmophobia was more closely studied in the first 241 (69.5%) migraine patients with osmophobia, with the following results.
Time of appearance of osmophobia in the patient's medical history (Fig. 1)
Osmophobia was present from the start of the first headache attack in 90.8% (219/241) of migraine patients: in 90.2% (185/205) of patients with MoA and in 94.4% (34/36) of those with MA. It appeared within 5 years in 6.2% (15/241) of patients: in 6.3% (13/205) of patients with MoA and in 5.5% (2/36) of those with MA. It appeared after 5 years in 2.9% (7/241): in 3.4% (7/205) of patients with MoA and in no patient affected by MA. In just one patient affected by MoA, osmophobia appeared after 10 years from the first attack.

Time of appearance of osmophobia in the patient history in relation to occurrence of headache (% of patients). ▪, Migraine without aura (MoA) + migraine with aura (MA); □, MoA;  , MA.
, MA.
Frequency of osmophobia during attacks (Fig. 2)
Osmophobia was present in 10 of 10 attacks in 52.9% (127/240) of migraine patients (one patient did not know how to describe the symptom): in 54.9% (112/204) of patients with MoA and in 41.7% (15/36) of those with MA. In 33.8% (81/240) of patients it occurred in five to nine of 10 attacks: in 32.3% (66/204) of patients with MoA and in 41.7% (15/36) of those with MA. In 13.7% (33/240) of patients it occurred in fewer than five of 10 attacks: in 13.2% (27/204) of patients with MoA and in 16.7% (6/36) of those with MA.

Frequency (% of patients) of osmophobia occurrence (n/10 attacks). ▪, Migraine without aura (MoA) + migraine with aura (MA); □, MoA;  , MA.
, MA.
Time of appearance of osmophobia in relation to the pain phase (Fig. 3)
Osmophobia was present before the start of the attack in 3.7% (9/241) of migraine patients: in 3.4% (7/205) of patients with MoA and in 5.6% (2/36) of those with MA; at the start of the attack in 23.2% (56/241) of patients: in 23.9% (49/205) of patients with MoA and in 19.4% (7/36) of those with MA; during the attack (after 10 min) in 73.0% (176/241) of migraineurs: in 72.7% (149/205) of patients with MoA and in 75.0% (27/36) of those with MA; in no case was the onset of osmophobia reported at the end of the pain phase or after it had concluded.

Time of appearance of osmophobia in relation to the pain phase (% of patients). ▪, Migraine without aura (MoA) + migraine with aura (MA); □, MoA;  , MA.
, MA.
Duration of osmophobia during headache attacks
Osmophobia stopped before the end of the pain phase in 1.7% (4/241) of patients, all with MoA (1.95%, 4/205); it stopped with the pain phase in 98.3% (237/241) of patients. In five patients (2.1%) affected by MA, it persisted longer than the pain phase.
Categories and types of smells
Categories
Osmophobia was reported for a single category of smells by 35.3% (85/241) of migraine patients: by 36.1% (74/205) of patients with MoA and 30.6% (11/36) of those with MA. Of these, 42 patients reported an annoyance for scents, 24 for food odours, 18 for smoke and one for other types of odour. A total of 43.2% (104/241) reported it for two odours: 41.9% (86/205) of patients with MoA and 50% (18/36) of those with MA. More than two odours were reported by 21.5% (52/241): 21.9% (45/205) of patients with MoA and 19.4% (7/36) of those with MA.
