Abstract
A temporary sensitization of central trigeminal neurones in migraine patients during acute attacks has been described in previous studies using the electrically evoked nociceptive blink reflex. The cornea is innervated by small myelinated A-delta and unmyelinated C-fibres only. Stimulation with air puffs activates peripheral nociceptors and allows the investigation of peripheral trigeminal nerve structures. Our objective was to investigate whether corneal reflex examinations with air puff stimulation detect abnormalities in migraineurs during their pain-free interval and if the corneal reflex may be modulated by the administration of an oral triptan. After validation of the nociceptive air puff technique by investigating the corneal reflexes before and after a local anaesthesia of the cornea, we recorded corneal reflexes in 25 migraineurs during their pain-free period and 25 healthy controls before and after the oral administration of 100 mg sumatriptan in a randomized, placebo-controlled, crossover study.
Baseline response areas under the curve (AUCs) and latencies of the R2 components of the corneal reflexes did not show any significant differences between patients and controls. Patients did not show any significant differences regarding their headache and non-headache side. The use of an oral triptan had no significant influence on latencies or AUCs in both patients and controls. Our data suggest that there is no facilitation of the trigeminal system in the headache-free interval among patients with migraine. The stable corneal reflexes after the oral administration of 100 mg sumatriptan suggest that there was no inhibition of the trigeminal system, both in patients during their headache-free period and in healthy controls.
Introduction
The trigeminal system and the processing of its nociceptive input play an important role in the pathophysiology of acute migraine attacks. One appropriate way of studying trigeminal function in humans is the study of the blink reflex (BR) (1). It is a trigeminofacial brain stem reflex leading to activation of the orbicularis oculi muscle after electrical or mechanical stimulation of the supraorbital region. The BR is used in the neurophysiological diagnostic routine to indicate the topographical level of putative brainstem lesions. Moreover, it is a useful tool in the evaluation of trigeminal transmission. The standard blink reflex response consists of an ipsilateral early R1 component and bilateral late R2/R3 components (2, 3).
In studies on trigeminal activity in headache syndromes, the focus should be mainly on nociceptive pathways. Approximately 90% of the R2 reflex response of a standard blink reflex recording depends on non-nociceptive Aβ−fibre input (4). Furthermore, present methods of blink reflex measurements only provide information for central neuronal structures due to the direct electrical depolarization of trigeminal axons (N. supraorbitalis). Peripheral receptors at the afferent nerve endings are bypassed this way.
The corneal reflex (CR) is a naturally occurring protective brain stem reflex and can be evoked by mechanical or electrical stimulation of the cornea causing contraction of the orbicularis oculi muscle similar to the blink reflex response. In contrast to the BR, the CR has no early ipsilateral R1, but only a late bilateral R2 response (5, 6). In studies on nociception, the corneal reflex has the possible advantage that the cornea is solely innervated by small myelinated Aδ and unmyelinated C-fibres (7, 8) but not Aβ-fibres. The nociceptive quality of the CR is defined by its above outlined anatomy and functionally by its naloxone-reversible suppression by fentanyl (9). In addition, the CR receives only little influence from cortico-pontine fibres (10, 11).
Whereas electric shocks directly excite nerve fibres bypassing the receptors of afferent nerve endings in the cornea, air puff stimulation does activate nociceptors and therefore allows the investigation of peripheral trigeminal nerve structures, whose activation may play a pivotal role in the development of the acute migraine attack (12). The stimulation of the cornea with air puffs has been described previously (13).
The results of previous studies using nociceptive blink reflexes suggest a temporary sensitization of central trigeminal neurones in migraine patients only during acute attacks and not in their headache-free period (4, 14). However, a potential facilitation of central trigeminal structures interictally has also been suggested (15–17). The question therefore arises whether the investigation of the corneal reflex with air puff stimulation is more sensitive in migraine patients for detecting abnormalities also during the pain-free interval. As a consequence, a further question is, whether the corneal reflex activity may be influenced by the administration of an oral triptan and if the examination of the CR, more than the BR, is suited to serve as a prediction model of the efficiency of acute or prophylactic migraine drugs in humans.
