Abstract
The second exteroceptive suppression period (ES2) of masseter or temporalis muscle activity may be reduced in adults with chronic tension-type headache. In adults with migraine, ES2 was found normal or tended to be protracted. To date, no studies on exteroceptive suppression in children and adolescents with headaches have been published. We investigated the exteroceptive suppression of masseter muscle activity in 14 migraineurs and 19 controls between 6 and 18 years of age. It was elicited by electrical stimulation at the labial commissure. No differences were found regarding the first suppression period, but ES2 was significantly longer in the migraine group than in controls. The results of the migraine group suggest overactivity of the interneurons of the reflex loop due to impaired inhibitory control from superior antinociceptive systems already at the beginning of this headache disorder.
Keywords
Introduction
While the parenchyma of the brain is insensitive to pain (1), intracranial and especially meningeal blood vessels are richly endowed with nociceptors (1, 2). Afferents to supratentorial nociceptors originate within the trigeminal ganglia (1, 2) and it is generally recognized that migraine headache is generated in the so-called trigeminovascular system (3, 4). Nevertheless, the very mechanism of migraine headache is still a matter of discussion (5–9). Moreover, pain perception is not the simple consequence of the activation of nociceptors (10), but is crucially influenced, for example, by sensitization processes (11) and by endogenous descending control systems of pain (12). Also in migraine, antinociceptive systems may play a pivotal role (5–9).
Migraine pathophysiology can be studied by electrophysiological techniques (13). Several studies have used the trigeminofacial orbicularis oculi reflexes (blink reflex and corneal reflex) (14) to investigate whether there are migraine-specific peculiarities of the trigeminal system. Blink reflex and corneal reflex are usually elicited by supraorbital or corneal electrical stimulation, the afferent impulses reaching the pons via the ophthalmic root of the trigeminal nerve, which also conveys meningeal afferents (14, 15). In adult migraineurs during the headache-free interval, the so-called R1 and R2 responses of the standard blink reflex (16–19) are normal; however, the response thresholds for the principally nociceptive (20) R3 response of the standard blink reflex (18, 21) as well as for the purely nociceptive corneal reflex (22) are lowered, and the habituation of the so-called ‘nociceptive blink reflex’, which is elicited by a nociceptive-specific stimulation electrode, is reduced (23). During migraine attacks, the area of R3 (24) and the area of the response of the nociceptive blink reflex (25, 26) are increased. This increased excitability of trigeminal nociception, it is suggested, is caused by central mechanisms rather than by peripheral sensitization of meningeal nociceptors (18–26).
Another reflex that may be elicited by stimulation of trigeminal afferents is the exteroceptive suppression (ES) of masseter or temporalis muscles activity, a trigeminotrigeminal reflex also termed masseter or temporalis inhibitory reflex. In persons voluntarily closing the mouth, electrical stimulation of the lower part of the face on either side elicits two successive transient suppression periods (ES1 and ES2) of the electromyographic (EMG) activities of the masseter and temporalis muscles bilaterally (15, 27, 28). ES responses are reckoned to be reduced by the activity of antinociceptive systems, and their study is considered to allow a look also at the influence of antinociceptive systems on trigeminal afferents (29, 30). Several investigators have studied ES in adults with episodic migraine and found ES2 duration normal (19, 31–38) or tending to be protracted (29, 39–41).
The incidence rate of migraine is highest in late childhood and early adolescence (42). However, ES has not yet been studied in children and adolescents suffering from migraine (43). The investigation of ES in juvenile migraine should provide insight into migraine pathophysiology at the beginning of this disorder, and possible peculiarities might be interpreted as a primary characteristic of migraine and not as the result of repeated attacks or of medication (43).
