Abstract
In anaesthetized rats, extracellular recordings were made from neurones of the spinal trigeminal nucleus, involved in the processing of nociceptive input from the dura. Blockers of voltage-gated calcium channels (VGCCs) were administered topically to the exposed brainstem. Blockade of N-type (CaV2.2) channels reduced spontaneous activity and responses of the neurones to cold and chemical stimuli applied to the dura, suggesting that N-type channels regulate excitatory synaptic activation. Blockade of L-type (CaV1) channels enhanced spontaneous discharges of the neurones. Blockade of P/Q-type (CaV2.1) channels slightly decreased responses to chemical and cold stimuli but markedly increased spontaneous activity, an effect which was absent during concomitant application of GABA to the brainstem. The data suggest that P/Q-type VGCCs regulate a tonic synaptic inhibitory control of the brainstem neurones. The risk of migraine by genetic modifications of P/Q-type channels may thus be sought in disturbed inhibition in the network that processes nociceptive dura input.
Introduction
Since the pioneering studies of Penfield and McNaughton in 1940 (1) and Ray and Wolff (2) it is thought that the meninges are the origin of headache during migraine and that the trigeminovascular system is activated during migraine pain. There is evidence that primary dural afferents are sensitized in the course of migraine (3) and that peripheral sensitization may be accompanied by central sensitization, i.e. the development of hyperexcitability in nociceptive neurones in the trigeminal nucleus and possibly at other sites (4–6). Nociceptive neurones in the trigeminal nucleus are synaptically activated by dural and other meningeal afferents, but they seem also to be under the control of local inhibitory interneurones and descending inhibition (7, 8).
Voltage-gated calcium channels (VGCCs) trigger the release of transmitters and control numerous neuronal functions such as neuronal excitability. Different types of voltage-gated calcium channels (VGCCs, new nomenclature Cavx.x, see 9) have been identified by cloning and electrophysiological techniques (10–14). P/Q- (Cav2.1) and N-type (CaV2.2) channels trigger the release of transmitter from presynaptic endings. N-type calcium channels and also L-type (CaV1) calcium channels are located on the cell soma and dendrites and may thus regulate cell excitability (10–12). Interestingly, the P/Q-type channel came in the focus of headache research because recent reports suggested that humans suffering from a familial hemiplegic form of migraine have genetic modifications of P/Q-type channels (11, 15). Mutations of the CACNA1A gene on chromosome 19p13 lead to alterations of the α1A subunit of the P/Q type VGCC with consequences for the channel gating causing altered Ca2+ influx (16, 17). Currently, it is difficult to relate this finding to a defined neuronal mechanism of headache because the functional role of P/Q- and other VGCC-types in the trigeminal nociceptive system has not been explored.
This lack of knowledge led us to address the potential functional role of N-, P/Q- and L-type VGCCs in the nociceptive processing in the trigeminal nuclear complex in the brainstem. This is the first site at which synaptic processing of dural input takes place and where P/Q-type and other VGCCs could play a role. As an experimental approach, we tested how the topical application of specific VGCC blockers to the brainstem would influence the firing of neurones in the trigeminal nucleus with input from the dura. Specifically we were interested to see different effects of the application of blockers of N-, P/Q- and L-type channels.
In the spinal cord, different effects of these blockers on neuronal firing have been observed. Typically, spinal application of N-type blockers reduces the responses of spinal cord neurones to noxious stimuli applied to peripheral tissue and is thus anti-nociceptive (18). By contrast, blockade of P/Q-type VGCCs produces variable effects; they are, for example, dependent on whether nociceptive spinal cord neurones are synaptically activated by primary afferents from normal or inflamed tissue (19). The literature on the involvement of L-type channels in nociceptive processing is controversial (see 18).
In the present experiments, we recorded extracellularly from neurones in the spinal trigeminal nucleus with input from the dura in anaesthetized rats, and we applied specific VGCC blockers (11, 12) topically to the brainstem. Blockade of N-type VGCCs reduced spontaneous discharges and responses evoked by chemical and cold stimulation of the dura. Blockade of P/Q-type VGCCs led to an increase of spontaneous firing but reduced responses to chemical and cold stimulation. Blockade of L-type channels mainly increased spontaneous discharges. These data indicate differences in the role of VGCC in the network of trigeminal nociceptive processing. Implications for the generation of headache will be discussed.
Methods
Animal preparation
The present study was approved by the animal protection committee and the regional government of Thueringen (reg. 02–09/01). Sixty-seven male Wistar rats (350–450 g) were used for the experiments. The rats were anaesthetized with thiopentone sodium (Trapanal, Byk Gulden, Konstanz, Germany; initially 100 mg/kg i.p.). During dissection, animals were not paralysed and depth of anaesthesia was checked by testing for nociceptive reflexes to squeezing the tip of the tail and by testing the corneal blink reflex. Absence of the corneal blink reflex and of nociceptive reflexes was maintained by supplemental doses of 20 mg/kg i.p. The trachea, the left femoral vein and artery were cannulated. The head of the animal was fixed stereotactically, and the skin above the skull was removed.
The dura mater was exposed over the left hemisphere from bregma to lambda (from midline spanning 4–6 mm laterally) using a mini-drill under saline cooling and kept moist with Tyrode throughout the experiment. The atlanto-occipital ligament and the underlying spinal dura mater were incised and moved aside to expose the medulla oblongata (20). Dental acrylic was used to build a wall around the exposed dura to prevent accidental overflow of solutions. The muscles of the neck were separated in the midline.
After having completed the dissection, the animals were paralysed with pancuronium bromide (Pancuronium Organon, Organon Teknika, Eppelheim, Germany; 1 ml/kg i.v.) and artificially ventilated during the recording period. To monitor depth of anaesthesia throughout the recording period, the mean arterial blood pressure was continuously measured, and supplemental doses of 20 mg/kg thiopentone sodium were given when blood pressure showed even slight changes to stimulation of the dura and the facial receptive field (see below). Body temperature was kept at 37°C by a feed back-controlled heating system.
