Abstract
In this work we have developed and characterized primary cultures of neonatal rat trigeminal ganglia neurones; calcitonin-gene-related-peptide (CGRP) released from cells was taken as a marker of neuronal function. A significant and consistent increase in CGRP secretion was elicited by non-specific (56 mM KCl or veratridine) or specific (capsaicin) depolarizing stimuli. This paradigm was subsequently used to investigate the effects of nociceptin, an opioid-like peptide involved in central and peripheral control of nociception. We found that the nociceptin analogue nociceptin (1–13)NH2 (NOC) did not affect baseline CGRP release, but it reduced in a concentration-dependent manner CGRP release induced by all tested stimuli. NOC-induced reduction was statistically significant from 0.01 nM onward and achieved maximal effects at 10 nM. Such effects of NOC were seemingly mediated by the activation of specific ORL1 receptors, as a well-known nociceptin antagonist, N(Phe1)nociceptin (1–13)NH2, was able to completely revert NOC inhibition of capsaicin-stimulated CGRP release.
Introduction
The heptadecapeptide nociceptin, also referred to as orphanin FQ (N/OFQ), has been identified as the endogenous ligand for the opioid receptor-like receptor ORL1 (OP4) (1, 2). Although structurally related to the opioid peptide dynorphin A, N/OFQ does not bind to µ-, δ- or κ- opioidergic receptors, nor are its effects antagonized by naloxone (3, 4). However, similarly to opioids, N/OFQ activates inwardly rectifying K+ channels and inhibits both voltage-gated (N/P type) Ca++ channels and adenylate cyclase activity (3), and, like dynorphin A, it exerts opioid modulating actions (5). Supraspinal injection of N/OFQ decreases morphine-induced analgesia (anti-opioid activity), and spinal administration induces analgesia or potentiates the analgesic effects of morphine (pro-opioid activity) (6–8), suggesting an involvement of this peptide-receptor system in the regulation of nociception. Indeed, in the central nervous system (CNS), N/OFQ has been found to exert both anti-analgesic, and analgesic and/or hyperalgesic effects, depending on the experimental models and route of administration (9–11). In the peripheral nervous system (PNS), N/OFQ mediates mainly antinociceptive effects similarly to classic opioids but via activation of its specific ORL1 receptor (12, 13).
The ORL1 receptor is widely expressed throughout the CNS in rodents and humans (14–16), supporting a role of this system in several CNS functions (17), such as the stress response, anxiety, learning and memory processes, and mechanisms of reinforcement and reward during drug addiction, besides pain control (18). An involvement of N/OFQ has been also hypothesized in the pathogenesis of migraine, in which the activation of the trigeminal afferents causes vasodilatation of dural vessels leading to neurogenic inflammation and pain. N/OFQ was shown to inhibit neurogenic dural vasodilatation in the rat (19); however, the exact mechanism of action underlying such an effect was not determined in this study. ORL1 mRNA expression and N/OFQ immunoreactivity have been detected in rat and human trigeminal ganglia, which indicates a possible role of N/OFQ in the control of trigeminal nociception (20, 21). Interestingly, low levels of N/OFQ were recently demonstrated in migraine patients, suggesting that deregulation of the opioid-like system may be associated with increased susceptibility to migraine (22). During migraine attacks, trigeminal ganglia innervation of meningeal vessels mediates the release of vasoactive peptides such as calcitonin gene-related peptide (CGRP) and substance P, followed by dural vasodilatation and subsequent activation of nociceptive trigeminal sensory fibres (23, 24). In fact, CGRP was found to be elevated in jugular blood samples of patients during migraine attacks (25), and effective antimigraine drugs, such as 5HT1B/D agonists (triptans), can reduce CGRP secretion from trigeminal nerve endings (26).
N/OFQ attenuates peripheral neuropeptide release in a variety of tissues, including the bronchi (27), vas deferent and skin (28, 29), as well as from peripheral sensory nerve endings in in vivo models of neuroinflammation (30, 31). Therefore, we hypothesize that N/OFQ can regulate trigeminal nociception via the modulation of neuropeptide release, particularly reducing CGRP secretion. To test this hypothesis, we used primary cultures of rat trigeminal ganglia neurones. This is a well-established tool to investigate the patho-physiological mechanisms of migraine, as well as to test putative antimigraine drugs. We have investigated the effects of a full ORL1 agonist, nociceptin (1–13)NH2 (NOC) (32), on CGRP-release from trigeminal neurones, under basal conditions and after stimulation by non-specific or specific depolarizating agents. In these experimental conditions, we have found that NOC reduced CGRP-stimulated release without affecting basal secretion. The inhibitory effects of NOC were mediated by the ORL1 receptor activation, and were almost completely reversed by the selective ORL1 antagonist, N(Phe1)nociceptin (1–13)NH2 (NPheNOC) (33). Altogether, evidence presented in this manuscript indicates that trigeminal nociception may be reduced by the activation of the N/OFQ-ORL1 system, and the latter may be thought of as a novel candidate target for antimigraine drugs.
Methods
Drugs
Veratridine and capsaicin were purchased from Sigma (Sigma Chemicals Co., St. Louis, MO, USA), and dissolved in 100% ethanol at 10 m
Trigeminal neurone cultures
Trigeminal neuronal cultures were prepared from 6- to 7-day-old Wistar rats as previously described (34), with some modifications introduced in our laboratory. The use of animals for this experimental work has been approved by the Italian Ministry of Health (licensed authorization to P. Navarra). In brief, animals were decapitated and, after removal of the brain, basal skulls were exposed and trigeminal ganglia from both sides were aseptically removed. Tissues were collected in a Petri dish containing 3–5 ml of ice-cold phosphate buffer saline without Ca++ and Mg++ (PBS w/o; Sigma), supplemented with antibiotics (100 IU/ml penicillin and 100 μg/ml streptomycin; Sigma) and
Because sensory ganglia consist of neuronal cells and satellite cells, the latter being part of a sheath that surrounds neurones (35), satellite cells may represent an important contaminant of neuronal cultures. These cells share several properties of glial cells (35), and glia tend to adhere to the bottom of uncoated culture flasks (36). Thus, we introduced into the standard procedure of isolation of trigeminal neurones (34) a preplating incubation step, and let non-neuronal cells adhere to the uncoated tissue culture flasks for a few hours. At the end of the preplating incubation, neurones were harvested from the flask and plated in 24-well tissue culture plates, previously coated with poli-D-lysine (40 µg/ml; MW 70000–130000, Sigma) at a density of 100 000 cells/well. The incubation volume was 1 ml/well of complete culture medium (see above), enriched with 50 ng/ml of 2.5 S murine Nerve Growth Factor (Alexis-Vinci Biochem, Vinci, Florence, Italy). The culture medium was changed within 24 h from seeding and 10 µ

