Abstract
Assignment of a diagnosis of migraine has been formalized in diagnostic criteria proposed by the International Headache Society. The objective of the present study is to determine the reproductibility of the formal diagnosis of migraine in a cohort of headache sufferers over a one-year period. The study was performed in a community cohort taking part in a long-term prospective health survey, the GAZEL study. Two thousand five hundred individuals reporting headache in the GAZEL cohort were sent two postal questionnaires concerning headache symptoms and features at 12-monthly intervals. Replies to the questions allowed a migraine diagnosis to be attributed retrospectively using an algorithm based on the IHS classification scheme. The response rate was 82% for the first questionnaire and 69% for both questionnaires. Of the 1733 subjects providing information at both time-points, the agreement rate for the diagnosis of strict migraine (IHS categories 1.1 or 1.2) was 77.7% (kappa = 0.48), with 62.2% of the patients with this diagnosis (IHS categories 1.1 or 1.2) at Month 0 retaining the same diagnosis at Month 12. When diagnostic criteria were widened to include IHS category 1.7 (migrainous disorder), the agreement rate of the diagnosis was similar at 77.6% (kappa = 0.52), but 82% of the patients with this diagnosis (IHS categories 1.1 or 1.2 or 1.7) at Month 0 now retained the same diagnosis at Month 12. In conclusion, the one-year reproducibility of reporting of migraine headache symptoms is only moderate, varies between symptoms, and leads to instability in the formal assignment of a migraine headache diagnosis and to diagnostic drift between headache types. This finding is compatible with the continuum model of headache, where headache attacks can vary along a severity continuum from episodic tension-type headaches to full-blown migraine attacks.
Introduction
For many years, the concept of migraine headache was poorly defined, leading to huge variation in the estimates of its prevalence and hindering the validation of appropriate treatments. However, 15 years ago, the International Headache Society (IHS) attempted to formalize the notion of migraine and other headache types in a structured classification system with simple dichotomous diagnostic criteria (1). Concerning migraine, these criteria require the presence of core symptoms, recurrence of headache and long headache attack duration, and can be accompanied or not by aura.
This initiative has had a significant impact on the study of migraine, notably in determining its prevalence and in facilitating the design of clinical trials. The prevalence of migraine headache as defined by the IHS criteria has been estimated at 9–17%, with a male : female sex ratio of 3 : 1 (2–5). The variation in prevalence rates between studies can largely be explained by different demographic structures of the study populations in terms of age and gender (6, 7).
As well as better defining the prevalence of migraine, the IHS diagnostic criteria for different types of headache has provided treating physicians with a tool for improving diagnosis and for orienting treatment. Since migraine is a chronic disease characterized by episodic headache attacks, it is important that the diagnosis of this condition be reproducible over time. As diagnosis requires matching of headache symptoms to the predefined IHS criteria, reproducible diagnosis relies on these symptoms being stable over time. This is important to assess, from a theoretical point of view to understand more completely the natural history of migraine, and practically in order to optimize long-term treatment, prophylaxis and secondary prevention of migraine. Although there have been several studies published (8–12) looking at long-term follow-up of juvenile migraine (showing principally that this tends to disappear with age), there is little published data on symptom stability in adults. A study of 81 adult migraineurs with aura followed up over a period of from 10–20 years provided evidence for a decrease in the frequency and severity of migraine attacks in many patients (13). The stability of individual migraine symptoms was, however, not reported in this study. In a cohort of 779 headache sufferers identified from primary care registers in the Seattle area of the USA, Stang & Van Korff (14) evaluated the stability of headache symptoms over a period of two years. Those which were the most persistent were the unilateral pain of the headache, its pulsatile quality and photophobia. In contrast, nausea and vomiting were unstable over time.
Since this study has shown that different migraine symptoms are more or less stable over time, the stability of the diagnosis will depend on which symptoms are used to define the condition. The IHS classification includes a strict definition of migraine (IHS categories 1.1 and 1.2), as well as a more broadly defined ‘migrainous disorder’ (IHS category 1.7). Such definitions have been used in many recent epidemiological surveys, including the most recent in France (5, 15, 16).
