Abstract
In this multicentre, randomized, double-blind, single-dose study a total of 374 patients generally suffering from migraine attacks suitable for treatment with non-prescription drugs, received either oral acetylsalicylic acid effervescent 1000 mg (ASAE) or effervescent placebo for the treatment of an acute migraine attack. Of the 343 patients fulfilling the criteria for efficacy analysis 169 patients took acetylsalicylic acid and 174 placebo. Response rates (reduction of headache severity from severe or moderate to mild or no pain at 2 h after administration) were 55.0% for acetylsalicylic acid and 36.8% for placebo (P < 0.001). Twenty-nine percent of patients in the active treatment group were pain-free after 2 h compared with 16.7% in the placebo group (P = 0.007). No headache recurred within 24 h post-dose in 84.6% of patients in the active group and in 85.1% of patients in the placebo group. Effervescent placebo reduced nausea and vomiting to the same degree as the active drug. Adverse events of acetylsalicylic acid (8.3%) were generally mild or moderate and comparable to those of placebo (2.9%). This study shows that oral ASAE is safe and effective for the treatment of acute migraine attacks.
Introduction
Surprisingly, despite the common usage of acetylsalicylic acid (ASA) in the treatment of acute migraine attacks the scientific literature is almost silent concerning adequate and well-controlled studies on the efficacy of ASA given as the single drug. Instead, during the past 17 years a total of nine studies on the action of ASA co-administered with metoclopramide (MCP) were published (1). The dosage of ASA or equivalent used in these studies varied from 650 mg to 900 mg and was thus below the single dose of 1000 mg recommended nowadays (2). Newly developed effervescent ASA tablets with an acid neutralization capacity of 11 mEq provide dissolved citrates which appear to promote gastric emptying and thereby enhance the onset of the analgesic effect of ASA (3, 4). The present study was designed to compare the safety, tolerability and efficacy of a single-dose administration of acetylsalicylic acid effervescent 1000 mg (ASAE; 2 × 500 mg) with placebo in the treatment of acute migraine attacks.
Patients and methods
Patients
From April 1998 to September 1998, suitable patients were enrolled by 36 medical practices throughout Germany. The study was conducted in accordance with the Declaration of Helsinki (1996), the German Drug Law, and ICH GCP standards and did not start before favourable IEC approval was obtained. Eligible patients had to be ambulatory male or female patients (18–65 years) requiring oral pain treatment for an acute migraine attack according to the classification of the International Headache Society (5). Prior to enrolment the patients gave their informed consent in writing. In order to be included into the study, patients had to suffer from migraine for more than 1 year with a minimum of one attack per month, but no more than six attacks per month. Patients meeting at least one of the following criteria were not included in the study: hypersensitivity to salicylates, peptic ulceration or gastric bleeding, impairment of renal, hepatic, endocrine, pulmonary, cardiac, neurologic or cerebral function, vomiting more than 20% of the time during migraine attacks, usually so incapacitated as to require bed rest during the attack, drug or alcohol abuse, pregnancy or lactation, intake of analgesic or anti-migraine drugs within 24 h prior to the intake of the study medication, participation in a clinical study within the previous month or previous enrolment into this study.
