Abstract
The effect of the antimigraine drug rizatriptan on the amplitude and habituation of the contingent negative variation (CNV) in healthy women was examined in a randomized, double-blind, placebo-controlled trial. The test persons were assigned either to a drug (n = 20) or a placebo group (n = 20). The CNV was recorded three times: before, directly after, and 24 h after drug or placebo intake. The CNV paradigm was presented in a standard, a cued and a choice version. Rizatriptan led to an increase of CNV amplitude that depended on the level of difficulty of the task. Whereas there was no drug effect in the standard version, an amplitude increase was obtained mainly in the choice task. The results are in line with the ceiling theory of migraine, which assumes a rise of CNV amplitude if the serotonin level is lowered.
Introduction
Brain excitability plays a central role in the pathophysiology of migraine. Attempts to explain the mechanisms of migraine have led to the development of two different accounts, one assuming cortical hyperexcitability, the other hypoexcitability (for review see (1) and (2, 3), respectively). The former view states that migraine patients have a high pre-activation level, primarily in the occipital cortex, which makes their brain susceptible to the occurrence of a neuroelectric and metabolic process similar to cortical spreading depression (CSD) found in animal studies (1). The CSD, spreading from the visual areas to the rest of the cortex, is thought to result in the development of migraine headache (4). In contrast, the latter concept states that migraine patients have a low cortical pre-activation level compared with healthy controls (2, 3). Having a higher pre-activation level, healthy individuals can quickly reach the maximum cortical negativity (the ceiling), which triggers the mechanism of habituation, allowing them to protect themselves against overstimulation (5). Because of their low pre-activation level, migraine patients cannot reach the ceiling and are therefore unable to habituate. The resulting overstimulation disturbs the metabolic homeostasis in the occipital cortex and triggers CSD (2).
A way frequently used to assess cortical excitability is the recording of the contingent negative variation (CNV). The CNV was first found as a slow electrical cortical wave in a simple task in which participants had to make a quick response to the second stimulus in a pair, and the interval within stimulus pairs was kept constant (6). The authors suggested that the CNV reflects contingency between the two stimuli, hence its name. It was subsequently shown that the CNV consists of at least two subcomponents, one of them early in the interval between the two stimuli, the other one later, just before the second stimulus (for review see (7).
Early studies of CNV in migraine showed that migraine patients have an increased CNV amplitude during the interictal phase (8–11). More recent studies (most of them carried out at the University of Kiel) have revealed during the interictal phase an increased early CNV component in migraine patients compared with controls (12–21). A few days before a migraine attack, a further increase of the CNV amplitude can be observed (14), followed by a decrease during the attack, when there is no difference between patients and healthy individuals (13, 14, 18, 19). Up to 3 days after an attack, patients still have a normal CNV amplitude (14). During the interictal phase, patients also show a lack of habituation mostly for the early CNV component (13, 15, 19, 20, 22), but the habituation returns to normal values during the attack (13, 19).
However, these findings could not be replicated in some studies. One study found no differences at all in CNV amplitude between the ictal and interictal phases in migraine patients (23). Another (24) compared three groups of migraine patients (a Cyclandelate treatment group, a placebo group, and a psychotherapy group), before and after treatment, with a healthy control group. Before preparation intake, the placebo group had significantly smaller CNV components, and the medicament and the psychotherapy groups had a smaller late CNV component than the control group. In the medicament and placebo groups, this remained true for both the ictal and interictal phases (the CNV of the psychotherapy group was recorded only interictally). Migraine patients did not habituate in any of the two phases, but nor did control subjects. After 3 months of treatment (which resulted in a significant decrease in the frequency of attacks), the differences in the amplitude of the interictal CNV between patients and controls disappeared. Still, no habituation was found in any subjects. A Dutch group (25) found during the interictal phase a tendency in migraine patients (P = 0.092) for a smaller late CNV component compared with healthy controls. In a previous study (26), these authors obtained in the ictal phase a tendency (P = 0.078 and P = 0.067) to a decrease of both CNV components, but only after sumatriptan intake. Hence, they suggested that the often registered ictal drop in CNV amplitude might partially be due to a low serotonin level due to medication with triptans.
The aim of the present study was to separate the effects of migraine and of triptans and thus to investigate the influence of rizatriptan (a second-generation triptan) on the amplitude and habituation of the CNV in healthy subjects in a double-blind, strictly controlled design. To explore further the selectivity of the effects of the serotonergic drug, the CNV was recorded at three levels of task difficulty. Also, the effects of rizatriptan on reaction time and error rate were addressed, because it has been shown that such effects can depend on the level of difficulty (27).