We have previously evaluated migraine headache in a large cohort of employees of the French national power company, the GAZEL cohort, who have participated in a large prospective health survey using postal questionnaires since 1989 (17). These studies have allowed us to assess quality of life and work absenteeism in migraine, as well as headache severity (18, 19).
The iterative nature of the GAZEL project provides a suitable framework for gathering data on the long-term stability of headache symptoms. We have therefore undertaken an analysis of the changes after 12 months in the diagnosis of migraine assigned from self-reported headache symptoms using an algorithm based on the IHS classification system. This parameter has been compared using either IHS categories 1.1/1.2 or IHS categories 1.1/1.2 with 1.7 (migraine disorder).
Methods
Subject selection
The GAZEL cohort is a French nationwide cohort of more than 20 000 volunters working in the ‘Electricité et Gaz de France’ company, which produces and supplies electricity and gas in France. In 1989, 46 244 workers aged between 35 and 50 years old were contacted to participate in a prospective health survey concerning 44 diseases (17). This survey involved provision of health data on a yearly basis using a postal questionnaire. The GAZEL cohort corresponds to the 20 325 (45%) employees who agreed to participate. After a run-in period to ensure satisfactory response rates (>90% over the first four years), a subcohort, the Hemicrania cohort was derived in 1993 in order to study specifically headache. This cohort included 2500 randomly chosen participants who had consistently reported headache over the run-in period and 2500 participants who had never reported headache. Men and women were equally represented in the Hemicrania cohort.
In 1993, all 2500 headache sufferers in the Hemicrania cohort were sent a questionnaire to be completed and returned by post. This included questions on symptoms of headache permitting diagnosis of migraine to be assigned retrospectively using the IHS criteria (1). Socio-demographic data was also collected. Twelve months later, an identical question-naire was again sent to all those individuals who had returned exploitable information from the first questionnaire.
Definition of migraine
Two definitions of migraine headache were compared in this study. Firstly, all subjects corresponding to IHS subgroups 1.1 and 1.2 (Definition 1) correspond to subjects with migraine on the strict sense. This requires individuals to meet four criteria relating to symptoms, frequency and duration of headache attacks. Definition 2 includes subjects with migraine (IHS subgroups 1.1 and 1.2) and those with migrainous disorder (IHS subgroup 1.7).
Nausea and vomiting were grouped as a single question at inclusion. Only fully completed questionnaires were retained for the analysis.
The frequency of the different symptoms necessary to assign diagnosis was compared at the two time points (Month 0 and Month 12), as well as the proportion of patients fulfilling the four IHS criteria for migraine, and the proportion of subjects meeting the two different definitions of migraine. From these data, an agreement rate, representing the stability of the parameter during the one year period, was calculated as the sum of the number of subjects presenting the symptom at both M0 and M12 and those symptom-free at both time-points, divided by the sample size, expressed as a percentage. Cohen's kappa statistic were used to test the agreement, corrected for chance, for each comparison. A value > 0.6 is set to indicate acceptable agreement.
Results
Among the 2500 headache sufferers sampled, 2051 (82%) returned useable data for the first questionnaire at Month 0, and were recontacted at Month 12, when 1733 (69% of the entire Hemicrania cohort and 84% of the respondants at Month 0) provided complete data. Men and women were equally represented and the average age of the study sample was 46.5 years (Table 1). The demographic profile of those patients recontacted at 12 months was essentially identical to that of the entire cohort at Month 0 (Table 1).
Socio-demographic characteristics of the study sample at inclusion and at Month 12

At Month 0, 561 (32%) subjects met migraine Definition 1 (fulfilling criteria for IHS migraine 1.1 or 1.2), and 1107 (64%) met Definition 2 (IHS migraine 1.1 or 1.2 or IHS migrainous disorder 1.7).
The concordance rate of headache symptoms has been assessed between Month 0 and Month 12 (Fig. 1; Table 2). The best Cohen's kappa statistic was found for the following symptoms: nausea/vomiting (0.67), photophobia (0.6), unilaterality (0.57) and duration of attacks with treatment (0.71).