Subjects and methods
Patients suffering from migraine were recruited from our paediatric out-patient clinics after soliciting patients and controls in a newspaper article. Eligible for inclusion were patients 6–18 years of age who had suffered from migraine for at least 1 year and who had had at least three attacks during the last 3 months. Migraine diagnosis was established according to the International Headache Society (IHS) criteria (44), which were documented in a structured diagnostic interview of the child and one of his or her parents (45). After publication of the revised IHS classification the diagnoses were checked according to the revised diagnostic criteria without any changes being necessary (46). In addition, patients kept an age-adapted prospective headache diary over an 8-week period. Excluded were children with any other headache subtype, with any other neurological disorder, with other serious underlying disease, or with a prophylactic headache treatment during the last half-year. Controls were classmates of the patients or those who had read about the study in the local newspaper. Only subjects reporting neither chronic or recurrent headaches nor other chronic or recurrent pain and—since other electrophysiological investigations found result differences from healthy controls to be similar in migraineurs and their family members (47, 48)—only subjects without any first-degree relatives suffering from migraine were included in the control group. Apart from this, controls were subject to the same exclusion criteria as were patients. The evaluation of subjects’ medical conditions was based on full medical history and complete physical and neurological examination.
Over a period of 18 months we included 15 patients with migraine and 19 controls. Clinical data of controls and migraineurs (Table 1) showed no significant differences by Student’s t-test or Pearson’s χ2 test. The migraineurs had suffered from headaches for 4.5 ± 2.3 years. Headache intensity was 6.7 ± 1.8 (scale 1–10); four migraineurs suffered from migraine more than three times a month. Nine migraine patients had migraine without aura and five had typical migraine with aura headache. In migraineurs with and without aura, the duration of headache disorder and frequency as well as intensity of headache attacks were comparable (Table 2). We analysed together the data obtained from patients suffering from migraine with and without aura.
Clinical characteristics of subjects

Characteristics of migraineurs without aura and with aura

Electrophysiological studies were conducted in the headache-free interval with at least a 72-h interval from the last and from the next attack. No child had to be excluded from analysis because of headache attacks during the 72 h after the investigation.
Exteroceptive suppression of masseter muscle activity was registered based on the standards recommended by the European Headache Federation (49), modified for application in children by reducing the number of stimulus repetitions.
Subjects were seated in a padded chair and, during the investigation, they were instructed to clench their teeth at maximal strength which was controlled by the technical assistant. A standard bipolar stimulatory electrode was placed at the left labial commissure. Four single stimuli were applied with 20 mA intensity and 0.2 ms duration. A minimum interval of 30 s was maintained between the stimuli. The EMG was recorded simultaneously from the masseter muscles bilaterally. The active electrodes were placed over the belly of the masseter muscles with the reference electrodes positioned over the mandibles. The children tolerated the investigation excellently.
Off-line measurement of all recordings was conducted by one examiner blinded to group affiliation. Full-wave rectification being unavailable, the alternatively recommended measurement method (28) was used: the beginning and the end of the ES1 and ES2 of either side were determined as ‘visual mean’ (35), i.e. with one single cursor when four responses were displayed superimposed on the screen (Fig. 1). A reduction of the EMG amplitude by at least 80% was defined as suppression. For further analysis we used the mean of the latencies of the left and the right side of each subject and calculated the duration of the suppression periods. One control had recordings without a detectable ES2 period, which was defined as ES2 duration of 0 ms in accordance with the literature (50). In an alternative analysis excluding this patient from evaluation, the principal results and conclusions remained unchanged.