Subjects
Only female subjects were chosen for this study, because migraine has a higher prevalence in women (28) and there are sex-related differences concerning serotonin synthesis (29, 30). Forty healthy women (age 19–36 years, mean 24.45) were assigned randomly either to a triptan group (n = 20) or to a placebo group (n = 20) and were matched according to age. One of the authors (W.L.), who controlled age matching and thus could know the identity of the subjects, did not participate in experiments and data analysis. All subjects were students enrolled at the University of Tübingen. All were right-handed according to the Edinburgh Handedness Inventory. The health check was done by a medical doctor. None of the participants was taking analgesics, antidepressants or β-blockers. Further exclusion criteria were pregnancy, epileptic seizures, cardiovascular disorders, liver and kidney disorders, smoking, drugs and alcohol abuse. The study was approved by the Ethics Committee of the Faculty of Medicine, University of Tübingen, Germany. The subjects received detailed information about the study and gave written consent. They received an expense allowance of €100 for their participation.
Methods
Experimental design and recording sessions
The study was designed as a randomized, double-blind, placebo-controlled experiment. Drug and placebo had the same size and coating and neither the subject nor the persons who conducted recording (R.Z.) and evaluated the data (R.Z., B.K.) knew what the preparation contained. The triptan group was given 10 mg rizatriptan (Maxalt®), whereas the control group received a placebo. The CNV recordings were conducted for each subject on two successive days. In order to avoid circadian and food influences (i.e. coffee), both appointments began at 09.00 h with a standardized breakfast (a sandwich, an apple, yogurt and fruit-tea).
On the first appointment a CNV recording was made (baseline), followed by medication or placebo intake. Since rizatriptan reaches its plasma maximum after 1–1.5 h, the second CNV recording was made 1 h after preparation intake. Approximately 24 ± 0.5 h after the intake, a third CNV recording session was carried out.
Each session was in three parts. The first part was the standard CNV paradigm. Each trial consisted of a warning stimulus (WS) followed by an imperative stimulus (IS). The WS was a bell sound lasting for 1000 ms. The IS was a sine tone of 1000 Hz. The interval between WS offset and IS onset was 3 s. The intertrial interval varied randomly between 3.5 and 5.5 s. Subjects had to interrupt the IS by pushing a button with the right thumb as fast as possible. If the reaction time (RT) was < 100 ms or > 1000 ms they received an acoustic feedback announcing an error, and the trial was automatically repeated. A total of 44 trials were registered.
The second part was a choice task with the same WS as before, but with two different IS. One of them was a 400-Hz sine tone and the other a 1000-Hz sine tone. The tones had to be interrupted with the left and the right thumb, respectively. Before this recording started, subjects had received six practice trials. A total of 88 trials were recorded (44 trials for each hand). If subjects responded too fast, too slowly or with the wrong hand, an acoustical feedback informed them about the error and the trial was repeated.
The third part of CNV recording was a cueing task. The two tones from the preceding part were used here as WS, whereas the bell became the IS. When the warning tone was of low or high pitch, subjects had to interrupt the IS with the left or right hand, respectively. After receiving six practice trials, 88 trials were recorded (44 for each hand presented randomly). If subjects responded too fast, too slowly or with the wrong hand, an acoustical feedback informed them about the error and the trial was repeated.
During sessions, subjects sat comfortably in an armchair in an acoustically isolated room. The auditory stimulation (70 dB) was produced by a loudspeaker situated 1.5 m away from the subject. In order to avoid ocular and jaw movement artefacts, participants were asked to keep their eyes closed during recording and to try not to blink or move their jaw. Closing eyes during auditory tasks can alleviate artefacts in event-related potential recordings (31). The EEG was recorded using Ag/AgCl electrodes attached at Fz and Cz, according to the International 10–20 System, with references placed on the earlobes and the ground electrode on the forehead. The two reference electrodes were on-line connected. An electro-oculogram was also recorded. Electrodes positioned above and below the right eye registered the vertical movements and electrodes positioned on the outer sides of the eyes registered the horizontal movements. Ocular artefacts were corrected off-line (32). Electrode impedance was kept < 5 kΩ. The EEG signals were amplified using a Brain Amp amplifier and registered with Brain Vision Recorder software. The recording was made using a sampling rate of 500 Hz, a time constant of 5 s and a high cut-off filter of 70 Hz.
