Abstract
The literature suggests that triptans are cost effective compared with older types of migraine treatment. However, which of the triptans that is most cost effective has not been established. We compared the costs and effects of triptan treatment from a Swedish societal perspective, using evidence from the literature. A probabilistic cost-effectiveness model was constructed to investigate the costs and effects of treating a single attack in a typical migraine patient. The end-point used in the base-case analysis was sustained pain free without any adverse events (SNAE). We searched the scientific literature for meta-analyses reporting the efficacy of oral triptans. All treatments except rizatriptan 10 mg and eletriptan 40 mg were dominated. The incremental cost per SNAE of rizatriptan 10 mg compared with eletriptan 40 mg was approximately €100. There was substantial uncertainty concerning the results, but probabilistic analysis showed that rizatriptan 10 mg and eletriptan 40 mg had the highest probability of being cost-effective.
Introduction
In Sweden, the Pharmaceutical Benefits Board (PBB) was instituted in 2002 with the purpose of contributing to a rational and cost-effective use of pharmaceuticals. The Board's assignment is to systematically, and in accordance with national prioritization guidelines, decide which pharmaceuticals should be included in the national pharmaceutical benefits scheme. Societal cost effectiveness is a key decision parameter. An important task of the PBB is to review the subsidy status of all products (approximately 3000) that were already in the pharmaceutical benefits system when the Board was instituted. These reviews serve two purposes. First, to inform the Board's decisions on subsidy status, and second, to help other decision makers in Swedish healthcare to use pharmaceuticals in a rational and cost-effective way. To meet both these objectives, the reviews need to meet stringent scientific standards and yet be delivered in a timely fashion.
The products have been classified into 49 groups (using three-digit ATC codes) and the first group to be reviewed was migraine. This paper reports the results of an economic evaluation of treating acute migraine with 5 HT-agonists, so-called triptans.
Migraine is a debilitating chronic disease affecting approximately one million individuals in Sweden (1, 2). At present, no cure for migraine exists and available treatments aim to control and reduce the intensity of the symptoms. In Sweden, like many other countries, guidelines suggest a stepped approach to migraine treatment, with non-pharmacological treatments being tested first, followed by more advanced (and expensive) drugs as the patient moves up the treatment ladder. The last step, involving the most modern class of acute migraine treatment, is triptan therapy. Generally, it has been suggested in the literature that triptans are cost effective compared with other types of migraine treatment (3). In Sweden, there are six different triptans available (seven globally) and they differ in both price and efficacy. Which triptan that is most cost-effective has not been established and the purpose of the present study was to compare the costs and effects of the six triptans available in Sweden when treating one attack.
A decision-tree model was developed, a societal perspective employed and the scientific literature was searched for systematic reviews and meta-analyses reporting the efficacy of oral triptans in order to populate the model.
Methods
Cost-effectiveness model
A decision-tree cost-effectiveness model was constructed to investigate the costs and effects of treating a single attack in migraine patients. The patients were assumed to be 40 years old, had 15 attacks per year, and were employed. Furthermore, it was assumed that the patients had reached the last step on the migraine treatment ladder where a triptan should be tested. Triptans come in different formulations such as tablets, injections and nasal spray. Some patients are unable to take tablets (e.g. because of nausea), but in the present study only tablet formulations of oral triptans, which are by far the most common, were considered. In the model it was assumed that nothing was known about patient characteristics that could influence treatment response or adverse events.
The model was made probabilistic, requiring probability distributions to be defined for parameters in the model. The probability distributions reflect the volume and quality of information available for model inputs such as clinical effects and the use of resources. Computer simulation was then used to propagate the uncertainty through the model and therefore the uncertainty in the cost-effectiveness results indicate the uncertainty in the decision to implement a treatment strategy rather than the uncertainty surrounding single model inputs (4, 5).
