Abstract
Uncontrolled increase in intracranial pressure (ICP) continues to be one of the most significant causes of early death in patients with acute liver failure (ALF). In this study, we aimed to determine the effects of indomethacin on ICP and cerebral perfusion pressure in twelve patients with ALF and brain edema (9 females/3 males, median age 49,5 (range 21 to 64) yrs.). Also changes in cerebral perfusion determined by transcranial Doppler technique (Vmean) and jugular bulb oxygen saturation (SvjO2) were measured, as well as brain content of lactate and glutamate by microdialysis technique. Finally, we determined the cerebral blood flow autoregulation before and after indomethacin injection. We found that indomethacin reduced ICP from 30 (7 to 53) to 12 (4 to 33) mmHg (P < 0.05). The cerebral perfusion pressure increased from 48 (0 to 119) to 65 (42 to 129) mmHg (P < 0.05), while Vmean and SvjO2 on average remained unchanged at 68 (34 to 126) cm/s and 67 (28 to 82) %, respectively. The lactate and glutamate in the brain tissue were not altered (2.1 (1.8 to 7.8) mmol/l and 34 (2 to 268) μmol/l, respectively) after injection of indomethacin. Cerebral blood flow autoregulation was impaired in all patients before injection of indomethacin, but was not restored after administration of indomethacin. We conclude that a bolus injection of indomethacin reduces ICP and increases cerebral perfusion pressure without compromising cerebral perfusion or oxidative metabolism in patients with ALF. This finding indicates that indomethacin may be valuable as rescue treatment of uncontrolled intracranial hypertension in fulminant hepatic failure.
Keywords
Swelling of astrocytes is a common neurohistological feature in patients with liver injury, which in severe cases of acute liver failure (ALF) may result in cerebral edema, high intracranial pressure (ICP) and serious brain damage (Norenberg and Bender 1994). Though the exact pathophysiological mechanism is essentially unknown, there is accumulating evidence that high circulating levels of ammonia resulting from the splanchnic vascular bed (Clemmesen et al. 2000) are a key mediator of these complications (Schenker et al. 1967). Ammonia easily crosses the blood-brain barrier by diffusion and through ion channels (Ott and Larsen 2004) and influences brain metabolism (Bessman and Bessman 1955) and neurotransmitter trafficking (Felipo and Butterworth 2002). Arterial ammonia concentration has also been demonstrated to correlate to brain water content and ICP in the experimental setting (Takahashi et al. 1990; Olafsson et al. 1995). Indeed, it has been shown that severe hyperammonemia also correlates to cerebral herniation in patients with ALF (Clemmesen et al. 1999; Strauss et al. 2001a; 2001b).
Exposure of isolated astrocytes to millimolar ammonia concentration induces metabolic disturbances with membrane depolarization, calcium influx, glutamate release, activation of lipases and proteases. These alterations may set the stage for astrocyte injury with cascades that involve free radicals, protein nitration and mitochondrial damage (Norenberg 2003). Besides these profound metabolic alterations there is accumulating evidence that an increase in cerebral blood flow (CBF) is of critical importance for development of brain edema and high ICP in patients with ALF (Durham et al. 1995; Larsen 1996; Blei and Larsen 1999). Although the exact mechanism behind ammonia-induced cerebral vasodilatation is not fully understood (Larsen et al. 2001) it has been reported that the gene expression of the enzyme cyclooxygenase (COX) is increased in brain during hyperammonemia (Song et al 2002). This finding may suggest that high CBF is mediated by increased ammonia-induced production of prostaglandins and epoxyeicosatrienoic acids, probably by mediating their effect by activation of potassium channels in arterioles (Harder 1997).
