Abstract
The present study examined the sensitivity to light of melatonin (MLT) secretion in familial migraine during a headache-free interval. Twelve female patients and 12 healthy controls were included in the trial. All subjects were studied twice. In each session, light exposure (300 lx) or placebo was randomly administered for 30 min between 00.30 and 01.00 h. Blood was sampled hourly between 20.00 and 24.00 h, and 02.00 and 04.00 h and every 15 min between 00.30 and 01.30 h. Plasma MLT levels were determined by radioimmunoassay. MLT suppression was more marked in the migraine group than in the control group [difference of area under curve (δAUC) = −53.8 ± 16.2 vs. 18.5 ± 12.7 pg/h/ml, P < 0.005; maximum of MLT suppression (δ) = −35.7 ± 10.2 vs.- 6.7 ± 5.8 pg/ml, P < 0.05]. These findings show a clear hypersensitivity to light in young female migraineurs during the headache-free period.
Introduction
The pineal secretion of melatonin (MLT), an indole compound derived from serotonin, displays a nyctohemeral variation, as shown by high nocturnal but low or undetectable diurnal plasma concentrations (1). Because the pineal gland plays an important role in maintaining homeostatic equilibrium in close relationship with changing environmental conditions, the possibility that the pineal hormone MLT is deficient in migraine has been proposed (2). In addition, hemicranial headache and unilateral orbital cephalalgia with or without sympathetic symptoms have been reported in pinealomectomized subjects (3). An interictal decrease in the levels of nocturnal plasma MLT (4) and an alteration of the urinary MLT excretion throughout the ovarian cycle have also been described in migraine patients (5, 6). Furthermore, seasonal variations in daily MLT secretion have been reported, patients with migraine being more likely to have headache during the bright Arctic summer season (7, 8).
In addition to perceptual vision, the mammalian eye is capable of detecting changes in environmental light resulting in non-image-forming light responses such as suppression or entrainment of pineal MLT production (9). Photic information involving this response is transmitted to the central pacemaker located in the suprachiasmatic nuclei (SCN) via the retino-hypothalamic tract (10, 11). The multisynaptic neural pathway from the SCN to the pineal gland passes first through the hypothalamic paraventricular nucleus, the upper thoracic region of the cervical spinal cord, which subsequently projects to the preganglionic cell bodies of the superior cervical ganglia (SCG) of the sympathetic chain. The neural cells in the SCG send projections to the pineal gland. The main neurotransmitter regulating the pineal gland is norepinephrine which is released at night, in response to stimulatory signals originating in the SCN.
A number of abnormalities in visual function have been described in migraine, some of which indicate hyperexcitability of the visual pathways (12, 13). Clinical observations suggest that hyperexcitability occurs not only during attacks, typically in the form of photophobia, but also between episodes. Furthermore, compared with headache-free subjects, migraineurs are more sensitive to light (14). As early as the second century AD, Aretaei Cappadocis had written: ‘fugiunt enim quodam modo lucem, tenebraeque his aegritudinem solentur’ (they avoid light by all possible means and the dark subsides their feeling of sickness) (15).
These data prompted us to test the hypothesis of light supersensitivity in migraine by comparing nocturnal MLT suppression to light in migraine patients during the between-episodes period and well-matched controls.
Subjects and methods
Subjects
The protocol was approved by the Ethics Committee of Hospices Civils de Lyon and the subjects gave written informed consent. Twenty-four female subjects (12 controls, 12 migraine subjects) were recruited through advertisements in the Faculty of Medicine and Pharmacy of Lyon and were remunerated for their participation. All potential participants took part in an extensive clinical interview including a detailed description of their health or headache history. Subjects of Caucasian origin were neither smokers nor strong alcohol consumers. They were within 10% of their ideal body weight and were free of any organic or psychiatric disease. Treatment with any antidepressant or hypnotic drugs was a basis for exclusion. Those excluded from the study were volunteers with ocular disease, including ametropia > 5 dioptries. No abnormality of colour vision and visual field was detected in our volunteers using suitable tests (visual field: Goldmann, Humphrey FastPac 24-2, FDT perimetry, Ishihara test, Farnsworth 28 Hue test). Associated with biomicroscopy of the anterior segment, intraocular pressure measurements and fundoscopy with blue light photography of optic nerve fibres rule out vascular disease of the retina and glaucoma. Migraine-free control subjects were 18–29 years old (median 22 years) and migraine subjects were 19–29 years old (median 22 years). Six subjects had a regular 28-day ovarian cycle (one control and five migraine patients) and 18 were taking an oral contraceptive (11 controls and seven migraine patients). Eight patients displayed migraine with aura (MA) and four patients migraine without aura (MO), according to the criteria of the International Headache Society Classification of Headache (16). They met strict criteria: long-standing migraine, no recent change in migraine characteristics and a family history component. They should have more than one attack per year and less than one per month, without relationship with menses (mean three attacks per year on average). They were free of antalgic or prophylactic medication for at least 1 month at the time of exploration. MLT secretion was previously evaluated at home, in domestic light, by determination of saliva MLT levels. Saliva was sampled with Salivette® (Sarstedt, Orsay, France) at 20.00, 21.00, 22.00, 23.00, 24.00, 06.00, 07.00, 08.00 and 09.00 h.