Type
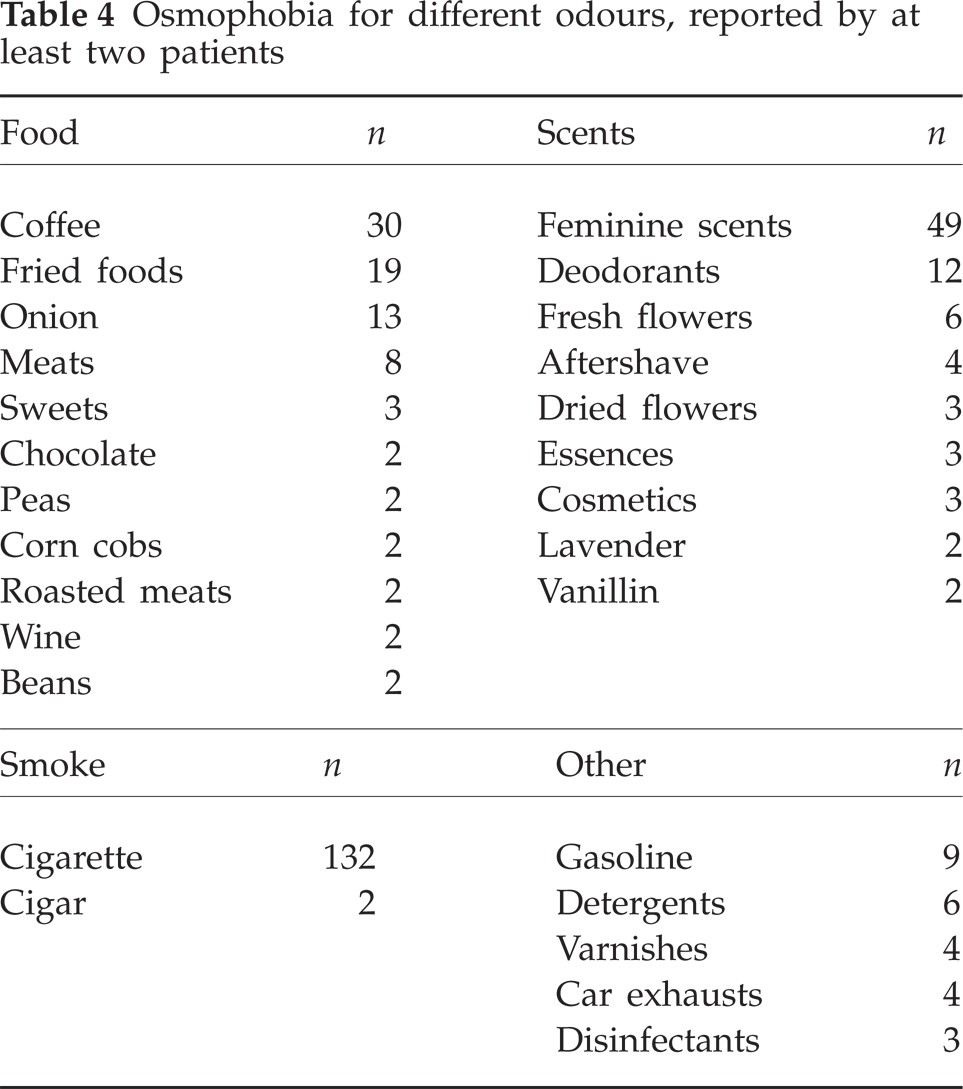
In 69.7% of patients with osmophobia (168/241) the type of odour involved was further specified: in 68.3% (140/205) of patients with MoA and in 77.7% (28/36) of those with MA. Scents (Table 3) were reported by 63.9% (154/241) of patients: by 62.9% (129/205) of patients with MoA and by 69.4% (25/36) of those with MA. Among the scents more often mentioned were feminine scents (cited by 49 patients, 39 female and 10 male), deodorants (n = 12), fresh flowers (n = 6), aftershaves (n = 4), dried flowers (n = 3), essences (n = 3), cosmetics (n = 3), lavender (n = 2) and vanillin (n = 2) (Table 4). The odour of food (Table 3) was cited by 55.2% (133/241) of migraineurs with osmophobia: by 54.6% (112/205) of patients with MoA and by 58.3% (21/36) of those with MA. Some of the more often mentioned smells within this category were coffee (n = 30), fried foods (n = 19), onion (n = 13), meat (n = 8) and sweets (n = 3) (Table 4). Cigarette smoke (Table 3) was cited by 54.8% (132/241) of patients: by 55.1% (113/205) of patients with MoA and by 52.8% (19/36) of those with MA; two patients also cited cigar smoke in addition to cigarette smoke (Table 4). Other odours were reported by 14.5% (35/241) of patients: by 14.6% (30/205) of patients with MoA and by 13.9% (5/36) of those with MA (Table 3). Some of the odours more often cited were gasoline (n = 9), detergents (n = 6), varnishes (n = 4), car exhausts (n = 4) and disinfectants (n = 3) (Table 4).
Osmophobia toward different categories of odours during migraine attacks

MoA, Migraine without aura; MA, migraine with aura.
Osmophobia for different odours, reported by at least two patients

Olfactory stimuli as triggers for attacks (Fig. 4)
Olfactory stimuli were reported to trigger attacks only in migraine patients who complained of osmophobia. Of these, it occurred in 25.3% (61/241) at least once in a lifetime: in 25.7% (52/202) of women and 23.1% (9/39) of men, and in 26.3% (54/205) of patients affected by MoA and 19.4% (7/36) of those affected by MA. Further categorizing these patients (Fig. 4), olfactory stimuli triggered attacks on less than one of 10 occasions involving contact with the same stimulus in 4.1% (10/241) and on at least one of 10 occasions in 21.2% (51/241). More detailed analysis showed that olfactory stimuli triggered attacks in 9.1% (22/241) on one of 10 occasions, in 5% (12/241) on two of 10, in 5% (12/241) in three to five, in 1.7% (4/241) in six to nine, and in only 0.4% (one patient) did an olfactory stimulus (the smell of a grilled chicken) trigger attacks on every occasion of contact. Thus, olfactory stimuli were reported to trigger migraine attacks in the total migraine patient population (with and without osmophobia) at least once in a lifetime in 10.7% (61/569), and in at least one of 10 attacks in 8.9% (51/569).

