Abstract
Altitude headache (AH) is the most common symptom of high altitude exposure. This prospective cross-sectional and analytical study, conducted in the Ecuadorian Andes, aimed to explore AH symptomatology, taking into account subjects' views. Thirty mountain climbers took part in the Questionnaire Elaboration. The symptoms were grouped into three categories: (A) pain dimension (PD) — intensity, location, quality, outset form, evolution, exacerbating and relieving elements; (B) concurrent symptom dimension (CSD); and (C) feeling/mood dimension (FMD). Ninety-eight mountain climbers comprised the sample for Field Research. Three evaluations were carried out: 4700-5000 m, n = 1, and 5700-5800 m, n = 2. Pearson's correlation coefficient was used to assess internal consistency. Scores between 0.7 and 1 were considered as significant to assess the strength of association between the PD and its different items and CSD and FMD. The following clinical features were found: holocranial 65.6%; pulsatile-burst type quality 75.3%; oscillating evolution 36.7%; increasing with exercise 49.5%; relieved by rest 41.8%; concurrent symptoms referred to, anorexia 26.8%, irritability 26.5%, and finally pessimism and anxiety feelings 33.2 and 29.5%, respectively. We believe that elements provided by us must lead to a new official AH diagnosis criterion.
Introduction
Headache is the most constant and frequent symptom of high altitude exposure. It may occur in isolation or in the context of acute mountain sickness (AMS) [1–4]. We found that in an 8-h rapid exposure to an altitude between 4800 and 5000 m, 100% of a cohort of 615 patients with AMS presented with headache [1]. It was described many years ago: ‘When we others arrived at the second camp (5020 m), we ourselves were in good condition; which was to be expected, as we had ridden most of the way; but in about an hour I found myself lying on my back, along with both the Carrels, placed hors de combat … The symptoms come under the three heads, headache … with all three of us was intense, and rendered us almost frantic or crazy …’) [5].
There are few studies asessing specifically the clinical characteristics of altitude headache (AH). Silber et al. [6] studied 60 subjects before and after going through a mean altitude of 4723 m, focusing on these clinical features. Early studies which mention this type of headache [4, 7, 8] stress the central understanding of its pathophysiology rather than analysing its characteristics. On the other hand, the study recently published by Silber et al. [6] is from a physician's point of view, with less emphasis on headache features from a patient's perspective.
Hyperfunction of the sympathetic system is considered [9] as embedded in the context of taking AH and AMS as part of an incipient or actual cerebral oedema [10].
The main objective of this study was to explore AH symptomatology, taking into account subjects’ views, and to describe these clinical characteristics in mountaineers with AH, i.e. those of life at 2800 m high, where International Headache Society (IHS) criteria become inapplicable. We present this prospective cross-sectional and analytical study on AH clinical characteristics conducted in the Ecuadorian Andes.
Patients, materials and methods
Part I: questionnaire elaboration
Thirty mountain climbers took part; 15 were amateur climbers and members of any of the mountain climber groups belonging to the Asociación de Excursionismo y Andinismo de Pichincha (AEAP); the others were high mountain guides, members of the Asociación Ecuatoriana de Guías de Alta Montaña (ASEGUIM). This group comprised 18 men and 12 women with a mean age of 28 years (range 16–45 years) and a mountain climbing experience of 9.5 years (range 2–27 years).
All subjects resided in the city of Quito (2800 m) and were individually instructed and asked regarding their last headache episode experienced at any altitude above 4500 m to note the following elements, grouped into three categories: (A) pain dimension (PD) comprising these items: (i) intensity. They were provided a visual analogue scale from 0 to 10 (0 = no pain, and 10 = maximum imaginable pain); (ii) location; (iii) quality; (iv) outset form (regarding time elapsed since arriving at the altitude until pain appeared); (v) evolution; (vi) elements worsening it; and finally (vii) elements relieving it; (B) concurrent symptom dimension (CSD); and (C) feeling/mood dimension (FMD).
With the exception of pain, variables had to be categorized as the most important at the time measured (for both frequency and subjective score given by researchers) from 1 to 5, with significance being as follows: minimum, 1; mild, 2; moderate, 3; high, 4; and great or extreme, 5. Subjects could only choose one answer which best matched their experience.