The pathophysiological changes within the brain induced by ammonia have important clinical implications for management of patients with ALF (Larsen and Wendon 2002). Several studies, experimental and clinical, support the idea that restriction of CBF is of critical importance to prevent edema and high ICP (Larsen 1996; Cordoba et al. 1999; Master et al 1999). In fact, it has been demonstrated in an experimental model that inhibition of COX by intravenous indomethacin restrict CBF and ameliorates development of brain edema (Chung et al. 2001). Certainly, this latter experimental finding raises the possibility that indomethacin increases cerebrovascular tone and reduce both cerebral blood volume and ICP. Such a CBF restriction, however, also raises the possibility that indomethacin may compromise cerebral oxidative metabolism (Larsen 1996; Clemmesen et al. 1997) and aggravate brain damage. In this clinical study, we determined if a bolus injection of indomethacin reduces ICP in patients with ALF complicated by brain edema. We also evaluated the possible adverse effects of indomethacin by measuring cerebral perfusion, jugular bulb oxygen saturation, and lactate concentration in the extracellular space of the brain cortex using a microdialysis technique. Since, two studies suggest that an increase in cerebral vascular tone induced by hyperventilation (Strauss et al. 1998) and by hypothermia (Jalan et al. 2001), restore the impaired CBF autoregulation in patients with ALF we also determined if indomethacin could restore CBF autoregulation.
PATIENTS AND METHODS
Patients
Twelve patients with severe ALF (9 females/3 males, median age 49.5 (range 21 to 64) yrs.) were included (Table 1). All patients had preserved pupillary light response during the study and all patients were maintained on mechanical ventilation (Servo 900, Siemens, Solna, Sweden). Nine patients with ICP above 20 mmHg in spite treatment with mannitol, mild hyperventilation and sedation as well as three patients with normal ICP received an IV bolus of indomethacin 25 mg. Cerebral microdialysate was collected before and until 2 hours after injection of indomethacin and was analyzed for extracellular concentrations of lactate and glutamate in 6 patients. The local Ethics Committee approved the study and informed consent was obtained from the patients (before development of encephalopathy) or the next of kin (KF 00/002).
Age, gender, and biomarkers of liver failure at the time of study for each of the indomethacin treated patients. Also complication and outcome are given
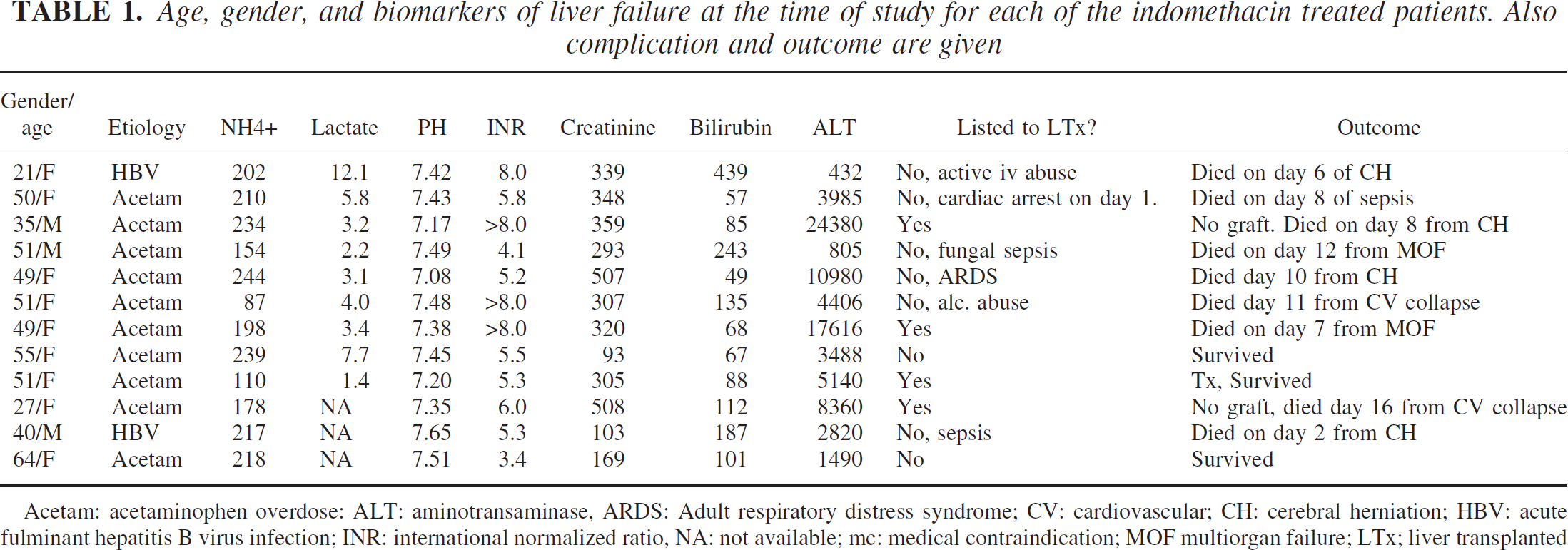
Acetam: acetaminophen overdose: ALT: aminotransaminase, ARDS: Adult respiratory distress syndrome; CV: cardiovascular; CH: cerebral herniation; HBV: acute fulminant hepatitis B virus infection; INR: international normalized ratio, NA: not available; mc: medical contraindication; MOF multiorgan failure; LTx; liver transplanted
Hemodynamic and metabolic variables. ICP
ICP-monitoring was performed in all patients by an intraparenchymal fiberoptic device (Camino Laboratories, San Diego, CA, U.S.A.) inserted into the posterior frontal cortex. Cerebral perfusion pressure was calculated as mean arterial blood pressure (MABP) minus ICP