Small amounts (100 µl) of Tyrode solution were applied slowly to the surface of the brainstem with a micropipette at the most possible distance to the recording electrode. Before applying a VGCC blocker, the Tyrode was carefully removed with a fine thread made from filter paper or cotton wool.
Recording from neurones in the spinal trigeminal nucleus
For extracellular recordings of action potentials from single brainstem neurones, carbon fibre microelectrodes (5–10 MΩ) were used (see Fig. 1a). Amplified action potentials were stored and further processed on PC using the Spike/Spidi software (21). As a searching stimulus, we used mechanical probing of the cornea and the skin around the eye. This searching procedure was used because the vast majority of neurones with dura input has convergent inputs from both the cornea and the dura (22), and because we wanted to keep mechanical stimulation of the dura at a minimum prior to the testing protocol. Once a neurone that responded to corneal stimulation was isolated we searched for a receptive field of the neurone in the dura by gently probing the dura surface with a glass rod. A neurone was finally taken for study if it responded in addition to the application of noxious cold (ice spray on cotton pad) and/or a mixture of inflammatory mediators (IM) to the dura. The mixture consisted of serotonin, histamine, prostaglandin E2, bradykinin at 10−5 M each (pH 6.8) as used in many studies on nociceptors (23). This selection of neurones was used because it was necessary to test the effects of VGCC blockers on the responses to stimuli of different modalities.
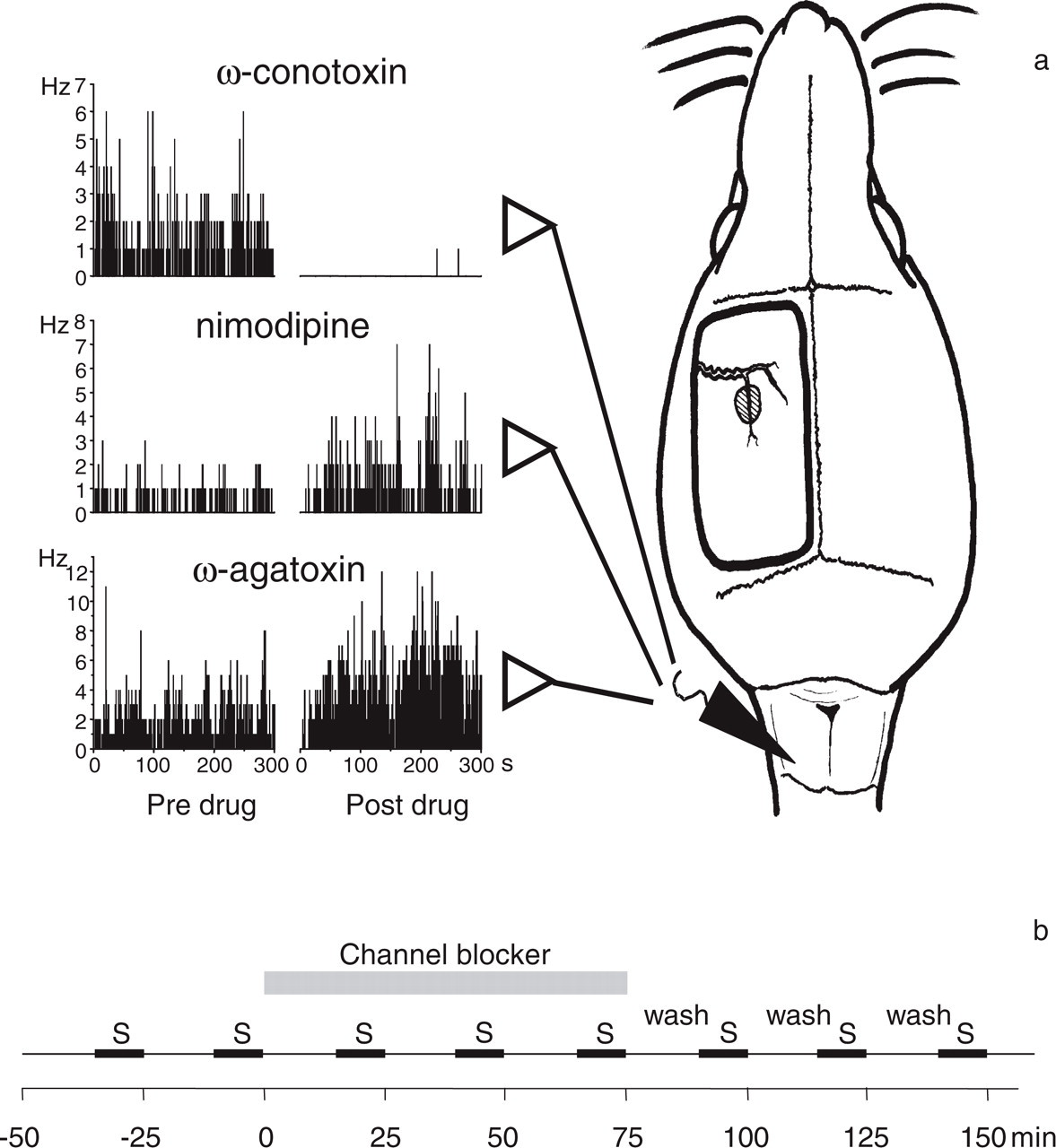
(a) Experimental set-up for recordings. For recordings of single cell activity, the brainstem was exposed and microelectrodes were inserted in the spinal trigeminal nucleus to monitor single cell action potentials. For stimulation of the dura, the bone was removed over the ipsilateral cortex and the dura was exposed. The receptive field on the dura of a neurone in the spinal trigeminal nucleus is shown by the shaded area. The traces at the left show peristimulus time histograms of the spontaneous activity of three different neurones and changes of the discharge rate after topical administration of calcium channel blockers to the surface of the brainstem. (b) Test protocol for the application of calcium channel blockers. S, stimulation protocol (see Methods).