Data analysis
The EEG signals were processed using the Brain Vision Analyzer software. Forty artefact-free trials per condition were averaged for each person. The zero point of CNV amplitude was determined as the average value of the 200-ms interval before the onset of the WS. The amplitudes of the early and late CNV components were computed according to a method frequently used in migraine research (12). For computing the early component, the highest peak within 550–750 ms after WS onset was determined. The early component was measured as the average amplitude within a 200-ms interval centered on this peak. The late component was defined as the average amplitude within the last 200 ms before the IS onset. Habituation was computed by averaging blocks of four successive artefact-free trials, resulting in 10 habituation blocks for the standard condition (40 trials) and 20 habituation blocks for the choice and the cued condition (80 trials). For each of these blocks, the early and late CNV components were computed.
Statistical evaluation
The data were normally distributed (Kolmogorov–Smirnov goodness-of-fit test) and a mixed-design analysis of variance was therefore performed (General Linear Model module in SPSS; SPSS Inc., Chicago, IL, USA) for the early and late CNV component amplitudes. For the standard task the analysis included a between-subject factor Group and within-subject factors Time (with three levels: baseline, intake, and 24 h after the intake) and Site (with two levels: Fz and Cz). The non-sphericity was evaluated for the factor Time by means of the Mauchly test. If the test was significant, the degrees of freedom (d.f.s) were corrected using Huynh-Feldt ε. In such cases, corrected d.f.s are shown in the text. For the choice and cued tasks, an additional within-subject factor Hand (left vs. right) was considered. Habituation was defined as a linear trend across the 10 blocks in the standard condition, respectively across 20 blocks in the choice and cued conditions. For each person the linear regression was computed with the variables block number on the x-axis and CNV amplitude on the y-axis. The slopes of this regression (33) underwent a mixed-design analysis of variance with the between-subject factor Group and the within-subject factor Time. A drug effect on the habituation would result in a significant Time × Group interaction. In order to assess the habituation for a given recording, the group regression lines were computed for every level of the time variable. Since the CNV amplitude has negative values, a positive slope would indicate habituation and a negative one potentiation. Furthermore, the linearity across blocks was tested with the runs test provided by the software GraphPad Prism Version 4.00 (GraphPad Software Inc., San Diego, CA, USA).
RT was considered as the time between IS onset and the button push. The mean RT for each person was computed after removing outliers using the non-recursive method (34). The RT underwent a mixed-design analysis of variance with the between-subjects factor Group and the within-subjects factor Time.
As errors were considered all RTs < 100 ms or > 1 s, in the choice and cued tasks also responses with the wrong hand were regarded as errors. Because the distribution of error rates strongly differed from normal distribution, the between-group differences were evaluated by means of a Mann–Whitney U-test.
Results
Behavioural data
There were no age differences (Mann–Whitney U-test, P = 0.382) between the triptan group (mean = 25.05, median = 24) and the placebo group (mean = 23.85, median = 23).
There were no significant differences between groups in terms of RT. In the third session the participants were significantly faster compared with the other two sessions in all tasks: standard (F 1.58,59.97 = 9.14, P = 0.001), choice (F 1.48,56.39 = 4.60, P = 0.023) and cued (F 1.52,57.67 = 5.32, P = 0.013). In the choice task the triptan group made significantly more errors than the placebo group after drug intake (see Fig. 1). In the cued task the placebo group made more errors in the first session (Z =−2.47, P = 0.014), but there were no differences thereafter.

Error rate in the choice task, in %.
CNV data
In the standard task, no effect on CNV amplitude or habituation attained significance. As can be seen in Table 1 and Figs 2 and 3, in the choice task the early CNV component was generally larger in the triptan group than in the placebo group. The small difference between groups in the first session increased in the second session and disappeared 24 h later, resulting in a significant interaction between Time and Group: F 2.76 = 3.23, P = 0.045. Because during the first session the amplitude was larger in the triptan group at Cz but larger in the placebo group at Fz, the interaction Time × Site × Group was also significant: F 2.76 = 3.80; P = 0.027 (see Fig. 2). The amplitude in the placebo group decreased after the intake of placebo (compared with the first session), whereas the amplitude in the triptan group remained constant after the intake of rizatriptan and decreased only on the next day.
Amplitude of the early contingent negative variation component in the choice task, in µV (SD in parentheses)


The amplitude of the early contingent negative variation component in the choice task.

The waveform of the contingent negative variation in the choice task. Negativity in this figure and Fig. 5 is plotted upwards.
In the cued task, also, the early CNV component was larger in the triptan group, particularly at Cz, yielding a significant interaction between Site and Group: F 1.38 = 5.19; P = 0.028 (see Table 2 and Figs 4 and 5). Like in the choice task, the between-group difference increased in the second session and decreased in the third one, the interaction Time × Site × Group approaching significance: F 2.76 = 2.72; P = 0.072 (see Fig. 4).