Each one of the variables was then individually grouped along with the importance assigned to it. Those with 50% validity were chosen [by multiplying the number of responses according to their importance, with a maximum theoretical score of 150 (30 participants × 5)]. From these a final questionnaire was prepared to be used in field study (Appendix 1).
Part II: field research
Ninety-eight consecutive mountain climbers presenting with headache made up the sample (none of them took part in preparing the questionnaire); all were students from different Ice and Snow Courses organized by the AEAP Escuela Provincial de Alta Montaña (EPAM) between September 1993 and December 1999. This group consisted of 63 men and 35 women with a mean age of 26.08 years (range 18–41 years) with an active mountain climbing experience of a mean of 9.04 years (range 3–21 years).
As primary requirements to be able to participate, all had to accomplish at least 10 high-altitude ascents (defined in Ecuador as those exceeding 5000 m), and also undergo a medical evaluation to ensure they were in optimum health.
For all of them a demographics interview structure and a survey were conducted on: (i) episodes of acute altitude sickness from the outset of their mountain-climbing activity, using the following previously validated criteria [11]: (a) the result of exposure to at least 4600 m of altitude; (b) after a minimum of 4 h and a maximum of 24 h of exposure; (c) accompanied by headache; (d) occurrence of at least three of the following signs/symptoms: lack of appetite, nausea, vomiting, irritability, sleepiness, apathy, abnormal fatigability; (ii) primary headache (migraine, chronic tension-type headache) under stringent IHS criteria [12], occurring over the last 12 months (this became an exclusion criterion); (iii) coexistence or absence of any concurrent pathology, information which was matched to the medical certification mentioned above. There was an additional inquiry regarding smoking, and alcohol use (had the subject consumed alcohol to the point of intoxication at least once per month over the last 12 months). Finally, the subject was asked if he practised mountain climbing as a sport on an independent basis for at least 120 min per week.
Snow and ice courses ended with a 4-day journey to the mountain (Chimborazo mountain, 6310 m high) including: (i) day 1: departure from the city by bus for a 4-h trip to a height of 4100 m; the ascent started from this location and after 4–5 h camp 1 was set up at an altitude of between 4700 and 5000 m (first night); (ii) day 2: ascent 6–10 h, to an altitude of between 5700 and 5800 m (second night); (iii) day 3: ascent to the top and altitude rescue activities for 6–8 h and then return to camp 2 to sleep (third night); (iv) day 4: descent from the mountain (4–6 h) and return to the city.
Activities on the mountain were as follows: (i) first evaluation, carried out the day after sleeping in the first camp (at approximately 10 h of exposure). Only mountain climbers presenting headache on this first evaluation are included in this study, which amounted to 98 as previously noted; (ii) second evaluation, performed in the second camp before night rest (some 12 h after the first one, and with an approximately 2-h accrued exposure to high altitude); (iii) third evaluation, carried out upon returning from activities and before night rest (about 24 h after the second one; and at approximately 46 h of accrued exposure to high altitude). All evaluations were administered using the questionnaire prepared, under direct supervision of the author.
With the exception of latency time elapsed between arriving at the camp altitude and developing headache, the other parameters were assessed on three occasions. Results for each parameter of the three evaluations were averaged to determine the individual variable frequency. Correlation between them was made to assess internal consistency using Pearson's correlation coefficient [13] recommended for evaluating qualitative ordinal variables [14]. Scores between 0.7 and 1 were considered as significant to assess the association strength between the PD and its different items and against CSD and FMD. To compare antecedents and habits, Yates’ corrected χ2 test was used; P < 0.05 was considered significant.
Results
The study group consisted of 63 men and 35 women, with no differences regarding age and years of mountain climbing. Among antecedents, the AMS was greater among men (P < 0.01), as was alcohol use (P < 0.05). The other variables weresimilar (Table 1).