Symptoms declared at the two date points. Data are presented as the percentage of respondants reporting the symptom at Month 0 (□), Month 12 (n = 1497; ▪). ADL, activities of daily living. Data are reported only for the 1733 subjects providing information at both time points.
Symptom variability over one year. Data are presented as numbers of patients presenting the symptom at M0, and the number and percentage of these patients also presenting the symptom at Month 12. Agreement rates (AR) and Cohen's kappa coefficients (κ) are provided. The total cohort size was 1733 patients

Individual symptoms were then clustered into groups corresponding to the four criteria of the IHS classification of migraine headache (Table 3). Overall, the concordance rate of each criterion was similar to that of its constituent symptoms. The best Cohen's kappa statistic was found for criterion B.
Variability of IHS criteria over time. Data are presented as absolute numbers of subjects (%) from the cohort of 1733 individuals providing information at both time-points. Agreement rates (AR) and Cohen's kappa coefficients (κ) are provided
In a next step, each patient was attributed a diagnosis on the basis of the IHS classification system, and compared this between Month 0 and Month 12. Overall, around 60% of subjects retained their diagnosis over 12 months (Table 4). Of those patients who were identified as having nonmigraine headache at Month 0, 6% fulfilled the criteria for migraine definition 1 (IHS categories 1.1 or 1.2) and 29.3% for migraine definition 2 at Month 12. Finally, we verified whether the initial assigned diagnosis was concordant between the two time points. Reassignment of the initial diagnosis at Month 12 was possible in 62% of subjects according to Definition 1, and in 82% of patients according to Definition 2 (Table 5). The κ coefficient for Definition 2 (the broader one) was slightly higher than for Definition 1.
Evolution of diagnostic category over 12 months. Migraine Definition 1 corresponds to IHS categories 1.1/1.2, Definition 2 to IHS categories 1.1/1.2 and 1.7 combined

Variability of migraine diagnosis over 12 months. Migraine definition 1 corresponds to IHS categories 1.1/1.2, migraine definition 2 to IHS categories 1.1/1.2 and 1.7 combined. Agreement rates (AR) and kappa coefficients (κ) are provided