The principal health outcome in the study was freedom from pain at 2 h without recurrence and no adverse events (SNAE), and results are reported as incremental cost per SNAE. Costs are in Euros and have, when necessary, been converted to Euros employing an exchange rate of €1 = 10 SEK. Cost-effectiveness acceptability curves are reported to illustrate uncertainty in the estimated cost effectiveness of different treatments (6, 7).
Treatment strategies
The primary interest of the study was to assess which triptan to try first once the patient had reached the step on the treatment ladder when a triptan should be tested. Response to triptan therapy is idiosyncratic and a patient often needs to test more than one in order to find a treatment with satisfactory effects and tolerability. Hence, only those triptans that are considered to be first-line treatment in Sweden (i.e. zolmitriptan 5 and 2.5 mg, eletriptan 40 mg, sumatriptan 50 and 100 mg, rizatriptan 10 mg and almotriptan 12.5 mg) were considered in this study.
Model structure
Given the purpose of the study, a relatively simple decision tree could be employed (8). Figure 1 illustrates the basic structure of the model. Following the onset of a migraine attack, the possible pathways hypothetical individuals can experience in the model are shown in Fig. 1. As indicated by the first (or leftmost) chance node (indicated by a circle) in the decision tree, patients may or may not experience some side-effect of treatment. Moreover, patients can be pain free at 2 h, or fail to respond to the treatment as shown in the second chance node. Patients who respond at 2 h may experience a recurrence of headache within 24 h, as indicated by the last (rightmost) chance node.

Model structure. PF2, Pain free at 2 h; PF2NR, PF2 with no recurrence; SNAE, sustained pain free with no adverse events.
Thus, the measure of symptom improvement is freedom from pain at 2 h, which is available for all triptans and considered to be a better measure than pain relief (9, 10). Recurrence of pain is quite common, is considered important by the patients (11) and varies between the triptans and was considered important to include in the model. Furthermore, similar reasoning warranted the inclusion of side-effects in the model. Hence, in the cost-effectiveness analysis freedom from pain at 2 h, no recurrence and no adverse events are combined into SNAE. The proportion of patients reaching this end-point is simply those following the pathway with a terminal node (indicated by a triangle) named SNAE in Fig. 1. It should be noted that in clinical trials, sustained pain free has a different definition, which includes no use of rescue medication. The reason that measure was not used is that it has not been widely used until recently and therefore many studies would have been excluded from the analysis, causing much important information to be lost. As the measure including the use of rescue medication was considered only marginally better, this was deemed a reasonable compromise. In the results, using pain free at 2 h (PF2) and PF2 combined with no recurrence (PF2NR) as outcomes are reported as sensitivity scenarios. These pathways are also shown in Fig. 1.
For each treatment strategy, the cost of the triptan is incurred. Furthermore, all the outcomes in the decision tree are associated with a cost due to productivity losses, with the highest cost being associated with the outcome of no treatment effect. The model does not include other healthcare costs than the actual drug cost (e.g. costs of adverse events). The main reason for omitting costs of adverse events was that these costs are likely to be small (12). This is further supported by improved handling of chest pains (a common adverse event), which used to cause several hospitalizations in the past, at least in the USA. Other healthcare costs than triptans are not included for similar reasons. These costs are small, and are often related to diagnosis rather than to treatment (3). Furthermore, two studies have pointed out that a substantial proportion of these other costs are related to comorbidities, e.g. depression, rather than migraine (13, 14). Also, little evidence supports that the triptans differ in this respect.
Effect data
We searched the PubMed, Cochrane and INAHTA databases for systematic reviews and meta-analyses reporting efficacy of oral triptans. Companies that market triptans in Sweden were also asked to submit relevant references.
