Abstract
Triptans are commonly used anti-migraine drugs and show agonist action mainly at serotonin 5-HT1B/1D/1F receptors. It is not known whether frequent or long-term treatment with these drugs would alter 5-HT receptor function. We investigated the effects of protracted (14-18 days) sumatriptan and zolmitriptan treatment in rats on 5-HT1 receptor mRNA expression and function in tissues related to migraine pathophysiology. RT-PCR analysis revealed that 5-HT1B/1D/1F receptor mRNA was reduced in the trigeminal ganglion after treatment with either triptan (reduction by: sumatriptan 39% and zolmitriptan 61% for 5-HT1B; 60% vs 41% for 5-HT1D; 32% vs 68% for 5-HT1F). Sumatriptan attenuated 5-HT1D receptor mRNA by 49% in the basilar artery, whereas zolmitriptan reduced 5-HT1B mRNA in this tissue by 70%. No change in 5-HT1 receptor mRNA expression was observed in coronary artery and dura mater. Chronic triptan treatment had no effect in two functional assays [sumatriptan mediated inhibition (50 mg/kg, i.p.) of electrically induced plasma protein extravasation in dura mater and 5-nonyloxytryptaminestimulated [35S]guanosine-5′-O-(3-thio)triphosphate binding in substantia nigra]. Furthermore, vasoconstriction to 5-HT in isolated basilar artery was not affected by chronic triptan treatment, while it was slightly reduced in coronary artery. We conclude that, although our treatment protocol altered mRNA receptor expression in several tissues relevant to migraine pathophysiology, it did not attenuate 5-HT1 receptor-dependent functions in rats.
Introduction
Anti-migraine drugs such as non-steroidal anti-inflammatory drugs (NSAIDS), ergot alkaloids and triptans, can lead to drug-induced headache in subjects with a primary headache disorder (e.g. migraine) (1, 2). Interestingly, patients respond favourably to these drugs prior to the development of drug-induced headache. This phenomenon accounts for 8–10% of patients in specialized headache clinics and about 1% of the general population, but its underlying pathophysiological mechanisms are not known (3). The most obvious possibilities are receptor desensitization, as demonstrated, for instance, in cancer pain following treatment with opioids, or altered receptor expression following recurrent agonist exposure (4). Both phenomena could lead to a vicious circle in susceptible individuals resulting in increased drug intake without pain relief. Epidemiological data from a Danish study in migraineurs showed a reduced response rate (33%) to sumatriptan in frequent users in contrast to low-peak users (56%), indicative of a loss of drug effect in the aforementioned population (5). In contrast, studies in episodic cluster headache patients with an overuse of sumatriptan (greater than three injections of 6 mg/day) did not reveal tachyphylaxis or tolerance in this population (6).
In a large retrospective study, sumatriptan has been shown to account for 10% of drug-induced headaches between 1993 and 1999 (7). While most commonly used anti-migraine substances bind to a variety of receptors, triptans such as sumatriptan bind preferentially to 5-HT1B/1D/1F receptors (8, 9) and thus offer the possibility of studying the effects of chronic agonist treatment on a limited number of potential targets. To our knowledge, no animal studies have been performed to investigate mechanisms which may contribute to the development of drug-induced headache, or to assess the functional effects of repeated treatment with abortive anti-migraine drugs.
Although widely and successfully used for the treatment of primary headache disorders, it is not known whether a frequent use of 5-HT1B/1D/1F receptor agonists can lead to reduced receptor mRNA expression, receptor desensitization or a change in receptor functional properties. Therefore, we investigated the effects of 5-HT1B/1D agonist treatment in rats injected repeatedly with zolmitriptan or sumatriptan and determined whether this treatment led to reduced 5-HT1B/1D/1F receptor mRNA expression in tissues related to migraine pathophysiology or pharmacotherapy, such as the dura mater, trigeminal ganglion and cerebral and cardiac blood vessels [for review see (10)]. To assess the functional consequences of frequent agonist treatment, we studied the contractile reactivity of the basilar and coronary arteries in an in vitro preparation, the efficacy of sumatriptan to reduce plasma protein extravasation, a well-established marker of trigeminal nerve activation in the dura mater and GTPγS binding in substantia nigra.
