Abstract
The nociceptive flexion reflex (NFR) of the lower limbs (RIII reflex) was examined bilaterally in 54 cluster headache (CH) patients suffering from episodic CH (ECH) and chronic CH (CCH). Fifteen ECH patients were examined in both remission and active phases. The RIII reflex threshold (Tr) and the threshold of pain sensation (Tp) were significantly reduced on the symptomatic side in patients with episodic CH during the bout. During the active phase of episodic CH an inverse correlation was found between the severity of CH (ratio: number of cluster periods/years of illness duration) and the Tp, which may suggest a role for secondary central sensitization in pain pathways. The lower Tr and Tp on the symptomatic side is in keeping with previous observations exploring pain mechanisms using different methods (i.e. corneal reflex, pain pressure threshold). On the whole, these data tie in with the view of an impairment of the pain control system, which parallels the periodicity of the disorder in the episodic form.
The pathogenesis of cluster headache (CH) is still far from clear. The temporal pattern of the attacks, the lateralization of the pain and the oculocephalic signs provide interesting aspects for exploration of the mechanisms involved. Both central and peripheral mechanisms have been suggested in order to explain the origin of the pain and the accompanying symptoms (1, 2); in particular, impairment of the trigeminal pain control system and/or trigemino-vascular system have been hypothesized in CH (3, 4), while recent data stress the pathogenetic role of hypothalamic pace-maker regions of the brain (5) which may play a permissive role, releasing or entraining the trigemino-vascular pain system.
Indirect instrumental and clinical evidence suggests central sensitization in the pain control system (at trigeminal level, in particular), lateralized to the affected side, which may be responsible for the allodynia (6) and hyperalgesia reported by some CH patients either on cephalic or extracephalic parts of the body, e.g. neck and shoulder ipsilaterally (7) or extended to the entire hemisoma (8).
An impairment of the pain transmission system outside the active period has been found and thus involvement of the central tonic pain mechanisms in the pathogenesis of CH has been suggested (9).
The present study aimed to explore, through the nociceptive flexion reflex (NFR) of the lower limbs (RIII reflex), the pain control system in CH patients in the active period and in remission. This neurophysiological method has been used in previous studies to investigate the mechanisms involved in the antinociceptive activities in several pain conditions, including primary headaches (10–14).
Moreover, since the hypothalamus is known to receive nociceptive afferents from the brainstem reticular system and to project heavily onto the periaqueductal grey (PAG) (15, 16), which exert inhibitory control on both the trigeminal and the spinal neurones involved in the transmission of nociceptive messages (17, 18), the NFR could yield information about the activity of these structures in CH.
Materials and method
Study population
Fifty-four patients diagnosed as having CH according to the classification of the International Headache Society (IHS) (19) were recruited from among the people seeking treatment at the University Centre for Adaptive Disorders and Headache (University of Pavia, Italy). Thirty-six patients were suffering from episodic cluster headache (ECH) (31 males (M) and five females (F); mean age 38.0 ± 13.2 years; mean illness duration 9 ± 6 years). Twenty-three tests were carried out in the ECH patients during an active period (ECHp) (19 M and 4 F; mean age 36.7 ± 13.0 years) and 28 tests in patients during a remission phase (ECHr) (25 M and 3 F; mean age 37.2 ± 13.2 years). In 15 ECH patients the examination was repeated in both a cluster phase and a remission phase.
The patients were considered to be in an active period when severe attacks had been occurring more than once daily for at least 10 days. The patients were considered to be in a remission phase when no attacks had occurred for at least 2 months. Eighteen chronic cluster headache patients (CCH) (16 M and 2 F; mean age 44.3 ± 12.7 years; mean illness duration 10.5 ± 8.2 years; frequency of attacks: 1.3 ± 0.7/day) underwent the same investigation. According to the IHS classification nine were chronic form ab initio and 12 secondarily chronic. Twenty-one healthy individuals (19 M and 2 F; mean age 33.7 ± 5.2 years) served as a control group.
All patients and controls were right-handed. At the time of testing, the subjects had been drug-free for at least 24 h, except for O2 inhalation. None of the patients had been treated with ergot-containing medications or with sumatriptan for a period of 2 weeks prior to the study. Prophylactic treatment was tapered off 6–32 weeks before testing. Two cases (ECH in active phase) received a single dose of steroids (desametasone 4 mg 1 fl i.m.) 7 and 10 days before the examination. In the active phase the patients were examined 6–18 h after the attack. All patients gave their informed consent before entering the study. The study was approved by the local ethics committee.