Direct comparator trials are the most reliable source of information regarding treatment efficacy, but in the area of acute migraine treatment most comparisons are missing. For this reason, the cost-effectiveness analysis was based on a meta-analysis of direct comparator trials and trials where drugs were compared with placebo. Our search of the scientific literature identified four meta-analyses reporting the efficacy measure PF2 for at least two different oral triptans (15–18). These were assessed according to a system used by the Swedish Council for Health Technology Assessment (SBU) (19). The meta-analysis by Ferrari and colleagues was judged to have the highest quality. This conclusion was supported by a systematic review (20). The assessment also showed that the meta-analyses by Oldman (17) and Belsey (16) were of acceptable quality, whereas the study by Gawel (18) was not. Therefore, the base-case analysis was based on Ferrari, but sensitivity analyses were performed using data from Oldman and Belsey.
Data on treatment effect are often reported both as an absolute value and a placebo-adjusted value. The placebo adjustment simply consists of subtracting the placebo effect from the absolute effect. It is a matter for discussion which of these measures should be used. On the one hand, adjusting for placebo effect reduces the impact of any differences in the study population of the treatment. On the other, the absolute effect is closer to the effect that the patient will experience in a real clinical situation, and hence affects symptoms, quality of life and productivity. As published meta-analyses were used to establish treatment effect, heterogeneity could not be fully accounted for and, when reported, placebo-adjusted effects were used in the model.
Table 1 shows the parameter estimates used to populate the model. In order to incorporate uncertainty in the parameters for the probabilistic analysis, β distributions were fitted. In order to define the parameters for the β distribution, methods of moment technique were used (4), employing the reported means and standard errors in Ferrari (15).
Parameter values used in the model
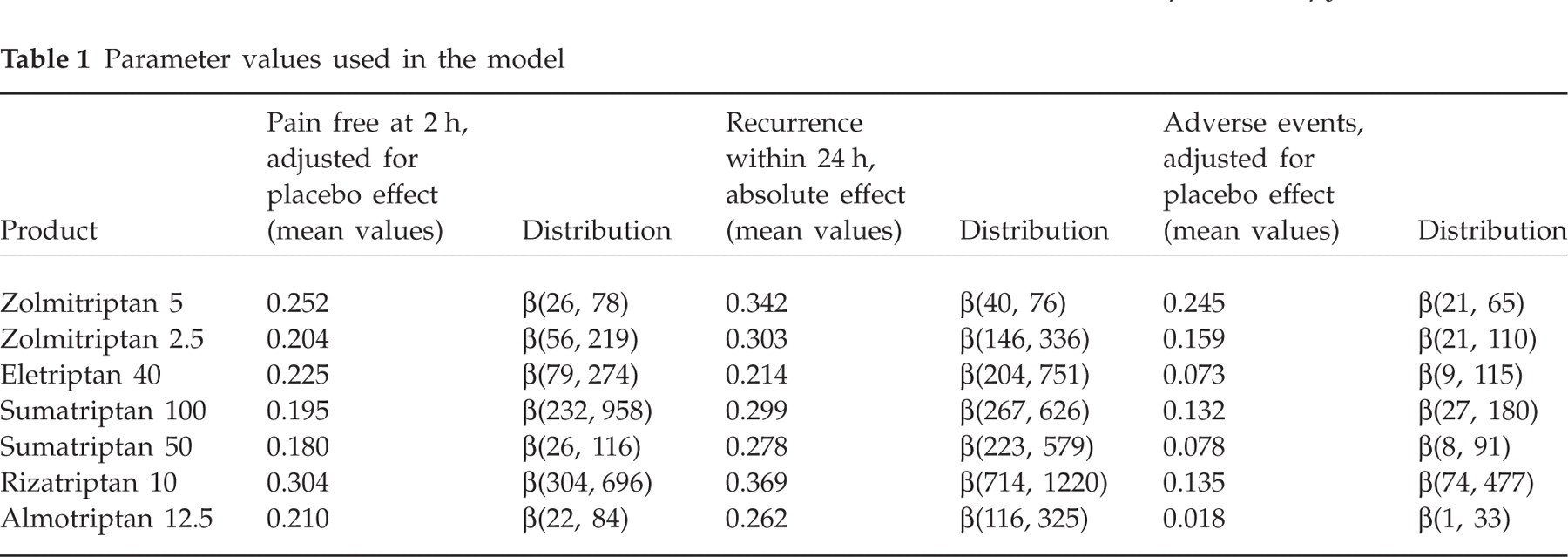
Two systematic reviews which compared different triptans were identified. In Hefland et al. (20), 31 direct comparator trials were described, but only eight were of acceptable quality. Of these eight, four reported data on PF2 for at least two triptans. García Monco et al. (21) identified six direct comparator trials of acceptable quality and they overlap with the studies in Hefland. The four studies identified in Hefland et al. (20) and García Monco et al. (21) were used as a validation exercise to the base-case model results.