Materials and methods
Experimental animals
Animal experiments were approved by the Massachusetts General Hospital, Committee on Research, Subcommittee on Research Animal Care. Male Sprague Dawley rats (150–170 g; Charles River, Wilmington, MA, USA) were injected intraperitoneally twice a day with either sumatriptan (300 µg/kg), zolmitriptan (100 µg/kg) or vehicle. All substances were dissolved in 25%β-cyclodextrin in saline (NaCl 0.9%) and a volume of 100 µl was injected each time. Although osmotic pumps would have resulted in stable drug levels we decided to inject the rats twice a day in order to mimic the typical treatment paradigm of a patient. We also chose this treatment regimen because these triptan doses have been found to fully inhibit neurogenic dural plasma extravasation and c-fos expression in the trigeminal nucleus, two models predictive for anti-migraine efficacy (11–13). Rats were weighted every third day to adjust the drug dose for weight change. The treatment was performed for 14–18 days and animals were not injected the evening before sacrifice. Sumatriptan was kindly provided by GlaxoSmithKline and zolmitriptan by AstraZeneca.
Rt-pcr
Rats were perfused transcardially with 250 ml cold normal saline, the dura mater, trigeminal ganglia (TG), basilar and coronary arteries were dissected out and immediately frozen in liquid nitrogen. Total RNA from vessels was isolated using a RNeasy Mini Kit (Qiagen, Valencia, CA, USA) while the RNA from TG and dura mater was isolated using the Trizol (Gibco, Carlsbad, CA, USA) extraction method according to the manufacturers’ instruction. Concentration and purity of RNA was determined by spectrophotometry (Beckman Spectrophotometer BU 640) and gel electrophoresis, respectively.
RNA was reverse-transcribed to first-strand DNA with an Omniscript™ kit kit (Qiagen), using random priming. One microlitre of cDNA was used for polymerase chain reaction (PCR). cDNA was amplified in a programmable thermal cycler (MJ Research, Cambridge, MA, USA). The reaction mixture contained 1 µl cDNA, 200 µM dNTP, 10 × reaction buffer, 25 U RedTaq DNA Polymerase (Sigma, St Louis, MO, USA), 18.75 µl H2O and 200 µM deoxynucleotides. The PCR primer sequences were: 3′-CCAG GTGGTCTGCGACTTCT-5′ sense and 5′-AGAG GACGTGGTCGGTGTTC-3′ antisense for r5-HT1B receptors, 3′-CTCTCCAATGCCTTCGTACT-5′ sense and 5′-GGCCATACAGGATAATGAGC-3′ antisense for r5-HT1D receptors and 3′-AGGATGAAAAC CAGTT-5′ sense and 5′-CATAATCCAACTCTCTCT CAC-3′ antisense for r5-HT1F. PCR was initiated for all receptors with a 5-min denaturation at 95°C for 5 min followed by cycles of 45 s at 95°C, 30 s at 60°C and 45 s at 72°C (40 cycles for 5-HT1B; 42 cycles for 5-HT1D; 41 cycles for 5-HT1F). PCR products were separated by gel electrophoresis using a 1% agarose gel in TAE buffer containing ethidium bromide and visualized with an imaging system (MCID M4, Imaging Research, St Catharines, Ont., Canada). The relative amounts of 5-HT1B/D/F mRNA under different conditions were determined by dividing the optical intensity of the bands for the 5-HT1B/D/F PCR products by the intensity of the band for the housekeeping gene GADPH. Primers for GADPH were 3′-TAAAAGGGCATCCTGGGCTACACT-5′ sense and 5′-TTACTCCTTGGAGGCCATGT-3′ antisense and PCR was initiated with a 5-min denaturation at 95°C and 29 cycles through 94°C for 30 s, 55°C for 15 s., and 72°C for 1 min.
PCR products from trigeminal ganglia tissue for all receptors investigated in this study were sequenced. PCR products were purified with Qiaquick PCR Purification Kit (Qiagen, Hilden, Germany) and sequenced with dye (Cy5) primer chemistry using Primer Cycle Sequencing kit (Amersham Biosciences, Braunschweig, Germany) in combination with an automated ALFexpress DNA sequencer (Pharmacia Biotech, Freiburg, Germany). The sequences were compared with entries in GenBank using the BLAST program. For each product, the correspondence to the targeted gene was confirmed (data not shown).
