Abstract
Experimental studies in humans have shown that nitric oxide (NO) may play an important role in initiation of primary headaches. It has been proposed that activation of L-arginine-NO pathway and increased endogenous production of NO may be responsible for NO induced headache. NO is synthesized from L-arginine and that reaction also yields citrulline. In the present study we aimed to investigate plasma levels of citrulline and arginine as markers of NO production after infusion of the NO donor, glyceryl trinitrate (GTN). We recruited 16 patients with chronic tension-type headache and 16 healthy controls. The subjects were randomly allocated to receive 0.5 μg/kg/min GTN or placebo over 20 min. Patients were examined on headache free days. Blood samples were collected at baseline and 60 min after start of infusion. Both patients and controls developed stronger immediate headache on the GTN day than on the placebo day (P = 0.008). The headache was more pronounced in patients than in controls (P = 0.02). Plasma levels of citrulline increased significantly 60 min after start of GTN infusion compared to placebo infusion in patients (P = 0.01) but not in controls (P = 0.50). Plasma levels of arginine were unchanged in both patients (P = 0.12) and controls (P =0.18). We suggest that GTN administration may trigger endogenous production of NO in patients with chronic tension-type headache resulting in activation of perivascular sensory afferents.
Introduction
An experimental headache model offers unique possibilities to study mechanisms responsible for headache. Infusion of the NO donor, glyceryl trinitrate (GTN), induces significantly stronger immediate and delayed headache in patients with chronic tension-type headache (1) and migraine (2) and than in healthy controls. Moreover, NO triggers tension-type headache in patients with chronic tension-type headache (1) and migraine in migraineurs (2). These data suggest that NO play a key role in initiation of primary headaches. The pathophysiological mechanisms of GTN-induced headache are not fully clarified. In particular, it is unclear whether infusion of GTN may lead to increased endogenous production of NO by activation of L-arginine-NO pathway. It is known that NO is synthesized from L-arginine and that the reaction also yields citrulline. In the present study we aimed to investigate plasma levels of citrulline as a marker of endogenous NO production and plasma levels of arginine as a marker of endogenous NO precursor during experimental GTN-induced headache in patients with chronic tension-type headache.
Methods
Subject
Sixteen patients with a diagnosis of chronic tension-type headache according to the criteria of the International Headache Society (IHS) (3) and 16 healthy controls were included (Table 1). The patients were recruited from the outpatient headache clinic at Glostrup Hospital. All patients underwent a general physical and a neurological examination and completed a diagnostic headache diary (4) during a four-week run-in period. Exclusion criteria were: a history of migraine or any other type of primary headaches; any kind of daily medication (including prophylactic headache therapy but not oral contraceptives); excessive use of analgesics (corresponding to >2 g of aspirin/day or to intake of simple analgesics on ≥15 days/month for >3 months); serious somatic or psychiatric diseases including depression (Hamilton Depression Score ≥ 17) (5). The controls never had migraine and had less than 12 headache days per year. Patients and controls were examined in random order and at comparable times on the day to avoid seasonal or diurnal variations. The subjects were not allowed to take any kind of analgesics 24 h prior to examination.