Amplitude of the early contingent negative variation component in the cued task, in µV (SD in parentheses)


The amplitude of the early contingent negative variation component in the cued task.
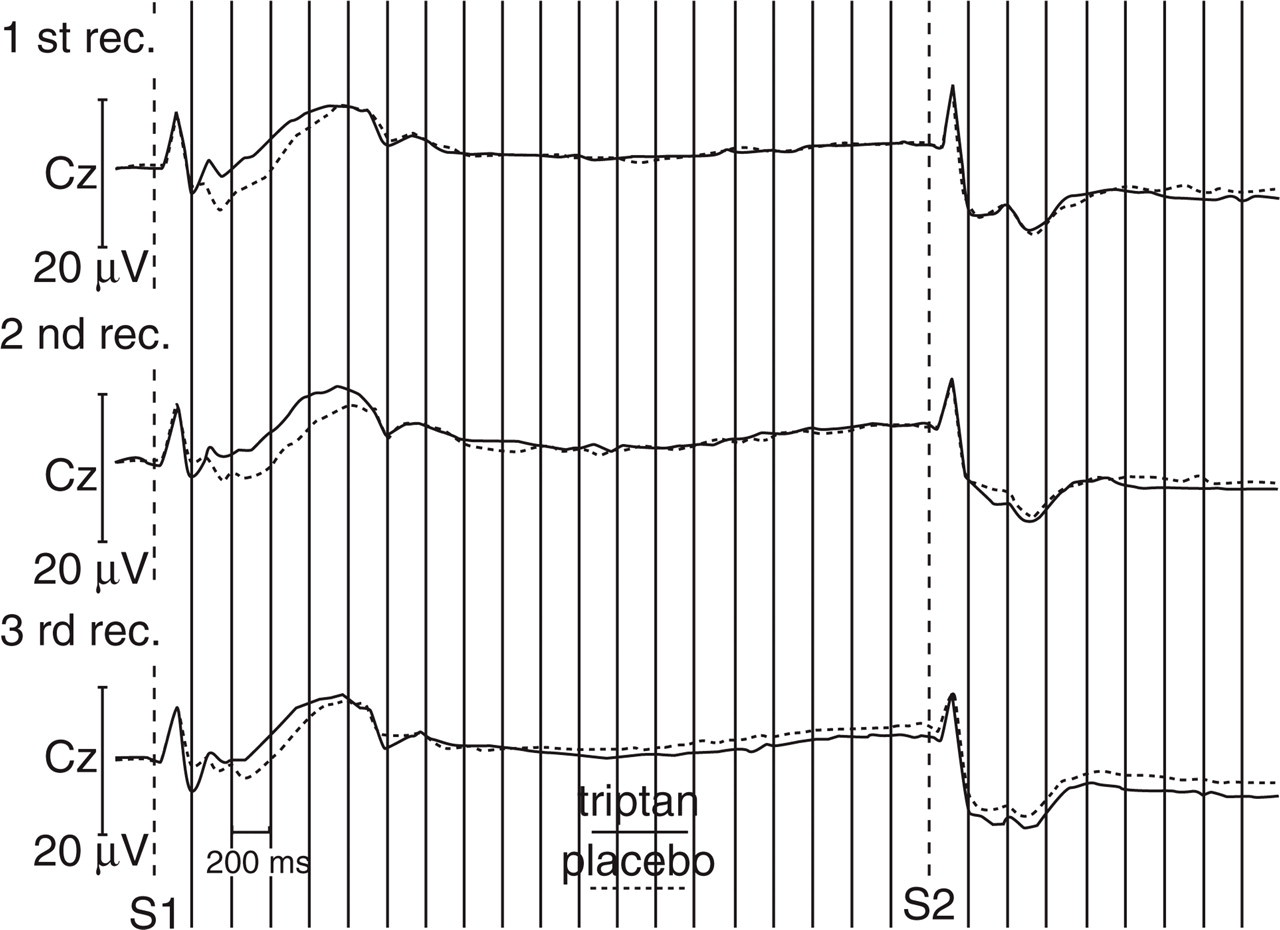
The waveform of the contingent negative variation in the cued task.
For neither task was there any difference between groups in the late CNV component at any time.