Protocol
Each subject was studied during two night sessions separated by exactly 4 weeks, at the beginning of the menstrual cycle. Subjects wore black goggles from 19.30 to 04.00 h, except during light exposure. During the sessions, they were allowed to rest or to pursue an intellectual or recreational activity. They could sleep after 01.30 h but only in a sitting position to eliminate a posture influence on MLT levels (17). Light was administered between 00.30 and 01.00 h with a helmet designed for depression treatment (Lumino®; Schreder Inc., Ans, Belgium). The optical block was individually adjusted to the head anatomy to deliver 300 lx at eye level. The intensity and duration of light exposure were chosen to obtain a significant degree of MLT suppression in controls and not to evoke an attack in migraineurs. Light and placebo (wearing of helmet with light off) were randomly assigned in a cross-over trial to a control and a migraine subject who were both explored in the same sessions, so that the same lamp was used for the whole trial. Blood was sampled every hour between 20.00 and 24.00 h and 02.00 and 04.00 h and every 15 min between 00.30 and 01.30 h. Blood was drawn through an indwelling catheter inserted into an antecubital vein. The i.v. line was kept patent with a slow drip of heparinized saline (5000 IU heparin/l). At each sample time, 3 ml of blood were collected through a three-way stop-cock, immediately transferred to heparinized plastic tubes, stored at 4 °C, and centrifuged at the end of the session. The plasma and saliva samples were kept frozen at − 20 °C until determination of MLT concentration using a radioimmunological method (18). The sensitivity of the assay was routinely 3 pg/ml. The intra-assay coefficients of variation were < 7% between 30 and 200 pg/ml and the inter-assay coefficients of variation were 8.7 and 7.9% (n = 18) for MLT concentrations of 55 and 115 pg/ml, respectively. All plasma sampled in one control and one migraine patient was simultaneously run in the same assay in order to reduce interassay variation.
Statistical analysis
For each subject, we calculated the reduction in total MLT secretion by calculating the surface area between the MLT curves obtained during light and placebo sessions [Δ area under curve (ΔAUC)] from 00.30 to 04.00 h and the maximum plasma MLT suppression (Δ) observed at a fixed time. ΔAUC and Δ were negative when there was MLT suppression. The effect of illness, time and light was assessed by applying to placebo and light profiles a multiple analysis of variance (ANOVA) for repeated measures, including Greenhouse–Geisser corrections (SPSS®, Paris, France). The simple comparisons of means were performed using Mann–Whitney's test to detect significant between-group differences. Results are given as mean ± SEM.
Results
Saliva MLT levels obtained at various times of the day from samples at home were similar between the control and the migraine groups (Fig. 1a) and no subject was excluded because of too low MLT levels. All subjects complied with the protocol, and in particular, light administration did not evoke any migraine attack. No side-effect occurred, so that it was not necessary to exclude any of the patients from the trial. Only one attack occurred in two migraine patients on the night following the light session, related to sleep deprivation. A difference between patients and controls was showed by ANOVA performed on the plasma MLT profiles obtained during the placebo session (Fig. 1b). ANOVA did not show a difference between plasma MLT profiles of MA and MO patients (F(1,10) = 0.26, P = 0.62). MLT suppression by light exposure was significantly different between the two groups (Fig. 2a,b) and was more marked in the migraine group than in the control group (ΔAUC =−53.8 ± 16.2 vs. 18.5 ± 12.7 pg/h/ml, P < 0.005; Δ=−35.7 ± 10.2 vs. − 6.7 ± 5.8 pg/ml, P < 0.05 for migraine patients and controls, respectively) (Fig. 3). When patients’ data were distributed between MA (n = 8) and MO (n = 4) subgroups, no between-subgroup difference was observed in MLT suppression.

(a) Saliva melatonin (MLT) profiles determined from samples obtained at home and (b) plasma MLT profiles determined during placebo sessions in controls (○) and patients (▪). ANOVA performed on plasma MLT profiles between 00.30 and 04.00 h showed a significant between-group difference (P = 0.015).

