Abstract
The aim of this study was to evaluate in a double-blind, randomized, placebo-controlled study the safety and efficacy of venlafaxine extended release (XR) in the prophylactic treatment of out-patients with tension-type headache (TTH) and no current depression or anxiety disorders. Sixty neurology and headache clinic out-patients meeting the International Headache Society diagnostic criteria for TTH were treated with venlafaxine XR (150 mg/day, n = 34) or placebo (n = 26) for 12 weeks. The primary efficacy variable was the decline in number of days with headache. At end-point, the venlafaxine XR group had a significantly greater decrease in the number of days with headache compared with placebo (P = 0.05). Differences with regard to secondary efficacy variables where not significant. The number needed to treat for responders (≥50% reduction in days with headache) was 3.48. Six patients in the venlafaxine XR group interrupted therapy due to adverse events, while no patients in the placebo group did so for the same reason. The number needed to harm was 5.58. This study provides preliminary evidence for the efficacy and safety of venlafaxine XR 150 mg/day in reducing the number of days with TTH.
Introduction
Tension-type headache (TTH) is a common and disabling condition. Based on monthly frequency it is classified as either episodic or chronic (1). A telephone survey conducted in the USA with 13 345 subjects revealed 1-year prevalence rates of 38.3% for episodic TTH and 2.2% for chronic TTH (2). Rates of >75% for episodic TTH have also been reported (3, 4). Both headache types are more common in women than in men, peak in occurrence between the ages of 30–39 and result in significant impairment in work, familial and social activities (2). Patients experiencing chronic headaches lose an average of 3.5 h per week of productive work time (5).
Treatment for TTH takes the form of acute symptomatic treatment or pharmacological prophylaxis. Over-the-counter analgesics are commonly used to treat acute symptoms. Paracetamol, aspirin and the non-steroidal anti-inflammatory drugs (NSAIDs) ibuprofen and naproxen are the most common (6, 7). Often, doses of 600–1000 mg of aspirin, 1000 mg of paracetamol, 375 mg of naproxen or 400 mg of ibuprofen are sufficient to provide rapid relief of TTH symptoms (8). For patients experiencing frequent attacks, the safety and tolerability of the long-term use of these medications may impede their daily administration. Analgesic-induced headaches (4, 9, 10) or other side-effects, such as gastric distress (11, 12), have been linked to the long-term use of these agents.
The tricyclic antidepressant (TCA) amitriptyline has been widely studied as a prophylactic treatment for TTH
The side-effects associated with the TCAs include weight gain, sedation, orthostatic hypotension, dizziness, blurred vision, dry mouth and constipation. In addition to troublesome side-effects, serious safety concerns are associated with TCA treatment (i.e. cardiotoxicity, death with overdose and a potential for dangerous drug–drug interactions). Amitriptyline treatment is associated with a number of side-effects (14–20).
Apart from amitriptyline, there is little evidence of the efficacy of other pharmaceutical agents in treating TTH. Thus, drugs with better safety profiles are needed to improve daily prophylactic treatment. Recently, mirtazapine, a serotonergic/noradrenergic antidepressant, has proved effective in TTH (21). This finding further supports the hypothesis that serotonin/norepinephrine play an important role in TTH.
The serotonin and norepinephrine reuptake inhibitor venlafaxine extended release (XR) is currently approved by the Greek National Organization of Medicines for the treatment of all types of depression and generalized anxiety disorder (22). Its efficacy and safety in the treatment of the somatic and emotional symptoms associated with major depressive disorder and generalized anxiety disorder (23, 24), as well as a variety of chronic pain syndromes (25–29), have also been studied. Additionally, studies suggest venlafaxine XR has an effect in in the prophylactic treatment of migraine and TTH (30–32). The mode of action of venlafaxine XR is believed to be similar to that of amitriptyline, but its more selective inhibition of 5-HT and NE gives it a more favourable tolerability and safety profile (33–35).
The aim of the present randomized, placebo-controlled study was to investigate the efficacy and safety of venlafaxine XR in the prophylactic treatment of TTH.