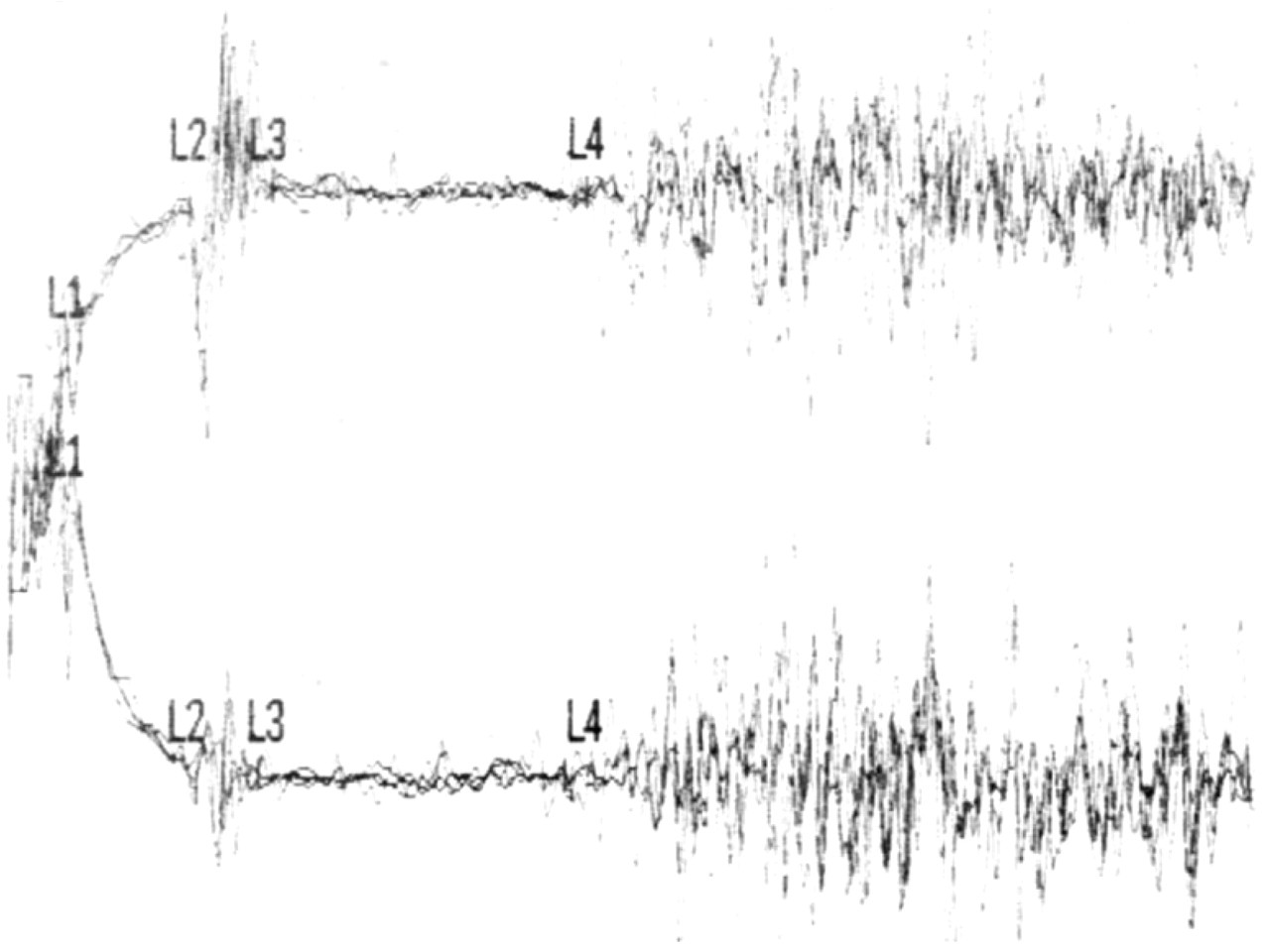
Registration of exteroceptive suppression of masseter muscle activity in a 9-year-old migraineur: simultaneous bilateral electromyographic recording; the responses to the four stimulations are displayed simultaneously. L1 and L2 mark the beginning and the end of the first suppression period (ES1), L3 and L4 mark the second suppression period (ES2).
Statistical evaluation was performed using Statistical Package for Social Sciences (SPSS Inc., Chicago, IL, USA). Data were summarized as mean ± SD. They were checked for normal distribution using the Kolmogorov–Smirnov test. The Levene test was used to check for homogeneity of variances. The latencies and durations of ES1 and ES2 in migraine patients and controls were compared by Student’s t-test and by general linear model (GLM) univariate analysis for each of the four measurement variables including ‘group’, ‘sex’ and ‘interaction of group and sex’ as factors and the subjects’ ages as covariate. A P-value <0.05 was considered to be significant.
