Abstract
The aetiology of primary cough headache (PCH) is obscure. The aim of this study was to investigate the magnetic resonance (MR) morphometric characteristics of the posterior cranial fossa (PCF) in patients with PCH. Eighteen consecutive patients with PCH (14M/4F, mean age 75.1 + 6.0 years) and 18 sex- and age-matched control subjects were recruited for study. Based on the midline sagittal MR images, parameters indicating posterior fossa crowdedness were measured. Compared with controls, patients with PCH had a similar size of hindbrain tissue area but a significantly smaller PCF area, resulting in a higher mean hindbrain/PCF ratio (0.78 + 0.04 vs. 0.73 + 0.06, P = 0.005). In addition, these patients also had a lower position of the cerebellar tonsillar tip, a shorter clivus length and shorter distances from the clivus to the mid-pons and from the basion to the medulla than the control group. Patients with PCH were associated with a more crowded PCF, which might be a contributing factor for the pathogenesis of this headache syndrome.
Introduction
Primary cough headache (PCH), previously termed benign cough headache, is a distinct syndrome of headache of sudden onset, lasting from one second to 30 min, brought on by and occurring only in association with coughing, straining and/or Valsalva manoeuvers, and not attributed to another disorder, according to the second edition of the International Classification of Headache Disorders (coded as 4.2) (1). It is relatively rare, but important to recognize as effective treatment, such as indomethacin or lumbar puncture, is available (2).
Until the mid 1950s, cough headache and exertional headache had been regarded as ominous symptoms. Then, a study in 1956 reported on 27 similar patients who demonstrated the occurrence of a brief headache that was precipitated by coughing, and often also by other exertions such as sneezing, straining during bowel movements, laughing or stooping (3). This group of disorders was labelled as ‘cough headache’. The study concluded that a syndrome of PCH existed in patients without evidence of structural lesions (3).
The aetiologies of symptomatic cough headache include Chiari malformation type 1 (CM-1), posterior fossa space-occupying lesions, platybasia/basilar impression, intracranial haemorrhage and brain tumours (4–6). Therefore, MRI of the brain must be performed in every patient with cough headache to exclude possible structural lesions (2). CM-1 is the most common of these intracranial abnormalities (6–8). For example, Pascual et al. (6) reported 30 patients with cough headache. Seventeen of them were symptomatic and all of them were secondary to CM-1. In addition, cough headache occurs in up to 30% of the patients with CM-1 (7, 8). Besides CM-1, posterior cranial fossa (PCF) lesion is another frequent cause of symptomatic cough headache (5).
The underlying mechanism of PCH is even more obscure. Although patients with PCH do not have tonsillar herniation, their posterior fossa has never been investigated. We hypothesized that patients with PCH might also be associated with a crowded PCF. Therefore, we conducted an MRI study to investigate the degree of crowdedness of the PCF in patients with PCH in comparison with age- and sex- matched control subjects.
Methods
Patients and control subjects
Eighteen consecutive patients with PCH (14 men, 4 women; mean age 75.1 ± 6.0; range 62–88 years) and 18 age- and sex-matched control subjects were recruited for this study from the Taipei Veterans General Hospital. The PCH patients’ demographics and headache profiles are illustrated in Table 1.