Study design
The objective of this multicentre, randomized, double-blind, placebo-controlled, parallel group study was to determine the analgesic efficacy of a single dose of ASAE (2 × 500 mg) in comparison with effervescent placebo (single dose) in patients with acute migraine attacks. The allocation to a treatment group was done at screening after the investigator had checked all inclusion and exclusion criteria and the patient's eligibility has been established. The patient was thoroughly instructed as to how to identify a suitable episode of a migraine attack so that he was able to self-medicate. Patients had to be aware that they should not take the study medication before their pain was at least of moderate intensity and that they had to take the medication within 6 h of onset of symptoms. The dose had to be taken within 2 months of the screening visit. The patient was also instructed by the investigator as to how to complete the diary card, given to him/her at the screening visit. Date and time of onset of symptoms was to be recorded as well as the time the medication was taken and the headache time course. Pain intensity and accompanying symptoms (nausea, vomiting, photophobia and phonophobia) of the migraine attack were to be reported by the patient just before intake of study medication, and at 0.5, 1, 1.5, 2, 3, 4, 5, and 6 h post-dosing on a verbal rating scale (severe, moderate, mild, none). In addition, patients had to record the recurrence of any migraine attacks within 24 h post-dosing as well as their opinion on the efficacy of the study medication, i.e. global assessment of pain therapy (categorical scale: poor, moderate, good, excellent) at 24 h. The use of another analgesic medication of the patient's choice (‘escape medication’) was allowed as of 2 h post-dosing and had to be recorded in the diary card. Additionally, adverse events were recorded over the treatment period. Patients had to contact the investigator after a treated migraine attack for their follow-up visit, ideally within 24–48 h after dosing.
Evaluation of efficacy and safety
For the evaluation of efficacy the two treatments were compared concerning the primary efficacy criterion ‘number of patients with pain reduction from severe or moderate to mild or no pain at 2 h after intake of study medication’. Secondary criteria were: number of attacks resolved within 2 h, modification of accompanying symptoms (nausea, vomiting, photophobia, phonophobia) within 2 h after intake of the study medication, number of recurrent attacks within 24 h after intake of the study medication, global assessment of treatment by the patient 24 h after intake of the study medication, adverse events during the treatment period.
Sample size and statistical analysis
The number of responders in both treatment groups was compared to test the hypothesis that the responder rates were independent of the administered study medication. A patient was defined a responder if the pain intensity after intake of the study medication changed from severe or moderate to mild or no pain. Assuming a responder rate of 30% in the placebo group, a total of 103 patients per treatment group was required for the detection of a clinically relevant treatment difference of 20% with a power of 1 − β = 80% (20% probability of a type II-error) if Fisher's exact test was used to test the H0-hypothesis with a significance level set to α = 5% (two-sided). Therefore, 206 patients were required to complete the trial according to the protocol. With an assumed total of 30% of patients dropping out before randomization or not taking the trial medication, a minimum of 300 patients was to be enrolled to ensure an intent-to-treat population of at least 206 patients. Fisher's exact test was used to test the above specified hypotheses on the chosen significance level. Ninety-five percent confidence limits for the difference of responder rates in both treatment groups were provided. Secondary efficacy variables were analysed by using descriptive methods to detect differences in treatment effects.
Results
Study population
The study was conducted in 36 study centres. A total of 374 migraine patients were recruited and randomized. Thirty-one patients were randomized but did not take the study medication. The intent-to-treat group which was used as the main population for efficacy analysis as well as for safety analysis covered all patients who took the study medication (n = 343), of whom 169 received ASAE and 174 effervescent placebo. There was no statistical difference between the two groups regarding demographic factors (Table 1). Migraine had been present for at least 1 year in all patients enrolled. All patients reported to experience at least one but no more than six attacks per month on average. Triptans were used previously by 13.4% of patients, while 37.3% used different analgesics; most of those were fixed combinations with ergot alkaloids. Of the total population, 83.4% were women. A total of 58.6% of patients in the active group and 63.8% of patients in the placebo group reported severe pain at entry.