Rat primary cultures of trigeminal neurones after 7 days in vitro. (a) Morphology of trigeminal neurones after maturation in vitro. Image taken in phase contrast microscopy (20×). (b) Cultures were stained with the antibody against the phosporilated neurofilament 200 (pNF200), a specific marker of sensitivity neurones. Scale bars are 50 µm in both panels.
CGRP release studies
For these studies, at the beginning of experiments, the incubation medium was replaced and cells were incubated with 300 μL of complete Ham's F12 medium (containing 10% FCS, 1 μM bacitracin, 10 μg/ml aprotinin, without NGF) at 37°C. In these conditions we assessed a basal release of CGRP, which could be increased by incubating the cultures with medium containing different stimulating agents. In the experiments in which KCl was used as depolarizing agent, 56 m
In the experiments with the selective ORL1 antagonist, a 5-min pre-incubation in the presence of this compound was performed. After pre-incubation, the medium was replaced with fresh release-medium containing testing drugs. At the end of the experiments, media were collected and stored at −35°C until the assays were performed.
CGRP radioimmunoassay (RIA)
CGRP release was measured by a radioimmunoassay technique validated in our laboratory. We generated the antiserum used in this assay by immunization of New Zealand white rabbits with synthetic human α-CGRP (hα-CGRP) coupled to BSA with carbodiimide as previously described (38). The cross-reactivity of the anti-hα-CGRP serum with several other peptides and CGRP analogues was tested by adding up to 1 µg/tube of hα-CGRP, hβ-CGRP, rat α-CGRP (rα-CGRP), rβ-CGRP, rα-CGRP (8–37), rα-CGRP (29–37), h-amylin, r-amylin, cholecystokinin-8 (CCK-8) sulphated, neurokinin A (NKA), neurokinin B (NKB), physalaemin, somatostatin, substance P (SP), thyrocalcitonin, and vasoactive intestinal polypeptide (VIP). Iodinated hα-CGRP was purchased from Amersham (Amersham, UK). The RIA was performed in a buffer containing 10 m
On the whole, these findings indicate that: (i) the polyclonal antibody is mainly directed towards the middle region of the peptide; and (ii) the presence of the amino-terminal sequence reduces the affinity of the antibody towards the peptide. The mean IC50 of the standard curve was 50.8 ± 2.0 pg/tube (n = 10), and non-specific binding of the labelled ligand was not different from the background of the γ-counter. The intra-assay (n = 6) and inter-assay (n = 10) coefficients of variation were, respectively, 1.27% and ±0.95% at the lowest (1.95 pg/tube) and 13.7% and ±12.6% at the highest (1000 pg/tube) levels of standard. The lowest concentration that could be measured with 95% confidence (i.e. 2
Immunocytochemistry
For immunocytochemical (ICC) characterization of neuronal cultures, cells were plated on glass coverslips coated with 1 mg/ml poli-D-lysine, at a density of 5000 cells/well, and kept in culture for 7 days. At this time, cells were washed three times with PBS containing Ca++ and Mg++ (PBS-w) and fixed in 4% paraformaldehyde for 20 min. After fixation, cells were washed twice with PBS-w and blocked with 0.5% BSA in PBS-w for 30 min. Primary antibodies were diluted in 0.1% BSA and 0.2% Triton X100 in PBS-w (anti-pNF200 1:400) and cells incubated overnight at 4°C. Secondary antibodies were donkey anti-mouse conjugated with Texas-red. Secondary antibodies were incubated for 1 h at 37°C and diluted 1:200 in PBS-w in 0.1% BSA. Coverslips were mounted using Vectashield mounting media. Images were obtained on a Nikon Eclipse TE300 inverted fluorescence microscope equipped with a Cool SNAP professional digital camera and LUCIA-G/F imaging software.
Statistical analysis
All data are presented as mean ±
Results
Primary rat trigeminal neurones release CGRP
In preliminary experiments, we characterized the release of CGRP-LI from primary cultures of rat trigeminal ganglia neurones. Cells were cultured for 7 days in vitro in the presence of 50 ng/ml NFG (as described in Methods), to reach complete maturation (Figure 1a). At this time, neurones highly expressed the specific marker for sensory neurones (39), pNF200 (Figure 1b). In these conditions, it was possible to detect CGRP-LI immunoreactivity in the incubation media. The basal secretion of CGRP ranged from 53.11 ± 3.29 pg/well after 10 min incubation to 76.26 ± 9.45 pg/well after 20 min incubation (data expressed as means ±

