Abstract
Triptans are efficacious for the acute treatment of migraine attacks. Yet, defining a concentration-effect relation for these compounds is difficult as the dynamics of the migraine attack are not thoroughly understood. The objective of this investigation was to develop a disease model to predict measures of headache in randomized placebo-controlled clinical trials investigating oral sumatriptan as a paradigm compound. A hidden Markov model based on the states of response (no relief, relief, and pain free) and headache scores (observed variable) was used in conjunction with population pharmacokinetics. Model parameters were capable of predicting the course of headache relief, pain-free status and headache recurrence. It was shown that sumatriptan shortens mean transit times between states by up to 5 h. The potency of sumatriptan (EC50) was 9 ng/ml. These findings demonstrate the value of combining pharmacokinetic and efficacy information to model disease and characterize time-independent drug properties in a population of migraineurs.
Introduction
Migraine attacks are self-extinguishing phenomena. Thus far, clinical evaluation of anti-migraine treatments has been rather descriptive, providing the relationship between dose and pain response at a predefined time after dosing; a comparison which is often confounded by differences in placebo response rate (1). This is partially due to the dynamics of a migraine attack, which causes time dependencies in the pharmacological response. Understanding of the dynamics of disease is therefore essential to optimize the evaluation of treatment effect. In addition, any attempt to develop new dosing regimens in migraine would benefit from knowledge of the underlying concentration–effect relation, particularly when comparing different compounds in clinical drug development.
Sumatriptan has proven effective in aborting migraine attacks. Oral doses of 50 mg and 100 mg are commonly used to treat migraine pain, but a clear concentration–effect relation has not yet been established (2). Therefore, it was aimed to develop a model for predicting the time course of efficacy measures in the absence and presence of treatment with sumatriptan, using disease-related parameters to characterize the migraine attack itself and pharmacological parameters to describe drug effect on the attack.
In evaluating acute therapies for migraine, efficacy and failure measures often used are pain relief, pain-free status and recurrence of attack (3). A model should be able to differentiate between the main phases of the migraine attack in order to capture the time course of any of these endpoints. However, a few limitations must be overcome to accomplish that goal. First, the initial phase of the migraine attack cannot be described in detail since most clinical efficacy studies are designed to start dose administration and pain assessment only after the attack is fully developed (4). Second, a model should recognize the subjective nature of pain rating scales by being capable of handling noise in efficacy data. Third, pharmacokinetic (PK) information should be incorporated into the model to define a concentration–effect relation (5–7). The latter requirement cannot be easily addressed since current clinical research practice does not always assess pharmacokinetics in conjunction with efficacy.
Previously, continuous-time Markov chains have been identified as an interesting approach to modelling processes related to migraine (8). In this type of model, the different phases of a disease process are represented by a number of states which are interconnected according to pathophysiology. The path of states in a model is governed by the Markov property, according to which transition to a successive state in any given process is determined only by the time spent on the current state. Hence, a Markov model presumes no influence of historical transits. Model parameters include the elements of a state-to-state transition matrix, presenting the chance of moving away from a particular disease state to another state.
However, this method focused on binary data (headache or no headache) and was therefore not able to predict pain relief or recurrence using clinical definitions. Moreover, the effect of sumatriptan was not quantitatively characterized, since no concentration–effect relation could be derived from the available data. In this paper we propose the use of a hidden Markov model (9), which, in addition to the benefits offered by the standard Markov chain model, is able to predict various measures of efficacy and quantify drug effect on a migraine attack.
Materials and methods
Efficacy data
To test the concept of hidden Markov chain in disease progression, a hidden Markov model (HMM) was developed to describe the course of individual migraine attacks, treated either with placebo or with oral sumatriptan. Based on data derived from three clinical efficacy studies [10], 1180 migraine attacks in 638 patients were analysed using this model. Headache intensity had been measured on a four-point verbal scale, where scores 0, 1, 2 and 3 mean no pain, mild pain, moderate pain and severe pain, respectively. Drug administration and pain assessment were started after the attacks had fully developed. Fifty-six per cent of patients had moderate pain intensity at the start of treatment (i.e. baseline); the other 44% had severe pain intensity. Three hundred and ninety-two attacks were treated with placebo, whereas 46, 44 and 698 attacks were treated with single doses of 25 mg, 50 mg and 100 mg oral sumatriptan, respectively. Though the smaller sample size of the lower two dose groups may cause some of the drug-related model parameters to be estimated less precisely, this is not thought to unbalance the model since all predictions are necessarily constrained between the responses associated with placebo and 100 mg data, which set the outer values of the covariate range. All patients included in the analysis were adults, age 39 ± 10 years, weight 66 ± 14 kg (mean ± SD). Eighty-nine per cent of patients were female.