CNV habituation data
One can conclude that the linear relationship between x and y found in a sample is also present in the population only if the slope of the regression line is significantly different from zero (35). As shown in Table 3, only a few slopes were significant in the standard task, although linearity was given for every line, as indicated by non-significant runs tests. In the placebo group, the late CNV component habituated in the second and third sessions (both at Fz and Cz). In the triptan group the late component habituated in the third session at Cz, and the early component significantly potentiated in the first session at Fz. The expected Time × Group interaction for habituation slopes was not significant for either CNV component calculated in the standard task (Table 3). Generally, however, the triptan group tended to show less habituation than the placebo group, the main effect of Group being significant for the early CNV component at Fz (F 1.38 = 5.85, P = 0.020) and approaching significance for that component at Cz (F 1.38 = 3.68, P = 0.063) and for the late CNV component at Fz (F 1.38 = 3.88, P = 0.056). Furthermore, in both groups the potentiation trend decreased, and the habituation trend increased, from the first to the third recording sessions, as shown by significant effects of Time for the early component at Fz (F 2.76 = 3.32, P = 0.041) and for the late component at Cz (F 2.76 = 5.55, P = 0.006).
Habituation of the contingent negative variation components in the standard task
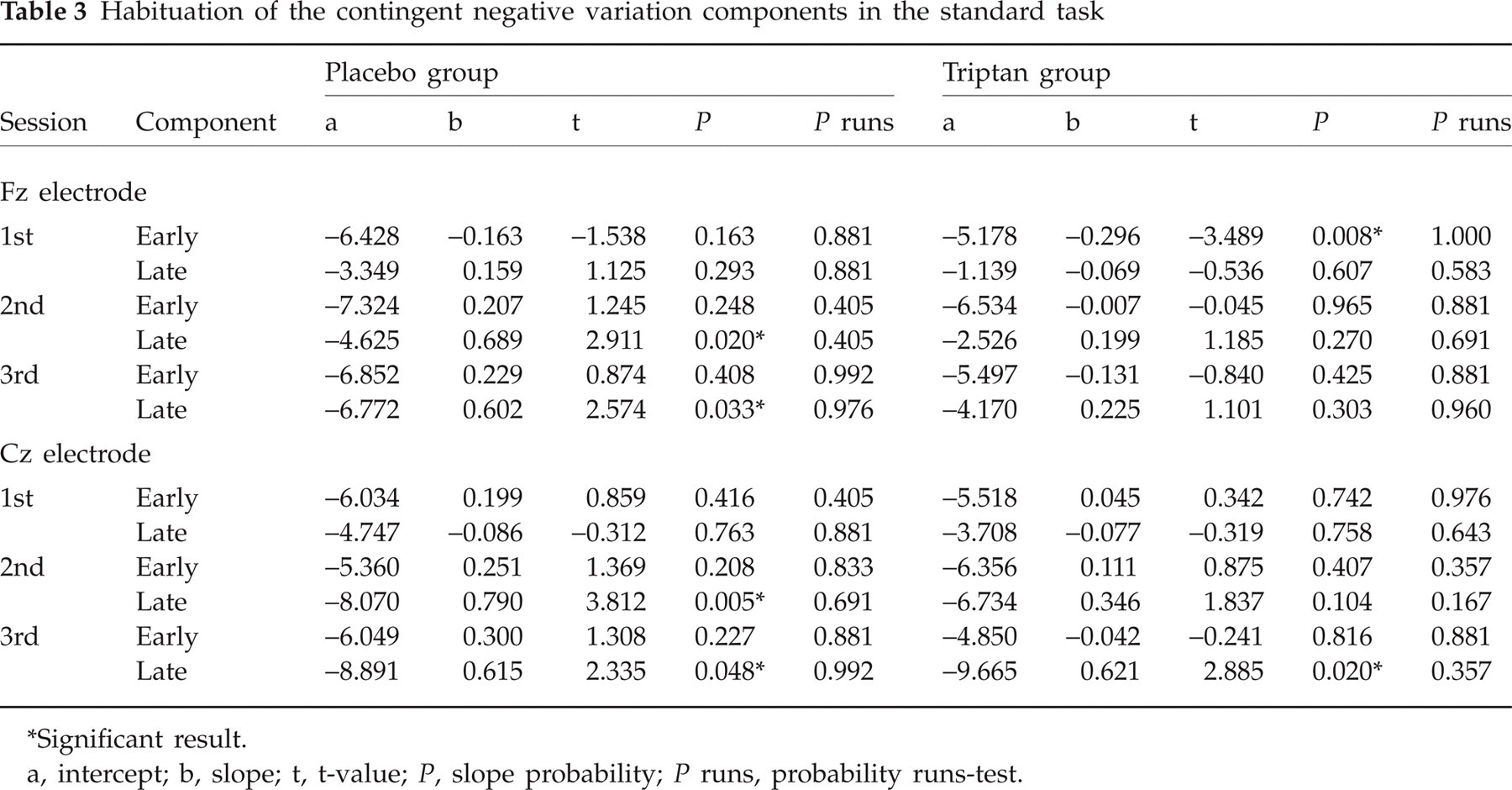
Significant result.
a, intercept; b, slope; t, t-value; P, slope probability; P runs, probability runs-test.