Clinical data on headache patients and healthy controls
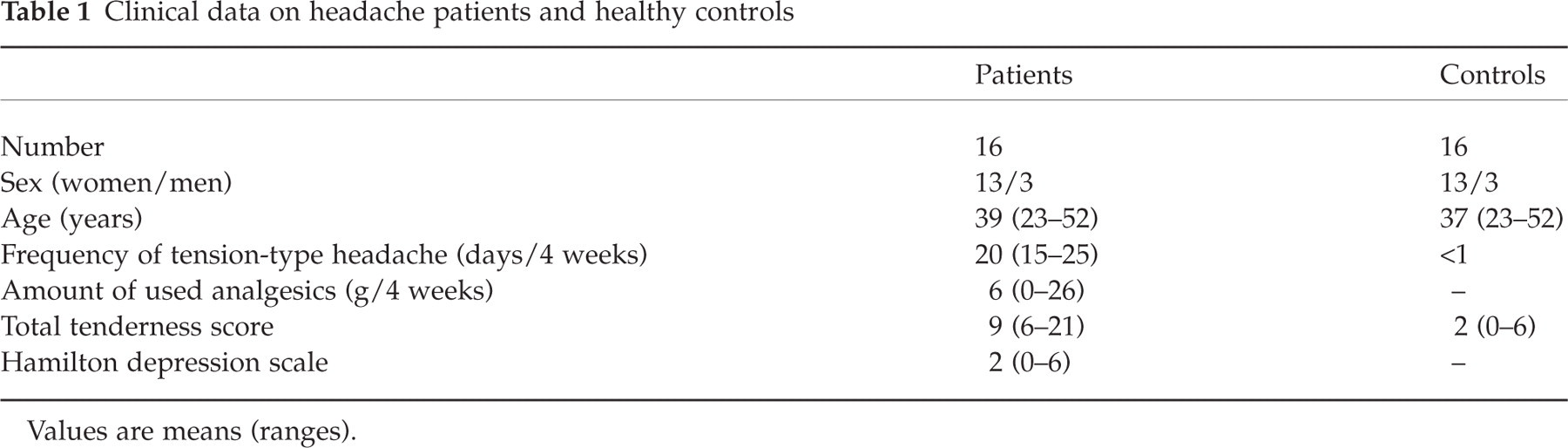
Values are means (ranges).
The study was approved by the local ethics committee and was undertaken in accordance with the Helsinki Declaration of 1975, as revised in 1983. All subjects gave informed consent to participate in the study.
Procedures
In a double-blind, placebo-controlled, crossover design the subjects were randomly allocated to receive 0.5 µg/kg/min GTN or placebo (isotonic saline) over 20 min on two days separated by at least one week. All subjects were examined on days without headache at 0900 h and they had not suffered from any type of headache for at least 12 h prior to examination. Headache intensity was measured on a Verbal Rating Scale (VRS) from 0 to 10 (0 – no headache; 5 – moderate headache; 10 – worst imaginable headache) before and after start of infusion. A cannula (Venflon®) was inserted into patients and controls in the antecubital vein on nondominant side. All subjects were rested supine for at least 30 min before blood sampling. Blood samples were collected at baseline and 60 min after start of GTN infusion. In the first part of the study we examined headache intensity, quality and time profile after infusion of GTN (1).
Quantification of amino acids by mass spectrometry
Amino acids were quantified by tandem mass spectrometry, essentially as described (6). In brief, 5-µl aliquots of plasma were dried and extracted with methanol containing stable isotope-labelled amino acids as internal standards. The extracts were derivatized with acidic butanol to form butyl esters, which were dried, reconstituted in acetonitrile-water-formic acid and were analysed in a Sciex API-365 tandem mass spectrometer. The following ion transitions were monitored: Arginine (231 > 70), 2H4 13C-arginine (236 > 75), citrulline (232 > 113), 2H2-citrulline (234 > 115). Concentrations were calculated by comparing the signal intensities of endogenous compounds with the signal intensities of stable isotope-labelled internal standards, which were present in known quantities.
Data analysis and statistics
Results are presented as median and quartiles. The primary end-point was the difference in changes of citrulline and arginine after GTN compared with placebo. Changes of plasma amino acids were calculated as the differences between the pretreatment value (baseline) and the post-treatment value (60 min). The difference variables for active treatment vs. placebo and baseline variable in the GTN arm vs. placebo arm were compared by the Wilcoxon Signed Ranks test. The differences in changes of plasma amino acids after GTN infusion between patients and controls were compared by the Mann–Whitney test. Five percent was accepted as level of significance. All analyses were done using SPSS®, version 11.5.0. software (SPPS Inc., Chicago, IL, USA).
Results
All subjects completed the study. The concentrations of citrulline and arginine were above the detection limit in all blood samples.