Frequency with which olfactory stimuli trigger attacks in migraine patients with osmophobia (n = 241). Results expressed as percentage of patients who present n attacks/10 occasions of contact with olfactory stimuli.
In the patients who reported olfactory stimuli as a trigger cause, general scents accounted for 54.1% (33/61); in particular, feminine scents accounted for 29.5% (18/61). In 31.1% (19/61) of cases the triggering odour was cigarette smoke, in 18.03% (11/61) food odour. Other smells reported to trigger attacks were: incense, lavender, coffee, melon, boiled or grilled meat, gasoline, varnishes, ammonia and detergents. A single odour triggered attacks in the majority of patients, 75.4% (46/61), whereas two or more odours, independently, could trigger attacks in 24.6% (15/61).
Discussion
The diagnosis of primary headaches is based on clinical-anamnestic criteria, since at the present time no biological or instrumental marker is available. Negative general and neurological examinations outside the crises are a constant in primary headaches and, together with the anamnestic-clinical picture, allow the physician to make the diagnosis in the majority of cases. Since 1988 the IHS (12) has introduced a classification system that codifies headaches on the basis of mainly anamnestic-clinical operational criteria; this classification has recently been updated (3). The most frequent forms of primary headaches, migraine, ETTH and episodic cluster headache (ECH), are characterized by a painful phase associated with neurovegetative phenomena. While ECH is characterized by a particular vegetative component and MA by the aura itself, less clear in this regard is the distinction between MoA and ETTH. In fact, IHS criteria allow diagnostic overlapping between MoA and ETTH (18–21), since both forms can be characterized by pain of moderate intensity and by a duration partially overlapping; moreover, both can be accompanied by neurovegetative symptoms, even if quantitatively different. According to various authors (19, 21), such diagnostic difficulty can be overcome by using the following IHS criteria for the diagnosis of MoA: unilaterality of pain, pulsating pain quality, and exacerbation with physical activity; moreover, grading of the accompanying symptoms can also be useful. The validation of IHS criteria of 1988, with the use of statistical analysis such asreceiver–operating characteristic (22), applied to a sample population from Zurich has identified the pulsating characteristic of pain and the symptom photophobia as the criteria yielding the best combination of sensitivity and specificity for the diagnosis of MoA. In addition to these two IHS criteria, the presence of osmophobia and difficulty in thinking during headache attacks has also increased the levels for sensitivity and specificity (22). The validity of semistructured questionnaires has been supported (21, 23), which can accurately investigate the medical history, reducing the subjective aspects of the clinical evaluation. Also, additional, complementary criteria have been proposed, such as the different instinctive manoeuvres adopted by migraine and ETTH patients (24). Moreover, a self-administered questionnaire has recently been made available, identifying the incapacitating intensity of the pain and the presence of nausea and photophobia as the more important clinical features in favour of the diagnosis of MoA (25).
Osmophobia is often reported during a migraine attack in association with phono- and photophobia. The Appendix of the new IHS classification (12) adds osmophobia among the alternative criteria for the diagnosis of MoA. Indeed, they differ from the original criteria only in point D, which requires the presence of at least two of the following: nausea, vomiting, photophobia, phonophobia and osmophobia. According to the diagnostic and therapeutic guidelines of the Italian Society for the Study of Headaches (26), the presence of osmophobia is reported among the additional clinical information in support of the diagnosis of migraine; moreover, the guidelines of the Canadian Headache Society recommend that the presence of osmophobia be assessed, since it has been considered a specific feature for the diagnosis of MoA (27).
Based on our data, the percentage of migraine patients who reported osmophobia (43%) was similar to that found by Blau and Solomon (13), and greater than that more recently reported by Kelman (14) in a patient sample numerically comparable to ours. Our study of ‘pure’ patient populations (MoA, MA and ETTH groups did not include patients with another concomitant form of headache) shows that osmophobia is reported during an attack only by migraine patients and not by those with ETTH. In this context, osmophobia has low sensitivity (43%) but high specificity (100%) for the diagnosis of migraine. With regard to the study of Kelman (28), we have found a greater sensitivity (43% vs. 23%) and specificity (100% vs. 87%). It should be mentioned that the evaluation of sensitivity and specificity in this instance considered a sample of migraineurs (IHS 1988: 1.1–1.6) in relation to a total patient population with 27% of patients diagnosed according to IHS 1988: 1.7 (Migrainous disorder not fulfilling above criteria); this fact, we believe, influenced the results reported above.