Changes in CBF was estimated by transcranial Doppler sonography (TCD) mean flow velocity (Vmean) using a 2 MHz pulsed Doppler probe (Multi Dop X, DWL, Ulden, Germany) in the middle cerebral artery. The TCD-probe was maintained in the right temporal region by a headband with insonation through ʻthe posterior temporal window' at a depth of 45 to 55 mm (Larsen 1996).
Microdialysis
A cerebral microdialysis catheter with a membrane length of 3 mm was also inserted. The microdialysis flow rate was 0.3 μL/minute The technique is previously described (Tofteng et al. 2002): in short the microdialysis catheter consists of a thin, double-lumen, 60 mm long plastic tube with a 10 mm long semipermeable membrane at the tip. After the catheter was inserted into brain cortex the catheter was connected to a micro infusion pump (CMA/106 Microinjection pump; CMA Microdialysis AB, Stockholm, Sweden) and continuously perfused with artificial cerebral spinal fluid at a rate of 0.3 μl/minute The perfusion fluid passes between the inner membrane and dialysis membrane. The dialysate was collected in microvials and contained each 10 ul for biochemical analysis. Subsequently the microdialysis samples were analyzed using a CMA 600 Microdialysis Analyser (CMA Microdialysis AB). Biochemical analysis of lactate and glutamate, were recorded 1 hours before and 1 hours after bolus injection of indomethacin.
Arterial pressure was measured with a catheter in the radial artery. Internal jugular bulb saturation (SvjO2) was monitored to help secure sufficient cerebral perfusion as part of a routine protocol. All patients received 20% glucose I.V., propofol IV (15 mg/h) and all were slightly mechanically hyperventilated (PRVC modus)(Servo 300, Siemens, Solna, Sweden) to ensure a slightly decreased carbon dioxide tension in arterial blood (PaCO2) without muscle relaxation. Respirator settings were not changed during the study.
Cerebral autoregulation was also determined prospectively in 7 patients (Table 1; patients 1 to 7) Vmean and cerebral perfusion pressure were both recorded before and during intravenous infusion of norepinephrine (2 mg 100mL-1, 0,05 to 0,15 μg min-1), to increase MABP more than 20 mmHg. Arterial blood samples were drawn anaerobically in heparinized syringes and immediately analyzed for oxygen saturation (ABL 625, Radiometer, Copenhagen, Denmark).
Statistical analysis
All data are presented as medians and range with a significance level at P < 0.05. For comparisons, the Mann-Witney and the Wilcoxon tests were applied for analysis of unpaired and paired data, respectively. For the evaluation of interdependencies the Spearmans test was used.
RESULTS
All patients suffered from severe ALF at the time of study with hepatic encephalopathy stage 4, a median arterial ammonia (NH4+) concentration of 206 (range 87 to 244) umol/l, arterial lactate concentration of 3.4 (1.4 to 12.1) mmol/l, International Normalization Ratio 5.7 (3.4 to 8.0), bilirubin 95 (49 to 278) mmol/l and amino transaminase (ALT) of 4195 (439 to 24380).