Cost data
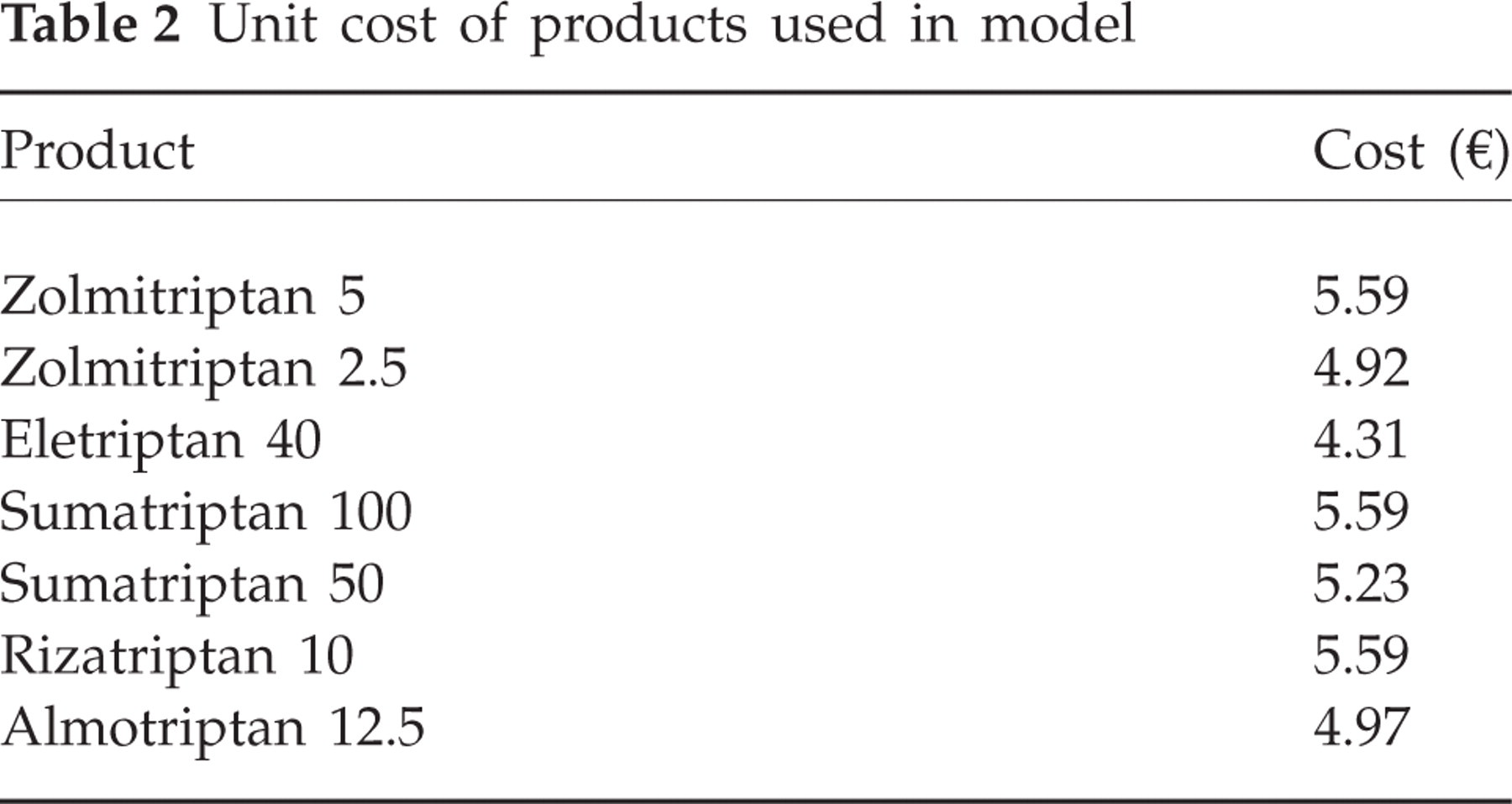
In the model, costs of triptans and productivity losses were included. Costs of triptans (retail price per tablet, based on the largest package) are shown in Table 2. Costs of triptans were treated deterministically in the model, as these can be considered not to be associated with sampling uncertainty.