Plasma protein extravasation
Rats were anaesthetized with sodium pentobarbital (50 mg/kg; i.p.) and prepared as described previously (14). Briefly, 50 µCi [125I]-BSA (New England Nuclear, Boston, MA, USA) were injected into the femoral vein. Rats were placed into a stereotaxic frame, the skull was exposed and two burr holes were carefully drilled above the trigeminal ganglia 2.8 mm posteriorly and 3.2 mm laterally to bregma. Two stainless steel bipolar electrodes were lowered 9.2 mm from the dura in the ganglia and the right trigeminal ganglion was stimulated for 5 min (5Hz, 5 ms, 1.2 mA, World Precision Instruments, Hartford, CT, USA). Animals were killed 5 min after stimulation by a 2-min transcardiac perfusion with normal saline at a constant perfusion pressure of 100 mmHg. The dura was dissected out, weighted and counted for radioactivity in a gamma counter (Cobra II, Packard Instruments, Downers Grove, IL, USA). Radioactivity (cpm/mg dura wet weight) was expressed as a ratio between the stimulated and unstimulated side.
In these experiments, sumatriptan was administered 15 min before ganglion stimulation. First, we determined a dose–response for plasma protein inhibition following sumatriptan pre-treatment (50, 75, 100 µg/kg i.v.; n = 5 per group) in naive rats. As 50 µg/kg sumatriptan reduced leakage submaximally, this dose was selected to investigate the effect of sumatriptan on plasma protein extravasation in chronically treated animals (n = 6 per group).
Isolated arteries
Rats were anaesthetized with chloroform (Fisher Scientific, Pittsburgh, PA, USA) and killed by decapitation. The dura mater and trigeminal ganglia were dissected out, frozen in liquid nitrogen and further processed for RT-PCR as described above. The brain and heart were removed immediately and immersed in physiological solution (composition, in mM: NaCl, 118; KCl, 4.6; NaHCO3, 25; MgSO4, 1.2; KH2PO4, 1.2; CaCl2, 2.5; glucose, 10; EDTA, 0.025; pH 7.4 at 37°C). The basilar and coronary arteries were dissected out under a microscope. Arterial segments (1.5–2-mm long) were threaded onto 40-µm stainless steel wires and mounted in a isometric myograph (610 M, Danish Myo Technology, Denmark). Each preparation was equilibrated unstretched, for 30 min, in physiological solution at 37°C and aerated with a gas mixture of 95% O2– 5% CO2. The normalized passive resting force and the corresponding diameter were then determined for each preparation from its own length-pressure curve, according to Mulvany and Halpern (15). Contractile responses were recorded using a data acquisition and recording software (Myodaq and Myodata, Danish Myo Technology). After normalization and 30-min equilibration in physiological solution, the preparations were stimulated with a 100-mM KCl-depolarizing solution (composition, mM: NaCl, 22.6; KCl, 100; NaHCO3, 25; MgSO4, 1.2; KH2PO4, 1.2; CaCl2, 2.5; glucose, 10; EDTA, 0.025, pH 7.4 at 37°C). After wash-out and 30-min recovery, they were exposed to cumulative concentrations of serotonin (5-HT, 1 nM– 10 µM) or the 5-HT1 receptor agonist 5-carboxamido, tryptamine (5-CT 1 nM– 10 µM; Sigma-RBI, Natick, MA, USA) (16).
Autoradiography
Autoradiography was performed in brain tissue from rats that were used for RT-PCR studies. [35S]GTPγS binding (New England Nuclear, Boston, MA, USA) was visualized as described (17). Brainstem tissue sections 10 µm-thick were incubated for 15 min at room temperature in 50 mM HEPES (pH 7.5) containing 100 mM NaCl, 1 mM MgCl2, 0.2 mM EGTA and 0.3 mM DTT and for a further 15 min in the same buffer supplemented with 2 mM GDP. Agonist-stimulated binding was determined by incubating the sections for 60 min at 30°C in buffer containing 2 mM GDP, 0.1 nM[35S]GTPγS and the appropriate concentration of the selective 5-HT1B/1D receptor agonist 5-nonyloxytryptamine (Sigma-RBI). Non-specific binding was assessed by including 10 µM unlabelled GTPγS in the incubation buffer and was not significantly different from film background (not shown). Slides were washed in ice cold 50 mM HEPES (containing 100 mM NaCl, 1 mM MgCl2, 0.2 mM EGTA), dipped in ice-cold distilled water, dried under a stream of cold air and exposed to Hyperfilm βmax for 48 h. [35S]GTPγS binding was assessed by measuring the optical density of the autoradiograms over selected brain regions using a computerized image analysis system (MCID M4, Imaging Res. Inc., Canada). 14C-polymer standards (Amersham, Piscataway, NJ, USA) were used for calibration. Agonist-induced [35S]GTPγS binding is expressed as percentage of basal binding.