Nine of the 12 patients fulfilled the Kings College Criteria for poor prognosis (Bernal et al 2002). Four of these 9 patients were listed for liver transplantation while 5 patients were not listed due to signs of sepsis, suspected cerebral ischemia after cardiac arrest, adult respiratory distress syndrome, active iv abuse or psychosocial reasons (Table 1). A liver graft did not become available for 3 of the 4 four listed patients. These 3 patients died of sepsis.
Ten of the 12 patients were considered at high risk to suffer from cerebral herniation, as arterial ammonia concentration exceeded 150 μmol/l (Clemmesen et al 1999). Four of these patients died from cerebral herniation 6 (2 to 8) days later (Table 1), four patients died from sepsis and two patients survived.
A second injection of indomethacin was given to 5 patients to control ICP 27.7 (3.5 to 44) hours after the first injection. In the first part of the study consisting of all 12 patients with ALF we found that indomethacin reduced ICP from 30 (7 to 53) to 12 (4 to 33) mmHg (P < 0.05) and that cerebral perfusion pressure increased from 48 (0 to 119) to 65 (42 to 129) mmHg (P < 0.05). In 7 patients Vmean was recorded by transcranial Doppler sonography and remained unchanged at 68 (34 to 126) cm/s after injection of indomethacin. Also SvjO2, as determined in 8 patients, remained unchanged at 67 (28 to 82)% (Fig. 1) and Table 2.
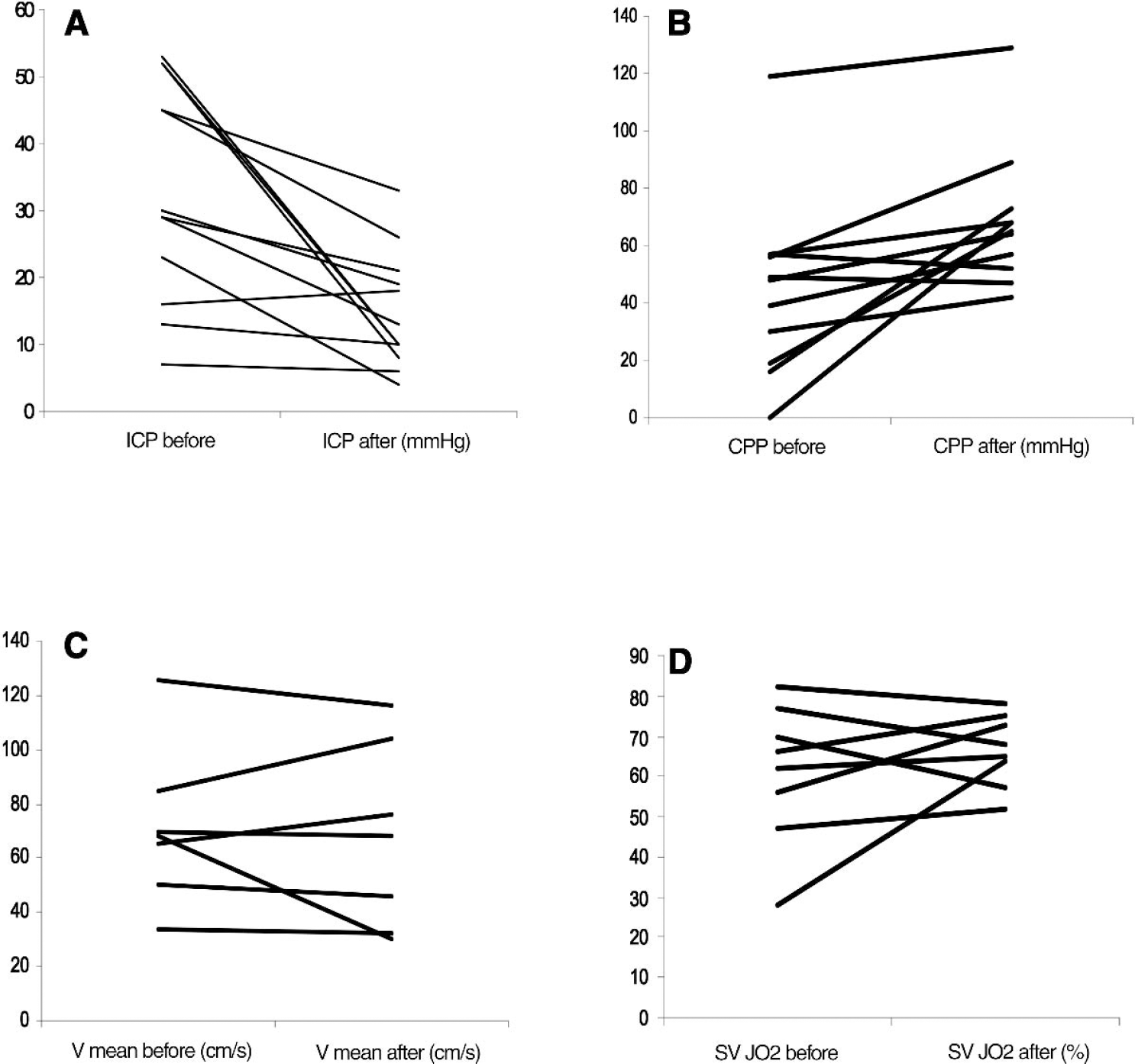
Changes in
Intracranial pressure (ICP), internal jugular bulb saturation (SvjO2), lactate (mmol/l) and glutamate (μmol/l) concentration in the cerebral extracellular space before and after injection of indomethacin in 9 of the 12 studied patients. In the 3 remaining patients only ICP and cerebral perfusion pressure were measured (see Fig. 1 and text)
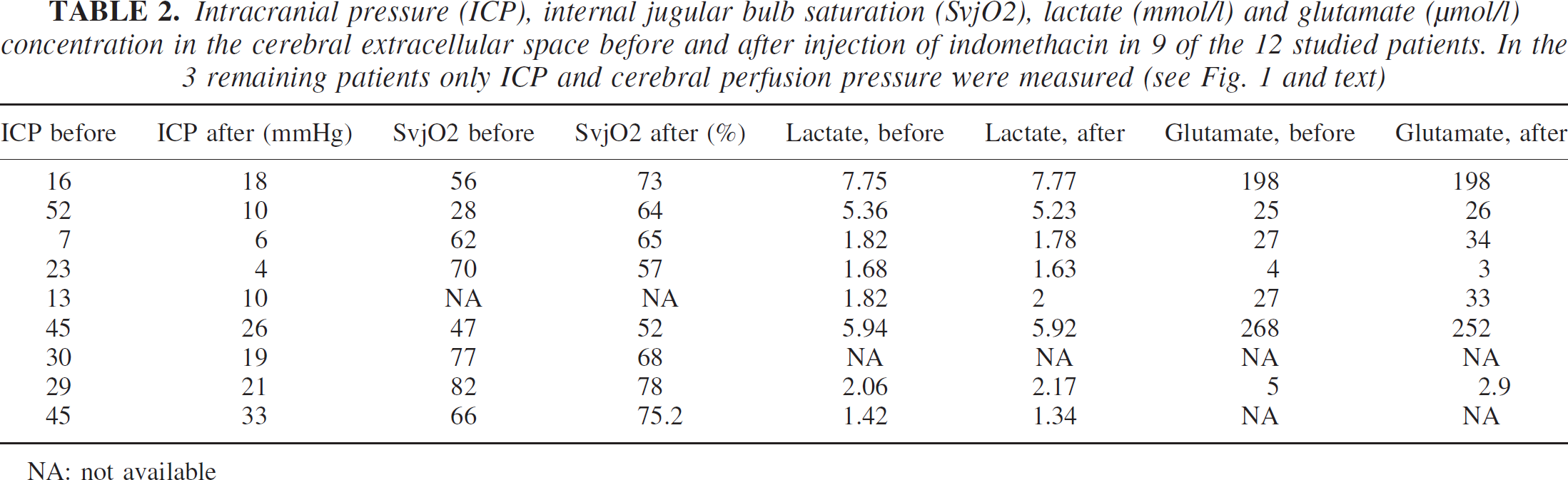
NA: not available
No changes in the brain tissue concentration of lactate and glutamate were observed after indomethacin injection (2.1 (1.8 to 7.8) mmol/l and 34 (2 to 268) μ mol/l, respectively).