Clinical presentations of the patients with primary cough headache
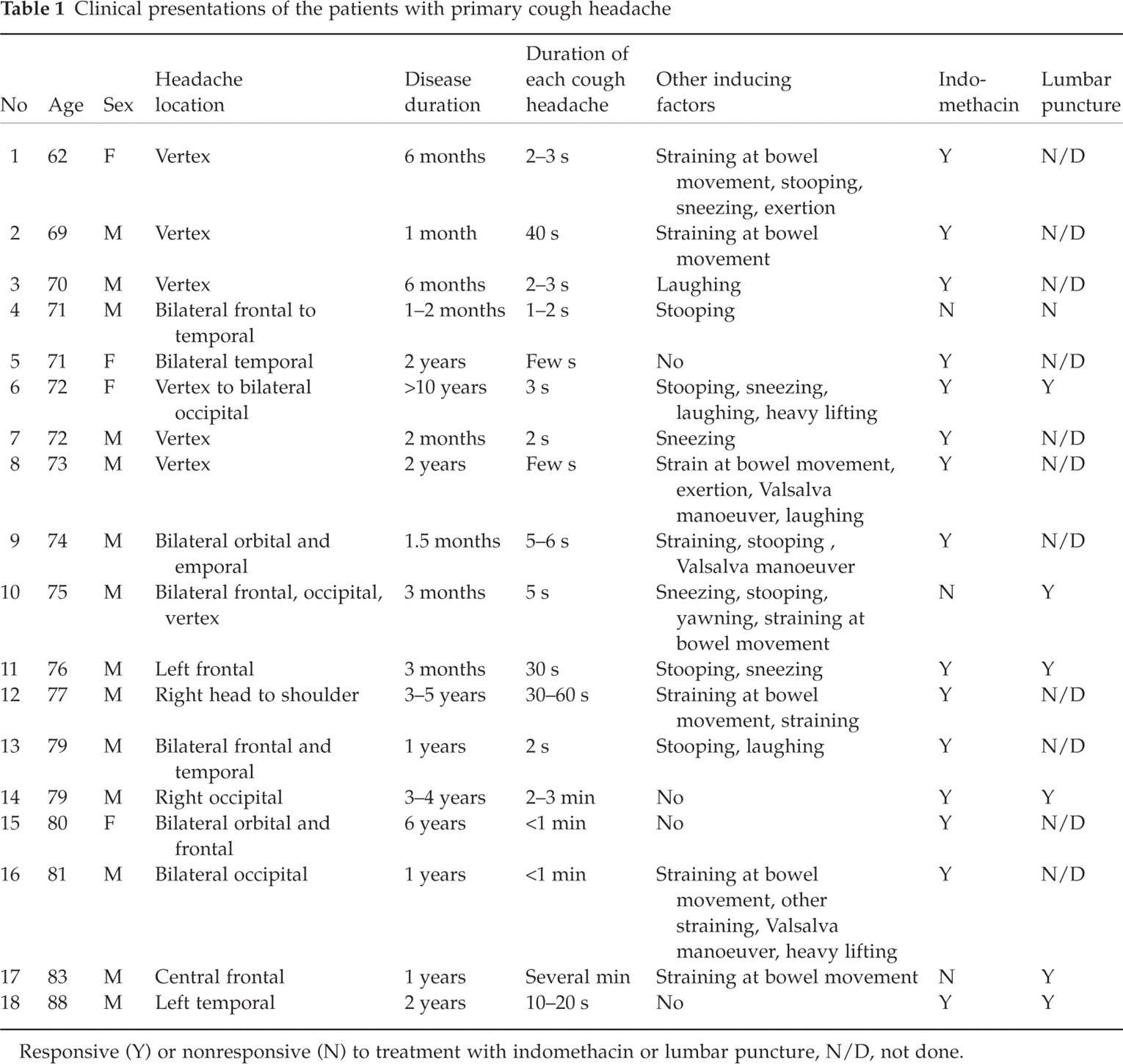
Responsive (Y) or nonresponsive (N) to treatment with indomethacin or lumbar puncture, N/D, not done.
The patients’ clinical presentations fulfilled the criteria of PCH proposed by the International Headache Society (1). Four patients had unilateral headaches and two patients had headaches persisted for longer than one minute. In addition to coughing, 14 (77.8%) patients had other exertional factors to induce headaches. None of the patients had CM-1, as indicated by tonsillar herniation ≥ 3 mm below the foramen magnum (9, 10), or other organic lesions demonstrated by their brain MRI. Fifteen of the 18 (83.3%) patients were responsive to indomethacin and 6 (85.7%) of 7 patients were responsive to lumbar puncture. Some patients have been reported in a previous study (11).
The control subjects were recruited from the health check-up subjects in the High-Tech Imaging Centre of the hospital. This imaging centre provides a series of whole body MRI studies including brain MRI and MRA as part of a routine health check-up. None of the control subjects had clinical histories of chronic headache including cough or exertional headache, stroke, dementia, hydrocephalus (Evan's index (12) (maximal width of frontal horns/maximal width of inner skull) > 0.30) or other significant CNS illnesses.
Magnetic resonance imaging
The structures of the brain were investigated using a 1.5 Tesla medical MRI system (General Electric, Signa CV/i, Milwaukee, WI, U.S.A; Siemens, Magnetom Vision, Erlangen, Germany). The data matrix was 256 × 256, the pixel size was 0.94 × 0.94 mm (corresponding to a field of view of 24 cm), and the number of acquisitions was 2. Spin-echo T1-weighted images were obtained using a repetition time of 500–600 ms and an echo time of 15–40 ms. Fast spin-echo T2-weighted images were obtained using a repetition time of 3000–4000 ms and an echo time of 80–100 ms. The axial, coronal and sagittal images were obtained with 5-mm slice thickness and 2.5-mm gap.