Methods
This prospective, double-blind, placebo-controlled study was conducted with headache and neurology clinic out-patients from six centres, aged ≥18 years, meeting International Headache Society (IHS) criteria for all subtypes of TTH, experiencing these headaches for >5 days/month, which result in severe disability: >21 in the Migraine Disability Assessment (MIDAS) questionnaire. All patients meeting these criteria entered a screening period of 4 weeks. All enrolled patients continued to meet the enrolment criteria at the end of the screening period (baseline evaluation).
Patients
Patients with a history of hypersensitivity to venlafaxine or use of venlafaxine in the month prior to the start of the double-blind treatment period were excluded from participating in the trial. Other exclusion criteria included: uncontrolled congestive heart failure or myocardial infarction within 6 months of the screening visit, a history or the presence of clinically significant hepatic (total bilirubin >2 mg) or renal disease (serum creatinine >2.5 mg/dl), a history or presence of seizure disorder other than a single childhood febrile seizure, an organic mental disorder, total score ≥12 on the Hamilton Anxiety Rating Scale, total score ≥12 on the Hamilton Depression Ratings Scale, meeting the criteria for any Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV) psychotic disorder, presence of any stomatognathic dysfunction, meeting IHS criteria for headaches of categories 1, 3, 4 and 5–12, pregnancy, currently breast-feeding, being of childbearing potential without a negative pregnancy test result, use of any investigational drug, antipsychotic drug or fluoxetine within 30 days, recent electroconvulsive therapy or use of any monoamine oxidase inhibitor or antidepressant drug within 15 days, history of DSM-IV drug or alcohol dependence within 2 years, and having any language or other problems preventing adequate cooperation in the assessment procedures. Specifically, patients with any history of migraine attacks were excluded from the study.
The only NSAIDs permitted during the treatment phase were acetylsalicylic acid and paracetamol at daily doses not exceeding 1 g. Any investigational drugs, psychotropic drugs or substances (including other antidepressants, antipsychotics, anxiolytics and sedative-hypnotics), and non-psychopharmacological drugs with psychotropic effects were prohibited. Drugs thought to inhibit or induce hepatic drug-metabolizing enzymes were prohibited. The introduction or change in intensity of non-pharmacological therapy was also prohibited. Other treatments were allowed provided they were recorded on the case report form, including drug name (trade or generic) or procedure name with start and stop dates.
Study design
Double-blind treatment lasted 12 weeks and was preceded by a 4-week screening and baseline evaluation period. At baseline, patients were randomly assigned to receive either venlafaxine XR or placebo. Study medications were taken orally once daily after morning meals. Venlafaxine XR and placebo were supplied as identically appearing capsules, with each venlafaxine XR capsule containing a 75-mg dose. The randomization and blinding were done by the drug company (Wyeth, Collegeville, PA, USA), whereas the drug was dispensed by the hospital pharmacies. Each patient was supplied with sufficient amounts of double-blind medication until the next evaluation visit. During the first week of treatment, patients took 1–75 mg venlafaxine XR capsule or a placebo capsule. Beginning with day 8, the number of capsules increased to 2–75 mg of venlafaxine XR (150 mg) or placebo. The 150-mg dose was chosen based on prior experience demonstrating the positive effect of this dose in treating TTH (30, 31). At the end of the active treatment phase, all patients underwent a 1-week taper period where the daily dose was reduced to one capsule. Patients were assigned to their treatment groups using balanced randomization with a random numbers table. A copy was sent to investigators in individually sealed envelopes. In case of an emergency, an envelope could be opened to identify the study medication for a particular patient. The calculation of the number of patients needed in each group was performed by assuming that placebo improvement was expected to be in the range of 30%, while venlafaxine improvement was approximated at 60%. In order for such a difference to be deemed statistically significant at the 0.05 level with 0.80 power, 44 patients were required for each arm. In order to allow for dropping out, 98 patients were initially recruited for the study. However, more than expected (38 patients) had to be excluded because of failure to complete the baseline evaluation (Fig. 1).