Stimulation protocol
To assess the responses of the neurones, we used a sequence of different stimuli. In order to test for the responses to mechanical stimulation of the facial receptive field, the tip of the nose, the whiskers, the snout, the skin in front of the eye, the nictitating membrane, the upper lid, the lower lid and the cornea were gently probed with a blunt glass rod. At each site the mechanical stimulus was repeated five times. Then we tested responses to mechanical stimulation of the dura. A blunt glass rod was gently pressed five times to the receptive field of the neurone. This stimulus evokes a phasic response of the brainstem neurone (the neurones do not respond tonically to sustained mechanical stimulation). Then we tested the responses to cold and to inflammatory mediators. These stimuli were applied to the dura by using cotton pads. Because the application of the cotton pad to the dura may stimulate the receptive field mechanically, we first laid a cotton pad that was soaked with Tyrode pH 7.4 to the dura for 15 s. For application of noxious cold, we then used an iced cotton pad for 15 s. Thereafter, the dura was stimulated with cotton pads soaked with Tyrode and with IM for 30 s, respectively. After each application of a test solution, the dura mater was rinsed three times with Tyrode pH 7.4. After having tested the responses to dural stimulation, the cornea was subsequently stimulated with noxious cold and IM in the same way. The whole testing protocol lasted approximately 10 min.
Test protocol for the application of calcium channel blockers
The times of the stimulation protocol and drug application are shown in Fig. 1(b). When a neurone had been chosen for recording, the spontaneous activity was recorded for 15 min. Then the first stimulation protocol (S) was performed, followed by a second stimulation protocol 30 min after the start of the first one. While the recording was continued, the Tyrode solution on the brainstem was replaced by 100 µl of a solution containing one of the VGCC blockers. The following VGCC blockers were used: the N-type VGCC blocker ω-conotoxin GVIA 10−6 M in 165 mM NaCl, pH 7.4 (Bachem, Heidelberg, Germany), the L-type VGCC blocker nimodipine 10−5 M in 165 mM NaCl, pH 7.4 (Research Biochemicals Inc., Natick, MA, USA), and the P/Q-type VGCC blocker ω-agatoxin IVA 10−6 M in 165 mM NaCl, pH 7.4 (Bachem, Heidelberg, Germany). After each stimulation protocol, the VGCC blocker solution was renewed.
Spontaneous activity was recorded for 15 min, followed by a stimulation protocol. This schedule was repeated twice. Afterwards, the drug was washed thoroughly from the brainstem and replaced by Tyrode solution. During the following wash-out period, three further stimulation protocols were performed (Fig. 1b). In control animals (n = 14) Tyrode instead of a calcium channel blocker was applied to the brainstem.
In a subset of experiments, a cotton pad soaked with procaine hydrochloride (Procaine® 2%, Jenapharm, Jena, Germany) was laid onto the dura or applied to the brainstem following ω-agatoxin application. In 11 animals, 100 µl of GABA 10−5 M (Sigma, Deissenhofen, Germany) was dripped to the surface of the brainstem after characterizing the neurones twice (baseline period) for three stimulation periods. Then a mixture containing of GABA 10−5 M and ω-agatoxin IVA 10−6 M was administered to the brainstem for a further three stimulation periods.
Data evaluation
The action potentials were discriminated by shape and amplitude with the Spike/Spidi software (21) to assure that activity was measured from one and the same neurone throughout the recording. Spontaneous activity was assessed by counting the action potentials of a neurone for at least 5 min prior to the start of each stimulation protocol. For each mechanical stimulus the action potentials within 1.5 s were counted and responses from the same region were averaged within one stimulation period. This mean value was corrected by subtracting the mean spontaneous neuronal activity of the time interval before this stimulation protocol. Responses to cold and chemical stimulation were quantified during the application period, and the activity during the preceding control stimulus (cotton pad soaked with Tyrode) was subtracted to exclude activity due to possible mechanical stimulation. If a neurone did not respond to a stimulus, this difference could be zero or even negative. Neurones which responded during searching to a certain stimulus but whose response to this stimulus in the baseline period (mean of responses prior to application of the VGCC blocker) had values ≤ 0 were not included in the data evaluation for this stimulus.
For normalization the baseline was subtracted from each response. Statistical comparisons were calculated between the baseline and the values during drug application. The first significant value after application of the VGCC blocker is indicated in the figures. The Wilcoxon's matched pairs signed rank test was used and significance was accepted at P < 0.05.
Results
Extracellular recordings in the spinal trigeminal nucleus
Fifty-seven neurones were recorded in the transition zone between the trigeminal nucleus caudalis and C1 (Vc/C1), and 11 neurones were recorded in the transition zone between the nucleus interpolaris and the nucleus caudalis (Vc/Vi). The neurones in the brainstem were recorded at a mean depth of 842 µm in Vc/C1 and of 1621 µm in Vc/Vi. All but three neurones were spontaneously active. All neurones included in the data evaluation responded to mechanical stimulation of the dura with a blunt glass rod as well as to the application of inflammatory mediators (IM) and cold stimuli to the dura. Additionally, the neurones had facial receptive fields of different size, but the eye was always included. Neurones of the same type had already been described in detail elsewhere (20, 24).