Unit cost of products used in model
The cost of productivity loss is based on the assumption that the average patient suffers 15 attacks per year (1, 22). Furthermore, the average person with migraine is assumed to lose 5 days of work per year without triptan treatment, i.e. 0.33 days per attack. This includes both being absent from work (absenteeism) and being at work but working with reduced productivity (so-called presenteeism). In the literature, there are estimates ranging from 0.1 days of loss per attack up to >1 day. These results are discussed in Ferrari (23).
If the treatment does not make the patient pain free at 2 h, it is assumed to have failed and productivity losses of 0.33 days are incurred. A patient who does respond at 2 h and does not have any recurrence is still assumed to experience a small productivity loss, 10% of the loss with no effect. This is because the patient may have migraine symptoms that are not helped by the triptan. If the patient is pain free at 2 h, but experiences a recurrence, the productivity loss is assumed to be 50% of the loss with no effect. The value of 1 day of lost production is €100 (24). In Table 3, these parameter values are shown together with the distributions used for the probabilistic analysis which are based on assumptions.
Parameter values of loss of production used in model

Analyses
Second-order Monte Carlo simulation was employed when analysing the model. In each simulation, parameter values were drawn randomly from the defined probability distributions and the cohort of hypothetical individuals was run through the model and mean costs and health outcomes calculated for all strategies. This procedure was repeated 5000 times, generating 5000 estimates of mean costs and mean effects of a cohort for both strategies (25). The model was programmed and analysed in Microsoft® Excel (Microsoft Corp., Redmond, WA, USA) and in TreeAge Pro (TreeAge Software, Inc., Williamstown, MA, USA).
Results
Cost-effectiveness
The base-case analysis showed that all treatments except rizatriptan 10 mg and eletriptan 40 mg were dominated (i.e. had higher cost and worse health outcome), as shown in Table 4. The incremental effect (cost) of rizatriptan 10 mg compared with eletriptan 40 mg was 0.0020 SNAE (€0.2), yielding a cost per SNAE of approximately €100. The results of the probabilistic analysis (Fig. 2) showed that there was a high degree of uncertainty concerning which triptan is the most cost effective. However, rizatriptan 10 mg and eletriptan 40 mg had the highest probability of being cost effective. Almotriptan 12.5 mg had a lower probability of being cost effective than rizatriptan 10 mg and eletriptan 40 mg, but was still more likely to be cost effective than zolmitriptan 5 mg, sumatriptan 50 mg, sumatriptan 100 mg and rizatriptan 2.5 mg, as the probability of these four treatments being cost effective was very low.

Cost-effectiveness acceptability curves. Note: sumatriptan 100 mg and rizatriptan 2.5 mg have a very small probability of being cost effective at every value of a health outcome and consequently those curves virtually lie on the abscissa.