From these data, it was possible to reconstruct the movement of subjects between the different diagnostic categories (IHS 1.1/1.2, IHS 1.7 and other episodic headache) over the 12-month period (Fig. 2). Approximately half the subjects changed diagnosis, with most of the diagnostic drift occurring in and out of the migrainous disorder (IHS 1.7) category. Less than 10 per cent of subjects evolved from IHS 1.1/1.2 migraine to other forms of episodic headache and vice versa.
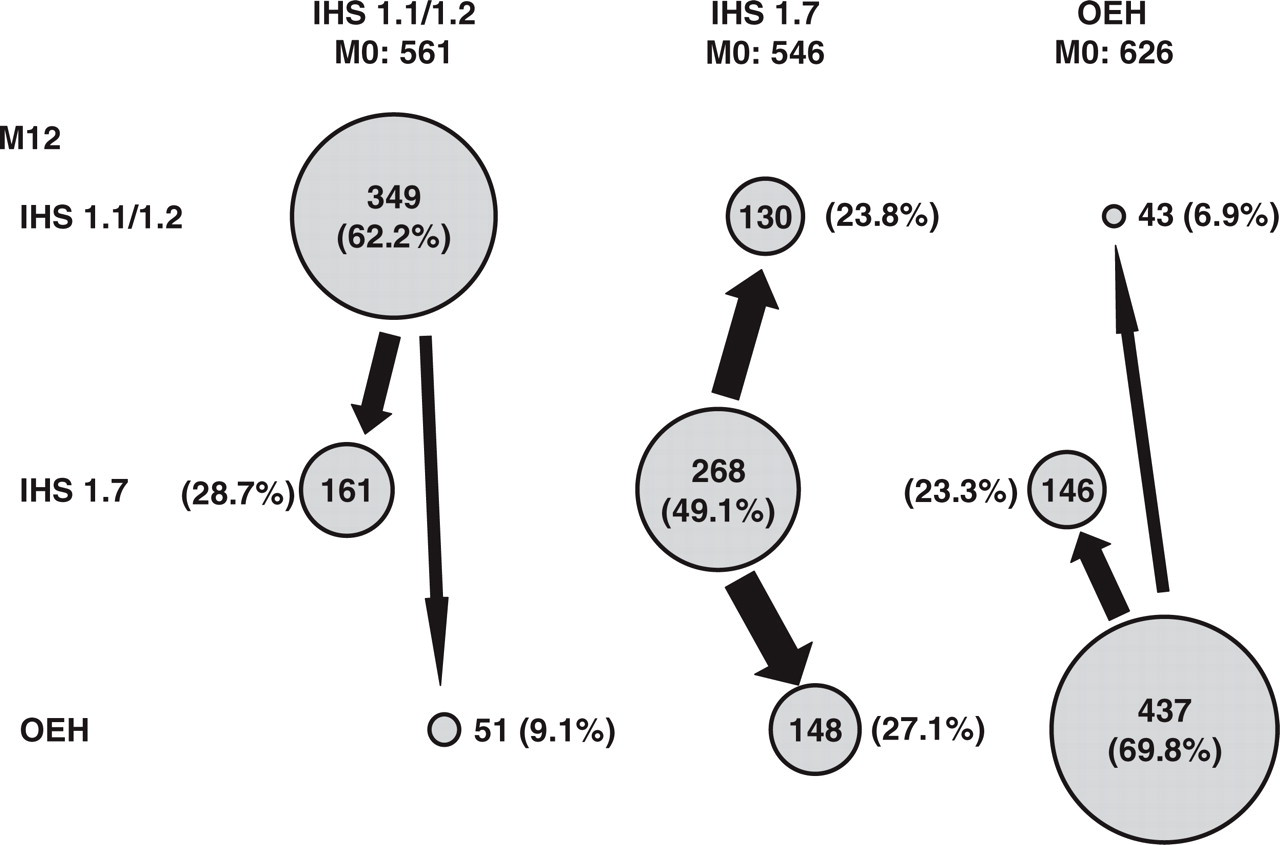
Diagnostic drift over a 12-month period. Data are presented as the number of subjects in each diagnostic category at M12, with the percentage referring to the initial number of subjects in the diagnostic category at M0. OEH, other episodic headache.
Discussion
This study has examined the stability of reported headache symptoms over a one-year period in 1733 long-term headache sufferers. There was considerable variability in this between symptoms. The symptoms that were the most stable contributed to Criterion A and B of the IHS classification. Headache duration, which alone constitutes Criterion C, was not particularly reproducible (73%). It has been suggested by Rasmussen et al. (20) that the duration of headache necessary for a diagnosis of migraine be reduced from four to two hours in future versions of the IHS headache classification. However, the results showed high agreement rate and low kappa. This paradox was discussed by Feinstein & Cichetti (21). Those authors found two main paradoxes in using the Cohen's kappa statistic. In a fourfold table showing binary agreement of two observers, the observed proportion of agreement, p0, can be paradoxically altered by the chance-corrected ratio that creates kappa as an index of concordance. In one paradox, a high value of p0 can be drastically lowered by a substantial imbalance in the tables's marginal totals either vertically or horizontally. In the second paradox, kappa will be higher with an asymetrical rather than symmetrical imbalanced in marginal totals, and with imperfect rather than perfect symetry in the imbalance.
The concordance in symptom identification between the two time points reflects not only the true stability of the symptom but also potential reporting error. This is a limitation of the method of data collection by the use of a postal questionnaire, without case ascertainment by a headache specialist. This is well illustrated by replies to the question on chronicity. Of 1469 subjects who claimed to have previously had more than five migraine-type headaches at Month 0, 178 (12%) denied this at Month 12. The importance of expert interview for collecting accurate data on migraine symptoms has recently been highlighted in a large epidemiological survey in France (22).