The study protocol was designed in accordance with the principles of the Helsinki Declaration. The protocol was approved by the ethics commission of the University of Heidelberg. Children and parents were instructed with regard to the study and signed informed consent was obtained from children and parents.
Results
Table 3 presents the results of ES1 and ES2 measurements in the study groups.
Exteroceptive suppression periods in relation to diagnosis
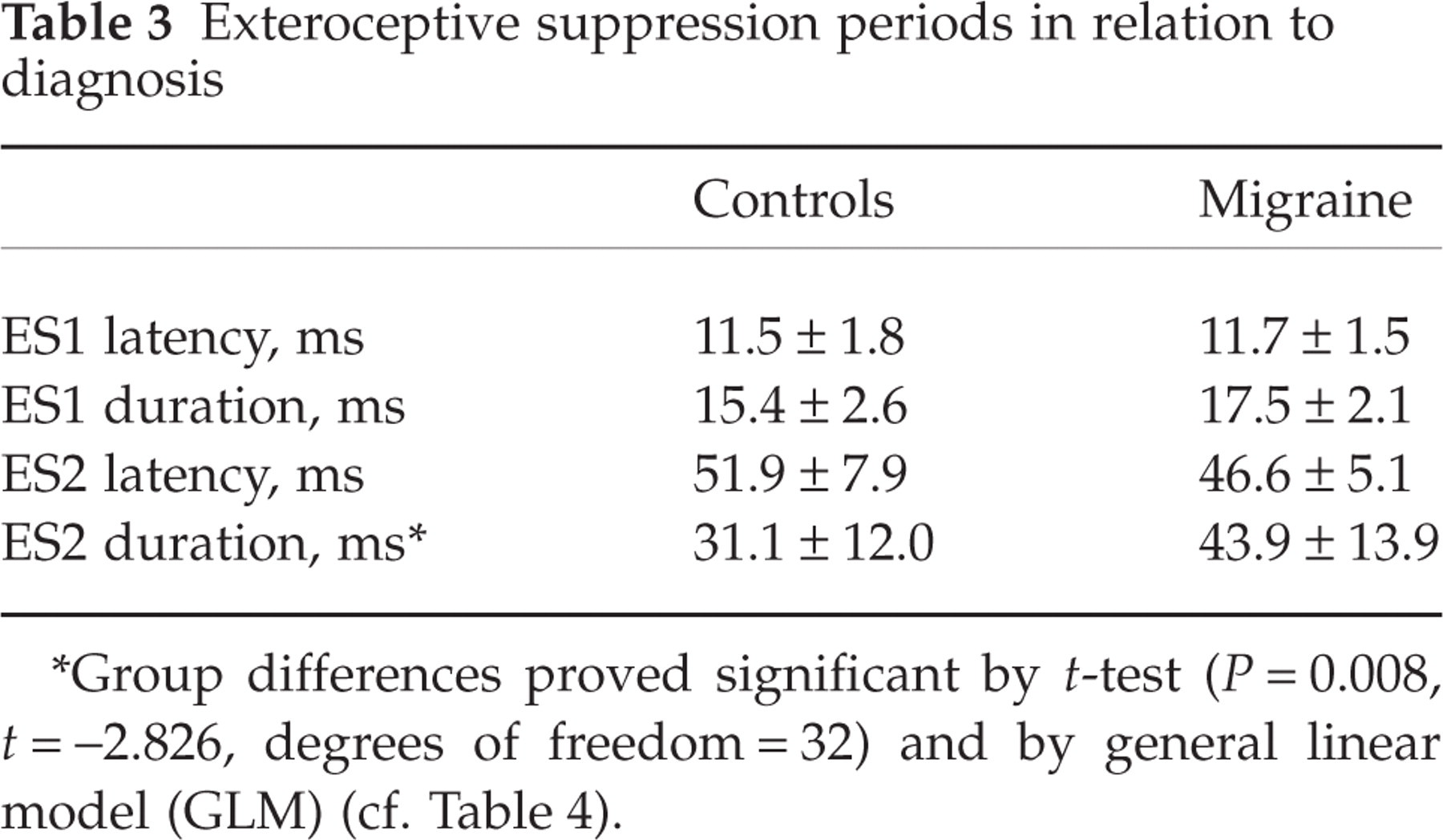
Group differences proved significant by t-test (P = 0.008, t =−2.826, degrees of freedom = 32) and by general linear model (GLM) (cf. Table 4).