Description and statistics of the sample
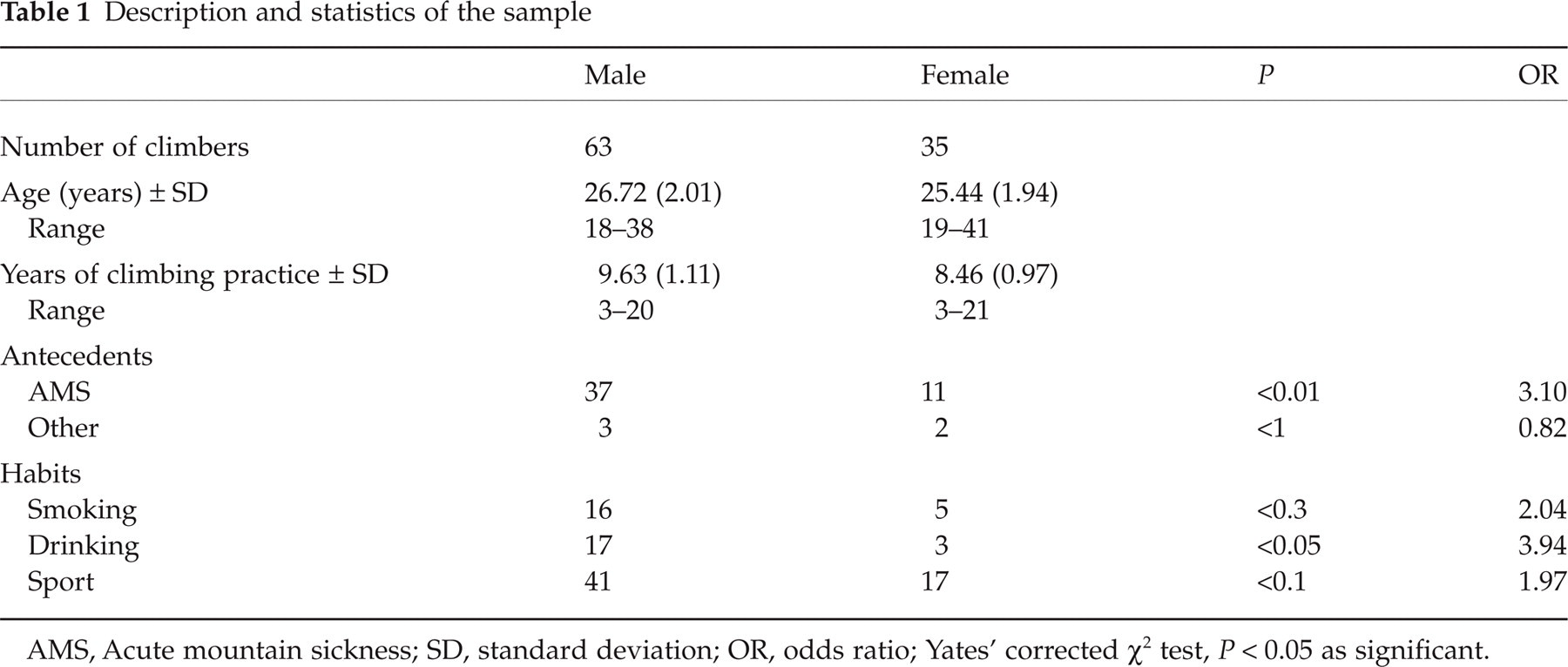
AMS, Acute mountain sickness; SD, standard deviation; OR, odds ratio; Yates’ corrected χ2 test, P < 0.05 as significant.
On the first evaluation 98 mountain climbers developed headache, 53 of them after > 8 h of exposure to this altitude; on the second evaluation headache persisted in 71 mountain climbers, and in 65 on the third survey. In all of them, intensity was always higher after the hours of night sleep. On average, the highest intensity (visual analogue scale 0–10) was found on the second evaluation, with a mean score of 7.84 (Table 2).
Pain intensity

SD, Standard deviation.
The following clinical features were found: (i) its location was holocranial in 65.6%; (ii) with a pulsatile-burst type quality in 75.3%; (iii) oscillating evolution in 36.7%; (iv) increasing with exercise in 49.5%; (v) relieved with rest in 41.8%; (vi) concurrent symptoms referred to were anorexia in 26.8, and irritability in 26.5; and finally (vii) pessimism and anxiety feelings in 33.2 and 29.5%, respectively (Table 3).