Experimental procedure
During the session, the subjects sat comfortably in an armchair, in order to achieve general muscle relaxation. The RIII reflex was elicited bilaterally (randomly first on the right or symptomatic side, then on the left or non-symptomatic side, or vice versa in each patient) according to a method described elsewhere (10, 14). The reproducibility of this method has been previously documented (20). Briefly, the sural nerve was stimulated with a pair of surface electrodes placed on degreased skin at the retromalleolar site. The stimulus consisted of a volley of 10 rectangular pulses (1 ms duration at 300 Hz), delivered by a constant current stimulator at random intervals (5–20 s). Muscular response was recorded electromyographically from the biceps femoris muscle (capitis brevis) using surface electrodes. The RIII reflex is a nociceptive reflex of the lower limb, that originates from stimulation of Aδ and C fibers, in particular. The minimal intensity of current needed to elicit a reflex response and its maintenance for 80–90% of the time of stimulation was taken as the reflex threshold (Tr). In addition, the threshold of the subjective pain sensation (Tp) was also noted, while recording the RIII reflex.
The Tp and Tr were determined using the staircase limits method following a standardized procedure (10). The Tp was determined as the minimal intensity of current needed to evoke a first perceived pain sensation (i.e. perceived by the patient as pain, and no longer as a tingling sensation). Both Tp and Tr are linear functions of the intensity of stimulation and high correlation coefficients have been shown in healthy subjects. The Tp/Tr ratio is, in fact, usually close to one in normal subjects (10, 14). The symptomatic and non-symptomatic sides were, respectively, compared with the left and right sides of the control subjects. The examination was carried out at the same time of day (10.00–12.00) in all subjects so as to avoid any influence related to circadian variation of the RIII reflex threshold (20).
Statistical evaluation of the results
The data are expressed as mean ± standard deviations. A one-way
Results