While ES1 latency, ES1 duration and ES2 latency did not differ between the study groups, ES2 duration was longer in the migraine group than in controls. This difference proved significant both by t-test and by GLM (Table 4).
General linear model univariate analysis
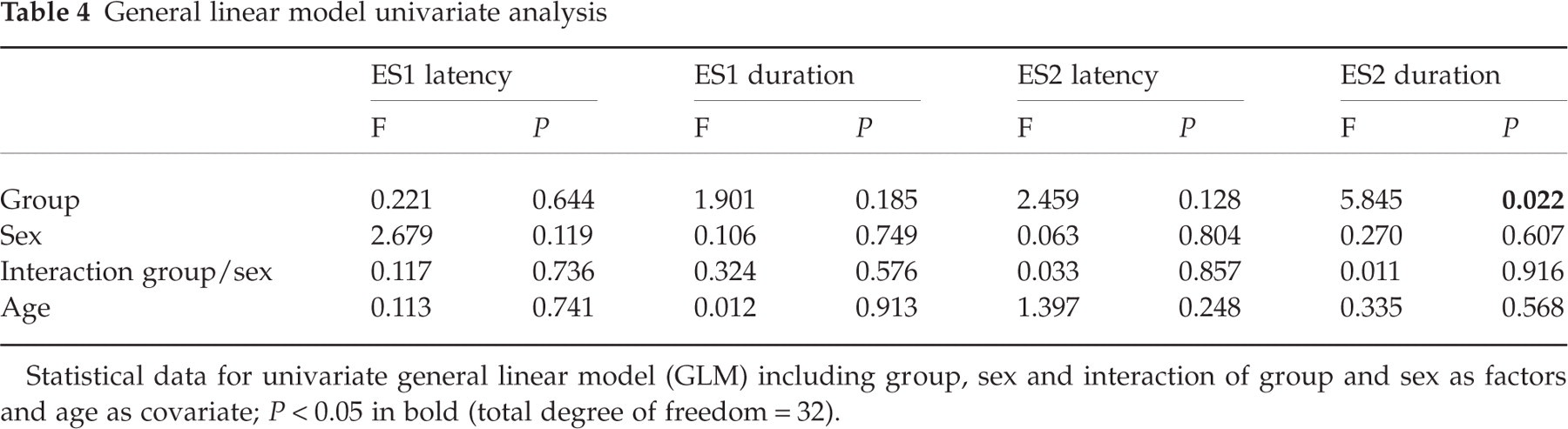
Statistical data for univariate general linear model (GLM) including group, sex and interaction of group and sex as factors and age as covariate; P < 0.05 in bold (total degree of freedom = 32).
Discussion
The present study, showing ES2 to be significantly longer in children and adolescents with migraine than in controls, is the first to investigate exteroceptive suppression of masseter muscle activity in paediatric headache disorder.
Though children tolerated the investigation very well, the prospect of electric stimulation to the face is a deterent from study participation. This is probably the very reason why there are scant published data on ES in children and adolescents. In fact, screening the literature revealed only one paper presenting paediatric data on this reflex (51).