Pharmacokinetic data
No concomitant PK information was available for most migraine attacks in the efficacy studies. To overcome this limitation and evaluate the link between sumatriptan effects and plasma concentrations, a population PK model was used to estimate and simulate population sumatriptan concentrations at the times of headache assessment in the efficacy studies. Simulations were based on PK parameter estimates, which were obtained by fitting concentration data from four sumatriptan phase I studies and one phase II study to a population model by Cosson and Fuseau (11). The studies contained data from 513 subjects who were administered single doses of sumatriptan, dose range 2.5–100 mg. Population models are capable of identifying parameters that are valid for the investigated population as a whole. Thus, although no individual concentration profiles were available, population-based concentrations could be predicted that reflect the pharmacokinetics of the dosing regimen used in the clinical efficacy studies. Fitting and simulation of sumatriptan concentrations was performed using the non-linear mixed-effects modelling software NONMEM V (Globomax LLC, Hanover, MD, USA).
Hidden Markov model
The pooled set of headache scores and simulated sumatriptan concentrations were analysed using a HMM (Fig. 1). The model consists of a hidden layer representing an unobserved disease process. The number of states in this layer was set to three, which is equal to the number of clinically identifiable response states: no relief, relief, and pain free. State-to-state transition dynamics within this layer are determined by transition rates. To allow for both recovery from headache and recurrence of headache to occur, both forward and backward transition rates were estimated. The hidden layer also represents the level at which sumatriptan exerts its activity. Estimation of treatment effect was based on the use of sumatriptan concentrations as a covariate in the model. The action of sumatriptan on forward transition rates was assumed to follow an E max model (Equation 1), the parameters in this model being drug potency and maximum effect (12).

Structure of the hidden Markov model. The hidden layer consists of a Markov model describing the dynamics of the migraine attack. A patient starts in state 1 and then can move back and forth between states, eventually ending in state 3. Transitions from state 1 to 2 and from state 2 to 3 (bold arrows) are affected by sumatriptan. Pain scores assigned to each state are drawn from distributions that constitute the open layer.
Equation 1 describes the rate r(t) x,y of drug-induced transitions in the HMM. r(0) x,y is the transition rate from a state xTo another state y in the absence of sumatriptan (placebo model). C(t) is the sumatriptan plasma concentration at time t. E max represents the maximum effect of sumatriptan on the transition rate. EC50 is defined as the sumatriptan concentration corresponding to the effect reached at half of the value of E max. The part of the equation in the exponent describes drug action and is known as the E max model.
A second layer in the model, the open layer, couples the response states to the observed headache scores. As there are four possible headache scores and three disease states, different distributions of scores will arise within each state. The transition rates and distributions of scores in each state are found by alternately optimizing the parameters in one layer while fixing the parameters in the other layer.
On the basis of headache scores (variable) and sumatriptan concentrations (covariate), parameters were estimated for both layers of the HMM using an open-code C software (13), which was operated from within S-Plus on a Linux workstation (SuSE Linux 7.2 Professional, kernel version 2.4.4-4GB-SMP). A user-written E max model defining the covariate effect was implemented in the model specification file. At the start of the parameter estimation process, the distributions of scores in the open layer were set uniform and maximum drug effect parameters were set to zero to avoid bias in the search performed by the algorithm. Parameter estimates are reported with their 95% confidence intervals (CIs). These were derived from the standard errors using the error propagation calculator program EPC (D. Kelley, http://epc.sourceforge.net/). This program calculates CIs based on a series of random perturbations of the mean and is especially useful when calculating CIs for non-linear equations such as the one in the current model.
The model was designed to predict measures of response on a population basis: individual time courses of headache progression cannot be obtained.
Results
Concentrations were simulated to match the times of headache assessment in the efficacy dataset. Since three different doses were present in the data, three concentration–time profiles were needed (Fig. 2). Concentration data and headache scores were combined and analysed in the HMM. Parameter estimates of the hidden layer and the open layer are summarized in Table 1 and Table 2, respectively. As can be seen in the open layer table, all ‘severe’ and most ‘moderate’ pain scores were clustered into the first state. ‘Mild’ scores and ‘no pain’ scores were placed separately into the second and third states.