Statistical analysis
Data are expressed as mean values ± S.D. for all data except results from isolated artery studies which are expressed as mean values ± S.E. Comparisons between groups were performed using one way ANOVA followed Turkey's protected t-test. Statistical significance was assumed when P < 0.05.
Results
Rt-pcr
The effect of daily injections with zolmitriptan (n = 9) and sumatriptan (n = 9) (twice a day for 14–18 days) on mRNA expression for 5-HT1B/1D/1F receptors was investigated in various tissues relevant to migraine pathophysiology and/or pharmacotherapy (Figs 1a–e). The PCR reaction products showed a single band of the expected size for each receptor. In the trigeminal ganglion, sumatriptan and zolmitriptan, significantly reduced mRNA expression for 5-HT1B/D/F receptors compared with vehicle treated animals (n = 8; P < 0.05). In basilar artery, 5-HT1B receptor mRNA was reduced following zolmitriptan, while sumatriptan treatment reduced the mRNA for 5-HT1D receptors; although sumatriptan also decreased 5-HT1B mRNA in the basilar artery, statistical significance was not reached (P = 0.07). Chronic sumatriptan or zolmitriptan application did not cause any significant changes in coronary arteries for the 5-HT1 receptor subtypes investigated. In dura mater 5-HT1F receptor mRNA could not be detected using two different sets of primers and 5-HT1D mRNA was increased after sumatriptan treatment compared with vehicle and zolmitriptan treatment.

5-HT1B/D/F mRNA expression in trigeminal ganglion (a), basilar artery (b), coronary artery (c) and dura mater (d) after a 14–18-day-treatment period with sumatriptan (300 µg/kg, i.p.; n = 9), zolmitriptan (100 µg/kg, i.p., n = 9) or vehicle (i.p., n = 8) twice a day. Data are shown as relative amounts of 5-HT1B/D/F mRNA, as determined by dividing the optical intensity of the bands for the 5-HT1B/D/F PCR products by the intensity of the band for the housekeeping gene GADPH; ♯ indicates statistical significance compared with control (P < 0.05). (e) Representative RT-PCR product images for each receptor studied (and the housekeeping gene GADPH) in the trigeminal ganglion after vehicle and triptan treatment.
Trigeminal ganglion stimulation
Unilateral electrical stimulation of the trigeminal ganglion increased leakage of [125I]BSA within ipsilateral dura mater in naive animals. The extravasation ratio between stimulated and control sides was 1.69 ± 0.13 (n = 6). Sumatriptan pre-treatment reduced the ratio dose–dependently to 1.25 ± 0.05 (50 µg/kg sumatriptan; P < 0.05; n = 6) and 1.07 ± 0.10 (100 µg/kg sumatriptan; P < 0.05; n = 6) in naive rats (Fig. 2). Because we expected desensitization of 5-HT1 receptors or reduced receptor expression following chronic triptan treatment, and thus a rightward shift in the response to acute sumatriptan administration after TG stimulation, a dose of 50 µg/kg sumatriptan was chosen for further experiment. Sumatriptan administration (50 µg/kg) resulted in a ratio of 1.30 ± 0.05 (n = 6) in animals chronically treated with vehicle, which was not different from the effects in naive rats. This ratio was not changed in rats treated chronically with sumatriptan [1.21 ± 0.06 (n = 6)] or zolmitriptan [1.27 ± 0.06 (n = 6)] (P > 0.05 for all acute sumatriptan treatment groups with respect to control; Fig. 2).