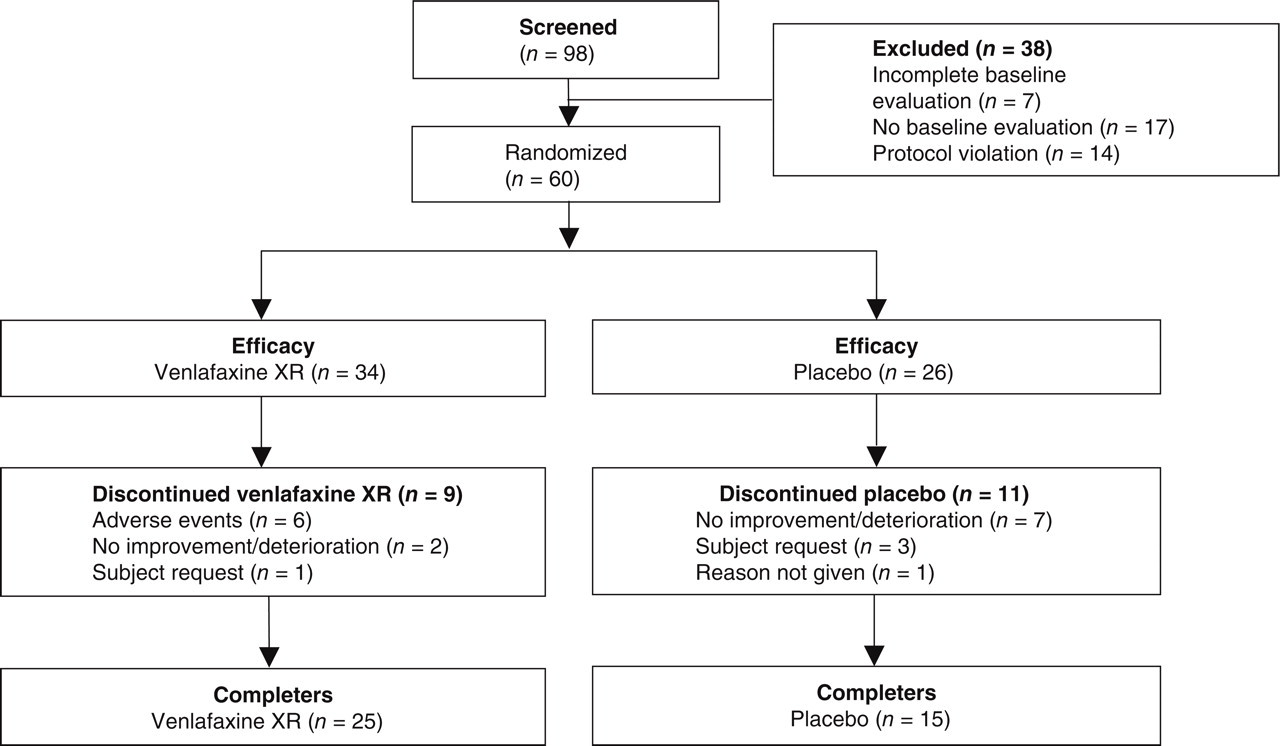
Study flow chart.
Evaluation criteria
Patients were provided diaries to keep track of their daily headache occurrence and intensity. On day 28 of the screening period, each patient's diary was evaluated to determine baseline values. Patients were evaluated at day 1 (baseline), 29, 57 and 85. Subjects maintained a record of each day with headache, the number of hours with headache and headache intensity.
The primary efficacy variable was the number of days with headache. The secondary efficacy variables were total hours with headache, daily headache intensity index, clinical global impressions (CGI) score and response to treatment, which was defined as a ≥50% reduction separately for number of days with headache, total hours and headache intensity. The daily headache intensity index (HII) was calculated by multiplying the hours with headache by the intensity of the headache. A 10-point visual analogue scale, for which 10 was the highest, was used to assess intensity. The maximum possible daily HII was 240 [24 h × 10 (highest intensity)]. The HII for each 28-day period was calculated by adding the HII per day and expressed as a daily average by dividing by 28. When a subject provided data for only part of a period, these data were adjusted appropriately. For example, if a subject kept their diary for only 7 days before dropping out, and recorded headache on four of those days, then the total days with headache for the 28-day period would be extrapolated as 4 × (28/7) = 16 days.