The most consistent habituation pattern was found in the choice task for the early component at Cz, where habituation was present at all time points except the second session in the placebo group (see Table 4 and Fig. 6). As can be seen in Fig. 6, the CNV amplitude in the second session/placebo group was low from the very beginning and, thus, did not decrease with repetition. In the first and third sessions this group, as well as the triptan group in all three sessions, started with a much larger CNV amplitude, which subsequently declined. Habituation in the cued task is shown in Table 5.
Habituation of the contingent negative variation components in the choice task

Significant result.
a, intercept; b, slope; t, t-value; P, slope probability; P runs, probability runs-test.
Habituation of the contingent negative variation components in the cued task
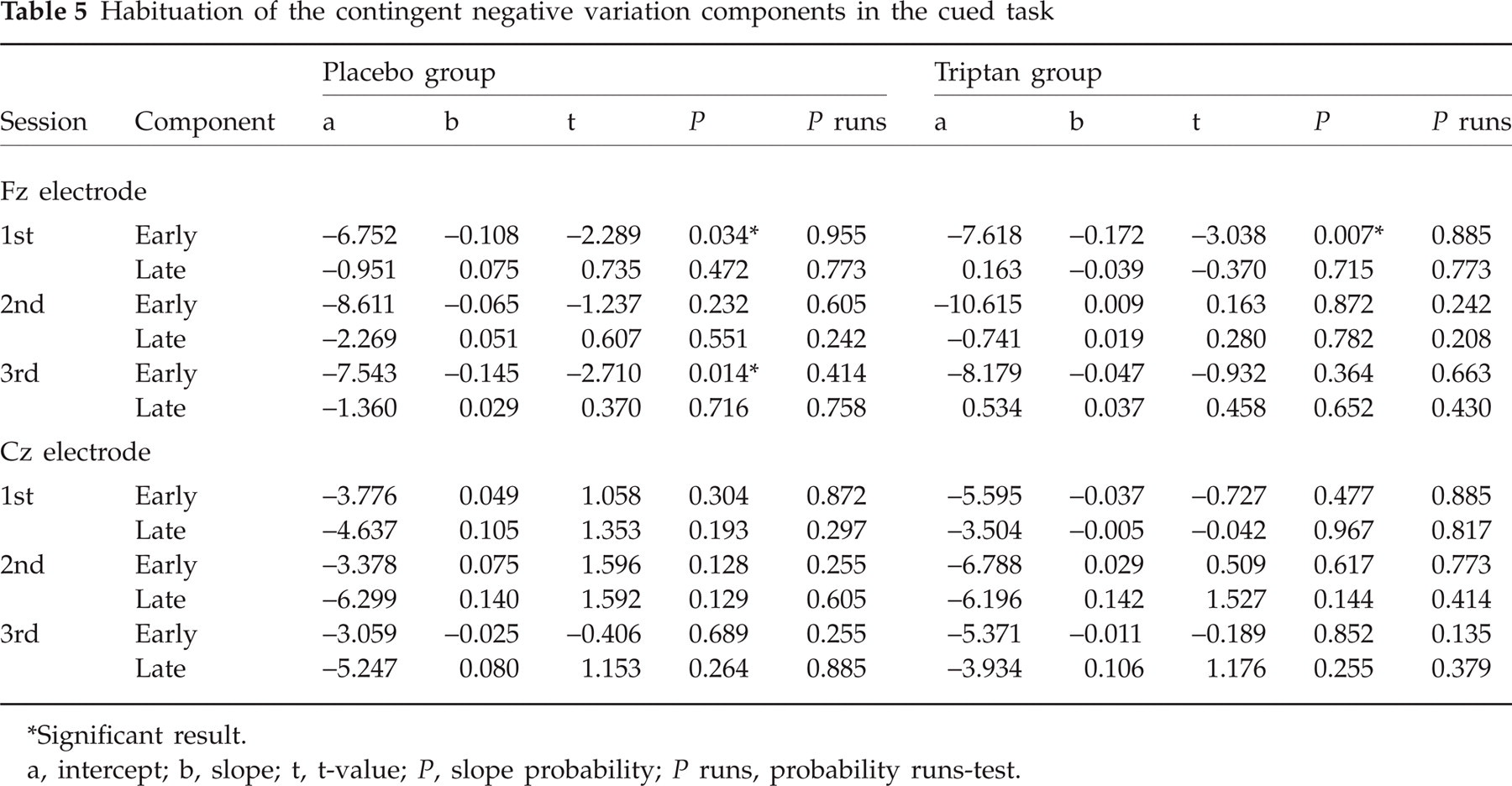
Significant result.
a, intercept; b, slope; t, t-value; P, slope probability; P runs, probability runs-test.