At first we validated the nociceptive stimulation with the air puff technique by investigating the corneal reflexes of seven healthy controls before and after a local anaesthesia of the cornea.
In the final study, we investigated corneal reflexes in a group of 25 migraineurs during their pain-free period and in a group of 25 healthy controls, each before and after the oral administration of 100 mg sumatriptan in a randomized, placebo-controlled crossover study, to answer the question of whether a sensitization of trigeminal transmission exists in migraine patients during their headache-free period and whether there is a difference between patients and controls regarding their response to treatment with triptans.
Subjects and methods
Subjects
Power analyses based on pilot data of corneal reflex recordings in healthy subjects revealed that a sample size of 22 patients should be sufficient to detect significant changes of blink reflex responses after drug administration (α = 0.05, β = 0.80, effect size = 30%).
For the preliminary evaluation, seven healthy participants were recruited from clinical staff (two women, five men, mean age 24.4 years).
For the final study, 25 patients (21 women, 4 men, age 19–43, mean age 23 years) with migraine were studied. Inclusion criteria were: age between 18 and 60 years and a diagnosis of migraine with or without aura according to the criteria of the International Headache Society (IHS) (18) for more than 5 years. Subjects had to be without a migraine attack for at least 24 h. Exclusion criteria were any other medical or psychiatric illness and any preventative medication and use of contact lenses. Twenty-five healthy volunteers (14 women, 11 men, age 20–49, mean age 26 years) without a history of a primary or secondary headache syndrome served as controls.
Informed consent was obtained from all patients and controls and the study was approved by the Ethics Committee of the University of Regensburg.
Electrophysiology
To stimulate the cornea, air puffs were used. These were delivered from a custom-built computer-controlled device. Air puff pressure was manually pre-selected with a high precision valve and duration computer controlled through an electromagnetic valve. All recordings were obtained in a supine position on a reclining examination chair. The left and right cornea was stimulated successively by air puffs ejected through disposable micropipette tips (opening diameter 0.5 mm) mounted inside custom-built goggles. The tip of the two ejection tips had a distance of 5 mm from the corneae. Air puffs were delivered with two different air pressures, which were the results of preliminary studies (data not shown). We used pressures of 60 mbar, which was the minimal threshold to provoke blink reflex responses in healthy volunteers. Moreover, we used pressures of 100 mbar, which was the supramaximal stimulus intensity and provoked stable reflex responses. From this stimulus intensity, no further increase of reflex responses had been observed in these studies. The duration of the air puffs was 25 msec for each stimulus. Surface electrodes for the recording of the electromyographic surface response reflex were placed infraorbitally below both eyes and at the root of the nose (19). Gain was 400 µV, bandwidth was 50–2500 Hz and sampling frequency was 5120 (1024 data points divided by 200 msec recording time). Sweep recordings were synchronized with the onset of the air puff via a shared TTL trigger pulse from a PC parallel port controlled by a custom-written program to provide the stimulus sequences. Two blocks of six sweeps each were recorded for stimulation on both sides. Air puffs were delivered at pseudo-random intervals to reduce habituation (between 8 and 15 s). PC-based off-line analysis was perfomed with custom-written software (Matlab 6.1, Mathworks, Natick, Massachusetts, US).
Analysis included the following steps. The first sweep of each block of six sweeps was excluded from further analyses to avoid contamination with EMG responses due to startle responses. From the remaining five sweeps we obtained (i) the onset latencies of the R2 components and (ii) the response areas (areas under the curve, AUCs) of the R2 components.
For the analysis of the response areas, the curves were demeaned, rectified and averaged. AUCs were calculated between 60 ms and 120 ms. The values given are the means of two measures (two blocks of five sweeps) of each side.