Clinical characteristics of altitude headache
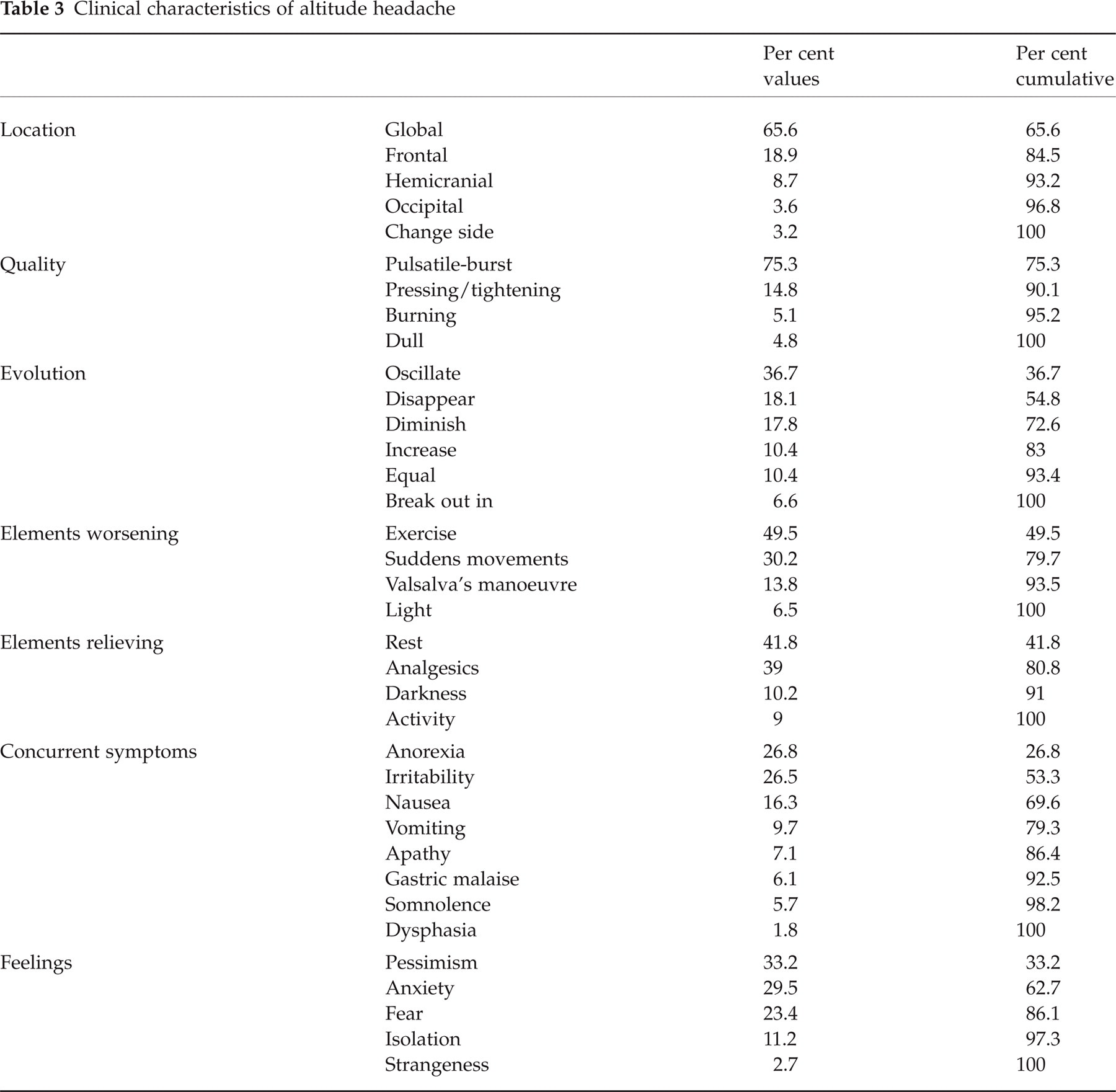
The most important correlations (scores of ≥ 0.7, which determine an important concordance as a determinant of consistency) for the different variables studied are listed in Table 4. From this we may conclude that our mountain climbers experienced headache holocranial in location, with a pulsatil-burst quality, whose evolution was oscillating in nature, increased with exercise, was alleviated with rest and analgesics, was accompanied by anorexia and irritability, by pessimism and anxiety feelings, and was of moderate-to-severe intensity.
Pearson correlation coefficient (scores between 0.7 and 1 to –0.7 are considered as significant to assess the strength of association)

Domain A, Pain dimension; Domain B, concurrent symptoms dimension; Domain C, feeling/mood dimension. Correlation between the most important items of each domain/dimension.
Only two mountain climbers had to descend after the second evaluation due to their headache intensity; (9/10 did not respond to analgesics, and there was a symptom complex that allowed us to categorize them as Grade 3 AMS sufferers according to the previously validated AMS score scale [11]).