Habituation of the early contingent negative variation (CNV) component at Cz.
Discussion
The explanations of CNV abnormalities found in migraine differ depending on which excitability theory is taken as a basis. Adherents of the hyperexcitability theory conceive of the lack of habituation as a result of a high cortical pre-activation level (36). Being overaroused, migraine patients are unable to habituate and to protect themselves against overstimulation when exposed to stressors. These authors (36) suggest that this overstimulation activates the orbitofrontal–brainstem pathway, as proposed by the hyperexcitability theory (37), thereby triggering a migraine attack. The increased CNV amplitude is thought to be a passive consequence of the lack of habituation. That is, initially the CNV amplitude does not differ from that in healthy controls, but in the latter this amplitude drops with repetition, whereas in migraine patients it does not (13, 22). The average difference is, therefore, a summation artefact.
From the point of view of the hypoexcitability (ceiling) theory of migraine, the increased interictal CNV amplitude is also seen as a summation artefact, but the reason for the lack of habituation is thought to be a low cortical activation level at rest, resulting in the inability to reach ceiling and thus to habituate under stimulation (2). In the interictal phase the amount of habituation of the visual evoked potentials (VEP) is negatively correlated with the amplitude in the first habituation block (38). This indicates that migraine patients initially have a lower arousal level compared with healthy controls (3).
Serotonin plays a key role in the hypoexcitability concept. In the interictal phase, migraine patients are thought to have a low cortical serotonin level, which increases during the attack (39). According to some authors (3), there is evidence that a low serotonin level leads to a decrease of cortical activation at rest, since migraine patients have been shown to have a high intensity dependence of the auditory evoked potentials (IDAP) (40), and the IDAP is negatively correlated to central serotonin level (41, 42). The interictally high IDAP normalizes during an attack (43). During the interictal phase, dexflenfluramine (which stimulates serotonin release) leads to the decrease of the IDAP, whereas zolmitriptan (which inhibits serotonin release) leads to an increase of the IDAP (44).
In the present study we have investigated the relationship, in healthy subjects, between the serotonin level lowered by the intake of triptan, and the cortical arousal as measured by CNV amplitude. The choice task in this study was similar in complexity to the task used in recent CNV studies related to migraine (e.g. the Kiel group used a Go/NoGo task with 32 Go trials and eight NoGo trials).
The drug effect manifested itself with increasing level of task complexity. No effect of drug was found in the standard task, the condition with the lowest task complexity. In the most complex choice task the differences between triptan and placebo after the intake were significant at both Fz and Cz, whereas in the less complex cued task these differences approached significance and were found only at Cz. In the choice task the amplitude of the early CNV component decreased from the first to the last recording session, but this decrease followed different patterns in the two groups. Namely, the amplitude dropped after the intake of placebo and remained, in the placebo group, at the same low level on the next day also. In contrast, after the intake of rizatriptan the amplitude did not change (or even slightly increased), and only after 24 h did it decrease dramatically, attaining about the same level as in the placebo group.
The effect of the triptan in the choice task was also confirmed by the higher error rate in the triptan group during the second recording session. Since CNV amplitude is an index for cortical arousal, the increased number of errors can be explained in terms of the Yerkes–Dodson law. The triptan group had a higher than optimal cortical arousal, which led to a lower performance.
Taken together, the triptan effects obtained in our study in 20 healthy subjects concur with the results of other studies (44–46) that used different physiological measures and found in similarly sized groups (n = 24 and n = 19, respectively) indicators of increasing cortical excitability in healthy participants taking triptans. Specifically, triptans were found to increase IDAP (44, 46) and to decrease the amplitude of P300 (45), which is frequently regarded as an indicator of cortical inhibition (47). The lack of effect of rizatriptan in the easiest task is similar to another study (23), which also did not find any effect of sumatriptan on CNV amplitude in a standard task.