As can be seen from analysis of the two groups of the population referred to our Headache Centre, it appears that osmophobia is peculiar to migraine (MoA and MA) compared with ETTH, in which it is not found. This fact underscores the importance of osmophobia as a diagnostic criterion for migraine and in the differential diagnosis between migraine and ETTH. We are presently carrying out a prospective study to confirm these data, thus eliminating any memory-related bias which may influence retrospective studies.
Moreover, none of the patients with ETTH reported olfactory stimuli as triggering attacks; this was a very important, clear-cut observation between the two groups of patients studied. In contrast, in another study (29), conducted on a smaller series and consisting of somewhat ‘prevailing’ migraine or ETTH cases (not ‘pure’ as in the present study), just a difference in the percentage of the olfactory trigger was found (respectively, 61% vs. 24% of the two groups).
In our patients, osmophobia was significantly more frequent in women than in men. This finding is in keeping with a study using functional neuroimaging, which showed greater olfactory sensitivity in women compared with men after exposure to the same odours (30). We could speculate that, phylogenetically, this important sense could have been favoured for conservation of the species (identifying food, determining if it was edible, choosing a partner), in the gender which is entrusted with the most delicate moments of reproduction, such as pregnancy and nurturing children.
Moreover, osmophobia during attacks is structurally integrated into the headache history of these patients. In fact, >90% of patients with osmophobia reported that this symptom was present at the beginning of their headache history (this information could be imprecise in patients with long history). In over half of patients it was present in all attacks; it appeared simultaneously with the pain phase in over two-thirds of patients; it persisted for the entire duration of the crisis in nearly the whole study population; and it did not attenuate in intensity or abate with time, as sometimes happens with other migraine symptoms, such as vomiting and nausea.
Nearly two-thirds of patients reported more than one category of odours as annoying during an attack. Those more frequently reported were scents, in particular, feminine scents; these were the odours more often involved also in triggering the attack. Overall, olfactory stimuli were reported as trigger factors at least once in a lifetime by about one-quarter of our population of migraine patients with osmophobia. Among these patients, >20% reported an olfactory stimulus as an attack trigger on at least one of 10 occasions involving contact with the stimulus. It should be emphasized that, in our series, all patients who reported odour as a trigger also reported osmophobia during attacks. Therefore, our data have identified those patients in whom olfactory stimuli can trigger crises as a particularly sensitive subpopulation of migraineurs who suffer from osmophobia, unlike the results reported by Kelman (14), who found that odours can trigger attacks in 45% of his patients, whereas osmophobia was limited to 25%.
It has been known for some time that the trigeminovascular system is activated during migraine attack (31). One might expect at least that odours reported as triggering attacks would have a distinct irritating component and thus lead to activation of the nasal trigeminal afferents; instead, neutral or pleasant odours are often involved. Therefore, it seems that when an olfactory stimulus triggers an attack, it only secondarily involves the trigeminovascular system.
Functional neuroimaging techniques, such as positron emission tomography (PET) and functional magnetic resonance imaging, have demonstrated that the cerebral areas that are activated with a selective olfactory stimulus (vanillin or hydrogen sulphide) are the pyriform cortex, orbitofrontal cortex, hippocampus, amygdala, insula, cingulate gyrus and the cerebellum (32–34). A recent PET study has shown that the perception of odourous compounds appears mediated by a series of regions that are in part distinct, depending on whether the olfactory stimulus is purely olfactory (vanillin) or olfactory with trigeminal stimulation (acetone). Whereas vanillin primarily activates the amygdala and the pyriform cortex, acetone also engages areas activated by painful stimuli, such as the primary somatosensory cortex and the posterior portion of the anterior cingulate gyrus (35). From these observations it appears that the study of osmophobia in migraine can provide new insights into the pathogenic basis of this form of headache.
In this sense, patients in whom olfactory stimuli provoke migraine attacks with high frequency (about 2% of our migraineurs with osmophobia report odour as a trigger on more than six of 10 occasions of contact with it) potentially constitute an important model. Indeed, their attacks could be timely induced for study by different methods, such as neurochemical, neurophysiological and, above all, with advanced functional neuroimaging.
In conclusion, our data show that osmophobia can be considered a peculiar symptom of migraine, both MoA and MA. Its presence adds a relevant element to the diagnosis of migraine in the differential diagnosis with ETTH. Consequently, we strongly support the inclusion of osmophobia among the diagnostic criteria for migraine, as proposed in the Appendix of the new IHS classification. Moreover, further studies are highly recommended to evaluate the possible role of this symptom in the differential diagnosis with the other forms of primary headaches and secondary headaches.
Acknowledgements
We thank Professor Michael A. Moskowitz, MD, Director of the Stroke and Neurovascular Regulation Laboratory, Department of Radiology, Massachusetts General Hospital, Harvard Medical School, for his kind, very helpful suggestions.