Statistical analysis
In the event of a patient's withdrawal from the study, statistical analysis was performed using the last-observation-carried-forward (LOCF) method. Quantitative measurements were compared between groups of patients by t-tests and within groups by paired t-tests after verifying that the quantity's distribution met the necessary assumptions; otherwise Mann–Whitney and Wilcoxon tests were used, respectively. Qualitative variables were compared between groups by the χ2 test, using Yates' correction in tables with 1 degree of freedom. Fisher's exact test was used when expected frequencies were low. The difference between the two groups in time to drop-out were evaluated by the log rank test. The percentage changes from baseline were compared between the two groups using the Mann–Whitney test. All tests were two-tailed.
Results
The study population included 60 subjects. Thirty-four (56.7%) patients received venlafaxine XR and 26 (43.3%) received placebo. See Fig. 1 for a population flow chart. The majority of participating subjects were female (venlafaxine XR 85.3%, placebo 76.9%, all subjects 81.7%). The mean and median ages were approximately 40 years for each group. The gap between the oldest subject (age 75 years) and the second oldest (age 66 years) was quite large. The mean age of the male subjects (34.6 ± 13.1 years) was on average slightly lower than that of the female subjects (42.3 ± 14.5 years), but the difference was not statistically significant (P = 0.12, t-test). The baseline and demographic characteristics are listed in Table 1. Although at baseline there were some differences between the two groups (higher MIDAS score combined with more hours with headache and higher headache intensity index in the placebo group— Tables 1 and 2—none of these reached a statistically significant level. The mean time since diagnosis of TTH was 6.0 years (median 2 years) in the venlafaxine XR group and 6.6 years (median 3 years) in the placebo group. The considerable variation between these two measures of central tendency was attributable to the broad distribution of scores in the two groups: 0–30 and 0–32 years, respectively. Fifteen subjects [10 (29.4%) in the venlafaxine XR group and five (19.2%) in the placebo group] had previously received antidepressant treatment. The median duration of therapy was 8 and 7 months, respectively.