Two methodological limitations of the present study should be mentioned. First, the overall activity level, and particularly that in the standard task, was rather low, given the instruction to relax and close eyes (31). This contrasts, for example, with the study of van der Post et al. (27), whose subjects had to solve visual and acoustic memory and recognition tasks that were parts of a computerized test battery. This difference may also explain the different results obtained in the two studies, e.g. the fact that rizatriptan significantly affected RT in healthy subjects in that study (27), but not in ours.
Second, we did not control the influence of menstrual cycle on the cortical activation. Although significant effects of the menstrual phase on CNV amplitudes were found in migraine patients (48, 49), healthy women demonstrated zero effects (49). As regards other indicators of cortical activity, the data are inconsistent. Some authors have reported increased amplitudes and longer latencies of VEP in the premenstrual phase of healthy subjects (50), whereas others found no changes in VEP (51) and auditory evoked potentials (52) across the menstrual cycle. One study reported a shortened P300 latency in the premenstrual phase (50), another a delayed P300 latency during the ovulatory phase (51), but mostly no latency change across the menstrual cycle is obtained in the healthy (52–54). To summarize, data in the literature do not support the menstrual phase having any large effect on the CNV results obtained in healthy women.
Both CNV and error rate changes obtained in the present study have confirmed one of the premises of the ceiling theory, i.e. that a low serotonin level leads to increased cortical activation. Serotonin is one of the neurotransmitters that regulate cortical excitability (55) and it is thought to maintain stable tonic baseline activation (41, 42). Triptans decrease the serotonin level by activating presynaptic autoreceptors that inhibit serotonin release (56, 57).
The finding that in our study the early and not the late CNV was affected is most plausibly related to the fact that the late CNV is specifically related to motor processes, i.e. preparation of the motor response. The overall level of cortical reactivity, i.e. the drop in firing threshold after perceiving a warning stimulus, is better expressed in the early CNV component.
Recent studies of CNV habituation (13, 15, 19, 20, 22, 24) in migraine have been conducted using a task (a Go/NoGo task with 32 Go trials and eight NoGo trials) whose complexity resembles the choice task in the present study. Available data are not consistent. Whereas the group of the University of Kiel (13, 15, 19, 20, 22) repeatedly reported habituation of the CNV in the healthy, Faran (24) found no habituation in the healthy in the same task. Another group (26) using a standard task did not find habituation differences between healthy and migraine patients (no statements regarding presence or lack of habituation were made). In the present data, the habituation in the standard task was significantly demonstrated only when data were collapsed over all 40 participants and three recording sessions; most habituation coefficients in single groups and in single sessions were not significant. There were no group differences in the Choice and Cued tasks. However, a consistent habituation pattern was found in the Choice task for Cz early component, where the placebo group lacked habituation at the second session. A possible explanation of this finding can be made by using the ceiling theory.
The interictally high IDAP and the increased amplitudes of evoked and slow cortical potentials in migraine are thought to be caused by the lack of habituation in patients (2, 3). According to the ceiling theory, a decrease of the baseline cortical activation leads to dishabituation in healthy individuals, and, conversely, an increase of the baseline activation leads to habituation in patients. Both have been demonstrated by means of repetitive transcranial magnetic stimulation (5, 58). In the present study the first and second sessions were done on the same day, separated by only 1 h. In Fig. 6 it can be seen that in the placebo group only a very slight dishabituation (i.e. amplitude increment) can be observed at the beginning of the second session. The impression is of the second session almost smoothly continuing the first one. In contrast, there was a clear dishabituation of the CNV amplitude at the beginning of the second session in the experimental group, which can plausibly be attributed to the effect of the drug. This dishabituation trend also indicates that taking rizatriptan resulted in an increase of cortical preparatory excitation as reflected in the amplitude of the CNV.
Three important conclusions can be drawn for the healthy persons who can serve as controls in migraine research. First, habituation in healthy is not a general phenomenon. In the present study it was shown that a certain amount of cortical arousal is needed in order to achieve a consistent pattern of habituation. This is in line with the ceiling theory of migraine, which states that a certain amount of cortical arousal (the ceiling) must be reached in the healthy before habituation occurs. Second, if the cortical serotonin level is lowered in the healthy, then their CNV amplitude increases, which confirms the assumption of the ceiling theory regarding the link between serotonin and cortical arousal. Third, a flat slope does not generally indicate lack of habituation. Our second session, placebo group, demonstrates that sometimes cortical responses cannot habituate (i.e. the activity level cannot go down) just because the cortex is already at a very low level of activity. The lack of habituation cannot, therefore, be unequivocally conceived of as a sign of cortical hyperactivation.