Sumatriptan dose-dependently (25, 50, 100 µg/kg i.v.; 15-min prior stimulation, n = 6 per group) attenuates plasma protein extravasation in rat dura mater induced by electrical stimulation of the trigeminal ganglion. Note that a dose of 50 µg/kg sumatriptan reduces plasma protein leakage by approximately 50%. The protracted sumatriptan (300 µg/kg; i.p.) and zolmitriptan (100 µg/kg; i.p) pre-treatment twice a day does not alter the abortive effect of sumatriptan (50 µg/kg; i.p.) on plasma protein extravasation (n = 6 per group). Data are expressed as a ratio of counts per minute per mg tisssue wet weight (cpm/mg) on the stimulated side and counts per minute per mg tisssue wet weight on the contralateral side. ∗P < 0.05 compared with control.
Isolated artery studies
The contractile response to 100 mM KCl did not significantly differ between groups.
Basilar arteries
5-HT and 5-CT evoked contractile responses in a concentration-dependent manner in all treatment groups (Fig. 3). Maximal contractile responses to 5-HT (n = 8 for all treatment groups) were slightly higher than that induced by 100 mM KCl, while the responses to 5-CT (n = 8 for all treatment groups) reached only about 50% of the KCl response. There were no differences in basilar arteries for the pD2 or the maximal contractile responses to 5-HT or 5-CT between vehicle, zolmitriptan and sumatriptan treated rats (Figs 3a and b).
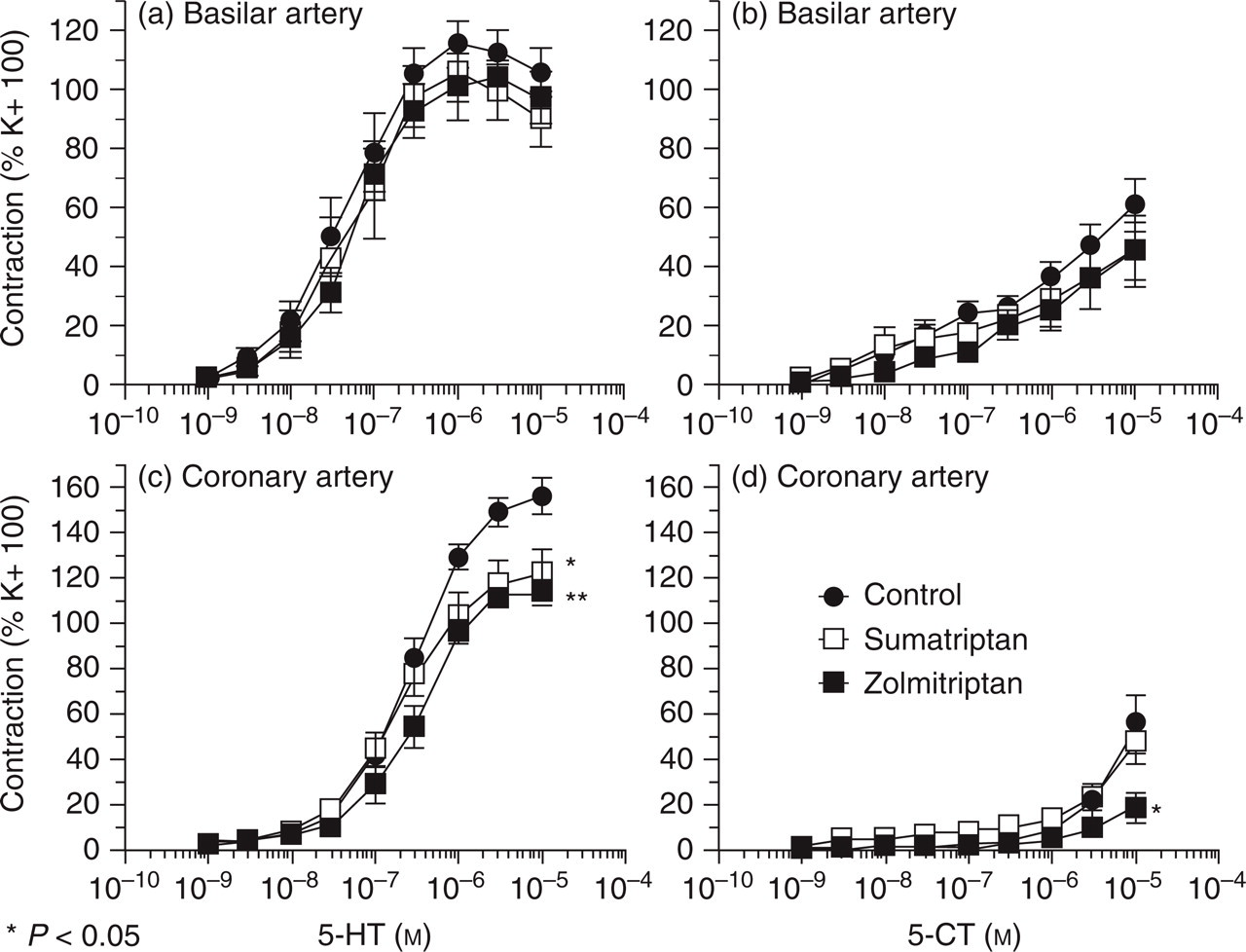
5-HT (a, c) and 5-CT (b, d) induced vasoconstriction in rat coronary and basilar artery after protracted sumatriptan (300 µg/kg; i.p.), zolmitriptan (100 µg/kg; i.p.) or vehicle treatment. In basilar artery, the contractile response to 5-HT and 5-CT is not different between treatment groups. In coronary artery, the maximum contractile response to 5-HT is attenuated after treatment with either triptan. In contrast, protracted zolmitriptan treatment leads to a reduced contractile response to 5-CT; (∗P < 0.05; vs control). Each curve represents the mean ± standard error of eight different preparations in basilar artery and 10 different preparations in coronary artery.