Spontaneous activity
In control rats, 14 neurones were recorded. All of them were spontaneously active (10.8 ± 12.2 Hz). During the whole recording period (200 min) the spontaneous activity of the neurones did not change significantly (Fig. 2a). The effect of ω-conotoxin applied to the brainstem was assessed in seven spontaneously active neurones. Following application of ω-conotoxin, the discharge frequency of the neurones declined progressively. Figure 1(a) displays the peristimulus time histogram of a neurone showing that after ω-conotoxin the spontaneous discharges had almost disappeared. Figure 2(b) displays the summarized data from all neurones. Each bar shows the averaged spontaneous discharges in intervals of 15 min, as per cent of the pre-drug baseline (set 100%). Between 50 and 65 min after application of ω-conotoxin, the mean spontaneous activity of the neurones had decreased by 56 ± 15% (mean ± S.E.M.). The lowest activity was found in the following wash period in which the reduction reached statistical significance (P < 0.05, Wilcoxon matched pairs signed rank test). Recovery was not observed, in line with many studies using ω-conotoxin (18). In the testing of another eight spontaneously active neurones nimodipine, a blocker of the L-type VGCC, was applied to the brainstem, and a rapid increase of the ongoing discharges was consistently observed. A typical example is shown in Fig. 1(a). Fifty to 65 min after nimodipine application to the brainstem, spontaneous activity had increased on average by 99.4 ± 50.6% (mean ± S.E.M.) (Fig. 2c). The predominant effect of the topical application of ω-agatoxin to the brainstem was a continuous increase of resting activity in neurones with dura input. A specimen is shown in Fig. 1(a), and Fig. 2(d) displays the result obtained from 11 neurones. On average, spontaneous activity increased by 164.9 ± 79.9% (mean ± S.E.M.) 50–65 min after ω-agatoxin.
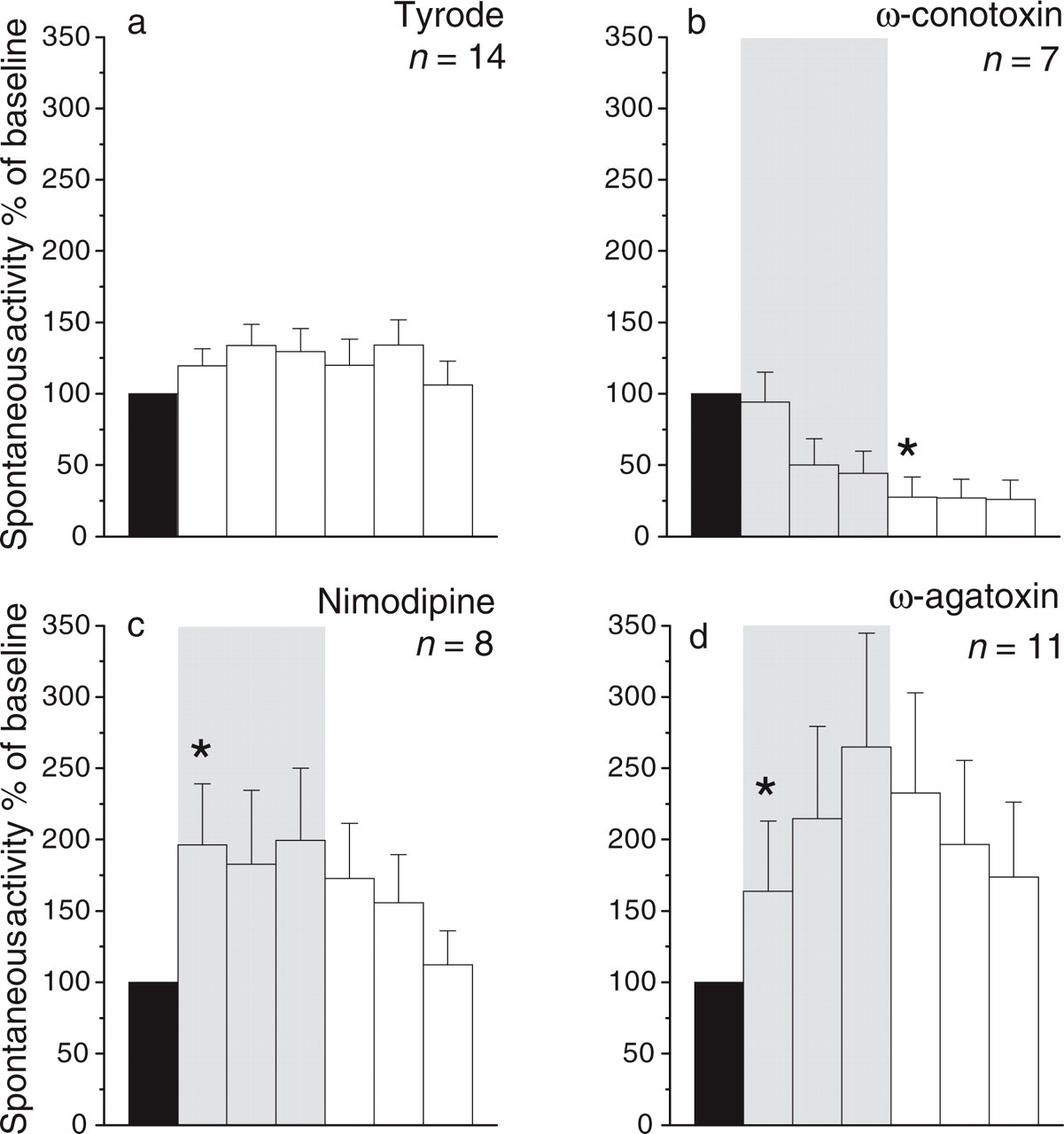
Spontaneous activity of brainstem neurones in control rats and in animals which were treated with VGCC blockers. The shaded area shows the application of a VGCC blocker to the brainstem. The discharge rate in the control period (black bar, mean ± S.E.M. of the spontaneous activity in the first two periods before VGCC blocker or vehicle application) was set 100% and the other values are expressed as per cent of the activity in this period. Each bar shows the average spontaneous discharges in periods of 15 min. (a) Spontaneous discharges in control rats between repetitive stimulation protocols. (b) Reduction of the spontaneous activity after ω-conotoxin. (c) Increase of the spontaneous activity after nimodipine. (d) Increase of the spontaneous activity after ω-agatoxin. Asterisks indicate significant changes compared with baseline (Wilcoxon's matched pairs signed rank test, P < 0.05); n, number of neurones tested.