Statistics
Statistical analysis was performed with SPSS for Windows, Version 11.1. No variables suggested deviation from normal distribution after visual inspection of the raw distribution and q-q plots. Two univariate analyses of variance for repeated measurements were used for latency- and AUC-analysis. Between-subject factors were ‘group’ (patients vs controls) and ‘substance’ (sumatriptan vs placebo). Within-subject factors were ‘side’ (headache side vs non-headache side in patients, right side vs left side in controls), ‘time’ (before vs after medication) and ‘pressure’ (60 mbar vs 100 mbar). Group differences of the initial R2 latencies or response areas between headache and non-headache sides in migraineurs or between right and left sides in controls were assessed with Student's t-tests. Comparisons of the corneal reflex variables of seven healthy subjects before and after the local anaesthesia with tetracaine-hydrochloride were done with analyses of variances for repeated measures. Within-subject factors were ‘side’ (right eye vs left eye), ‘time’ (before vs after anaesthesia) and ‘air puff length’ (10, 15, 20, 25 and 30 msec). The level of significance was set to P < 0.05.
Design
Preliminary study
Stimulation of the cornea of both eyes was done using an increasing air pulse length (from 10 ms to 30 ms, in steps of 5 ms) at a constant air pressure of 100 mbar. Two drops of tetracaine-hydrochloride (TH) (Ophtocain-N®, Fa. Winzer, Olching, Germany) were applied to the right cornea in each of the subjects, and recordings of the corneal reflexes of both eyes were repeated after 5 min. To differentiate a possible habituation effect from the effect due to the local anaesthesia, corneal reflexes were recorded bilaterally.
Main study
The design of the study was randomized, placebo controlled and crossover. None of the patients suffered from acute headache during testing. None of the patients reported having suffered from a migraine attack within the last 72 h. Subsequently, all subjects received either sumatriptan (Imigran® 100 mg, Fa. Glaxo-Smith-Kline, Munich, Germany) or identical placebo tablets (both reformulated as gelatine capsules). After 2 h, according to the t-max of sumatriptan, the corneal reflex recordings were repeated. After 1 week the subjects were re-examined in a crossover fashion. The patients were followed-up on the next day of both examinations to exclude a premonitory phase of their migraine.
Results
Preliminary evaluation
After the topical administration of tetracaine-hydrochloride on the right cornea in seven healthy subjects, the areas under the curve of the ipsilateral R2 components of the corneal reflexes were reduced by more than 90% compared with the baseline response area on the same eye. On the non-anaesthetized eyes the corneal reflexes remained stable (Fig. 1). Analysis of variance showed a significant effect of both factors ‘side’ (F = 8.21; d.f. = 1/6; P = 0.03) and ‘time’ (F = 10.19; d.f. = 1/6; P = 0.02) and their interaction (F = 8.38; d.f. = 1/6; P = 0.03). The factor ‘air puff length’ (F = 4.88; d.f. = 1/6; P = 0.09) or its interaction with ‘side’ and ‘time’ (F = 0.58; d.f. = 1/6; P = 0.68) had no significant influence on the corneal reflex areas (Fig. 2).

Effect of the topical administration of tetracaine-hydrochloride on the corneal reflex. Curves show one representative CR before and during local anaesthesia (LA).

Mean response areas (AUCs) of right and left corneal reflexes in seven healthy subjects before and after local anaesthesia (LA) of the right cornea with tetracaine-hydrochloride. Error bars represent the 95% confidence interval. Differences of AUCs before and after LA are significant regarding the right eye from an air puff duration of 15 ms or longer (∗). Air puff pressure was 100 mbar each.
Main study
Twenty-one migraineurs in the main study suffered from a strictly unilateral headache (13 on the right side, eight on the left side). The remaining four patients suffered from unilateral but side-changing headaches. The corneal reflexes of two patients and three controls had to be excluded from the final analyses, because no reproducible CR responses were obtainable at baseline. Initial response areas and latencies of the R2 components of the corneal reflexes did not show any significant differences between patients and controls. Furthermore, both groups did not show any significant differences regarding their headache and non-headache side, or their right and left side, respectively. When 100 mbar air puffs were compared with 60mbar, the results indicated a tendency towards shorter latencies and larger response areas in either patients or controls but these changes did not reach significance (Table 1a, b).