Baseline demographic characteristics

XR, Extended release;
Time of diagnosis unrecorded for one subject.
Efficacy data
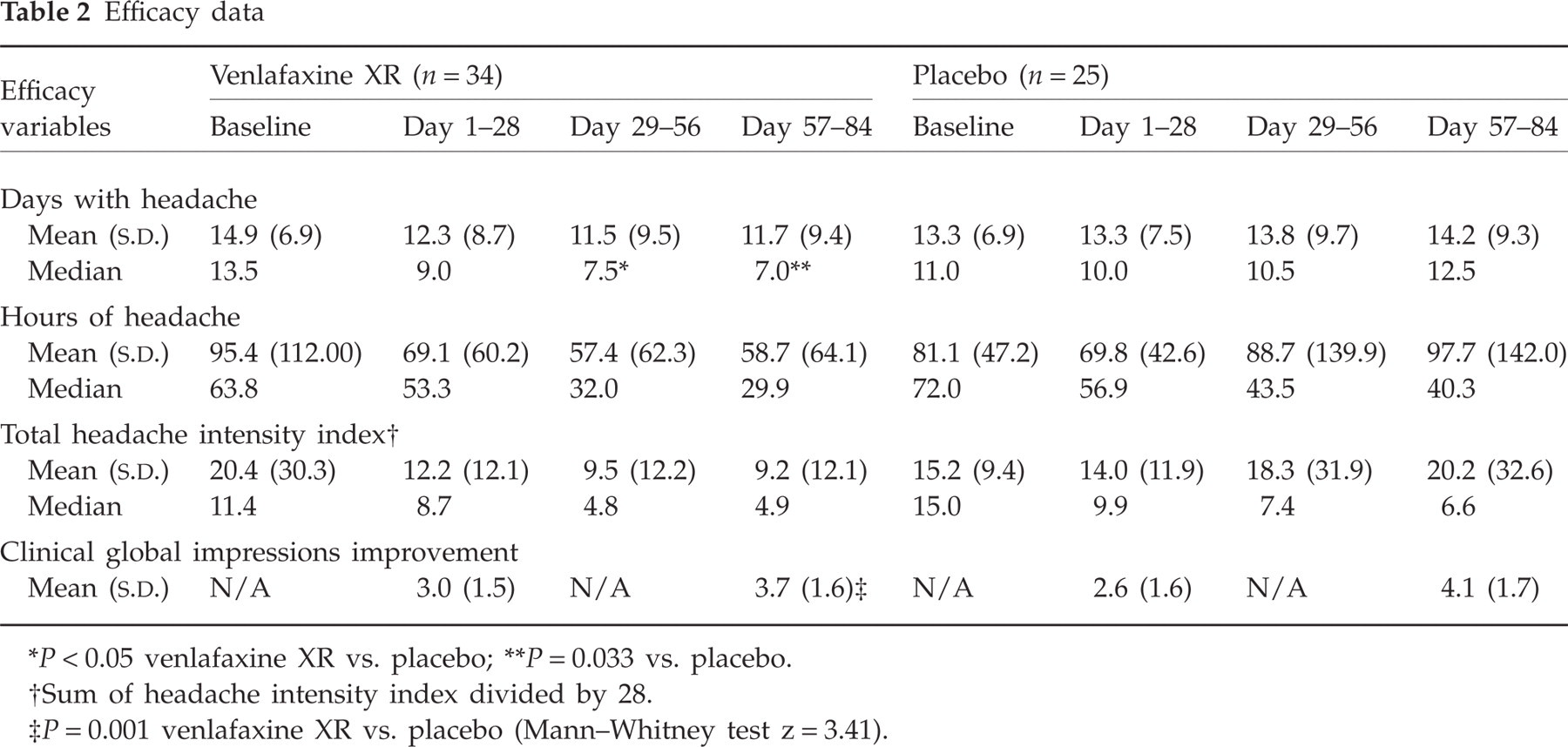
P < 0.05 venlafaxine XR vs. placebo;
P = 0.033 vs. placebo.
Sum of headache intensity index divided by 28.
P = 0.001 venlafaxine XR vs. placebo (Mann–Whitney test z = 3.41).
No patient met the criteria for analgesic-related headache. Six patients in the venlafaxine XR group (17.6%) and nine patients receiving placebo (34.6%) received approved analgesics (≤1 g) during the double-blind treatment period. The total duration of simple analgesic intake during the study period varied between 1 and 3 days. Use of analgesics as acute prophylactic therapy was prohibited.
Diary completion rates for each study period are presented in Table 3. Out of the 34 patients in the venlafaxine group, 25 completed all diaries (per protocol) and nine were evaluated as per LOCF. The respective figures for the placebo group were 15 (per protocol) and 11 (LOCF) (Table 3).
Diary completion rates for each 28-day period

Efficacy
Three sets of evaluations were made: (i) comparisons between the two groups, (ii) percentage decline from baseline in each group and comparison of these percentages between groups and (iii) percentage responders in the three variables (days with headache, hours with headache, index) in each group. The definition of a responder was ≥50% reduction in the rating of a specific variable, compared with baseline.
Comparison of the means/medians between the two groups
The mean number of days with headache during each 28-day observation period declined in the venlafaxine XR group, but not in the placebo group. The median number of days with headache decreased from baseline in the venlafaxine XR group (13.5) but not the placebo group (11.0) in period 2 (7.5 vs. 10.5, respectively; P = 0.05, Mann–Whitney test) and period 3 (7.0 vs. 12.5, respectively; P = 0.033, Mann–Whitney test). The difference between treatment groups in period 1 was not statistically significant (Mann–Whitney test). These data are presented in Table 2 and Fig. 2. Both treatment groups showed a decline in hours with headache and HII and the differences between the two groups were not statistically significant during any period.