In this study injection of a bolus dose of indomethacin did not influence urine output in 3 patients with preserved kidney function before indomethacin injection. Two of these patients survived spontaneously and 1 patient died.
Platelet was low in all the studied patients before indomethacin injection. In addition to the compromised secondary hemostasis that is always severely compromised in ALF, impairment of primary hemostasis induced by indomethacin could theoretically be feared to result in bleeding episodes. However, no bleeding episodes or oozing from invasive lines developed. Nor did we observe any bleeding from the tube in trachea, the gastrointestinal and the urogenital systems.
In the second part of the study we tested cerebral autoregulation before injection of indomethacin (Fig. 2). During infusion of norepinephrine MABP increased from 70 (64 to 85) to 100 (90 to 116) mmHg, and cerebral perfusion pressure increased from 57 (44 to 80) to 88 mmHg (75 to 104). Vmean increased from 67 (34 to 124) to 85 (58 to 193) cm/s an increase of 26%. The increase in Vmean exceeded 10% in all patients, indicating loss of CBF autoregulation (Strauss et al. 1997). ICP did not change (10 (5 to 26) vs. 14 (5 to 24) mmHg (NS)).
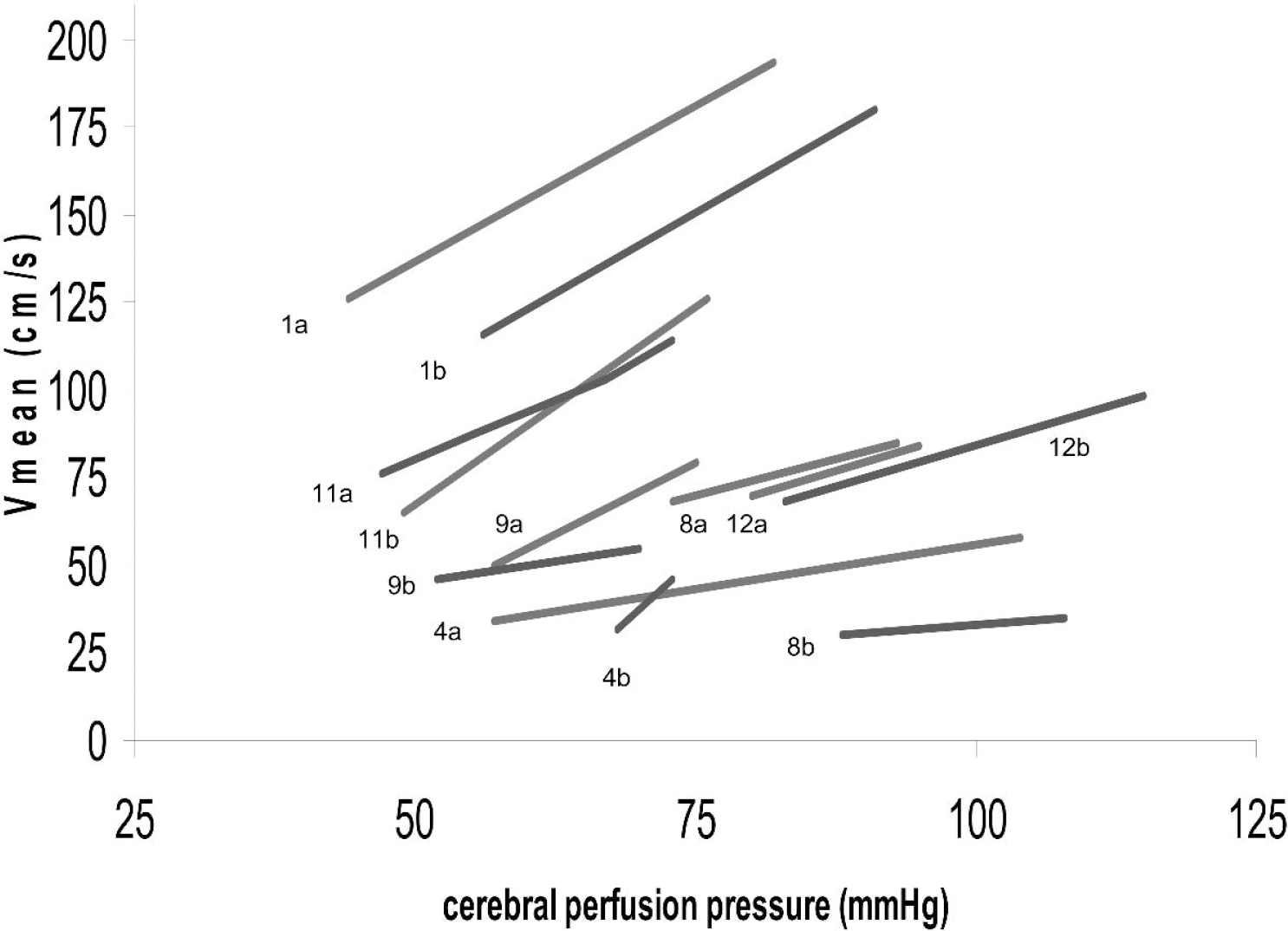
The relationship between cerebral perfusion pressure and cerebral blood flow velocity before (“a”) and after (“b”) injection of indomethacin for each individual patient studied. Patients are numbered in the figure according to the number the patient is listed in Table 1.
After indomethacin administration autoregulation was tested again. During norepinephrine infusion MABP increased from 73 (65 to 93) to 104 (80 to 120) mmHg and cerebral perfusion pressure increased from 62 (47 to 88) to 82 (70 to 115) mmHg. Vmean increased from 57(30 to 116) to 77 (35 to 180) mmHg, an increase of 32%, the increase in Vmean again exceeded 10% in all patients still indicating loss of CBF autoregulation. The increase in Vmean was not statistically different from the increase in Vmean measured before indomethacin injection. ICP did not increase on average 8 (5 to 18) to 10 (5 to 27) mmHg. The average increase in MABP during norepinephrine infusion was identical before (43%) compared to after (34%) administration of indomethacin (NS) (Fig. 2).