In the migraine and in the control groups the majority of subjects were male, which is in contrast to previously published studies on ES. In GLM we found no influences of the subject’s sex.
While it is usually recommended to study 10 responses (15, 28, 40, 49), some investigators used lower stimulation numbers such as three (52) or five (39). To increase acceptance of the investigation in children and adolescents, we decided to study only four responses.
Studies from different investigators show different normal values for ES2 duration depending on sometimes subtle differences in the method applied. In the investigation of headache disorders an between-stimuli interval of 10 s was more sensitive than an interval of 2 s or 0.5 s (41). For reasons of acceptability we used between-stimuli intervals of at least 30 s. In our control group ES2 duration was 31.1 ± 12.0 ms. Although in studies comparing different stimulation rates ES2 duration decreases with increasing stimulation rate (40, 41), interstudy differences of ES2 duration in healthy controls may not easily be explained by different stimulation rates (29, 32, 34, 35, 37–39, 50, 52–57).
Because it seemed unreasonable to ask children to participate in the study during a headache attack, we studied them in headache-free periods. Because electrophysiological results during the headache-free period may be modified by close temporal relationship to an attack (58, 59), we required a 3-day interval from an attack.
Schoenen and coworkers were the first to study ES in headache disorders. They found ES2 abolished or shortened in patients with chronic tension-type headache (CTTH) (29). While several further studies have corroborated these findings (cf. 30), others failed to do so (e.g. 19, 50, 54). The few studies that took into account whether the patients suffered from headache during registration (38, 50) or whether they were headache free (19, 37) gave inconsistent results. Most CTTH studies were conducted in middle-aged adults with long-term headache histories. Studies in young adults with CTTH did not disclose any modifications of ES2 (33, 36, 37) and ES2 duration was found to decrease with increasing duration of CTTH (35). Thus ES2 modifications may develop during the course of CTTH (33, 35–37, 60). In patients with other chronic pain disorders, ES2 reduction was also found in chronic secondary headaches (38), lumbago or neuralgias (52), though not in fibromyalgia (56). A further study found no ES2 differences in different symptomatic headaches (postlumbar puncture headache, meningitis, subdural haematoma, benign intracranial hypertension, cervical headache) but made no mention of headache duration (39). ES2 reduction was detected also in several (32, 40, 61), though not all (54) studies in patients with mood disorder. Mood disorder may be encountered in a high percentage of CTTH patients (62). Thus, ES2 reduction may be a non-specific consequence of long-lasting pain disorder or perhaps of associated mood disorder (40, 63).
In adults with episodic tension-type headache (ETTH), one study demonstrated shortened ES2 (41), while other investigators found normal ES2 (19, 31, 34, 36). It was speculated that ES2 reduction in ETTH might be due to headache presence during testing (31), but only two studies have specified the actual pain status (19, 34). We studied ES2 also in six children and adolescents with ETTH during the headache-free period and found no differences from controls, but this group is too small to allow reasonable conclusions.
In adults with episodic migraine, most studies were conducted during a headache-free period, although the interval from an attack was not noted. One study found interictal ES2 reduction (53), whereas in all other studies interictal ES2 was normal (19, 33, 34, 37, 38) or tended to be protracted (29, 39, 40). In studies not specifying the actual headache status, ES2 was also normal (31, 32, 35, 36) or showed a tendency to protraction (41). Our study in juvenile migraineurs confirms the results in adults, clearly interictally demonstrating a significant prolongation of ES2.
During a migraine attack, trigeminal pain processing differs from that outside the attack (23, 64). The interictal lack of habituation of blink reflex (23), evoked cortical potentials (59) and contingent negative variation (58) normalizes during and close to an attack. There are few published data on ES2 during migraine attacks: ES2 was found normal (40) or it was shorter than in controls and than in migraineurs studied during the painless period (38). Further investigations are needed to judge whether ES2 results during and outside a migraine attack differ substantially.