Days with tension-type headache (TTH), last observation carried forward. ▪, Venlafaxine XR (n = 34); □, placebo (n = 26). ∗P ≤ 0.05 venlafaxine XR vs. placebo; ∗∗P = 0.033 venlafaxine vs. placebo.
Percentage decline from baseline
At the end of period 3 the median percentage change from baseline in the number days with headache was a 44.8% decline in the venlafaxine XR group and a 15.7% increase in the placebo group (P = 0.023, Mann–Whitney test). The total hours of headache and HII showed similar trends in their results, but the differences between the two groups were not statistically significant (Table 4). More particularly, if the index is calculated by dividing by 28, a higher reduction is observed in the venlafaxine XR group compared with placebo (Fig. 3). However, if the HII is calculated by dividing by the number of days with headache during each period, the percentage decline of the index in the placebo group becomes marginally higher (48.7% vs. 44.3%) (Table 4). This also corresponds with the higher percentage of index responders in the placebo group (Fig. 4). The difference is explained by the higher number of days with headache in the placebo group. None of these differences was statistically significant.

Percentage decline from baseline in headache intensity index (calculated as the sum of the daily recordings of headache duration × headache intensity and divided by 28) (median values). Differences not statistically significant at any point. ▪, Venlafaxine XR (n = 34); □, placebo (n = 26).

Responders (≥50% reduction in above criteria), last observation carried forward (number of responders over total). †P = 0.037 venlafaxine XR vs. placebo. ▪, Venlafaxine XR (n = 34); □, placebo (n = 26).
Percentage reductions from baseline in headache frequency, duration and intensity (median values)

Divided by 28 (duration in days of each study period).
Divided by the number of days with headache during each study period.
The fact that the difference between the active treatment and the placebo groups was much smaller with regard to HII, when dividing by the number of days with headache, than with regard to days with headache, may indicate that the drug mostly eliminated the days with mild headache.
Responders
When examining the number of subjects who met the criteria of responder at the final evaluation, the difference between the groups was significantly different for the days with headache (44% in the venlafaxine group vs. 15% in the placebo group; P < 0.05, Mann–Whitney test), but not for hours with headache or HII. These results are provided in Fig. 4. The number needed to treat for days with headache responders was 3.48.
CGI
Improvement scores were recorded on days 29 and 85 for all but three subjects who dropped out early. The difference between groups at day 29 did not reach the level of statistical significance (P = 0.080, Mann–Whitney test), but the difference at day 85 was found to be significant (P = 0.001, z = 3.41, Mann–Whitney test). All primary and secondary efficacy data are presented in Table 2.
Safety
Forty (66.7%) subjects completed the study to 84 days. Nine (26.5%) of the venlafaxine XR-treated patients and 11 (42.3%) of the placebo group withdrew from the study. This difference was not statistically significant (P = 0.31, χ2 = 1.03). The median time to withdrawal was 24 days (range 6–44) in the venlafaxine XR group and 30 days (range 16–57) in the placebo group (P = 0.27, log rank test). The reasons for withdrawal varied between the groups and are presented in Table 5. Six of the nine subjects withdrawing from venlafaxine XR treatment did so because of adverse events, whereas none of the placebo subjects discontinued treatment for this reason (P = 0.004, Fisher's exact test). Withdrawal from the study was at the subject's request or because of failure to improve in 10 of the 11 subjects on placebo but only three of the nine subjects receiving venlafaxine XR (P = 0.027, Fisher's exact test). Adverse events were recorded in 10 venlafaxine XR and three placebo-treated patients and are reported in Table 6. The adverse events leading to discontinuation in the venlafaxine XR subjects were gastrointestinal symptoms (i.e. vomiting, gastralgia, nausea) in five patients and loss of libido/absence of orgasm in one female patient. All of these were deemed by physician opinion to be related to therapy. The symptoms of gastralgia, loss of libido and absence of orgasm were regarded as intense, whereas the rest were labelled as moderate. No specific treatment was provided and the symptoms disappeared shortly after the subjects ceased taking the medication. The number needed to harm was 5.58.