Demographic baseline characteristics

Primary efficacy variable
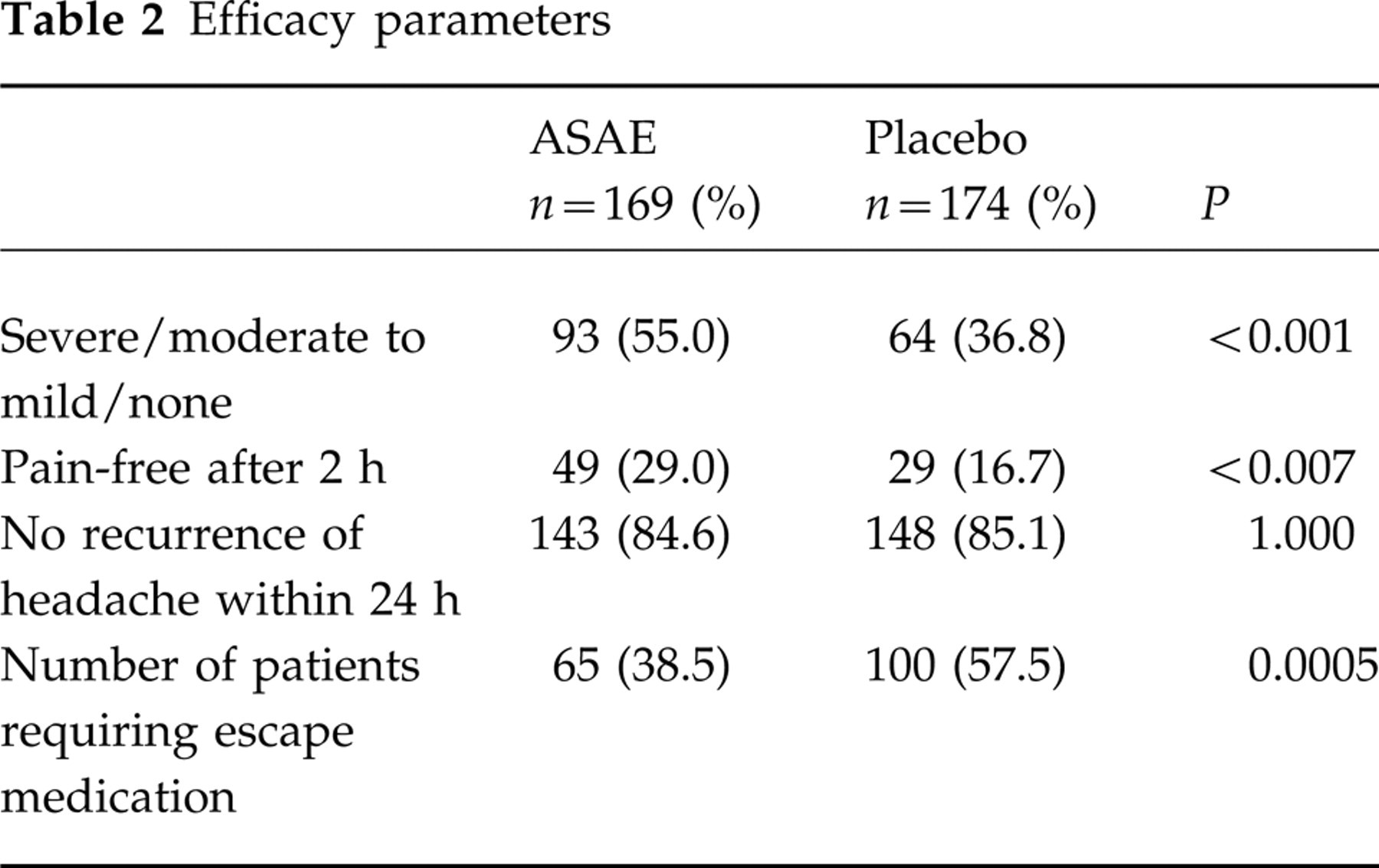
There was a significant difference (P < 0.001) in efficacy as expressed by reduction in headache severity from severe or moderate to mild or no pain at 2 h after administration of the active medication compared with placebo (Fig. 1). Response rates were 55.0% (93/169) for acetylsalicylic acid effervescent 1000 mg and 36.8% (64/174) for effervescent placebo (P < 0.001) (Table 2).