Headache intensity
In patients and controls, the area under the headache curve (intensity × duration) was significantly higher on the GTN day than on the placebo day (P < 0.01) as previously reported (1) (Fig. 1). In patients, GTN infusion resulted in a biphasic response with immediate headache and more pronounced delayed headache several hours later. The patients developed significantly stronger headache than healthy controls both during the first hour (immediate headache) (P = 0.02) and during the subsequent 11 h (delayed headache) (P = 0.008).
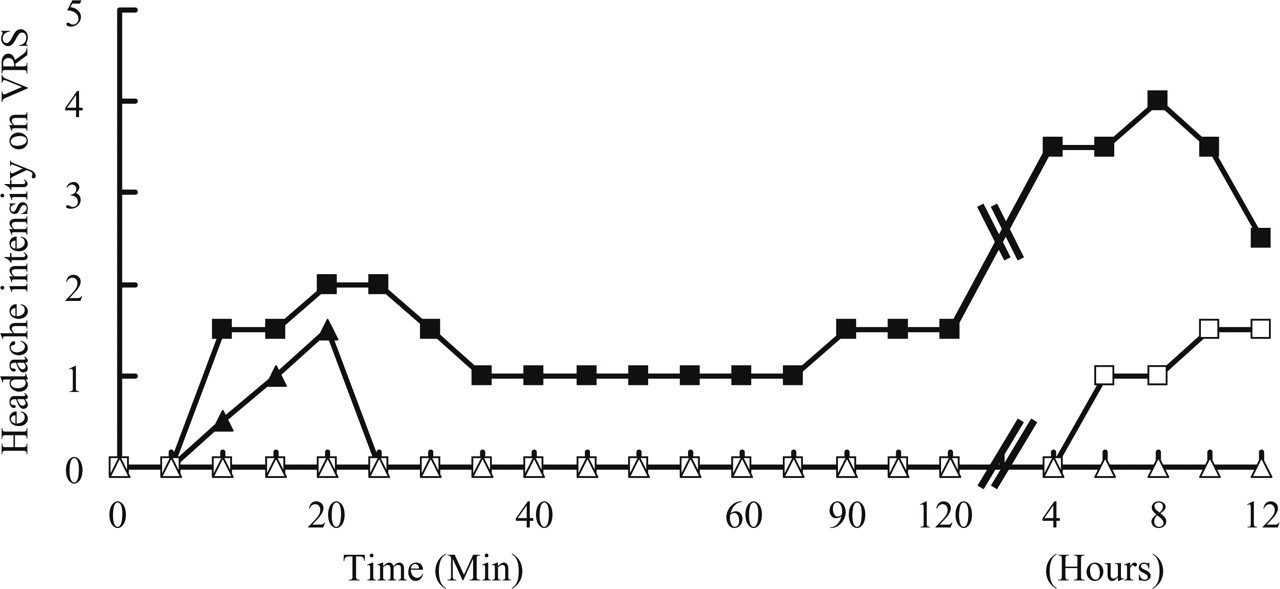
Median headache intensity over time during (20 min) and after infusion of glyceryl trinitrate (GTN ▪,▴) and placebo (□,▵) in 16 patients with chronic tension-type headache (▪,□) and in 16 healthy subjects (▴,▵). Headache was scored on a 10 point verbal rating scale (VRS). In patients and controls, the area under the headache curve (intensity × duration) was significantly higher on the GTN day than on the placebo day (P < 0.01). The patients developed significantly stronger headache than healthy controls both during the first hour (immediate headache) (P = 0.02) and during the subsequent 11 h (delayed headache) (P = 0.008). (Modified from Ashina et al. 2000 (1), by permission of Oxford University Press)
Citrulline and arginine levels in patients
Plasma levels of citrulline increased significantly after infusion of GTN compared to placebo (P = 0.01). Plasma levels of arginine were unchanged (P = 0.12) (Table 2). There was no difference in baseline values on GTN day vs. placebo day (P = 0.15) (Fig. 2a).