Stimulation of the dura
In control rats, the stimulation of the dura with inflammatory mediators produced an insignificant transient increase of the responses which returned to values below baseline at the end of the recording period (Fig. 3a). By contrast, ω-conotoxin caused a significant reduction of responses to chemical stimulation which outlasted the wash period (Fig. 3b). After nimodipine, responses increased similarly as in control experiments, reaching significance at one time point (Fig. 3c). ω-agatoxin caused a continuous decline of the responses which was not reversed by washing the brainstem (Fig. 3d). VGCC blockers exhibited a similar pattern of effects on the neuronal responses when the dura was stimulated with noxious cold (Fig. 4a–c). However, after ω-agatoxin, the reduction of the responses was only temporary and was followed by an increase of the responses in the wash period (Fig. 4d).

Effect of VGCC blockers on responses of the neurones to stimulation of the dura with inflammatory mediators (IM). The baseline responses were set 0, and the other values show the responses (action potentials per response, mean ± S.E.M.) during application of VGCC blocker or vehicle compared with baseline. (a) Responses in control rats to repetitive stimulation with IM. (b) Effects of topical administration of ω-conotoxin to the brainstem (shaded area). (c) Effect of nimodipine. (d) Effect of ω-agatoxin. Asterisks indicate first significant change compared with baseline (Wilcoxon's matched pairs signed rank test, P < 0.05); n, number of neurones tested.

Effect of VGCC blockers on responses of the neurones to stimulation of the dura with noxious cold (same display as in Fig. 3).
Responses to repetitive mechanical stimuli of the dura were unchanged in control rats and after application of ω-conotoxin and ω-agatoxin (Table 1). Like the responses to chemical or cold stimulation, the responses to mechanical stimulation of the dura increased during application of nimodipine.
Responses (number of action potentials as mean ± S.E.M.) of the brainstem neurones to chemical and cold stimulation of the cornea (a) and mechanical stimulation of the dura and cornea (b) pre- (Baseline) and during application of the substances indicated in the left column(a)

Numbers in bold give significant differences to baseline (Wilcoxon's matched pairs signed rank test, P < 0.05).
n, number of neurones.
Stimulation of the cornea
As indicated in Table 1, ω-conotoxin and ω-agatoxin decreased the responses of brainstem neurones to application of inflammatory mediators or noxious cold to the cornea during the application period. This resembles the results of the responses to dura stimulation. In contrast to the effects with dura stimulation, nimodipine decreased responses to stimulation of the cornea with inflammatory mediators as well as cold. Responses to mechanical stimulation of the cornea were more or less unaltered by ω-conotoxin and ω-agatoxin, whereas nimodipine caused increases (Table 1).
Testing for central mechanisms involved in the effect of ω-agatoxin