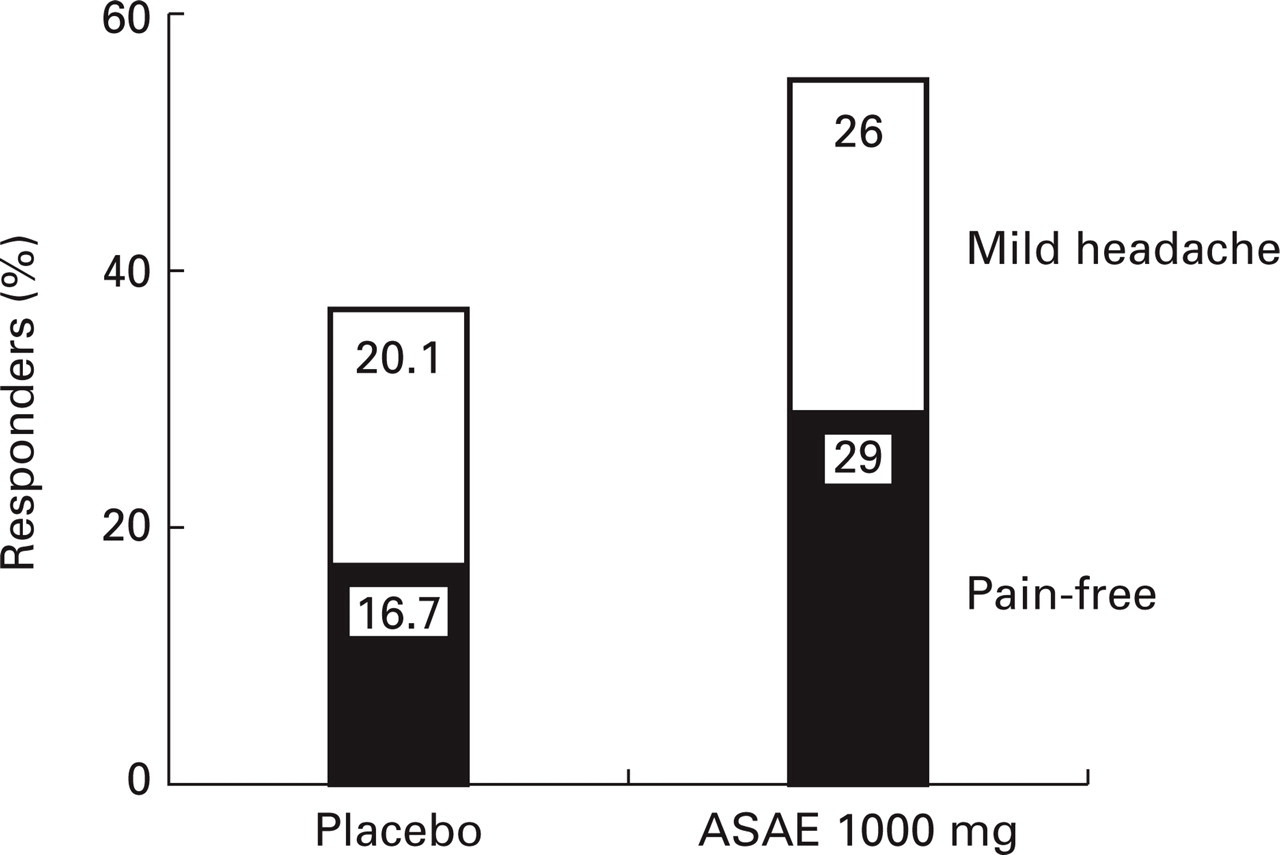
Percentage of patients with improvement from severe or moderate headache to mild headache (upper part) or no headache (lower part) 2 h after treatment.