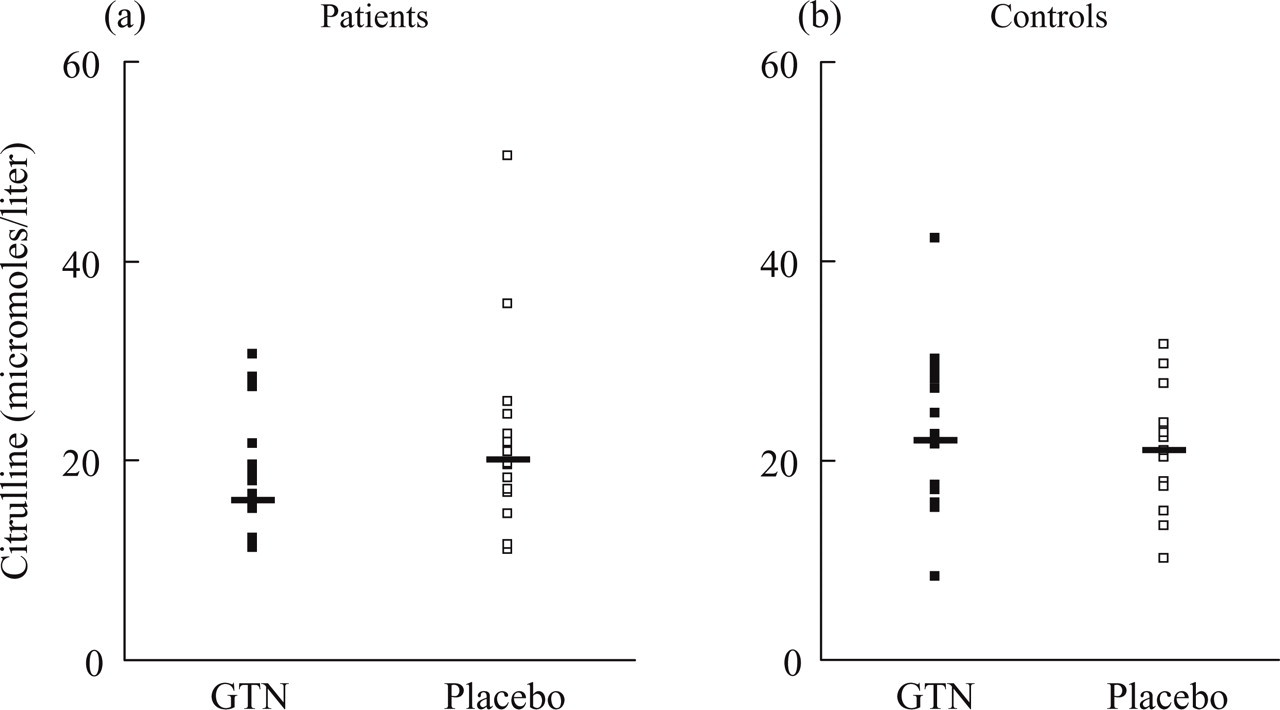
Plasma levels of citrulline at baseline on GTN (▪) and placebo (□) days in (a) patients and (b) controls. We found no difference in baseline values on GTN day vs. placebo day in either patients or controls (P > 0.05). Thick line shows median concentrations.