The effects of ω-agatoxin were further studied. We administered ω-agatoxin to the brainstem and tested whether the spontaneous discharges were influenced by procaine applied to either the dura or to the brainstem. Application of procaine to the dura did not reduce the spontaneous activity, but procaine to the brainstem largely reduced the discharges (Fig. 5a). The application of procaine to the dura reduced the responses to cold stimulation, showing that procaine was principally working at the dural site (Fig. 5b). Thus, the increase of spontaneous activity in brainstem neurones after ω-agatoxin was generated in the brainstem and not elicited by dural stimulation.
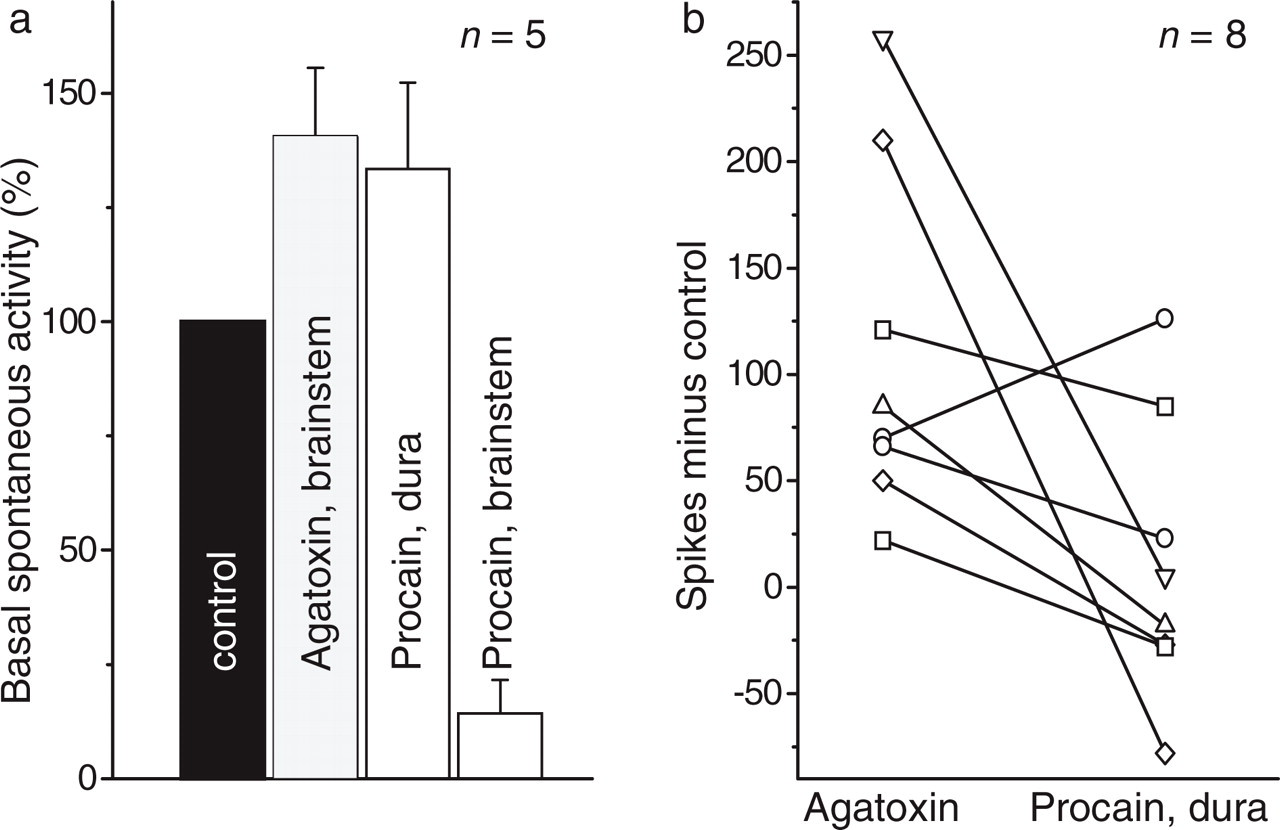
Applications of ω-agatoxin and procaine to the brainstem or to the dura. (a) Application of ω-agatoxin to the brainstem followed by application of procaine to the dura or to the brainstem. The bars show the average spontaneous activity of the neurones in the spinal trigeminal nucleus. (b) Reduction of the responses of brainstem neurones to cold stimuli applied to the dura, after application of procaine to the dura. The control values were from the last stimulation protocol during ω-agatoxin application.
Because we reasoned that the increase of the discharge frequency might reflect a disinhibition of the neurones, we tested in a further group of animals (n = 10) whether the effect of ω-agatoxin on neuronal activity is still present when GABA has been applied to the brainstem. Application of GABA alone reduced spontaneous activity, stimulus-evoked discharges were not significantly altered. When ω-agatoxin was subsequently given together with GABA, it did not increase spontaneous activity, and stimulus-evoked discharges declined (Fig. 6; Table 1).
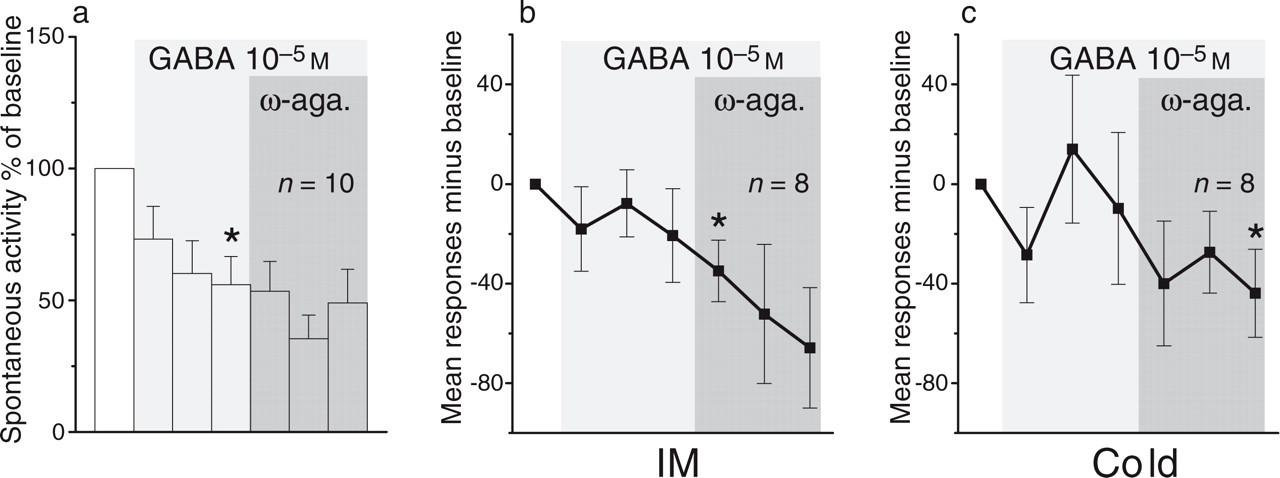
Effect of ω-agatoxin during application of GABA to the brainstem on spontaneous neuronal activity and stimulus-evoked responses. (a) Spontaneous activity. (b) Effect on the responses to stimulation of the dura with inflammatory mediators. (c) Effect on the responses to stimulation of the dura with noxious cold. Same display as in Figs 3 and 4.
Discussion
This study shows that the blockade of different types of voltage-gated N-, L-, and P/Q-type VGCCs in the brainstem causes different patterns of changes in the discharges of neurones with dural input in the spinal trigeminal nucleus. The blockade of N-type calcium channels reduced ongoing spontaneous activity and also slightly, responses to chemical and cold stimulation of both the dura and the cornea. After nimodipine, spontaneous activity increased but nimodipine effects on stimulus-evoked responses were variable. By contrast, the blockade of P/Q-type channels mainly caused an increase in ongoing discharges but it reduced the responses of the neurones to chemical and cold stimulation of the dura and the cornea. Spontaneous activity was not increased after ω-agatoxin when the brainstem had been pre-treated with GABA. Application of specific VGCC blockers did neither influence the mean arterial blood pressure nor the vasculature at the surface of the brainstem that was observed through a binocular microscope (Stemi 2000, Zeiss, magnification 20 ×). Dripping of these substances had no mechanical effect on spontaneous neural activity.