Efficacy parameters
Secondary efficacy variables
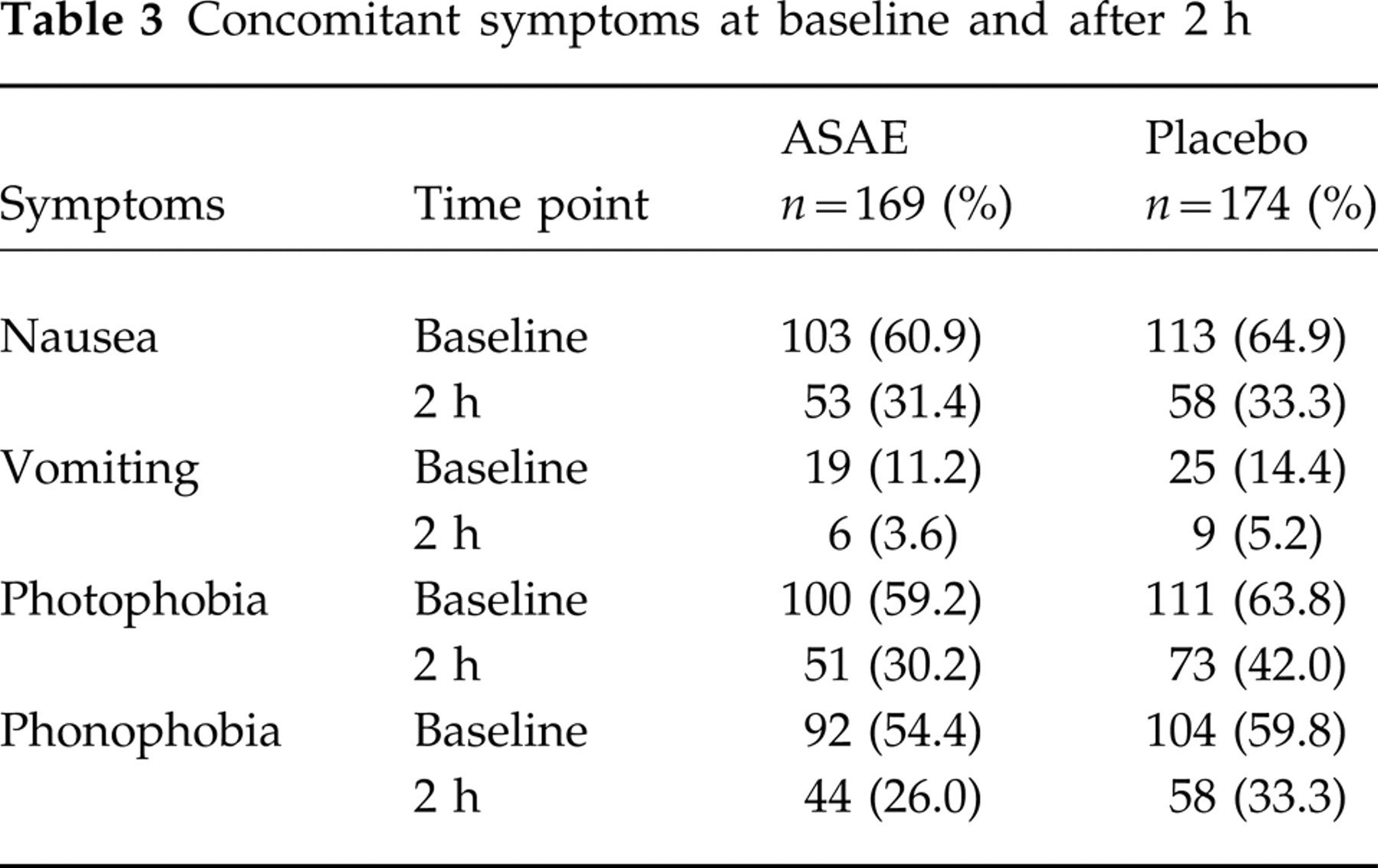
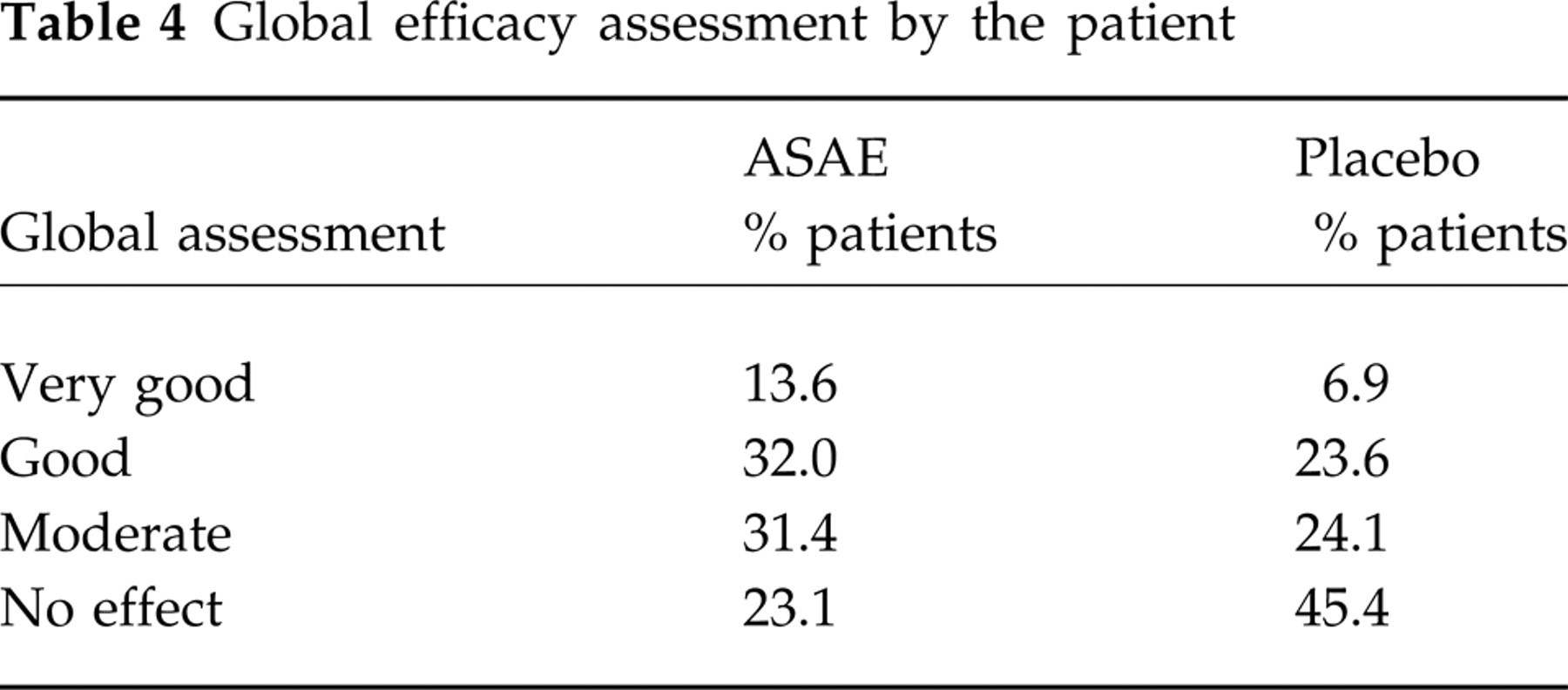
In the active treatment group 29% of patients reported to be pain-free after 2 h compared with 16.7% of patients in the placebo group (P < 0.007) (Table 2, Fig. 1). There were no statistically significant differences between the treatments concerning the frequency of nausea, vomiting or phonophobia (Table 3). However, the patients' sensitivity to light was remarkably reduced after ASAE treatment when compared with placebo treatment. In both treatment groups the percentage of patients having experienced no further migraine attack during the 24 h after intake of the study medication was about 85% (Table 2). Even though the number of patients with more than one relapse was different in both treatment groups (two of the ASAE patients and six of the placebo patients), this finding was considered to be not expressive due to the low number of cases. The global assessment of efficacy revealed notable differences between the treatment groups (Table 4). The proportion of patients assessing the therapeutic effectiveness to be very good or good was about 46% in the ASAE treatment group vs. 30% in the placebo treatment group. The number of patients who could not observe any therapeutic effect was also remarkably different between the two treatments (ASAE 23% vs. placebo 45%). Overall, the patients' assessment of the therapeutic efficacy was clearly more favourable in the ASAE treatment group.