Latencies and response areas of the air puff stimulated blink reflex responses
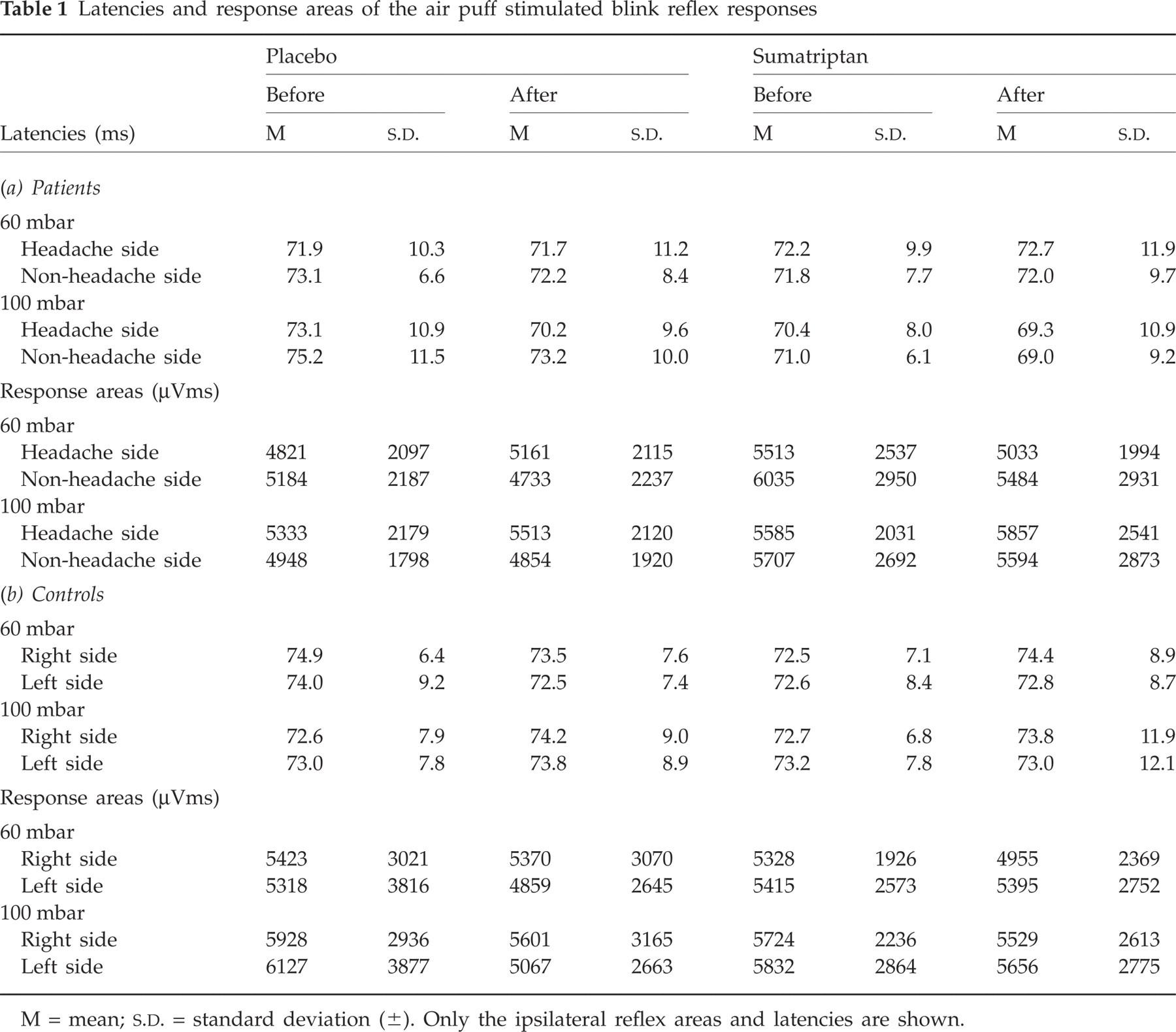
M = mean;
Analysis of variance showed that none of the factors included in the model (the side of headache, the pressure of air puff stimulation, the use of an oral triptan) or any of the interactions between these factors had a significant influence on R2 latencies or R2 reflex areas in both patients and controls (Table 2).
Analysis of variance for R2-latencies and reflex areas (AUCs) of air puff stimulated blink reflex responses

Between-subject factors = group (patients, controls) and substance (sumatriptan, placebo). Within-subject factors = side (headache side, non-headache side in patients, and right side, left side in controls), time (baseline, after oral medication), pressure (60 mbar, 100 mbar air flow). F
= effect size. d.f. = degrees of freedom (error/model). P
= level of significance. Only the ipsilateral reflex answers are shown.