Reasons for withdrawal
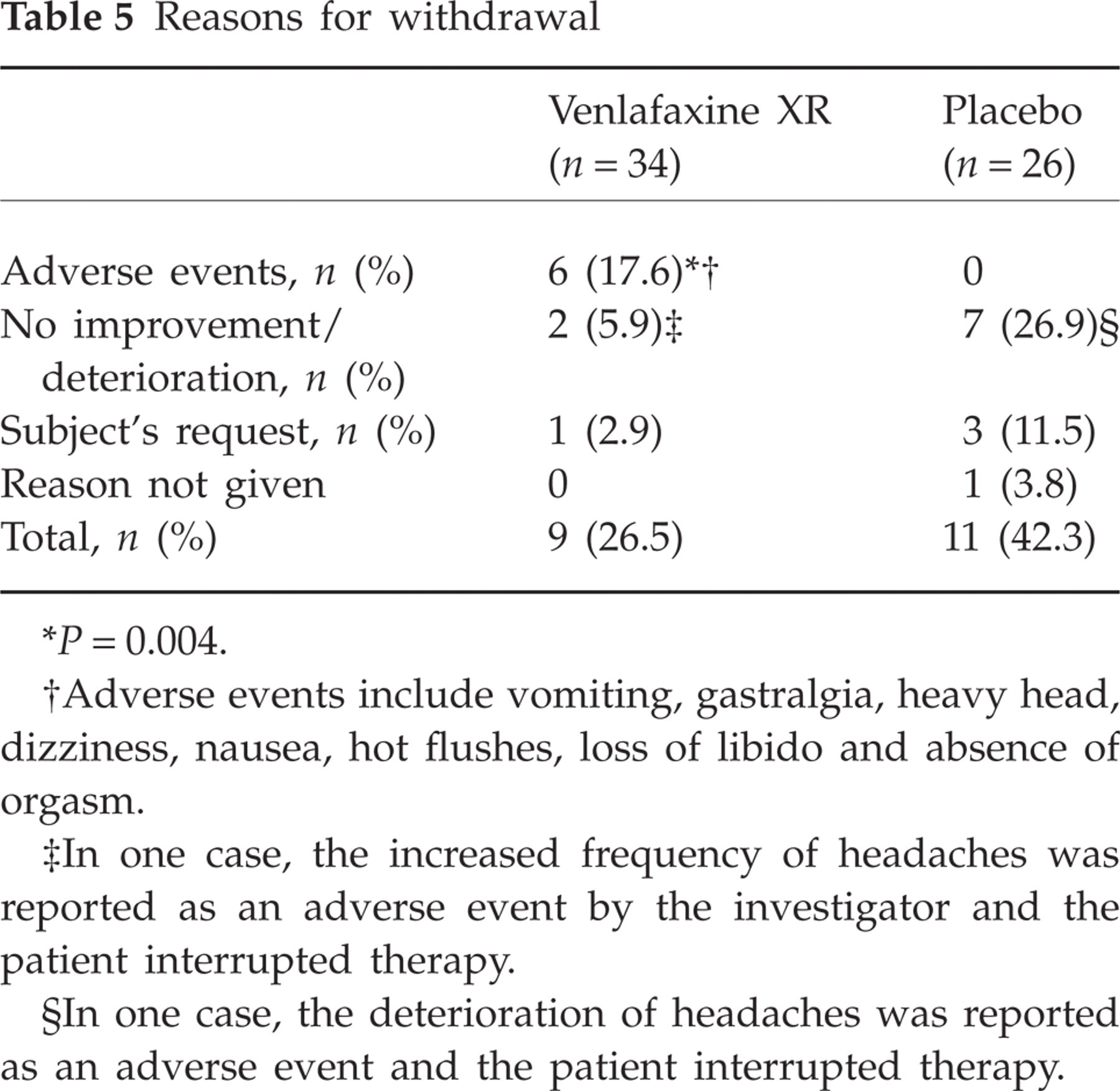
P = 0.004.