The mean age and duration of the disease of patients with CCH and ECH were not significantly different (one-way
When evaluating the side-to-side differences, the Tp and Tr values in ECHp and in CCH were significantly (paired t-test; P < 0.05) reduced on the symptomatic as compared with the non-symptomatic side. No significant (paired t-test; P > 0.1) side-to-side differences were found for ECHr and controls (Figs 1 & 2).
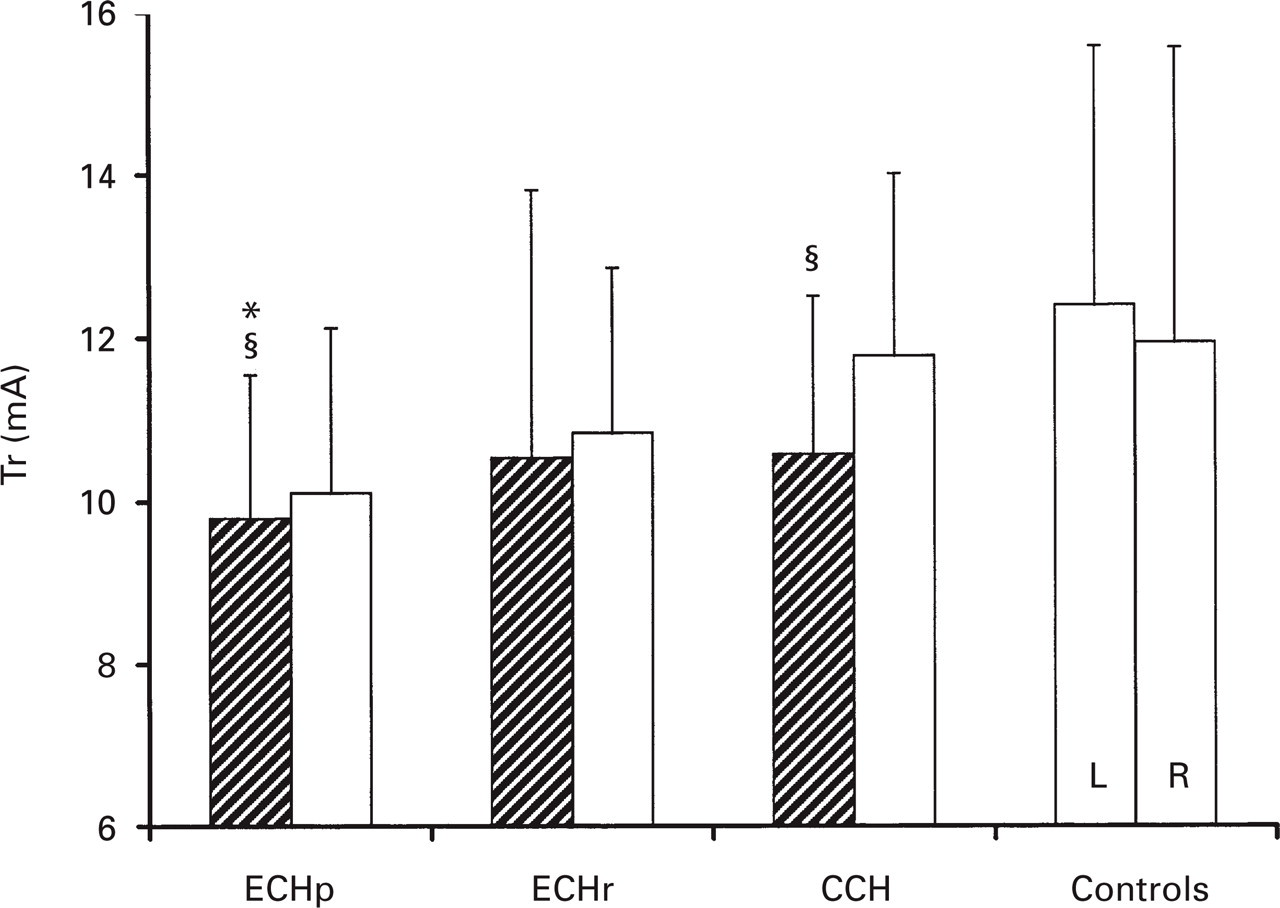
RIII reflex threshold (Tr) in episodic cluster headache in the active period (ECHp), in remission (ECHr), in chronic cluster headache (CCH) and in controls; (mean ± SD).∗P = 0.04 vs. controls (one-way
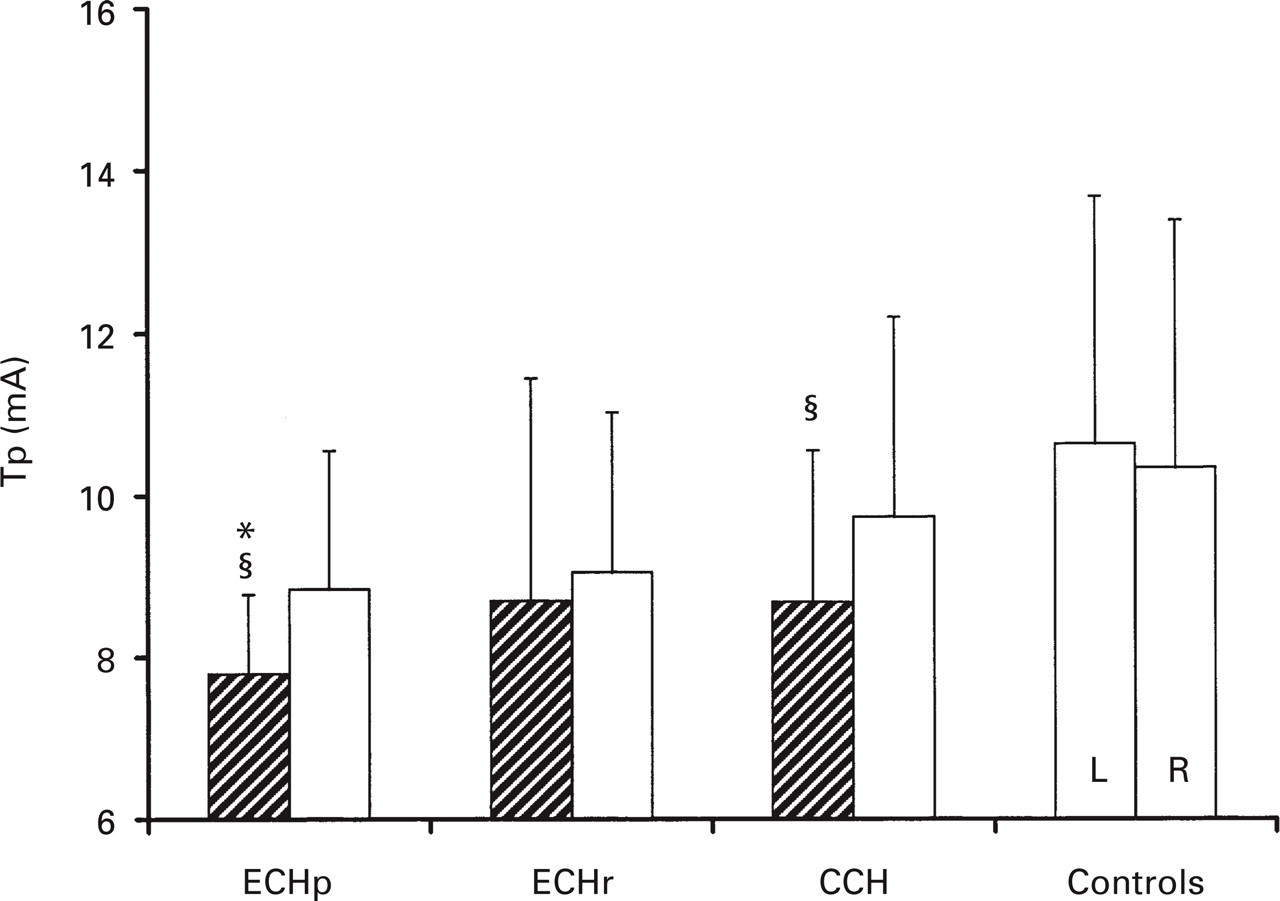
Subjective pain perception (Tp) in episodic cluster in the active period (ECHp), in remission (ECHr), in chronic cluster headache (CCH) and in controls; (mean ± SD).∗P < 0.005 vs. controls (one-way
ECHp patients showed a significantly (one-way
When ECH patients (n = 15) were tested both in the active period and in remission, the Tr on the symptomatic side was significantly (paired t-test; P < 0.05) lower in the active period than in remission (Table 1). The Tp values were also significantly (paired t-test, P < 0.05) lower in the active period than in remission.
RIII reflex threshold (Tr) and subjective pain threshold (Tp) values in 15 episodic cluster headache (ECH) patients. The same subject has been evaluated both in the active period and in remission (mean values ± SD)

P < 0.05 vs. the corresponding value in remission (Student's paired t-test).
The difference in Tp and Tr between CH patients suffering from headache on the left (n = 28) and right (n = 26) sides was not significant.
The Tp/Tr ratios are reported in Table 2. No significant difference was observed between the sides in ECH and CCH and in ECH between remission and the active phase, and when compared with controls.
Tp/Tr ratio values in episodic cluster headache (ECH) patients in period and in remission, in chronic cluster headache (CCH) patients and controls. There are no significant differences between sides, groups and between period and remission phase