Plasma levels of citrulline and arginine in patients after infusion of GTN and placebo
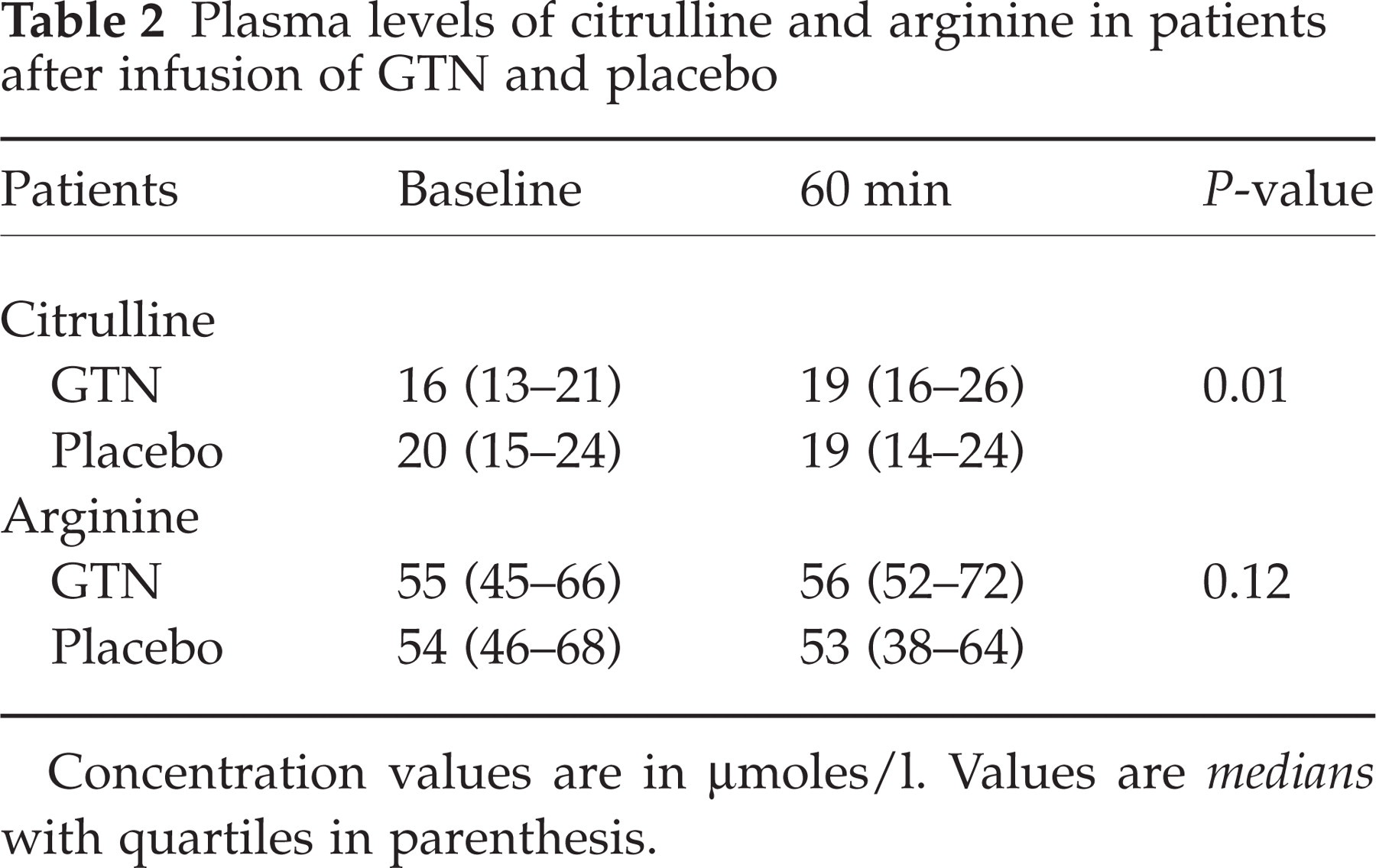
Concentration values are in µmoles/l. Values are medians with quartiles in parenthesis.
Citrulline and arginine levels in controls
There was no difference in changes of plasma citrulline (P = 0.50) and arginine (P = 0.18) after infusion of GTN compared to placebo (Table 3). There was no difference in baseline values on GTN day vs. placebo day (P = 0.41) (Fig. 2b).