Control rats
In non-treated control rats, spontaneous activity remained nearly unchanged in the course of the protocol. Upon repetitive stimulation, the responses to chemical and cold stimulation of the cornea and the dura showed an initial increase that was followed by a decrease of responses. The initial increase of responses upon repetitive stimulation suggests some sensitization of peripheral and/or brainstem neurones. This may be produced by the chemical and cold stimuli themselves or by secondary reactions in the dura (e.g. neurogenic inflammation) (3, 4). The subsequent decrease of the responses may reflect desensitization of neurones (e.g. by internalization of receptors for inflammatory mediators (25, 26)), and/or actions of inhibitory systems.
The next paragraphs will discuss the course of ongoing activity and stimulus-evoked responses after application of VGCC blockers. Figure 7 displays our hypothesis about the possible localization and relative importance of the different types of VGCC, and in the Discussion we will refer to this model. The neurone in the trigeminal nuclear complex is the second-order neurone recorded from. This neurone receives inputs from primary afferent fibres, inhibitory descending projections and from inhibitory interneurones, according to a wiring model proposed by Millan in his review about descending control of pain (27). Because the distribution of VGCCs in the brainstem has not been studied before, we used general information on the localization of VGCCs in neurones and data on VGCCs in the spinal cord (28).

Hypothetical model of the localization and relative importance of VGCCs in the network of nociception in the trigeminal nuclear complex. Recordings were made from a neurone in the trigeminal nuclear complex that is driven by primary afferent fibres from the dura. The neurone is also under the control of descending inhibitory projections and local inhibitory interneurones, and the latter are influenced by excitatory interneurones. The wiring diagram was constructed according to Millan (27). Glu, Glutamate; ?, possible actions at these localizations might be hidden in the net effect.
Blockade of N-type calcium channels in the brainstem
Compared with neurones in control rats, the neurones in rats with ω-conotoxin application showed a different response pattern in the course of the recording period. Firstly, after ω-conotoxin ongoing discharges showed a progressive decrease. Secondly, responses to cold and chemical stimulation did not show any increase, but only a slow decrease during the recording protocol. Thus, ω-conotoxin influenced the magnitude of ongoing discharges and evoked responses in the same direction, namely reduction of activity. Because N-type VGCCs are localized in presynaptic endings and in the soma of cells, the reduced neuronal firing of a brainstem neurone after ω-conotoxin may result from reduced transmitter release from excitatory synaptic endings (e.g. primary afferent neurones) and/or from reduced excitability of the neurone itself.
The effects of ω-conotoxin in the brainstem are similar to those in the spinal cord. Spinal application of N-type calcium channel blockers reduced nociception in behavioural experiments (29–32) and ongoing discharges and stimulus-evoked responses of spinal cord neurones in single unit recordings (33, 34). Typically, and similar to the spinal cord, the reduction of activity by ω-conotoxin was only partial (34). Numerous in vitro studies have also shown that the blockade of N-type channels rarely blocks synaptic transmission completely and that other, unidentified types of VGCCs are involved as well (see 34). Furthermore, similar to the spinal cord, recovery from the effect of ω-conotoxin was not routinely seen. In most in vitro studies the preparations did not show recovery from the effects of ω-conotoxin. It is not known under which conditions a recovery occurs (for discussion see 18, 34).
Surprisingly, ω-conotoxin did not reduce responses of the neurones to brief mechanical stimuli applied to the dura and cornea, although it reduced responses of spinal cord neurones to mechanical stimulation of the knee joint (34). Because responses of neurones in the spinal trigeminal nucleus to mechanical stimuli applied to the dura and cornea are typically only transient and small (24, 35) we might have been unable to detect weak effects of ω-conotoxin.
Blockade of L-type calcium channels in the brainstem
VGCCs of the L-type are mainly located on the soma and dendrites of neurones (10, 12) and hence they mainly regulate cellular events other than transmitter release such as excitability. Effects of L-type channel blockers on nociception are contradictory, i.e. some studies have found anti-nociceptive effects, while others failed to disclose analgesic effects (18). In the present study, nimodipine enhanced ongoing discharges. After nimodipine, the stimulus-evoked responses followed the pattern of the controls, and this was clearly different to the effect of ω-conotoxin and ω-agatoxin. Thus, nimodipine had only a small effect on stimulus-induced responses. Because Ca2+-influx through L-type channels usually enhances excitability of neurones (36), blockade of L-type channels in the soma and dendrites should reduce excitability and thus decrease neuronal activity (37). An increase of activity after nimodipine could result from disinhibition of the recorded neurone by reduction of an inhibitory input. This may happen directly at the inhibitory neurone or at an excitatory neurone driving the inhibitory one (Fig. 7).
Blockade of the P/Q-type channels in the brainstem
Effects of ω-agatoxin were different from those of ω-conotoxin and nimodipine. First, we found a substantial increase of the ongoing discharges, but at the same time we observed a reduction of the responses to chemical and cold stimulation on the other side. Thus, ongoing discharges and responses to stimulation were influenced in opposite directions. This is possible because ongoing discharge is not necessarily coupled only to the primary afferent input but may also depend on postsynaptic excitability and interneurones (see Fig. 7).
P/Q-type VGCCs are located on presynaptic endings and thus regulate transmitter release (10). Importantly, ω-agatoxin can reduce release of transmitters from excitatory as well as inhibitory neurones (38, 39). Given that a neurone has both excitatory and inhibitory inputs, and that the release of the excitatory as well as the inhibitory transmitter depends on the activation of P/Q-type channels, the blockade of P/Q-type channels by ω-agatoxin may cause increases and decreases of activity. It is conceivable, therefore, that the reduction of the responses to dural and corneal stimulation shows reduced release of excitatory transmitters from primary afferents, and that the increase of ongoing discharges is due to a reduction of inhibitory transmitter release from an inhibitory interneurone (see Fig. 7). Unfortunately, the precise localization of P/Q-type channels in the brainstem is not known. In the spinal cord, however, P/Q-type channels have been found in presynaptic endings, and in some behavioural and electrophysiological experiments a reduction of nociceptive responses was observed (19, 30, 32, 33, 40).