The ratio of the number of cluster periods/duration of illness was used to obtain an overall clinical parameter on the severity of CH in the episodic form. A negative correlation was calculated between the above ratio and the Tp in ECHp (r = − 0.56; P < 0.05), while no linear correlation was found between the number of attacks per day in CCH and Tp values (r = − 0.29 P > 0.1).
Discussion
The typical lateralization of pain and autonomic signs in CH has prompted several studies seeking a pathogenic interpretation of this strictly unilateral headache. While in the past, the autonomic dysfunction was investigated in order to clarify the pathogenesis of CH (21), more recently the focus of attention has shifted to the role of the trigeminal pain control system (2, 3). A trigemino-vascular connection and an ortho/antidromic conduction in the trigeminal fibers in particular (4) can account for both a peripheral and a central cause of headache. In a previous study, we documented a significant reduction of the corneal pain threshold (more evident on the pain side) in the active period, and a significantly reduced threshold of the nociceptive muscular response (corneal reflex) on the symptomatic side in CH during the active period (22).
These data suggest reduced inhibitory descending control on trigeminal nuclei in CH during the cluster period. More recent findings also suggest a hyperexcitability of the spinal trigeminal nucleus, as well as deficient descending inhibition by the hypoactive reticular nuclei, possibly related to hypothalamic disturbance and reduced central opioid activity (23). Involvement of the central tonic pain mechanisms in the pathogenesis of CH has also been suggested by a SPECT study during the cold water pressure test (9).
Since mid-brain and brainstem descending pain modulatory influences are common to trigeminal and spinal levels (18), it was of interest to study the threshold of the nociceptive flexion reflex (NFR) of the lower limbs in CH as indirect evidence of suprasegmental abnormalities. The RIII reflex in CH patients, both episodic, during active and remission phases, and chronic forms, were investigated in the present study.