Plasma levels of citrulline and arginine in controls after infusion of GTN and placebo
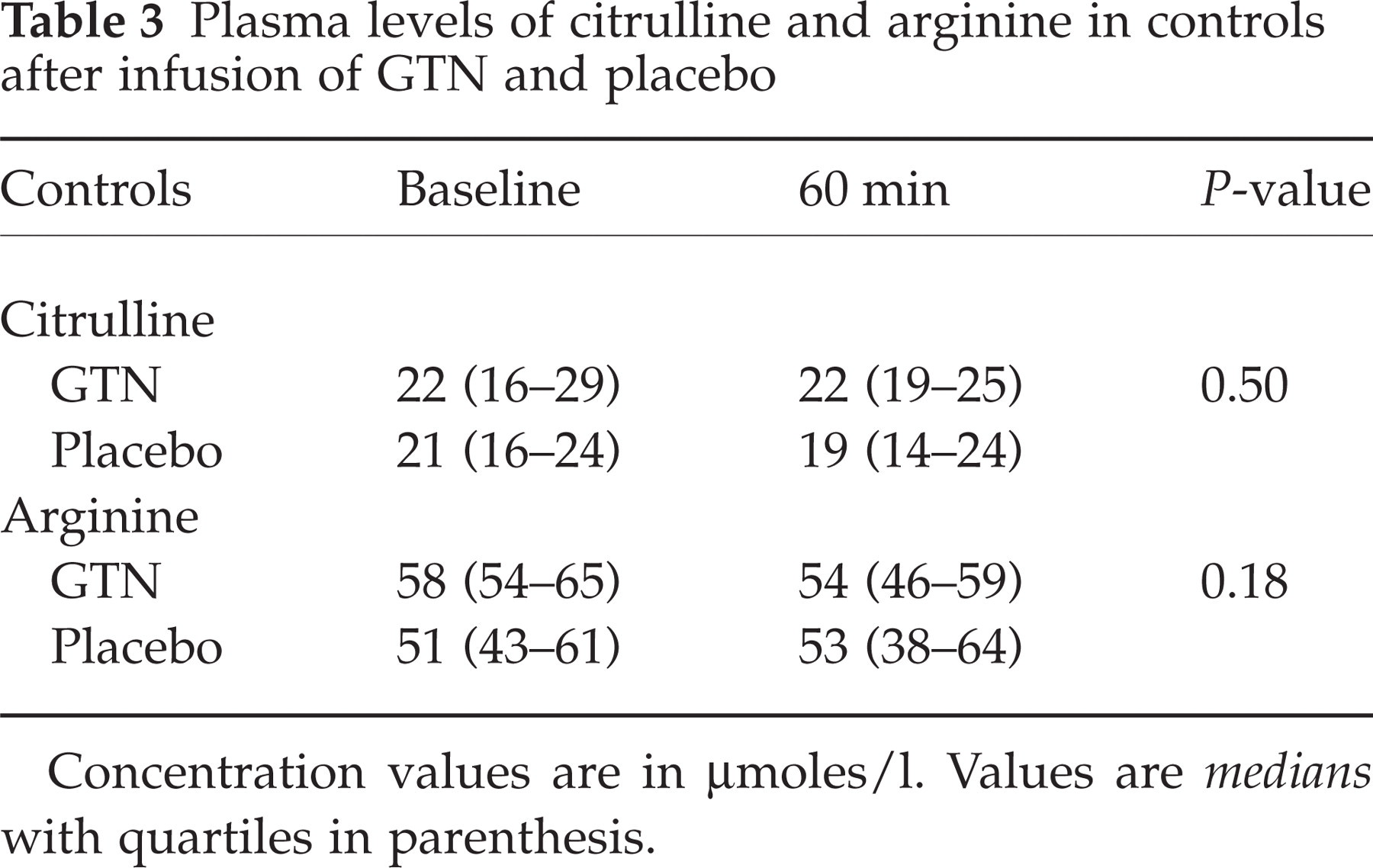
Concentration values are in µmoles/l. Values are medians with quartiles in parenthesis.
Citrulline and arginine levels in patients vs. controls
There was a trend toward difference in changes of plasma citrulline (P = 0.12) after infusion of GTN and a significant difference in changes of plasma arginine (P = 0.047) between patients and controls.