Simulated population concentration–time profiles after 25 mg, 50 mg and 100 mg sumatriptan oral dosing were used to provide pharmacokinetic information that was not available from the efficacy studies.
Parameter estimates of the hidden layer in the hidden Markov model
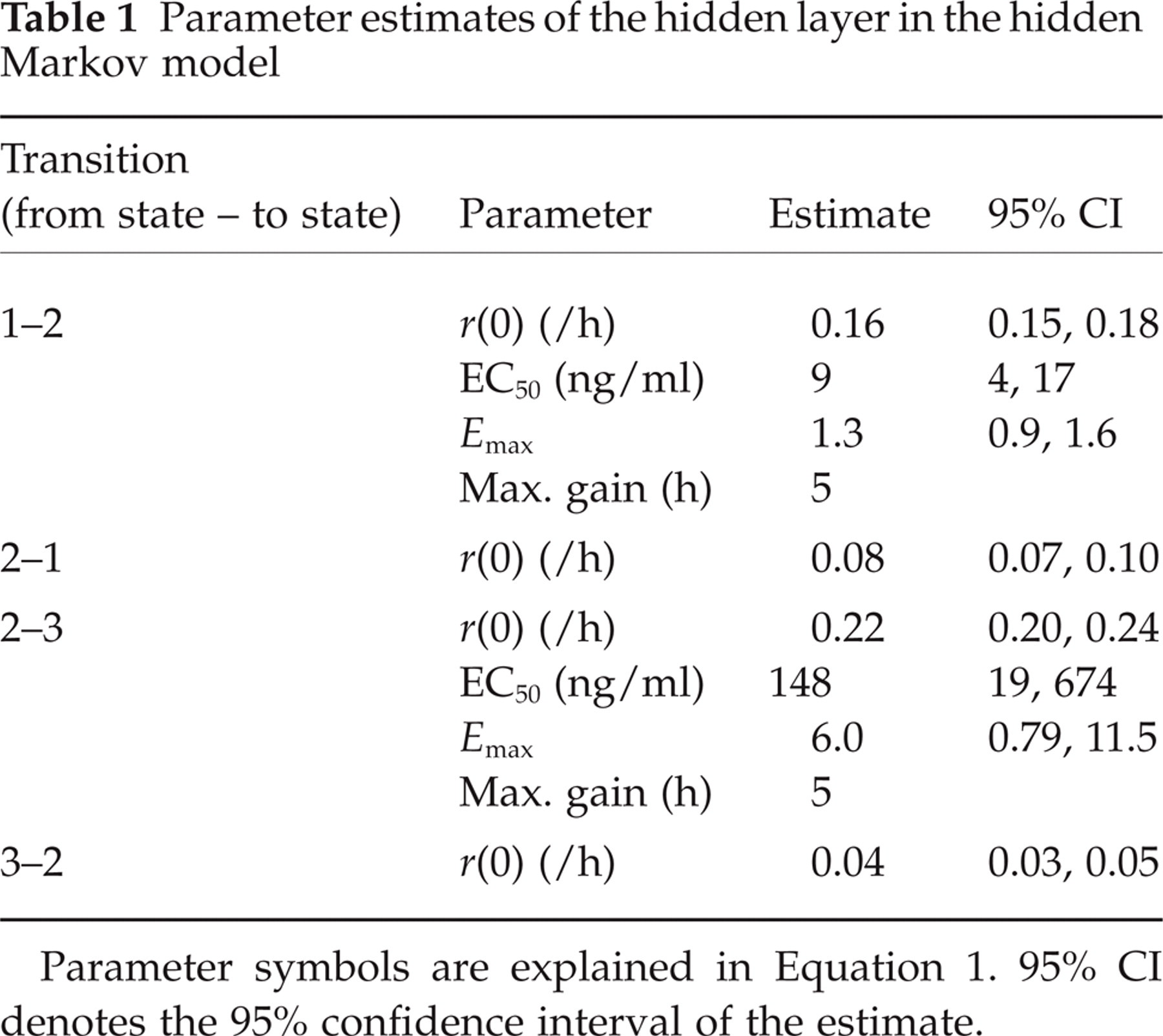
Parameter symbols are explained in Equation 1. 95% CI denotes the 95% confidence interval of the estimate.