Coronary arteries
In vehicle-treated rats, the maximal contractile response to 5-HT was 156.7 ± 7.7% to that induced by 100 mM KCl (n = 10). The maximal contraction to 5-HT in zolmitriptan-(114.9 ± 5.1%; n = 10) and sumatriptan-treated rats (123.4 ± 9.5%; n = 12) was significantly reduced compared with vehicle-treated rats (n = 10; P < 0.05 for both treatment groups; Fig. 3c). However, the pD2 values (nM) were not different between vehicle and triptan treatment groups (vehicle: 6.6 ± 0.1, zolmitriptan 6.6 ± 0.1, sumatriptan 6.7 ± 0.1).
The contraction to 5-CT application on coronary arteries was weak and resulted in a maximal contractile response of 55.6 ± 12.7% (n = 10, vehicle) and 49.4 ± 8% (n = 10, sumatriptan) compared with the 100 mM KCl response. In zolmitriptan-treated rats, the response to 5-CT was markedly reduced (18.5 ± 6.7% of KCl; n = 10) compared with vehicle and sumatriptan treatment (P < 0.05 vs vehicle and sumatriptan; Fig. 3d). The pD2 values for 5-CT were not different among the treatment groups.
In vitro autoradiography
Increasing doses of the 5-HT1B/1D receptor agonist 5-nonyloxytryptamine stimulated [35S]GTPγS binding in substantia nigra in a concentration-dependent manner in all treatment groups (Fig. 4). The maximal stimulatory effects in vehicle-, sumatriptan- and zolmitriptran-treated groups were 81 ± 9%, 97 ± 10% and 69 ± 8% with respective pEC50 values of 6.6 ± 0.2, 6.8 ± 0.2 and 6.6 ± 0.2 (– log nM). Chronic treatment with zolmitriptan or sumatriptan did therefore not significantly alter maximal [35S]GTPγS binding and EC50 values in substantia nigra (n = 6 each group).

Effect of increasing concentrations of the 5-HT1B/1D receptor agonist 5-nonyloxytryptamine on [35S]GTPγS binding in substantia nigra. Data are expressed as per cent increased [35S]GTPγS binding over basal binding (error bars, mean ± standard deviation values observed between different animals (n = 6 per group). Maximal stimulations of [35S]GTPγS binding are 81 ± 9%, 97 ± 10% and 69 ± 8% in rat substantia nigra following vehicle, sumatriptan and zolmitriptan treatment, repectively (P > 0.05).