Discussion
The major outcome of the present study is increased plasma levels of citrulline after GTN infusion in patients with chronic tension-type headache but not in controls. Our findings suggest that exogenous NO leads to enhanced NO production in patients with chronic headache. No significant changes in arginine were observed within the groups. If changes were solely due to activation of the NO pathway, an increase of the citrulline concentration would be expected to be mirrored by an identical molar decrease in arginine. However, haemolysis can cause the release of erythrocytic arginase, which catalyses the conversion of arginine to ornithine. Varying degrees of haemolysis might thus explain the failure to detect significant differences between GTN and placebo in the levels of arginine. It would seem that the most reliable amino acid marker of activation of the NO pathway is citrulline. Studies in rats and in humans have shown that after intravenous administration of GTN very little drug remains in the blood and that the majority of the GTN is distributed to tissues (7). In anaesthetized cats, intravenous infusion of GTN (0.25 mg/kg/min over 20 min) induces a significant increase of pial artery diameter and a prolonged (60 min) increase of NO in the brain (pial middle cerebral artery perfusion territory) (8). In another study, subcutaneous administration of GTN in rats induced an increase of soluble guanylyl cyclase (sGC) expression and activity in dural blood vessels after 20–30 min (9). The authors concluded that an up-regulation of the NO target enzyme contributes to the pathogenesis of GTN induced immediate headache. Interestingly, the time profile of up-regulation of sGC corresponds with the time of peak headache intensity during GTN induced immediate headache in patients and healthy controls. Reuter et al. (10) reported dose-dependent inducible nitric oxide synthase (NOS) up-regulation in dura mater beginning at 2 h and an increase in the corresponding protein expression at 4, 6 and 10 h after intravenous infusion of GTN. In addition, Pardutz et al. (11) reported that subcutaneous GTN produced a significant increase of NOS in the cervical part of trigeminal nucleus caudalis in rats after 4 h. Thus, these data indicate that GTN infusion may trigger increased endogenous production of NO in the CNS by activation of L-arginine-NO pathway. Furthermore, data from animal studies suggest that we would have observed further increase if samples had been collected 6 or 8 h later, i.e. during delayed headache.
The chronic pain state in patients with chronic tension-type headache may be due to central sensitization at the level of the spinal dorsal horn/trigeminal nucleus induced by prolonged nociceptive input from pericranial myofascial tissues (12). Experimental studies in animals have demonstrated a prolonged increase of NO levels in the spinal dorsal horn during central sensitization (13). During central sensitization both endogenous and exogenous nitric oxide induce c-fos (an immediate-early gene), which can further activate the production of other substances in the CNS (14). It has been demonstrated that inhibition of NOS results in reduction of spontaneous headache in patients with chronic tension-type headache (15). These data suggest that enhanced activation of NO pathway is already present in presensitized patients with chronic tension type-headache and that further increase activation of NO pathway after endogenous NO would be detectable only in patients. It has been shown that NO may activate or sensitize nociceptors around blood vessels directly (16) or by dilatation (17). Lambert et al. (18) reported that intracarotid arterial infusion of GTN lead to sensitization of first and second order neurons of craniovascular sensory pathways. Cumberbatch et al. (19) showed that dilatation of meningeal blood vessels in rats causes sensitization of central trigeminal neurons and facilitation of convergent sensory responses. Taken together these data indicate that sustained NO-induced vascular nociception may lead to central sensitization and subsequent convergence of nociceptive input from blood vessels and myofascial tissue. It is therefore possible that excessive vascular nociception caused by GTN may gradually augment the sensitizing effect of pre-existing myofascial input in chronic tension-type headache sufferers (20). In this case pre-existing facilitation of distinct nociceptive central pathways in chronic tension-type headache sufferers (myofascial pathways) may be enhanced by NO-induced dilatation and sensitization of sensory afferents around vessels. This could explain why NO induces stronger immediate headache in patients than in healthy subjects when no pre-existing sensitization is present.
In summary, we suggest that intravenous infusion of GTN may trigger enhanced endogenous NO production in patients with chronic tension-type headache resulting in NO-induced arterial dilatation and activation of perivascular nociceptive afferents.
Footnotes
Acknowledgements
We would like to thank Mrs Hanne Andresen and Neel Honoré for skilful technical assistance. The University of Copenhagen supported the study financially.