Basal and 56 m

Effect of veratridine on CGRP-LI release. Data are expressed as pg of CGRP-LI/well/10 min, means ±

Effect of capsaicin on CGRP-LI release. Data are expressed as pg of CGRP-LI/well/10 min, means ±
Nociceptin reduces CGRP-LI release
After the initial characterization, we tested the effects of N/OFQ on CGRP release, using the selective ORL1 agonist NOC. In resting neurones, NOC, given in the range 0.01 n

Effect of NOC on KCl-evoked CGRP-LI release. Data are expressed as pg of CGRP-LI/well/20 min, means ±
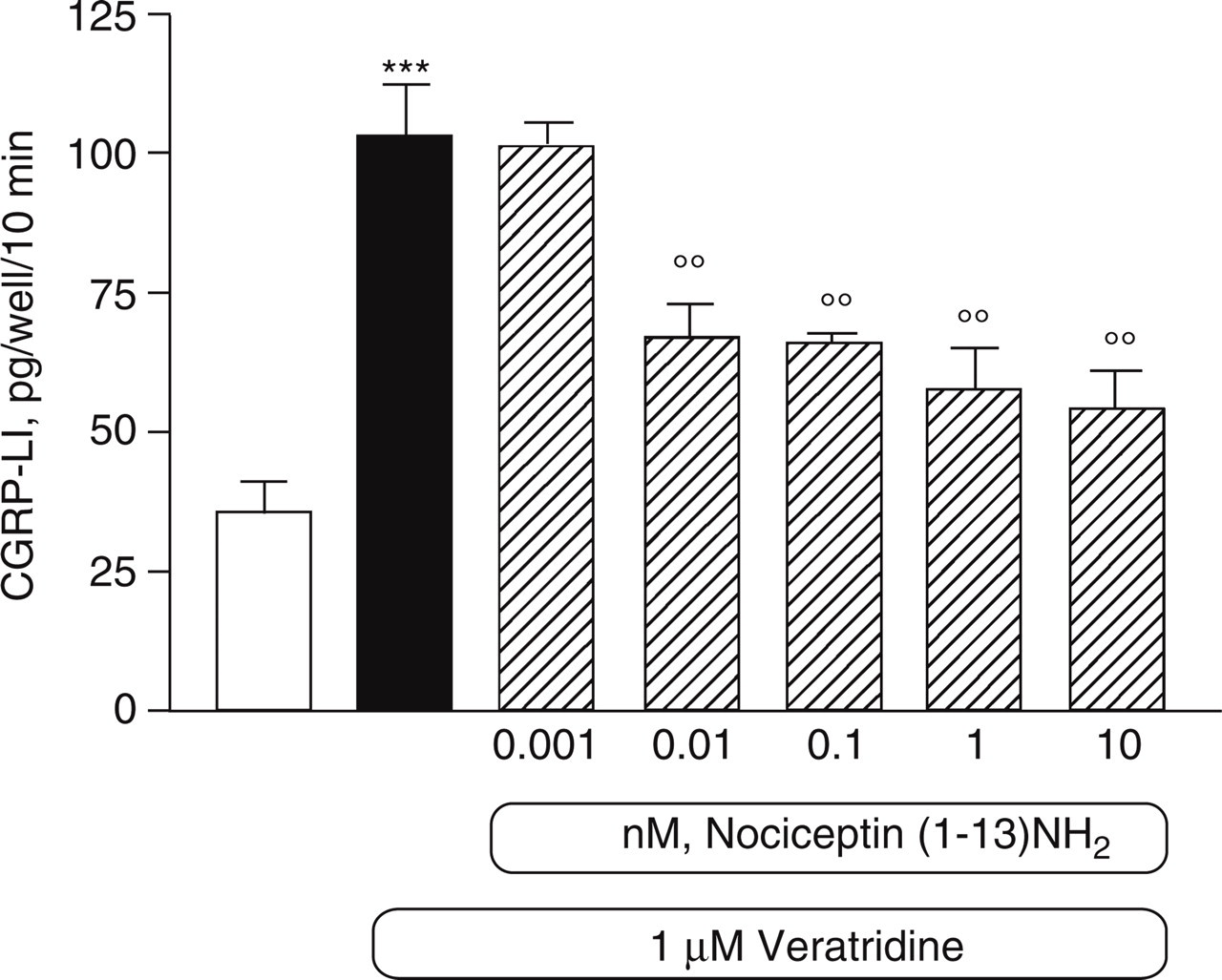
Effect of NOC on veratridine-evoked CGRP-LI release. Data are expressed as pg of CGRP-LI/well/10 min, means ±