Discussion
Our data suggest that there is no facilitation of the trigeminal system in the headache-free interval among patients with migraine. Neither the onset latencies nor the areas under the curve of the R2 component of the corneal reflex in migraineurs showed any differences compared with healthy controls. After the oral administration of 100 mg sumatriptan, corneal reflex latencies and response areas of patients and controls remained stable without any significant changes, suggesting that there was no inhibition of the trigeminal system, both in patients and controls. Power analyses suggested that the sample size of 25 patients and controls should be sufficient to detect differences greater than 30%.
One may argue that air puff stimulation of the cornea is not sensitive enough to detect minor abnormalities or subtle changes in trigeminal transmission. However, the use of stimuli near the thresholds for eliciting the corneal reflex results (60 mbar air pressure) might have revealed subtle changes in central or sensory trigeminal transmission, whereas the standard blink reflex, due to its high current intensities, leads to a near maximal saturation of the afferent pathway with a reduced sensitivity towards minor changes in trigeminal transmission (4).
Moreover, the suppression of corneal reflex activities after the local anaesthesia with tetracaine demonstrates that indeed receptors of nociceptive afferent nerve endings in the corneae had been stimulated. The stability of reflex responses in the repeated recordings of the un-anaesthetized cornea indicates that the suppression after the topical administration of TH was not due to a general habituation effect.
Using nociceptive blink reflexes, no facilitation was found in the headache-free interval (20) and using the standard blink reflex, also no significant differences in migraineurs compared with controls were observed in the headache-free interval (14). However, in another study differences were found, but exclusively during the headache attack (21).
The corneal reflex has been described as originating from the brainstem, being predominantly nociceptive in nature (2, 22–24). In contrast, the standard R2 blink reflex, evoked by electrical stimulation of the supraorbital region, is extremely susceptible to facilitatory and inhibiting impulses from cortical centres (3, 25). Therefore, the investigation of CR enables the examination of direct trigeminofacial circuits, whereas in standard blink reflex studies additional central pathways due to polysynaptic relays have to be taken into consideration.
Sumatriptan is a serotonin agonist, which acts directly on meningeal and cerebral vessels, causing vasoconstriction. Furthermore, sumatriptan inhibits the release of proinflammatory neuropeptides through an action at 5-HT1D receptors on peripheral trigeminal sensory nerve terminals in the meninges (26). Thirteen out of 15 patients suffering from an intractable postoperative eye pain, originating from nociceptor activation in the first trigeminal division, reported rapid relief after the oral application of 100 mg sumatriptan. This could suggest a peripheral neuronal mode of action of sumatriptan (27). Hence, we were interested in the question of whether the peripheral excitement of a receptor by air puff stimulation can uncover differences between the patients and healthy volunteers after the oral administration of 100 mg sumatriptan in case of a predominantly peripheral active mechanism of the drug. However, other findings support the hypothesis of a central origin of trigeminal facilitation, for example the increased scalp tenderness or an extracranial allodynia and hyperalgesia in some patients during their acute attacks (28–30). One refractory cluster headache patient was described, who continued to have attacks and moreover continued to respond to sumatriptan after a complete trigeminal nerve section (31). This finding would suggest an entirely central mode of action of sumatriptan. However, we cannot exclude that a more centrally acting triptan could have an impact on the trigeminal system outside the migraine attack, that sumatriptan, due to its limited blood brain penetration, may have not at all or exclusively during the acute migraine attack.
The lack of trigeminal inhibition by sumatriptan in our patients may point to a normal function of the trigeminal system in the headache-free interval. Therefore, in summary, the question regarding the 5-hydroxytryptamine(5-HT) receptor agonists, and whether the clinical efficacy in migraine attacks is linked with the action at the central level or at the peripheral one, cannot be answered from our present data.
In contrast, during the acute migraine attack, the nociceptive blink reflexes were described as being abnormal (4). The CR behaviour after air puff stimulation and the triptan response may change under these pathophysiological circumstances.