Results of base-case analysis

SNAE, Sustained pain free with no adverse events.
Results from the analyses of the four direct comparator trials are shown in Table 5. For example, rizatriptan 10 mg dominates the comparators (naratriptan 2.5 mg, rizatriptan 5 mg, sumatriptan 100 mg and zolmitriptan 2.5 mg) in the three analyses where it is included. Furthermore, rizatriptan 5 mg, zolmitriptan 2.5 and 5 mg are all inferior in all analyses where they are included.
Results using data from comparator trials

SNAE, Sustained pain free with no adverse events.
Sensitivity analyses
A number of sensitivity analyses were performed to test the robustness of the results. Some of these results are summarized in Table 6. Eletriptan 40 mg and rizatriptan 10 mg remained the most cost-effective treatment, even if a different health outcome was used, PF2 or PF2NR. The issue of whether or not results were placebo adjusted had a substantial impact on the results. Without placebo adjustment, almotriptan 12.5 mg dominated all other treatments if SNAE or PF2NR was used (Table 6). If the outcome was PF2, rizatriptan 10 mg had an incremental cost-effectiveness ratio of €4 compared with almotriptan 12.5 mg.
Results of sensitivity analyses

PF2, Pain free at 2 h; PF2NA, Pain free at 2 h, no AE; SNAE, sustained pain free with no adverse events.
Data from two meta-analyses other than Ferrari (15) were also used for sensitivity analyses. First, data (both with and without placebo adjustment) from Oldman (17) were used. Unfortunately, Oldman does not report data on recurrence, so data from Ferrari (15) were used for that parameter. Both with and without placebo adjustment, rizatriptan 10 mg dominated all other alternatives. However, almotriptan 12.5 mg was not included in Oldman's analysis. Second, data from the meta-analysis from Belsey (26) were used. In this case, data on recurrence and adverse events had to be taken from Ferrari (15) as they were not reported by Belsey (26). The differences in the effects reported by Belsey and Ferrari were very small and consequently, cost-effectiveness results were very similar to the base-case scenario. With placebo adjustment, rizatriptan 10 mg dominated all other treatments, and without placebo adjustment almotriptan 12.5 mg dominated the other alternatives, regardless of outcome in both cases (details are available from the authors).
Discussion
In the base-case analysis, all treatment strategies except rizatriptan 10 mg and eletriptan 40 mg were inferior (i.e. had higher costs and poorer health outcome). The incremental cost per SNAE of rizatriptan 10 mg compared with eletriptan 40 mg was approximately €100. If the societal willingness to pay for the defined health outcome is >€100, rizatriptan should be the recommended treatment for a patient testing a triptan for the first time. If, however, the societal willingness to pay is <€100, eletriptan should be the preferred choice. Probabilistic analysis indicated substantial uncertainty concerning the estimated cost-effectiveness. However, three treatment strategies, rizatriptan 10 mg, eletriptan 40 mg and almotriptan 12.5 mg, all appear to have a higher probability of being cost effective than the other four strategies included in the analysis.
As noted in Methods, direct comparator trials could be regarded as the most reliable source of information concerning treatment efficacy. However, as most comparisons are missing, these studies were used mainly for validation purposes. The most important conclusion from this analysis was that rizatriptan 10 mg was the dominant strategy in all analyses incorporating data from direct comparison trials, hence supporting the results from the base-case analysis. However, it should be pointed out that the ordering, according to the outcome of the model, of some of the inferior strategies from the base-case analysis was reversed when data from the direct comparator trials were used. This does not change the main conclusions of this study, but it should be recognized that the direct comparator trials may contain different information regarding treatment effects. All analyses performed with data from direct comparator trials are, on the other hand, based on single studies, whereas the meta-analysis contains much more information, as several trials of treatment effect on each triptan were included.