Effect of NOC on capsaicin-evoked CGRP-LI release. Results are expressed as pg of CGRP-LI/well/10 min. Data are the means ±

Effects of the NOC selective antagonist NPheNOC. (a) NPheNOC does not modify capsaicin-stimulated CGRP-LI release. (b) NPheNOC counteracts NOC inhibition of capsaicin-stimulated CRRP-LI release; representative of two independent experiments with similar findings. Results are expressed as pg of CGRP-LI/well/10 min. Data are the means ±
Discussion
In the present study, primary cultures of rat trigeminal ganglia neurones were used to investigate the involvement of the N/OFQ-ORL1 system in the regulation trigeminal nociception. In preliminary experiments, we have first characterized the in vitro model. As a specific marker of neuronal functions we assessed in the incubation medium the release of CGRP. Interestingly, this is a critical vasoactive peptide, which mediates in vivo neurogenic inflammation; therefore the assessment of CGRP in vitro also represents a useful tool to investigate the pain mechanisms involving trigeminal ganglia. In our cultures, we were able to detect a baseline CGRP secretion, which was significantly and consistently increased by different types of stimuli, namely 56 m
Experiments with the N/OFQ antagonist indicate that NOC inhibitory effects are indeed mediated by the activation of its specific ORL1 receptors. NPheNOC is a selective and competitive antagonist to ORL1 (33). NPheNOC binds to ORL1 and prevents the effects of N/OFQ in the CNS and PNS. In our model, NPheNOC did not have any effect per se, but it was able to reverse NOC inhibition of capsaicin-evoked CGRP release when given at 10 µ
More recent evidence also suggests a possible role of the N/OFQ-ORL1 system in the modulation of trigeminal nociception. N/OFQ and ORL1 were found expressed in human trigeminal ganglia, colocalized with substance P and CGRP (21); in small trigeminal ganglia neurones from the mouse, N/OFQ was able to inhibit Ca++ channel currents (45). Finally, CGRP is the major constituent of sensory neurones and is considered a marker of activation of the trigeminal-vascular system (25). Bartsch and colleagues have demonstrated that N/OFQ is able to reduce neurogenic dural vasodilatation in the rat (19), suggesting that this effect could be mediated by the inhibition of CGRP release from trigeminal fibres innervating meningeal vessels. Our findings represent a strong in vitro correlation to the above hypothesis and shed more light on the mechanisms of N/OFQ antinociceptive activity in peripheral pain. In fact, most of the previous findings involving N/OFQ (30, 31) were obtained in experimental models where the observation was limited to nerve endings, thereby missing nociceptin effects on the neuronal cell body. In contrast, here for the first time we were able to investigate the effect of N/OFQ on trigeminal neurones taken as a whole; our findings indicate that the N/OFQ-ORL1 system is definitely involved in trigeminal pain transmission and may represent a new target for antimigraine therapeutic agents.
Acknowledgements
This work was supported in part by Grünenthal–Formenti.