Adverse events include vomiting, gastralgia, heavy head, dizziness, nausea, hot flushes, loss of libido and absence of orgasm.
In one case, the increased frequency of headaches was reported as an adverse event by the investigator and the patient interrupted therapy.
In one case, the deterioration of headaches was reported as an adverse event and the patient interrupted therapy.
Adverse events

Some patients in the venlafaxine group had more than one adverse event.
In the final physical examination no significant changes from baseline were observed with regard to body weight. Reductions in systolic blood pressure (BP) (mean −6.3 and −6.7 mmHg) and diastolic BP (mean −2.7 and −2.9 mmHg) in the venlafaxine XR and placebo groups, respectively, were recorded.
Discussion
This randomized, double-blind, placebo-controlled clinical trial meets the majority of the IHS guidelines for drug trials in the treatment of TTH, but there are some limitations worth noting. Most notably, discrepancies exist between the populations of the active comparator and placebo groups. The small number of patients randomized to each group makes detecting clinically meaningful responses difficult. More subjects had previously received treatment with antidepressants in the venlafaxine XR group. Additionally, higher severity and fewer days with headaches in the placebo group at baseline add to the further confounding of results. Patients with headache episodes of <4 h were also allowed in the study, although their number was small. A result of not randomizing in small blocks was the uneven number of evaluable patients in the two groups. Finally, it should be stressed that, despite its limitations, this is the first randomized, double-blind, placebo-controlled trial demonstrating a significant improvement in days with headache with venlafaxine XR compared with placebo in the treatment of TTH.
The results of the present study, taken in conjunction with previous trials showing that venlafaxine XR and amitriptyline have a positive effect on TTH and other chronic pain syndromes, provide support for the role that 5-HT and NE play in treating pain symptoms.
Investigators have theorized that NE terminals are necessary for 5-HT-induced pain suppression to take effect (36), which may help to explain the lack of efficacy of the selective serotonin reuptake inhibitors (SSRIs) in the treatment of pain syndromes (18, 37). In a head-to-head study comparing amitriptyline and citalopram for the prophylactic treatment of chronic TTH, amitriptyline was determined to be more effective than citalopram (19). The level of 5-HT reuptake inhibition is not believed to be an indicator for the analgesic effect of an antidepressant (38). Additionally, in a study conducted by Singh and Misra (39), sertraline treatment did not significantly separate from placebo in the reduction of frequency or intensity of chronic TTH. Furthermore, in a randomized, single-blind study comparing fluoxetine, an SSRI, and desipramine, a selective noradrenaline reuptake inhibitor, in the treatment of chronic TTH, no differences between the two active treatments were found in any of the outcome variables (40). Although the authors attribute these results to the indirect effect that depressive symptoms have on TTH, these results could just as easily be interpreted as supporting the synergistic effect that 5-HT and NE have in treating TTH. In the present study, depressed and anxious patients were excluded during the screening phase, thereby removing this correlative relationship between depressive and TTH symptom improvement.
This point is further supported by the results of a small, double-blind, placebo-controlled trial, comparing mirtazapine, a noradrenergic and specific serotonergic antidepressant, with placebo. In that study mirtazapine proved more effective than placebo in TTH (21).
Until a head-to-head study is conducted between amitriptyline and venlafaxine XR, no direct comparisons of efficacy can be made between these two agents in treating TTH. However, in terms of tolerability and safety, venlafaxine XR may have some advantages. In a randomized, double-blind, crossover study comparing amitriptyline and venlafaxine in the prophylaxis of migraine, both agents were found to be comparably effective but the side-effects associated with amitriptyline were more severe (31).
In conclusion, treatment with venlafaxine XR was generally safe and, compared with placebo, resulted in less days with TTH. Larger trials are needed to demonstrate more fully the efficacy of venlafaxine XR in the prophylaxis of TTH.
Acknowledgement
The authors would like to acknowledge Dennis A. Stancavish MA for his editorial assistance.