ES1 and ES2 (65) as well as its animal analogue the jaw opening reflex (JOR) (66) are provoked by stimulation of nociceptive and non-nociceptive afferents (15, 67, 68), which via the mandibular or maxillary root of the trigeminal nerve (27, 28) finally reach neurons of the subnuclei oralis, interpolaris (ES1) and caudalis (ES2) of the spinal trigeminal complex (68, 69). These neurons act directly (ES1) or via interneurons (ES2) on neurons inhibitory to trigeminal motoneurons (70). By eliciting the JOR, antinociceptive brainstem systems are also activated (71). Conversely, stimulation of brain structures playing a crucial role in the endogenous control of pain, such as the periaqueductal grey (PAG) (72–75), the nucleus raphe magnus (73–76) or particular forebrain structures (74–77), will result in suppression of this reflex, and ES2 reduction following limb stimuli is attributed to activation of these antinociceptive systems (78). Thus ES2 reduction in CTTH has been explained by deficient activation of the interneurons of the reflex loop and by excessive effect of antinociceptive systems (29, 30, 40). Conversely, ES2 prolongation in juvenile migraineurs demonstrated in this study points to overactivity of the interneurons of the reflex loop due to impaired inhibitory control from superior antinociceptive systems.
As mentioned above, antinociceptive systems may play a pivotal role in migraine (5–9). Trigeminovascular afferents responsible for migraine headache (1–4) activate PAG neurons (79), while, conversely, stimulation of the PAG inhibits these afferents (80). Functional imaging during migraine attacks has shown activation of brainstem regions including the PAG which persists after pain relief (81, 82). Stimulation electrodes implanted into the PAG (83, 84) and anatomical lesions of the PAG (85–87) can produce migraine-like headache disorder in previously headache-free patients. Furthermore, in patients with episodic or chronic migraine iron accumulation in the PAG has been demonstrated (88). Thus it was hypothesized that abnormal PAG function might offer a primary mechanism for migraine headache (5–9).
Serotonin (5-HT) mediates the suppression of the JOR by the PAG (89). Agents increasing the 5-HT level reduce ES2 while the 5-HT receptor antagonist methysergide prolongs ES2 (90–92). The role of 5-HT in migraine has long been studied (93). While plasma 5-HT is lowered interictally, it is elevated during attacks (94, 95). Although the mechanism of these changes is unclear, the findings concerning the role of 5-HT in ES and in migraine suggest that low plasma 5-HT between migraine attacks corresponds to prolonged ES2, as demonstrated in this study. Increased plasma 5-HT during migraine attacks might be associated with reduced ES2, as found by others (38). Also, the normalization of evoked potentials during migraine attacks has been explained by enhanced serotonergic activity of brainstem neurons (59).
In conclusion, the ES2 prolongation we observed in juvenile migraineurs between attacks may be the consequence of abnormal descending control of the ES2 from antinociceptive brain structures such as the nucleus raphe magnus, PAG or forebrain structures, mediated in particular by serotonergic afferents.
There are shortcomings to this study. The sample sizes are small, limiting comparisons. It was therefore not possible to compare migraine with aura and migraine without aura, or boys and girls. Moreover, the number of four stimulations which we chose to increase acceptability may have reduced the reliability of measurement results. ES2 may change during the menstruation cycle, especially in menstrual headaches (55), and in further studies the number of menstruating females and the day of the menstrual cycle should be noted.
In summary, while results in adults give rise to the supposition that long-lasting chronic pain disorder may bring about ES2 reduction, the mechanism of which is still unclear, the interictal ES2 protraction in juvenile migraineurs points to overactivity of the interneurons of the reflex loop due to impaired inhibitory control from superior antinociceptive systems, not the result of long-lasting migraine but to be found already at the beginning of this disorder.
Footnotes
Acknowledgements
This work was supported by the Pain Research Programme of the Medical Faculty, University of Heidelberg, Germany. The technical assistance of B. Goor is gratefully appreciated. I thank the unknown referees for their helpful consideration.