Mean plasma melatonin (MLT) profiles determined during placebo (○) and light (▪) sessions in controls (a) and migraine patients (b). ANOVA for repeated measures performed on plasma MLT profiles between 00.30 and 04. 00 h showed an effect of time (P < 0.001) and of light treatment (P = 0.05) and interaction between light treatment and migraine/control group (P < 0.02) and between light treatment and time (P < 0.01).
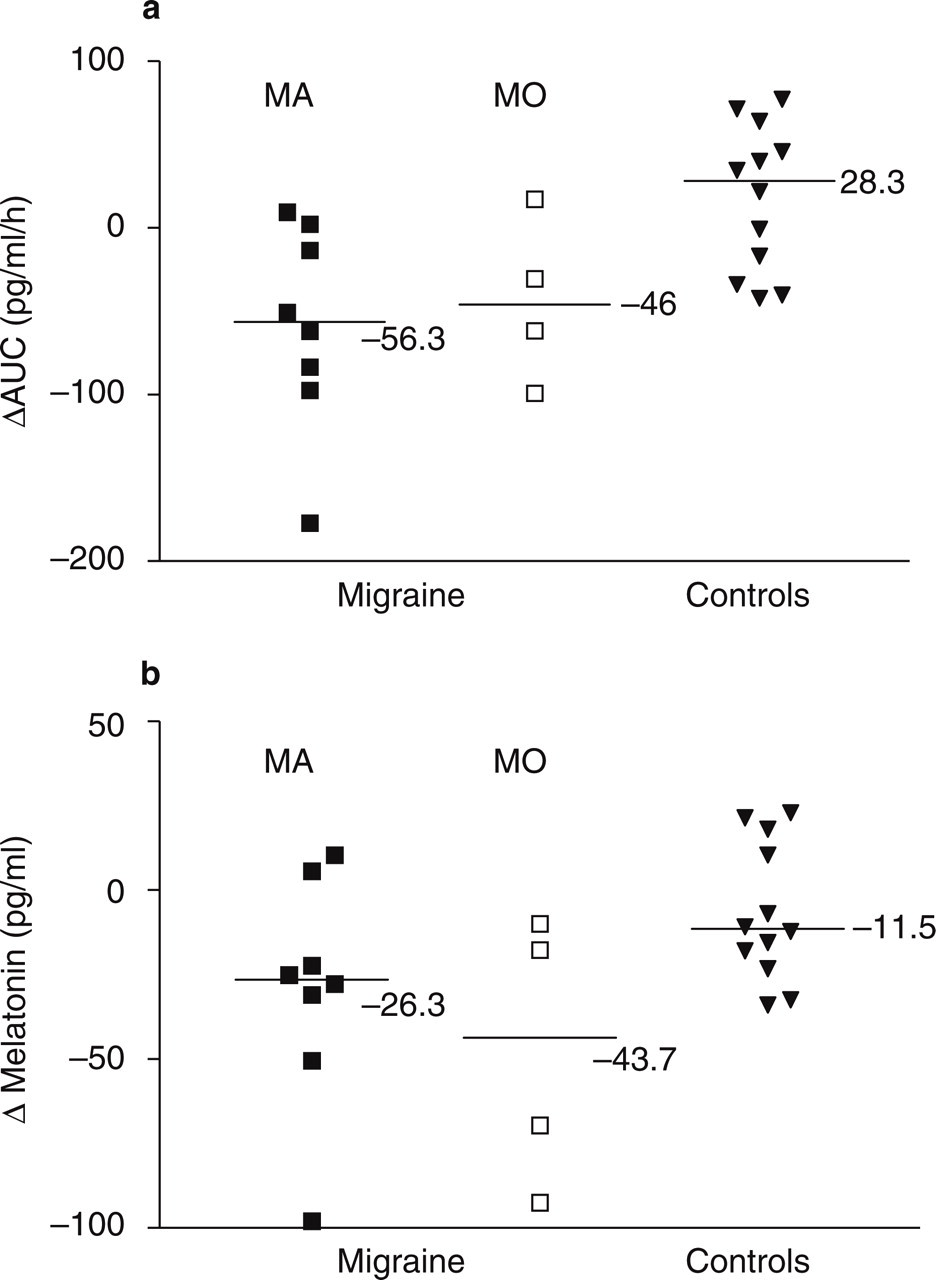
>(a)Individual and median surface areas between the melatonin (MLT) curves obtained during light and placebo sessions (ΔAUC) from 00.30 and 04.00 h.>(b) Individual and median maximum plasma MLT suppressions (Δ). Mann–Whitney's test showed a significant between-group (migraine vs. controls) difference for ΔAUC and Δ (P < 0.005 and P < 0.05, respectively). MA, Migraine with aura; MO, migraine without aura.