Discussion
Our sample was made up of a group of active mountain climbers used to ascending above 5000 m, residing at an altitude of 2800 m. For accuracy, those presenting with some sort of primary headache were excluded, for it is recognized that migraine, particularly, may be triggered by high altitude [15, 16], which could give misleading results.
In addition, all subjects submitted to an identical schedule that consisted of departing from 2800 m by bus, and reaching 4100 m after a 4–5-h journey. From this altitude they started an ascent until reaching an altitude of 4700–5000 m in 4–5 h, depending on climate and ground conditions. Hours elapsed until occurrence of headache was taken into consideration from arrival at this location.
Except for the first evaluation performed after the first night at high altitude, the other two assessments were made before night rest. The reason for this was because it is known that sleeping at high altitude favours headache development [1, 17–21]. It is also known that sleep aggravates AMS [18], causing intracranial vasodilation, particularly during REM [19]. In addition, at high altitude sleep causes hypoxia and Cheyne–Stokes breathing rate [17].
At 20 h of high altitude exposure, haematocrit and blood viscosity increase [20], promoting sleep apnoea [21]. We therefore conclude that the factors mentioned aggravate poor cerebral compliance resulting from oedema and worsen headache intensity. We therefore believe that a more realistic description of headache severity has been achieved. We have also prevented the problems created by sleep at high altitude from playing a role when answering the questionnaire, and even though the difficulty of sleep is often taken as a criterion for the syndrome of AMS, persistence of sleep disorders has been found even in acclimatized mountain climbers [22].
Similarly, the subjects ascended up to an altitude of 5700–5800 m on that day, which took them between 6 and 10 h; they slept at this altitude, and on the following day after 6–8 h they reached the altitude of 6310 m, then they descended and spent the night again at that altitude. This homogeneity of mountain climbers and altitudes in making evaluations, and the number of hours of exposure to these conditions, allow us to conclude that the data are from a cohesive sample.
It has been proposed [23] that headache is due to an incipient cerebral oedema, and its occurrence, resolution or evolution towards more severe forms such as high altitude cerebral oedema will depend upon an individual's susceptibility to compensate for this cerebral oedema. A defect in cerebral irrigation self-regulation does not appear to be the cause, for Jansen et al. [24] found no differences in this mechanism in subjects either adapted or not to altitude.
We believe that it should be taken into account that headache per se may be the cause of some symptoms of the AMS syndrome [23]; developing headache does not necessarily imply being an AMS sufferer, even with the most severe forms of headache. The latter has been clearly shown during the ascent without oxygen to Broad Peak (8121 m) [25].
Experiencing headache without developing AMS appears to be of utmost importance, for under IHS criteria [26] resolution of headache 8 h after descent (criterion D) is taken as an essential criterion. This implies that we could not diagnose an AH for mountain climbers staying at high altitude in spite of their headache.
Another problem existing in both the IHS classification [26] and a recent proposal by Silber et al. [6] is that they take ascent above 2500 m of altitude as an essential element, which makes these criteria inapplicable for those living at this altitude.
On the other hand, unlike the criteria of Silver et al. [6] and those proposed by the IHS [26], the most significant quality of pain in our mountain climbers was that instead of a dull or pressure-like quality, they presented a pulsatile-burst quality (Table 4).
We believe that the data in this study reflect the high consistency attained between the different variables assessed, which were previously obtained from a questionnaire consistently administered to active mountain climbers experienced in high latitudes. Silber et al. [6] include 19 trekkers in their analysis of clinical characteristics. It is our view that these elements, which have not been achieved before, provide greater robustness to our data.
The questionnaire used also impresses with acceptable measurement, i.e. there is a high convergent correlation (values of 0.8 and 0.9) between items of the same domain, and a divergent correlation against items from different domains. It is evident that because headache is holocranial it does not necessarily correlate to irritability (scores 0.1–0.5). Exceptions are items ‘rest’ and ‘analgesics’, which in spite of belonging to PD do not highly correlate with CSD and FMD items. On the other hand, CSD and FMD items correlate highly to each other, which allows the conclusion they are elements of the same domain or dimension, i.e. FMD would be part of CSD. Rest and exercise are items that did not correlate, because, we believe, they are mutually excludive variables.
In conclusion, we believe that elements provided by us must lead to new official AH diagnosis criteria.