Overall, the reproducibility of diagnosis for migraine (IHS class 1.1 or 1.2) was limited, with only two-thirds of subjects being reassigned this diagnosis after 12 months. When the definition was extended to subjects fulfilling criteria for migrainous disorder (IHS class 1.7) as well, the diagnosis was reassigned to 82% of the subjects. The two definitions differed in which patients were diagnostically most unstable. For definition 1, these corresponded to those who fulfilled the definition (only 62.2% retained the same diagnosis), whereas for definition 2, the most unstable were those excluded by the diagnosis (only 70% retained the diagnosis of non migraine according to definition 2). This is because the patients with migrainous disorder (IHS classification 1.7, excluded from definition 1 and included in definition 2) are responsible for much of the diagnostic drift. Similar percentages of subjects (23.3–28.7%, Fig. 2) evolve in and out of the migrainous disorder category both from IHS 1.1/1.2 migraine and from other forms of episodic headache. In contrast, diagnostic drift between IHS 1.1/1.2 and other forms of episodic headache was much more limited.
Those results could be explained by the limited specificity of the questionnaire for identifying migraine headaches. In a previous validation of the questionnaire and algorithm used in this study, diagnostic assignment by the algorithm was compared with expert diagnosis during an interview with a neurologist (16). Although the sensibility of the algorithm was excellent (95%), the specificity was slightly lower (78.5%). Moreover, if symptom presentation is incomplete in individual migraine attacks, or variable between attacks, fulfilment of diagnostic criteria for IHS migraine 1.1/1.2 may not be reproducible. This would lead to apparent diagnostic drift between definition 1 and definition 2 in patients who are in fact diagnostically stable but have incomplete or variable symptom presentation.
This could lead to an inaccuracy in responses, and thus could be a confounding factor of our study which cannot be dissociated easily from real instability of symptoms or criteria. Moreover, the potential reporting error may vary between each individual question, leading to an erroneous ranking of symptom stability. It's difficulty to separate error in reporting (i.e. reliability of self-reported information) from variation in experience over time.
Taken at face value, however, this finding is compatible with the continuum model of headache, where headache attacks can vary along a severity continuum from episodic tension-type headaches to full-blown migraine attacks (23–25). Our data are also consistent with the notion, as proposed by Rains and colleagues (26), that migrainous disorder (IHS 1.7) is heterogeneous (atypical migraine). Indeed, a marked overlap between atypical migraine (IHS category 1.7) and incomplete tension-type headaches (IHS category 2.3) has been suggested by others (12, 27).
The reason why the diagnostic criteria for what is seen as a chronic disease are not stable over time in a significant minority of subjects may be due to several factors. Firstly, the nature of headache attacks may vary with time, as suggested by the Spectrum Study. Migraine is a heterogeneous disease that causes symptoms that vary both among individuals and within individuals from attack to attack (28). Alternatively, the instability may reflect differences in therapeutic control of migraine attacks, with either the use or the effectiveness of migraine treatments evolving with time. It is also possible that the severity of migraine-type headaches changes of the course of the disease. A decline in the prevalence and severity of migraine has indeed been reported over a 10–20 year period in an Italian cohort (13). On the other hand, severity and chronicity increase with time in the case of transformed migraine, possible in response to analgesic medication overuse (29). However, it is unlikely that these long-term trends in the natural history of migraine can contribute significantly to symptom instability over a one-year time-frame.
A potential limitation is that subjects who no longer had headaches at the time of the Month 12 may not have returned the second questionnaire, leading to an over-estimation of the stability rate. Another limitation is the mean age (46.5 years) in this sample, which is higher than the median age of migraineurs in the general population (5). However, there is no a priori reason why the results obtained should not be extrapolated to the entire sample range of 30–54 years. On the other hand, the study has the advantage of representativity (at least of headache sufferers working in the French national power industry) and a satisfactory response rate (89% at Month 0 and 62% at Month 12). Moreover, the study included a community-based sample, and not just headache sufferers under treatment, who may have more severe headaches and potentially greater symptom stability.
In conclusion, the one-year reproducibility of migraine headache symptoms is only moderate, varies between symptoms, and leads to instability in the formal assignment of a migraine headache diagnosis.