Concomitant symptoms at baseline and after 2 h
Global efficacy assessment by the patient
Safety
Adverse events (AE) occurred in 14/169 patients (8.3%) of the ASAE treatment group and in 5/174 patients (2.9%) of the placebo group (Table 5). Additionally, seven of the 31 patients (22.6%) who were randomized but did not take the study medication reported AE. In the placebo treatment group four of the AE in one patient were reported to be of severe intensity, whereas no severe AE occurred in the ASAE treatment group. In the patients treated with acetylsalicylic acid effervescent the body systems most frequently affected by adverse events were body as a whole (n = 5), digestive system, nervous and respiratory system (n = 3 each). In the placebo group body as a whole was the body system (n = 3) most frequently affected by AE. Two serious AE were reported for one patient who was randomized but not treated with study medication. The overall incidence of AE was numerically higher in the ASAE group compared with the placebo group. However, due to the low number of AE in each body system no clinically relevant difference between ASAE and placebo and untreated patients was observed.
Overview of adverse events (AE)

Investigator's opinion.
Discussion
With a response rate of 55% this study proved the efficacy of oral acetylsalicylic acid 1000 mg in the treatment of acute migraine attacks when administered as a buffered, effervescent formulation. Nausea, as the most prominent accompanying symptom during a migraine attack, was reduced by ASAE by about 50% after 2 h. Since there was no difference to the effervescent placebo in this parameter, it is reasonable to assume that the anti-nausea and anti-emetic effects are attributable to the buffering capacity of the dissolved citrates provided (3, 4), though this needs to be confirmed in a further study. As to nausea and emesis reduction, the effervescent placebo tablet in fact appears to act similar to the active drug. Interestingly, a similar finding was already obtained as early as in 1984 when Tfelt-Hansen (6) showed that ASA was significantly better than (effervescent) placebo for pain but not quite significant for nausea. It can be concluded that ASA effervescent 1000 mg will certainly be a safe and effective choice for those patients who are used to self-medicating their migraine attack.
Footnotes
Acknowledgements
The authors would like to thank the following investigators for their valuable contribution: Dr Anwar Ansari, Einbeck; Dr Ulf Bauerfeld, Miltenberg; Dr Gisela Baum, Leipzig; Dr Renate Bork-Kopp, Mainz; Dr Manfred Deterding, Bochum; Dr Fuat Edgü, Hammelburg; Dr Maria Silke El Tobgui, Frankfurt; Dr Monika Esser, Leipzig; Dr Ursula Fleischmann, Fürth; Dr Hans-Michael Foerster, Duisburg; Dr Markus Föh, Fulda; Dr Helmut Harant, Schweinfurt; Dr Dezsö Heim, Nürnberg; Dr Mariana Heim, Nürnberg; Dr Nikolaus Hoffmann, Schweinfurt; Dr Ingrid Hunecke, Leipzig; Dr Barbara Kademann, Leipzig; Dr Roland Maeumbaed, Höchstadt; Dr Ingrid Marxen, Heidelberg; Dr Peter Mattes, Mannheim; Dr Bernhard Müller, Northeim; Dr Gerhard Neumann, Delitzsch; Dr Gernot Raab, Frankfurt; Dr Uwe Rahnefeld, Leipzig; Dr Oda Richter, Lützen; Dr Karin Seifert, Bitterfeld; Dr Bastian Steinberg, Hamburg; Dr Hans-Joachim Taenzer, Köln; Dr Ralf Töle, Gleichen-Rittmarshausen; Dr Jürgen Wachter, Mannheim; Dr Ernst Wassermann, Rimpar; Dr Hans-Gerd Weber, Borna; Dr Sabine Winkelmann-Laue, Köln; Dr Henning Wiswedel, Fürth; Dr Waltraud Wyborski, Werne; Dr Barbara Zahn, Wilhelmsfeld.