Discussion
In this study, we investigated the effects of repeated triptan administration on 5-HT1 receptor function and mRNA expression in tissues related to migraine pathogenesis. We chose a 2-week treatment period because this regimen has previously been found sufficient to show alterations of other 5-HT receptor subtypes (18–20). We have shown that sumatriptan and zolmitriptan administration can affect the expression of 5-HT1B/1D/1F mRNA in these tissues. In contrast, none of the 5-HT1 receptor-mediated functions was altered after the treatment period (14–18 days), with the exception of 5-HT1-mediated constriction of coronary arteries. In particular, inhibition of plasma protein extravasation by sumatriptan was not attenuated following recurrent 5-HT1B/D receptor agonist treatment. Mean contractile responses of basilar arteries to 5-HT or 5-CT, and agonist induced GTPγS binding in substantia nigra (an index of central 5-HT1B receptor coupling to G proteins) were likewise not altered. Our findings indicate that a protracted sumatriptan or zolmitriptan treatment does not alter the functions of 5-HT1B/1D/1F receptors, and therefore would not result in a loss of clinical efficacy.
Blood vessels
Basilar artery
RT-PCR studies found a consistent trend to a decrease in 5-HT1 receptor mRNA after chronic triptan treatment (see Fig. 1b), whereas the contractile properties of basilar arteries to 5-HT and the more selective 5-HT1 receptor agonist 5-CT were not altered. 5-HT elicits vasoconstriction in rat basilar arteries by stimulation of a mixed receptor population of 5-HT2 and 5-HT 1B receptors (21) in agreement with our findings that the selective 5-HT1 receptor agonist 5-CT only constricts rat basilar artery to a fraction of 5-HT's maximal contractile response. Moreover, a similar discrepancy between 5-HT1 receptor mRNA and 5-HT1-mediated effects has been recently observed by Hansen-Schwartz et al. in vessels maintained in organ culture. In this study, staurosporine inhibited the culture induced up-regulation of the response of 5-HT1B/1D receptors, but had no significant effect on the mRNA levels of these receptors (22).
There could be two likely explanations for the fact that the observed decrease in 5-HT1B mRNA was not accompanied by a loss of contractile activity. The first one is that the loss of mRNA was not paralleled by a corresponding decrease in receptor protein. It is also possible that the stimulation of only a fraction of the vascular 5-HT1B receptors by full agonists such as 5-HT and 5-CT results in maximal response in control basilar arteries (receptor reserve). If this is the case, a preserved contractile response would be observed, even in the presence of a reduced density of 5-HT1B receptors after protracted triptan treatment.
Coronary artery
Vasoconstriction in rat coronary arteries is inhibited by the 5-HT2 receptor antagonist ketanserin, but not by the 5-HT1 receptor antagonist l-propranolol, indicating that this response is mostly 5-HT2 receptor mediated (23). In line with a 5-HT2-dependent mechanism, the EC50 of 5-CT was lower in coronary than in basilar artery, and much lower than that of 5-HT. Treatment with either triptan reduced the maximal contractile response to 5-HT, while the maximal response to 5-CT was only decreased after chronic zolmitriptan. The effect of triptan administration on 5-HT-mediated coronary constriction is unlikely to be mediated by direct action at 5-HT2 receptors, because triptans have virtually no affinity for these sites (12, 24). An action at another receptor, such as 5-HT1A, which triptan can activate at therapeutic doses (25), is also unlikely as 5-HT1A receptor mediated constriction only accounts for a few per cent of the 5-HT mediated contriction in rat coronary artery (23). Because zolmitriptan is a more potent constrictor than sumatriptan in the coronary artery of several species (26, 27), we speculate that zolmitriptan may have a higher potency to desensitize the re-ceptor subtype (probably 5-HT1B), mediating cons-triction to 5-CT in this vessel. Interestingly, a desensitization to 5-HT was detected after treatment with both triptans. This could be because the contractile response to this agonist was more robust, rendering the effect of chronic treatment more detectable. It should be noted that chronic administration of neither triptan resulted in a detectable reduction of 5-HT1B/D/F receptor mRNA. Therefore, if changes in coronary contractile response to 5-HT1 receptor agonist are indeed mediated by 5-HT1B receptors, these alterations are likely to occur via post-translational mechanisms, or at the level of receptor coupling. As recurrent triptan administration should have led to chronic coronary constriction, it is also possible that the treatment resulted in the down-regulation of one or several molecules involved in the signalling of vasoconstricting agents.