Discussion
Eight patients with aura and four patients without aura were included in the study, although MA is less prevalent in the general population. This could be the result of selection bias (recruitment in the medical field of migraineurs with familial history displaying more than one attack per year and less than one per month) and the nature of the trial involving light. We observed a hypersensitivity to moderate light levels in migraine at the interictal period, as expressed by an alteration of plasma MLT profiles following light exposure in migraine patients compared with controls. Patients displayed mean plasma MLT levels consistently below those of placebo sessions, whereas in controls the suppression of MLT levels was transitory and followed by a slight rise, leading to positive AUC values. Such plasma MLT profiles observed in controls suggested a phase delay of MLT secretion, although additional samples after 04.00 h would be necessary to confirm this. During the placebo sessions, we observed plasma MLT levels significantly higher in patients than in controls, which is contrary to a previous result which reported decreased plasma MLT levels obtained in a single nocturnal (23.00 h) blood sample during the interictal period (4). In addition, the overall nocturnal urinary MLT excretion was decreased throughout the ovarian cycle in menstrually related migraine (5, 6); Murialdo et al. observed that urine MLT levels were significantly lower at the time of attacks, compared with those obtained during the pain-free period (5). In these reports, however, nocturnal light was not controlled, patients were much older and seniority of illness was higher as well as their attacks more frequent. In these cases, infraclinical inflammation thought to obliterate the venous outflow from the cavernous sinus on one side could be involved, thus injuring the traversing sympathetic fibres of the intracranial internal carotid artery, especially those projecting to the pineal gland, with as a consequence an alteration of MLT secretion. Saliva MLT profiles of migraine patients determined at home were not significantly higher than those of controls, although there was a tendency for levels to increase. This result was probably related to a lack of sampling over the complete night-time period and the presence of domestic light, whereas the higher plasma MLT levels observed in migraine patients during the placebo session was probably related to the absence of masking effect of light exposure (dark goggles) on MLT secretion (19). We did not observe any difference between MA and MO subgroups for basal MLT levels and MLT suppression. Due to the small sample (eight MA and four MO patients), this aspect needs further investigation.
The hypersensitivity of patients to light in this study is in agreement with the concept of central neuronal excitability in migraine (12). Electrophysiological investigations suggest that this hyperexcitability occurs at the cortical level, as a result of a direct increase of excitatory mechanisms, either at the level of the glutaminergic thalamocortical synapse, or at the level of recurrent excitatory circuits known to operate within the visual cortex (20, 21). Alternatively, since MLT secretion is inhibited during the day through the retinohypothalamic pathway (of which a main transmitter is glutamate) and the SCN is also involved in rapid changes of light-induced pineal metabolism, we suggest that hypersensitivity of MLT suppression to light in migraine is at least in part the result of an increase of glutamate transmission of the input pathway. These two observations are in agreement with results showing that GABA agonists which modulate glutamate-mediated neural transmission are effective in the treatment of migraine as well as the glutamate antagonist Lamotrigine (22, 23). Recently, retinal ganglion cells innervating the SCN were shown to intrinsically respond to light (24, 25). These melanopsin-containing cells are candidate photoreceptors for the photic entrainment of circadian rhythms, because the sensitivity and slow kinetics of the light response are compatible with those of the photic entrainment mechanism (25). Furthermore, this system appears to send photic information not only to the endogenous clock in the SCN, but also to other brain areas involved in irradiance detection, such as light-activated pupil response. An abnormality at the level of this irradiance detection system could thus be suspected as a contributing factor in migraine.
Given that migraine attacks are triggered by both light and changes in living habits, our data suggest a pivotal role of the SCN, a structure involved both in long-term phase adjustments and rapid changes of light-induced pineal metabolism (26). In conclusion, we show hypersensitivity to light of MLT suppression during the interictal period in young women suffering from migraine attacks. Migraine could be an attractive model for the study of entrainment pathways or mechanisms in humans.
Footnotes
Acknowledgements
This study was supported by a European grant (Biomed2 BMH4 CT97 2327). Hospices Civils de Lyon were the promoter of the clinical trial. We thank Dr Howard Cooper (INSERM U371, Lyon, France) and Dr Alfred Lewy (Department of Psychiatry, Portland, OR, USA) for helpful comments on the manuscript.