DISCUSSION
In this study, we found that a single bolus injection of indomethacin reduced ICP and increased the cerebral perfusion pressure in patients with severe ALF complicated by brain edema. As expected such a beneficial effect upon ICP and cerebral perfusion pressure did not compromise cerebral oxygenation or the lactate concentration within the extracellular space of the brain. The rapid effect of indomethacin on ICP indicates that indomethacin increases the tone of cerebral arterioles. Alternatively, it could be speculated that the decrease in ICP is mediated by a decrease in the cerebral venous blood volume. The cerebral vascular resistance is calculated as cerebral perfusion pressure divided by CBF and our data indicates that the cerebral vascular resistance actually increased after indomethacin injection, i.e. cerebral arteriolar tone increased. Experimental studies also show that both CBF and ICP are controlled by prophylactic administration of indomethacin (Chung et al. 2001). Thus, we find support for the assumption that indomethacin rapidly and potently reduce ICP in patients with ALF by increasing the cerebral vascular resistance and decreasing cerebral blood volume.
The effect of indomethacin was persistent in 7 of the 12 patients, i.e. indomethacin was not administered again. In the remaining 5 patients ICP increased again, which required a new injection of indomethacin. The effect of indomethacin was equal efficient in reducing ICP at this second time of injection in 4 of the patients, while the last patient maintained a relatively high ICP (>20 mmHg) for 3 hours.
Although this safety study only studied 12 patients, we expected 10 of these patients to suffer from cerebral herniation as arterial ammonia concentration exceeded 150 μmol/l (Clemmesen et al 1999; Strauss et al. 2001b). We found, however, that only 4 of these 10 patients died from cerebral herniation 6 (2 to 8) days later (Table 1). Two of the 10 patients survived without neurological deficits. The remaining 4 patients was also successfully treated for high ICP but died several days later from sepsis and multiorgan failure. This experience stresses that rescue treatment with indomethacin for high ICP is efficient in prolonging survival while waiting for definitive therapy, i.e. acute liver transplantation.