Ongoing discharges showed a robust increase during and following ω-agatoxin which was independent of peripheral input, because the application of procaine to the dura did not reduce the effect. To our knowledge, ω-agatoxin does not directly stimulate neurones. In this case, as mentioned above, an increase in spontaneous activity of a neurone is likely to be caused by the reduction of release of an inhibitory transmitter from an inhibitory neurone. Indeed, in cerebellar Purkinje cells the release of GABA is regulated by P/Q-type channels (41). A recent ionophoretic study has shown that application of the GABAA antagonist bicuculline caused an increase of spontaneous activity in neurones of the spinal nucleus caudalis (42), suggesting that GABAergic neurones exert tonic local inhibition. Furthermore, ionophoretic muscimol, a GABAA agonist, reduced firing of these neurones elicited by glutamate (43).
In the present experiments, we could provide some evidence for an interaction between ω-agatoxin and GABA. We reasoned that when exogenous GABA is applied, subsequent inhibition of GABA release from neurones by ω-agatoxin will not have an effect on spontaneous activity. Indeed, spontaneous activity was not increased by ω-agatoxin after previous and concomitant application of GABA. Just recently, Knight et al. (44, 45) have reported that blockade of P/Q-type VGCCs in the periaqueductal grey (PAG) facilitates trigeminal nociception suggesting a function of P/Q-type VGCCs in the descending inhibitory system. These authors observed, however, that the application of bicuculline, a GABAA receptor antagonist, into the PAG inhibited responses of neurones in the trigeminal nucleus before as well as after application of ω-agatoxin into the PAG, and they concluded that the effect of P/Q-type channel blockade in the PAG is not necessarily linked to GABAergic functions (46). It seems possible, therefore, that in the trigeminal nucleus also an inhibition of glutamate release by P/Q-type channel blockers (15, 47, 48) may increase spontaneous activity if the excitatory neurone drives an inhibitory neurone.
Surprisingly, GABA did not reduce responses to stimulation. This suggests, firstly, that GABA does not markedly influence the responses to the stimuli used and, secondly, that effects of GABA on stimulus-evoked responses and spontaneous discharges are different. The dissociation between the effects of GABA on stimulus-evoked and spontaneous discharges also became obvious when ω-agatoxin was applied following GABA. While ω-agatoxin had no effect on spontaneous activity after GABA it still reduced responses to stimulation.
Possible role of P/Q-type channels in migraine
The P/Q-type calcium channel is thought to be of particular interest for migraine because patients with hemiplegic migraine have mutations of the CACNA1A gene on chromosome 19p13 that lead to increased allele sharing in the CACNA1A gene region on chromosome 19p13 in families with increased risk for migraine (49). Furthermore, migraineurs may exhibit alterations of the motor system and the cerebellum, and P/Q-type VGCCs are involved in the functions of these systems (50). However, in different mutations of the P/Q-type calcium channel different changes of the Ca2+ influx have been observed, and therefore it is difficult at this stage to define precisely which functional change in the P/Q-type channel function is relevant to migraine (51). Tottene et al. have analysed calcium currents through human P/Q-type calcium channels in isolated and transfected cells, and they conclude that all mutations have in common that single channel Ca2+ influx is increased and that maximal Cav2.1 current density in neurones is decreased. These authors believe that the increased Ca2+ influx through Cav2.1 channels in the cortex is likely to be relevant for migraine aura because this alteration may increase cortical excitability, making the cortex more susceptible to spreading depression, the putative substrate of migraine aura (51, 52). However, the authors speculate that a decreased Ca2+ current through Cav2.1 channels in the PAG may trigger migraine pain because inhibition of P/Q-type calcium channels in the PAG facilitates trigeminal nociception (see above). The voltage-dependency of the P/Q-type VGCCs could explain the episodic character of migraine attacks: only if an activation of these channels occurs will the processing system be disturbed.
The data of Knight et al. (44–46) and the present data converge to the idea that the pathophysiological role of P/Q-type calcium channels in the brainstem has to be sought, presumably in the instability of tonic inhibition. Whether local inhibitory interneurones in the vicinity of the excitatory neurones, or descending inhibitory systems such as the system descending from PAG or both are important, needs to be established. Several reports propose a possible role of the PAG. Welch et al. (53) have shown that iron homeostasis in the PAG is impaired in patients with migraine, and this finding led them to suggest that reduced inhibition may be crucial for the generation of headache. Weiller et al. showed in a PET study that the brainstem of a patient, possibly the PAG, exhibited higher activity during migraine attacks (54). A recent fMRI-study showed a vascular malformation in the midbrain adjacent to the PAG and contralateral to the side of migraine headache in a patient suffering from chronic migraine (55).
In summary, we have shown different patterns of effects of the blockade of different VGCCs. While blockade of N-type VGCCs reduced both spontaneous and stimulus-evoked responses, the blockade of P/Q-type VGCCs seemed to alter spontaneous activity differently, presumably by changing tonic inhibition, and stimulus-evoked responses, and presumably by reducing synaptic activation by excitatory afferents. These data may contribute to the discussion of how P/Q-type VGCCs are involved in migraine. We propose to consider that abnormal P/Q-type channels may have effects on inhibitory systems in the first place. Hence, altered functions of P/Q-type VGCCs may lead to changes in the central processing of input from the meninges, even if they do not significantly influence synaptic activation of trigeminal neurones by the afferent input itself.
Footnotes
Acknowledgements
We thank Mrs Müller for excellent technical assistance. The work was supported by the Interdisciplinary Center for Clinical Research (IZKF) Jena (project B378-01012, TP 1.3).