Sensitivity analyses indicated that using outcomes without placebo adjustment leads to almotriptan 12.5 mg dominating all other treatments. The reason for this is the strongly performing placebo arms in the trials of almotriptan 12.5 mg included in the meta-analysis. This should perhaps mainly be regarded as a problem with the model of correctly estimating the absolute effect of each treatment. However, the relative effect of treatments, the principal concern of this study, are determined in a more relevant way by employing placebo-adjusted values.
Another important issue concerns how to measure effect. There is probably no entirely accurate measure of the effect of migraine treatment. An ideal measure would probably include effect on pain and other symptoms, time to onset of effect, whether the effect lasts and is consistent, functional status, patient satisfaction and preference, health-related quality of life, long-term effects and side-effects. However, the measures that are available for most triptans are pain relief and freedom from pain at different points in time, recurrence, effect on some other migraine symptoms (e.g. nausea), use of rescue medication and consistency of effect. All of these are interesting and relevant for the patient. However, the single most important measure is likely to be freedom from pain, which is also the most frequently used in recent clinical trials (10). A recent study has shown that patients, general practitioners and neurologists all agree that effect is more important than tolerability and reliability of migraine treatment (11). PF2 is considered intuitive and robust (15). Using a measure of pain free at 1 h is possibly even better, considering patients' preferences for fast pain relief. However, this outcome is not widely available. There are no data supporting any oral triptan as having a faster onset of action than any other.
Recurrence of headache is difficult to interpret. There can only be recurrence if the drug has been effective initially, which means that a less effective drug has lower absolute rates of recurrence. Also, a drug with better effect may be more effective in patients with more severe migraine and it is plausible that those patients may have more recurrences.
Because of these complications there is a possibility that attributes that are important for the patient and to society have been left out of the cost-effectiveness analysis. In particular, patients' health-related quality of life may not have been included sufficiently. The measures that have been used are certainly correlated to quality of life, but are unlikely to cover the concept entirely. For this reason, it may have been desirable to use quality-adjusted life years as outcome measure, but given that the cost effectiveness of treating one attack was investigated in this study, such a measure would add little to the conclusions. Quality-adjusted life years would, however, have simplified the interpretation of the results. Given the results of this study, a decision maker must form an opinion on the value of giving one more patient sustained freedom from pain without side-effects. With quality-adjusted life years it would have been easier to relate the value of migraine treatment to the value of treating other diseases. An alternative way to assess the patient's utility gain from a treatment would be to use willingness to pay. Searching the scientific literature for studies of willingness to pay for migraine treatment indicated that some evidence is available (27–31), but it is difficult to draw any conclusions from these.
Another important issue when model validation is considered is to ensure that errors in programming or analysis are avoided. To control for these types of errors, the model was programmed and run in separate software by different investigators. Reassuringly, the results were consistent in both analyses.
When interpreting these results it should be stressed that the reviews produced by the PBB need to be delivered in a timely fashion and hence have a very real budgetary constraint. Yet this must be balanced against stringent scientific standards, meaning that the resources of the PBB must themselves be used in a cost-effective way. Not every aspect of migraine treatment can be covered and various shortcomings of the analyses have been discussed and should be kept in mind. For example, a complete evidence synthesis of all available trials (direct comparator trials and placebo-controlled trials) would require that the PBB perform a full systematic review, a very costly task. Moreover, some costs that are normally included in a health economic evaluation when taking a societal perspective have been left out in the present analysis. These include costs of side-effects and other direct medical costs than triptans. However, the most important costs are productivity loss and costs of triptans and, most likely, the results would change only marginally if other costs were included.
Patients and doctors now have a large group of triptans from which to choose. Our analyses leave many interesting questions unanswered, e.g. how many triptans would be optimal, is stepped care or stratified care the most cost-effective strategy? However, our analyses suggest that, given the relative prices currently prevailing in Sweden, eletriptan 40 mg or rizatriptan 10 mg (depending on the valuation of an SNAE) will be the cost-effective choice when triptan treatment is tested for the first time.