NFR has proven to be an interesting neurophysiological tool for studying spinal and supraspinal pain processes in humans (11, 14).
A decrease in the RIII reflex threshold and the subjective pain threshold on the symptomatic side during the active phase in episodic CH compared with controls is the main result emerging from our investigation. Interestingly, during the remission phase the RIII reflex was within normal values, suggesting that a functional process underlies the pathogenesis of CH. Furthermore, the negative correlation between severity of disease and Tp values we found in ECHp suggests a predisposing condition to enhanced nociception and/or reflect central sensitization during the active period in those subjects who are more severely affected. These data agree with previous investigations concerning the instrumental exploration of autonomic responses to painful stimulation in this disease (25, 26). The data, suggesting that a region of the hypothalamus is activated only during the headache period (5, 27), may also explain why the RIII reflex was within normal values during the remission phase. However, an impairment of the pain transmission system outside the active period has also been reported (9).
Thyrotropin-releasing hormone (TRH) is known to produce changes in flexion reflex excitability (28), while reduced response of prolactin to TRH has been found in female subjects in both phases of cluster headache and in chronic cluster headache (29). However, probably no relationship exists between the two phenomena, since a neuroendocrine influence should produce a bilateral effect in contrast with our results.
Since a decreased Tp/Tr ratio, as observed in tension-type headache, is considered related to a subjective amplification of pain perception (14), parallel changes in the Tr and the Tp observed in CH patients, indicate that psychological factors do not play a role in the pain perception changes observed in our subjects. An impairment in descending inhibitory controls could account for these concomitant changes in Tp and Tr values.
An asymmetry between the two sides is another important finding in our study. Abnormalities (increase in latency or reduction in amplitude) on the painful side have been described in CH patients during the active phase when studying the brainstem auditory evoked potentials (30) and visual evoked potentials (31), suggesting that a central mechanism is prominent in CH pathogenesis.
No difference between the left and the right side was observed in our subjects. These data are in agreement with the finding that the right-lateralized central processing observed in cluster headache patients during nitroglycerin-induced attacks was independent of the pain side (32).
The concept of a central mechanism (most probably dysfunction in the region of the hypothalamus) in CH pathogenesis is increasingly accepted, as a purely vasogenic cause cannot explain all the features of the disease (27). The circadian and circannual occurrence of the symptoms as well as altered hormonal/autonomic rhythms (33–37) may also indicate a centrally located dysfunction, even though haemodynamic mechanisms have also been suggested to explain these findings (2).
Recently, activation of the trigemino-vascular system in patients with acute spontaneous attacks has been postulated on the basis of the observation of an increase in level of calcitonin-gene related peptide (CGRP) and vasoactive intestinal peptide (VIP) in blood from the external jugular vein (38). Activation of a brainstem reflex, whose afferent arch is the trigeminal nerve, may account for these data (38). However, several central structures can be activated by the neural processing of craniovascular pain (39). Central neuroplasticity change or nociceptive sensitization might explain the phenomena, including hyperalgesia, which follow peripheral inflammation (40).
Even though the oculocephalic symptoms are the dominant feature of a CH attack, extracephalic pain can be observed in some patients, while cutaneous and deep hyperalgesia homolateral to the symptomatic side has also been described in CH patients (8, 36).
In the multiple comparison we failed to reveal significant abnormalities of Tr and Tp in chronic CH, even though we found a significant decrease on the pain side compared to the non-symptomatic side. Clinical heterogeneity (41) and the long-term use of prophylactic medications (i.e. lithium salts is known to interact with serotonergic and opiate receptors) (42), which may have induced changes in the sensitivity of receptors in the pain signalling system could explain the less evident alteration in chronic form.
In conclusion, our data confirm, by means of a neurophysiological method, the existence of a reduced pain threshold on the painful side also in extracephalic parts of the body in episodic CH patients during the active phase, indicating an involvement of the pain control system in this disorder.
Footnotes
Acknowledgements
This work was partly supported by a grant from the Ministry of Health No. ICS 57.2/RC90.1277 and from CNR No. 95.02305.CT04. We are grateful to Miss Mirella Merli for her technical assistance and to Mrs Catherine Wrenn for language editing.
Professor Ivan Milanov and Professor Andrei Danilov participated in this study during their research stages. The stages were kindly supported by CIRNA Foundation and University Center for Adaptive Disorders and Headache.