Estimates of the open layer: distributions of pain scores per state

Percentages per state (columns) add up to 100%.
In the hidden layer, both estimates of the E max parameters are significantly larger than zero, implying that both forward transition rates are faster in the presence of sumatriptan compared with placebo treatment only. To aid in the interpretation of the E max parameters, they are shown as the maximum gain in transit time from one state to another relative to placebo treatment. Whereas the potency (EC50) on the first transition could be well identified by the model, the potency on the second transition could not be precisely estimated.
Based on these parameter estimates, the time course of headache relief and pain-free status (Fig. 3), as well as the rate of recurrence (Fig. 4), were simulated for placebo treatment and 100 mg sumatriptan after oral dosing. The predicted time course of pain relief and pain-free status were compared with those found in the original data. Ninety-five per cent CIs were constructed based on standard errors in the transition matrix (manuscript in preparation).
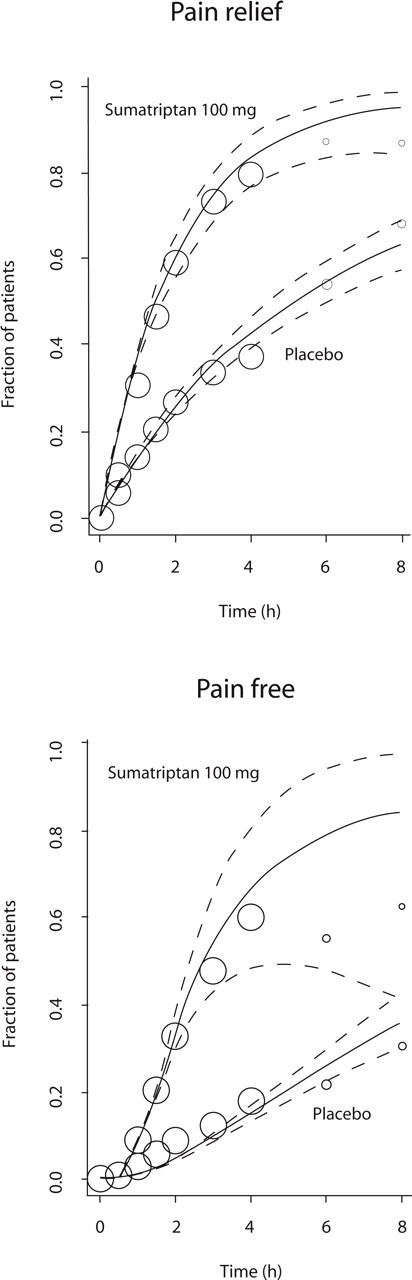
Predicted means (solid lines), corresponding 95% confidence intervals (dashed lines) and observed values (markers) of pain relief and pain-free response vs. time. Hairlines represent placebo treatment, thick lines represent treatment with 100 mg sumatriptan. Marker size reflects the relative number of observations available at each sampling time.

Predicted recurrence vs. time for placebo and 100 mg sumatriptan expressed as the fraction of patients having experienced recurrence. Predictions to the right of the vertical line are outside the model scope and thus extrapolated.
The properties of the concentration–effect relation found for orally administered sumatriptan can be conveniently summarized by relating sumatriptan concentration to the pain relief response at 1 and 2 h postdose (Fig. 5). Note that the value of parameter EC50,1−2 as given in Table 1 cannot be inferred from this graph. Headache relief in a fraction of patients is the sum of all paths leading to ‘mild pain’ or ‘no pain’, whereas this parameter refers to a single transition. The differences in the shape of the two curves show the effect of underlying disease progression mechanisms on the pharmacology.

Concentration–effect relation for pain relief at 1 h (solid line) and 2 h (dotted line) after administration of oral sumatriptan.
Discussion
In the current investigation, we have used sumatriptan clinical data to evaluate the feasibility of applying a HMM to characterize concentration–effect relations and the dynamics of disease in migraine. In contrast to the currently used measures, which rely on arbitrarily defined assessment intervals after dosing [14], a model-based approach can account for time dependencies in treatment effect, enabling accurate comparison of different compounds or formulations in clinical development. To demonstrate this paradigm, the model was designed for orally administered sumatriptan only. Since in most clinical studies efficacy measures are not assessed simultaneously with pharmacokinetics, sumatriptan plasma concentrations were simulated in order to estimate its concentration–effect relation.