Measurements
Mid-sagittal T1-weighted MR images, which clearly demonstrated the aqueduct, anterior and posterior commissures, were selected for measurement. The parameters indicative of posterior fossa crowdedness that were measured were: PCF area, area of the hindbrain tissue, lengths of the supraocciput and the clivus, position of the cerebellar tonsillar tip, distances from the clivus to the mid-pons and from the basion to the medulla, and the angles between the cerebellar tentorium to the Twining’s line and to the supraocciput. All measurements were performed on a workstation with the input of the patient imaging data transmitted through optical discs or an intranetwork.
The area of PCF in the mid-sagittal MR image was defined as the area encircled by the tentorium, clivus and occipital bones, the line drawn from the opisthion to the basion, and the line drawn from the top of the dorsum sella to the top end of the tentorium (Fig. 1). The area of the hindbrain tissue in PCF was defined by encircling the brain tissue inside the posterior fossa area, including the forth ventricle. The ratio of the areas of hindbrain tissue vs. PCF was used to represent the crowdedness of the PCF (Fig. 1).

Measurements of posterior cranial fossa (PCF) characteristics using T1-weighted sagittal magnetic resonance (MR) imaging. The area indicating PCF is encircled by the outer line. The area of the hindbrain in PCF is encircled by the inner line.
The position of the tonsillar tip was measured from the lower tip of the cerebellar tonsil to the line drawn from the opisthion to the basion (foramen magnum). A designation of positive indicated that the cerebellar tonsillar tip descended below the line. A designation of negative meant that the tonsillar tip ascended above the line. The length of the supraocciput was defined by measuring the line length from the internal occipital protuberance to the opisthion. The length of the clivus was similarly calculated, by determining the distance between the top of the dorsum sellae and the basion. The distance of the prepontine and premedullary cistern spaces was defined by measuring the distances between the mid-pons to the clivus, and from the medulla to the basion (Fig. 2).
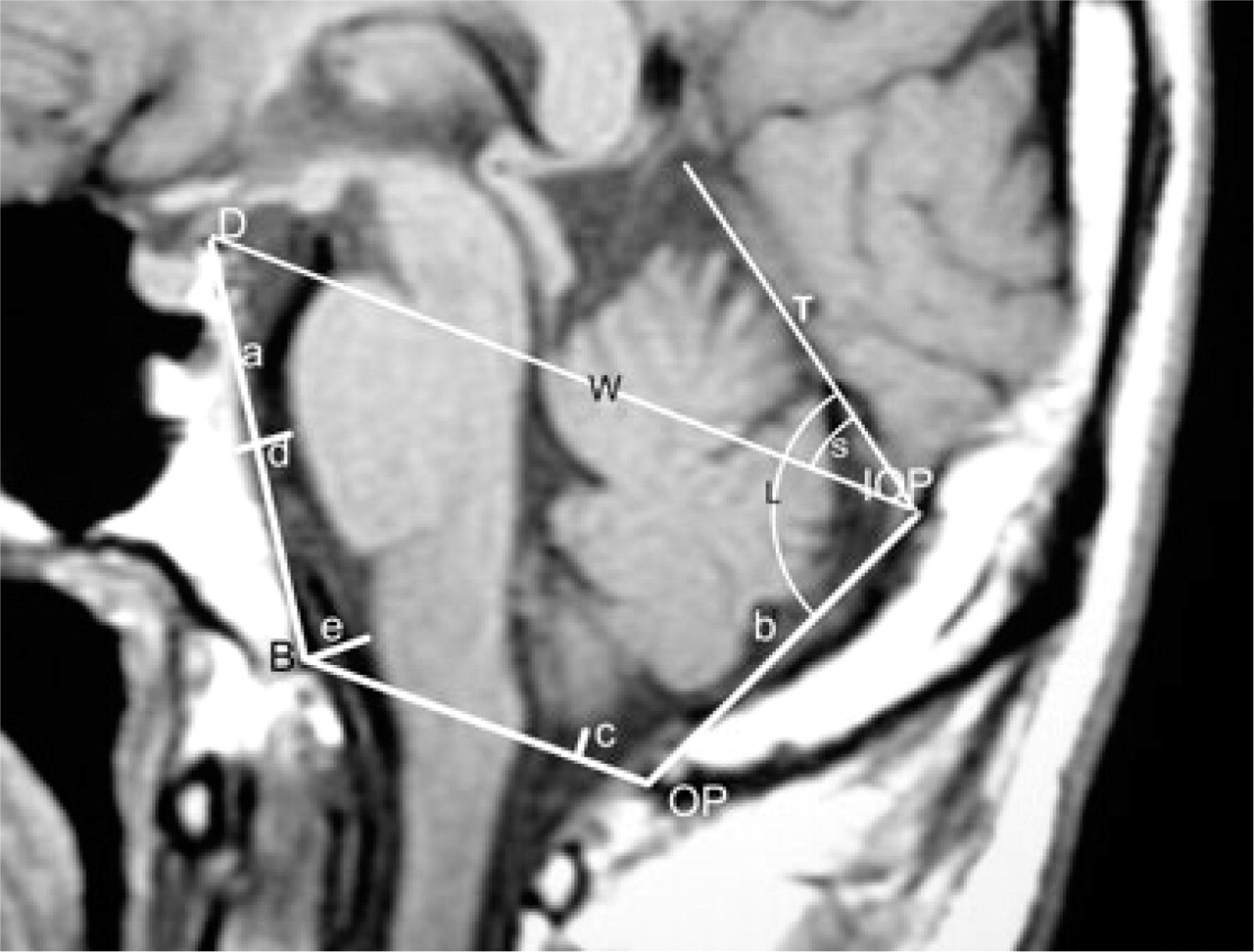
Positional measurements. The length of the clivus (a) was measured along a line drawn from the top of the dorsum sella (D) to the basion (B). The length of the supraocciput (b) was measured along the line from the internal occipital protuberance (IOP) to the opisthion (OP). The descent of the cerebellar tonsil (c) was measured by the distance of the cerebellar tonsillar tip above (recorded as negative) or below (recorded as positive) the line from opisthion to basion. The distances of prepontine (d) and premedullary (e) spaces were defined as the distance from mid-pons to the clivus, and from medulla to the basion, respectively. The slopes of the tentorium were the angles between the cerebellar tentorium (T) and the Twining's line (W) (angle S), and between the cerebellar tentorium (T) and the supraocciput (angle L).