Some concerns of the potential side effects of indomethacin should be addressed. Indomethacin injection could theoretically result in bleeding episodes as the platelets function and primary hemostasis is impaired. However, we did not observe any bleeding episodes or oozing from invasive lines in any of our patients.
Although indomethacin in this study did not influence urine output in three patients with preserved kidney function one concern is that extra-cerebral organ blood flow may be critically restricted; especially renal perfusion. However, such reservations should be put into the context of an otherwise life threatening condition with an uncontrolled, critical high ICP. Furthermore, the large majority of patients suffering from high ICP also have renal failure. Another concern could be that indomethacin compromises cerebral oxidative metabolism. We found that cerebral perfusion remained unchanged, and that the cerebral lactate-to-oxygen-index in the jugular vein remained unchanged after indomethacin injection (data not shown). To further determine if indomethacin compromise oxidative metabolism we also measured lactate in the extracellular space of the brain using cerebral microdialysis. We found that lactate concentration was not increased by indomethacin. These data do not support the possibility that indomethacin compromise cerebral energetics.
Cerebral blood flow velocity decreased in 5 and increased in 2 patients after indomethacin injection. In both healthy volunteers (Jensen et al. 1993) and in experimental studies of hyperammonemia indomethacin reduces CBF, and might have been expected in all our studied patients. It is possible that the increase in 2 patients could result from inaccuracy of the transcranial Doppler technique to reflect changes in CBF (Clemmesen et al. 1997, Strauss et al. 2001a; 2001b) after administration of indomethacin. However, this is unlikely as SvjO2 changed in parallel with blood pressure velocity. A more complex series of events is considered to be operative. Indomethacin increases cerebral perfusion pressure. Since, autoregulation is absent in patients with ALF (Larsen et al 1996), CBF would increase. However, indomethacin also increases the cerebral arteriolar tone (i.e. cerebrovascular resistance), which will tend to counteract an increase in CBF. Such a delicate passive balance between perfusion pressure and resistance is not directly operative in healthy man (Paulson et al. 1990) or in hyperammonemia models (Hirata et al. 1995) because of normal CBF autoregulation.
We found that the autoregulation of CBF was impaired before injection of indomethacin in all the studied patients. This finding is in accordance with a number of previously clinical studies (Strauss et al. 1997; 1998; Jalan et al. 2001; Larsen et al. 1996). The loss of CBF autoregulation in ALF is assumed to be associated with a gradual loss of arteriolar tone. It has previously been demonstrated that interventions such as hyperventilation and hypothermia restores the CBF autoregulation in patients with ALF, and has been assumed to result from an increase in cerebral vascular resistance. If the mechanism behind loss of autoregulation in ALF results from a simple loss of arteriolar tone, indomethacin would also be expected to restore it. We found that indomethacin did not alter the slope between cerebral perfusion pressure and Vmean, i.e. it did not restore autoregulation. The explanation for the fact that hyperventilation and hypothermia restores autoregulation, while indomethacin has no effect remains elusive. Yet, our finding indicates that the impaired CBF autoregulation in patients with ALF is not solely explained by vasodilatation. Other factors, independent of cerebrovascular tone seem to be involved. We speculate that the substance that are responsible for loss of autoregulation in ALF is not released from the brain endothelium but rather is released from structures localized on the abluminal side of the blood-brain barrier. One such factor could be the organic osmolyte glutamine that is known to be severely elevated both in the brain and in the systemic circulation in ALF (Norenberg and Bender 1994; Tofteng et al. 2003; Clemmesen et al. 2000). Indeed, experimental data provided by Okada et al. (2000) suggests that cerebrovascular reactivity is blunted by a high concentration of glutamine.
CONCLUSION
In conclusion, indomethacin reduced ICP without influencing cerebral perfusion, lactate or glutamate concentrations within the brain tissue. Although it was expected that indometacin would restore the cerebral autoregulation, as the cerebral vascular resistance was increased this was not the case indicating that loss of cerebrovascular reactivity is not simply caused by arteriolar dilatation. The potent and safe effect of indomethacin upon ICP, cerebral perfusion and metabolism indicate that COX-2 inhibition may be valuable, as a novel important rescue treatment of uncontrolled intracranial hypertension in ALF.