Model-derived parameters showed that sumatriptan is effective both in the process leading to pain relief (transition from first to second state) and in alleviating mild pain (transition from second to third state). Depending on plasma concentrations, these processes could be shortened by up to 5 h after sumatriptan treatment. The potency related to the maximum effect on the first transition was 9 ng/ml.
It is important to note that the use of population estimates instead of individual patients’ concentrations makes the calculation of drug-related parameters more difficult. The pharmacokinetic data used in this exercise contained only three (one for each dose) different concentration–time courses. As a consequence, one cannot assess how variability in systemic exposure affects treatment response. Moreover, the model cannot detect whether any variability in the pain assessment was caused by clinically significant differences in individual pharmacokinetics. Awareness of the role of pharmacokinetics is growing in migraine research, but it is not enough to ensure sparse pharmacokinetic sampling as a standard practice. On the other hand, earlier analysis has shown that differences in clinical effects cannot be directly explained by between-patient differences in pharmacokinetics (15). These findings imply that the use of population concentrations is not likely to bias the estimation of drug-related parameters.
The structure of this disease progression model was defined by three states and bidirectional connectivity between states, which allowed for the characterization of various measures of response, including headache recurrence. It was assumed in this model that recurrence mechanisms are independent from treatment type (i.e. placebo or active). Recurrence during active treatment may reflect recurrence in a mixture of patients, some of whom have had a placebo response, and others who experienced the actual drug effect. Under the assumption of identical mechanisms, this phenomenon does not affect the predicted recurrence rates since both types of recurrence are governed by the same transition rates (i.e. those from state 2 to 1 and from state 3 to 2).
At the same time, this structure was simple enough to maintain identifiability of the parameters. Although a four-state model could be considered to describe migraine attacks, this would imply the estimation of two additional drug-related parameters. Apart from the problem of identifying these parameters, they would also be more difficult to interpret: a transition from severe to moderate pain has no clinical correlate such as pain relief or resolution. Moreover, by distinguishing between a state for moderate scores and a state for severe scores one assumes that these scores represent genuinely different events in the migraine attack. It is possible however, that these two scores merely reflect an interpatient or interattack difference in pain perception. This is supported by the observation that a migraine attack at its worst can be either severe or moderate in intensity.
A two-state model, on the other hand, as developed by Hassani and Ebbutt (8), can only predict a single response (pain relief or pain resolution), as one state is reserved for the starting condition and the other for the response state. Consequently, it is not possible to detect changing disease conditions and drug characteristics in a model with only one transition.
The drug-related parameters E max and EC50 on the second transition were less precisely estimated, which is visualized by the broad CI for the pain-free response in Fig. 3. This is probably due to the lack of data for the lower dose groups (25 mg and 50 mg) and at late sampling times. However, the poorer precision of drug-related parameters in the second transition may also reflect a pharmacological effect that is qualitatively different from the first transition. Sumatriptan readily mediates pain relief (first transition) in most patients through blockade of neuropeptide release from the central terminals of meningeal nociceptors. Complete resolution of pain (second transition), on the other hand, is not easily achieved in patients with allodynia since in these cases pain has largely become dependent on the intrinsic activity of central neurons (16). It has been estimated that 79% of patients develop allodynia at some point during their migraine attack (17). The efficacy of sumatriptan has in this context been described as a race against the development of allodynia (18). Since it is likely that patients both with and without allodynia are represented in the second transition, sumatriptan efficacy cannot be unambiguously quantified.
A further motive that supports the number of states chosen is disease-related. The phase a patient is in when entering the clinical study may differ between patients and between attacks and cannot always be distinguished, i.e. the attack may not be fully developed yet or, if fully developed, it may have either moderate or severe intensity. In the three-state model, the estimation algorithm clusters these phases into a single state and assigns separate states to phases representing relief and pain-free status. In doing so, it acknowledges the physiological differences between the disease states.
In conclusion, a hidden Markov model was applied that includes disease and drug-related parameters allowing characterization of the natural history of migraine attacks and of treatment effect following administration of oral sumatriptan. This approach can be helpful in optimizing dosing regimens and comparing efficacy of different drugs in the treatment of migraine. Moreover, it can be adapted to include covariates and other determinants of response in migraine.