The tentorium slope was presented by the angle between the cerebellar tentorium and the Twining's line (13), and the angle between the cerebellum tentorium and the supraocciput (14) (Fig. 2).
Test-retest reliability
The parameters for PCF were measured by one neuroradiologist (LJF) who was blind to the clinical information. Test-retest reliabilities were investigated in a pilot study of 16 subjects with measurements taken two days apart. The reliability was good for each parameter, with a determined Pearson correlation coefficient (r) = 0.87–0.93 (P < 0.001).
Statistical methods
Student's t-test and χ2 test were used for comparisons between the patient and control groups. Pearson correlation was used to examine the test-retest reliabilities and the correlations of these measured parameters for posterior fossa crowdedness. Two-tailed P-value of less than 0.05 was considered statistically significant.
Results
Posterior cranial fossa area
Patients with PCH had a significantly smaller mean PCF area (3164.9 ± 233.8 mm2) as compared to the control group (3510.6 ± 250.4 mm2) (P < 0.001) according to the mid-sagittal T1 MR images. In contrast, the mean hindbrain area in the PCF did not differ significantly between the patient group (2463.1 ± 200.0 mm2) and the control group (2553.3 ± 217.0 mm2) (P = 0.20). Therefore, the patients with PCH had a significantly higher ratio of hindbrain to PCF area (0.78 ± 0.04) than did the controls (0.73 ± 0.06) (P = 0.005) (Fig. 3).

Comparisons between the patients with primary cough headache and controls. (a) The area of hindbrain in the PCF and the area of PCF. (b) The ratio of the hindbrain/PCF. (c) Two tentorium angles: between tentorium-supraocciput, and tentorium-Twining's line. (d) The position of the tonsil tip (+ below, – above the line of foramen magnum). (e) The lengths of the suprapocciput and the clivus (f) The distances between the mid-pons to the clivus (prepontine cistern) and the medulla to the basion (premedullary cistern) (Bars are mean ± SD).
Clivus and supraocciput length
Clivus length was shorter in patients with PCH (41.9 ± 2.7 mm) in comparison to the control subjects (46.6 ± 3.7 mm) (P < 0.001). However, there was no significant difference in the length of supraocciput between the two groups (PCH patients: 41.1 ± 5.5 vs. controls: 41.8 ± 4.6 mm, P = 0.68). Patients with PCH had a significantly lower position of the cerebellar tonsillar tip above the line of foramen magnum (1.3 ± 2.4 mm) than did the control subjects (4.4 ± 4.5 mm) (P = 0.012) (Fig. 3).