Plasma protein extravasation
We have previously shown that, in rats, prejunctional receptors of the 5-HT1B subtype block dural plasma extravasation after electrical stimulation of the trigeminal ganglion (11). As demonstrated previously, sumatriptan dose-dependently inhibited plasma protein extravasation in dura mater following electrical trigeminal ganglion stimulation (14). Because we expected a down-regulation of 5-HT1B receptors after recurrent triptan administration, we chose to test a single submaximally effective dose of sumatriptan (50 µg/kg). Inhibition of protein leakage was unchanged between all groups, indicating that repeated triptan administration did not affect the efficacy of sumatriptan to reduce plasma protein leakage. This finding is in line with clinical observations showing the efficacy of sumatriptan in multiple headache attacks, even when used repeatedly in a single day as observed in cluster headache patients (6).
The 5-HT1B/1D receptor mRNA expression was not reduced in dura mater after chronic treatment, which is again in accordance with the result of the functional study. In contrast to other tissues (e.g. trigeminal ganglion), we were not able to detect 5-HT1F receptor mRNA in dura mater using two different published primer pairs. We conclude that this receptor in the rat is expressed by trigeminal neurones, and not by meningeal blood vessels or other cell types present in this tissue, in agreement with previous findings (28, 29).
Both triptans reduced 5-HT1B/D/F mRNA in the trigeminal ganglion. In this and other tissues, it is possible that reduced mRNA did not result in significantly reduced protein expression as a result of slow turnover of the receptor protein. This possibility, or the existence of spare receptors, might explain why we observed an intact inhibitory effect of sumatriptan in the plasma extravasation paradigm in the presence of decreased 5-HT1B receptor mRNA.
[35S]GTPγS binding
In a third functional assay analysing neuronal 5-HT1B receptor coupling in substantia nigra, we did not detect any change in 5-nonyloxytryptamine-induced [35S]GTPγS binding following treatment with both triptans. The trigeminal nucleus would have been a brain region more relevant to migraine pathophysiology, as it is likely to be another important site of action of triptans (13, 30). However, this structure is not suitable for this assay because of its high level of basal [35S]GTPγS labelling (17). The substantia nigra was therefore selected as a surrogate brain region because of its large content in 5-HT1B receptors and its high signal-to-noise ratio in the [35S]GTPγS autoradiography technique (17). Although it could be argued that the lack of effect of sumatriptan treatment on 5-nonyloxytryptamine-induced [35S]GTPγS binding was due to its poor ability to penetrate the blood–brain barrier (BBB) under physiological conditions (31), zolmitriptan is known to cross the intact BBB (32).
The failure to detect a change in 5-HT1-receptor-dependent functions following triptan administration could be due the insufficient duration of the treatment. Although other studies indicate that a 2-week treatment period is sufficient to detect alterations in receptor densities or coupling properties (see above) (18, 19), we cannot rule out that a more protracted treatment would have resulted in detectable alterations of 5-HT1 receptor functions relevant to migraine therapy. Indeed, a 30-day treatment in rats with the 5-HT1A/5-HT1B receptor agonist RU 24969 significantly reduced the density of [125I]iodocyanopindolol-labelled 5-HT1B sites in cortex, hippocampus, striatum, brainstem, and spinal cord (33). The non-selective agonists (m-trifluoromethyl-phenylpiperazine [TFMPP], but not [1-(3-chlorophenyl)piperazine] (m-CPP) reduced the B max of cortical 5-HT1B sites. It is, however, unclear whether these effects were caused by a direct action on 5-HT1B receptors or by activation of another receptor subtype.
In summary, our findings do not provide evidence for a change in 5-HT1 receptor properties after a protracted triptan treatment (14–18 days) in intra- and extracerebral tissues important to migraine pathophysiology. These results are in agreement with the finding that cluster headache patients respond reliably to triptans, even after a treatment period of several months. In addition, the development of drug-induced headache in certain individuals does not appear to be accounted for by a desensitization of the trigemino-vascular 5-HT1 receptors to anti-migraine drugs, or endogenous inhibitory transmitters, at least not during the time frame we examined.
Footnotes
Acknowledgements
This study is supported by the International Headache Society (IHS)-Research Fellowship 1999 (UR), the Deutsche Forschungsgemeinschaft (DFG; Re-1316), and the CNR-NATO advanced fellowship (SS; 11.215.31, 28/05/98). CW is supported by the NIH grant NS043216.