Distance
Patients with PCH had a shorter distance from the clivus to the mid-pons (5.2 ± 1.5 mm) than did the controls (7.2 ± 1.5 mm) (P = 0.001). Likewise, PCH patients displayed a shorter distance from the basion to the medulla than did the control subjects (6.6 ± 2.1 vs. 8.2 ± 1.6 mm, P = 0.014) (Fig. 3).
Tentorial angles
There were no significant differences in the degree of the two tentorial angles (the cerebellar tentorium to the Twining's line and to the supraocciput) between two groups.
Correlation between hindbrain/PCF ratio and other parameters
The correlation analysis showed that the hindbrain/PCF ratio was correlated with PCF area (r = −0.49, P = 0.002), the degree of tonsillar descent in relation to the foramen magnum(r = 0.61, P < 0.001), the clivus length (r = −0.40, P = 0.015), and the prepontine distance (r = −0.54, P = 0.001)
Others
The mean Evan's ratio did not differ between the patient and control groups (patients: 0.26 ± 0.02; controls: 0.27 ± 0.03, P = 0.225).
Discussion
Our study showed patients with PCH had comparable size of hindbrain tissue area but had a smaller PCF area, as compared with control subjects. This smaller area produced a higher hindbrain/PCF area ratio. The observations of a shorter clivus length, lower tonsillar tip, and narrower prepontine and premedullary cisterns also support the contention that PCF was more crowded in patients with PCH. Similar findings have been reported in patients with CM-1 (13, 14). Previous studies have indicated that patients with CM-1 have an underdeveloped small PCF (14–19), normal developed hindbrain (13, 14), and a higher hindbrain/PCF volume ratio and other crowdedness parameters (13, 14). In the present study, the degree of the PCF crowdedness, CSF space decrease and displacement of the hindbrain in the PCH patients were not as severe as those observed in CM-1 patients. This might explain the discrepancy of the severity of the symptoms between the two groups of patients.
The pathophysiology of cough headache in patients with CM-1 has been proposed. Craniospinal pressure dissociation following the Valsalva maneuver has been demonstrated as a pathophysiology for cough headache in patients with CM-1, which was resolved after suboccipital decompression (20). Sansur et al. (21) suggested that cough headache in patients with CM-1 is related to the absolute value of intrathecal pressure reached during coughing. They suggested that the sudden increased intrathecal pressure was caused by ‘obstruction’ to the free flow of CSF in the subarachnoid space (21). Several morphometric studies have shown that a small size of the posterior fossa is a common feature in patients with CM-1 (14–19). One study of a large group of ‘symptomatic’ patients with suspected CM-1 found an obliteration of the CSF spaces posterior and lateral to the cerebellum was the most constant finding (100%) followed by tonsillar herniation (91%) (14). Therefore, posterior fossa crowdedness, CSF space decrease, as well as obstruction of the CSF flow at foramen magnum might play a role in such headache syndrome in patients with CM-1.
Unlike CM-1, the underlying mechanism of PCH is much more obscure. Few hypotheses have been proposed for the mechanisms of cough pain in patients with PCH (2, 22). It seems intuitive that cough pain is associated with a transient increase of the intracranial pressure (ICP) during cough (2). PCH patients might have a lowered threshold for pain associated with the increased ICP normally caused by coughing (2, 22). The response to indomethacin in patients with PCH might result from the decrease of ICP (2, 22–24). Raskin (22) proposed that PCH might be provoked by a sudden increase in ICP and alleviated by a sudden decrease in ICP that accompanies lumbar puncture. The nature of the receptors that are sensitive to such ICP alterations is probably key to understanding PCH (22). Our study provides a new approach to investigate the underlying mechanism of PCH and offers the first evidence of a possible relationship between PCH and CM-1. However, in patients with PCH, there is no complete CSF obstruction at the foramen magnum. Moreover, it is presently unknown if there is craniospinal pressure dissociation, and whether the dissociation could underlie cough headaches. Based on the evidence presented, we offer the suggestion that the crowded posterior fossa in patients with PCH might cause a ‘relative obstructive CSF pathway’ around the craniocervical region. This, in turn, could contribute to a higher ICP during cough.
In conclusion, the present study supports the contention that a structural anomaly, in the form of a crowded PCF in some patients with PCH, might be a contributing factor to the development of CSF disturbances during coughing.
Footnotes
Acknowledgements
This study was supported in part by a grant from Veterans General Hospital-Taipei (VGH 331).
