Abstract
Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) is a hereditary small-vessel disease caused by mutations in the NOTCH3 gene on chromosome 19. On magnetic resonance imaging (MRI), subcortical white matter hyperintensities and lacunar infarcts are visualized. It is unknown whether a decrease in cerebral blood flow or cerebrovascular reactivity is primarily responsible for the development of white matter hyperintensities and lacunar infarcts. The authors used phase-contrast MRI in 40 NOTCH3 mutation carriers (mean age 45 ± 10 years) and 22 nonmutated family members (mean age 39 ± 12 years), to assess baseline total cerebral blood flow (TCBF) and cerebrovascular reactivity after acetazolamide. Mean baseline TCBF was significantly decreased in NOTCH3 mutation carriers. In young subjects, baseline TCBF was significantly lower than in nonmutation carriers (mean difference 124 mL/min). Furthermore, baseline TCBF did not differ significantly between mutation carriers with minimal and mutation carriers with moderate or severe white matter hyperintensities. No significant difference in mean cerebrovascular reactivity was found between mutation carriers and nonmutation carriers. This study suggests that a decrease in baseline TCBF in NOTCH3 mutation carriers precedes the development of white matter hyperintensities.
Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) is a hereditary small-artery disease, clinically characterized by transient ischemic attacks, strokes, progressive subcortical dementia, migraine with aura, and mood disturbances (Chabriat et al., 1995a; Dichgans et al., 1998; Desmond et al., 1999). Mutations in the NOTCH3 gene are associated with CADASIL (Joutel et al., 1996). On pathologic examination, characteristic depositions of granular osmiophilic material within the media of the small and medium-sized leptomeningeal and long perforating arteries of the brain are found. This vasculopathy results in destruction of vascular smooth muscle cells and fibrous thickening of the arterial wall (Baudrimont et al., 1993; Brulin et al., 2002). Magnetic resonance imaging (MRI) reveals symmetrical white matter hyperintensities (WMH), often associated with small lacunar infarcts in the white matter and basal ganglia (Skehan et al., 1995; Chabriat et al., 1998; Yousry et al., 1999).
Cerebrovascular reactivity reflects the compensatory dilatory capacity of cerebral arterioles to a stimulus such as carbon dioxide or acetazolamide. In elderly persons, impaired cerebrovascular reactivity is associated with WMH and lacunar infarcts (Bakker et al., 1999; Molina et al., 1999; Cupini et al., 2001). In patients with CADASIL, alterations in cerebral blood flow and cerebrovascular reactivity have also been shown, with a significant reduction in baseline cerebral blood flow and cerebrovascular reactivity in areas of WMH (Mellies et al., 1998; Chabriat et al., 2000; Pfefferkorn et al., 2001; Bruening et al., 2001).
The aim of the present study was to further explore the correlation of flow disturbance and WMH. We hypothesized that flow disturbance is the primary event that gives rise to the development of WMH. To address this issue, we assessed baseline total cerebral blood flow (TCBF) and cerebrovascular reactivity using phase-contrast MRI in CADASIL patients with minimal WMH, in CADASIL patients with moderate or severe WMH, and in nonmutated family members.
MATERIALS AND METHODS
Subjects
Sixty-three members of 15 CADASIL families participating in an ongoing study of CADASIL agreed to undergo MRI. NOTCH3 mutation screening was performed for all subjects by direct sequencing analysis (Oberstein et al., 1999). Family members who proved not to have a NOTCH3 mutation served as a control group. Participants were considered symptomatic when they had a history of neurologic deficits or cognitive decline. In addition to the standard MRI protocol, 38 participants gave permission for the administration of acetazolamide. The institutional medical ethical committee approved the study.
Magnetic resonance imaging
All imaging was performed on an MR system operating at a field strength of 1.5 T (Philips Medical Systems, Best, The Netherlands). Conventional T1-weighted spin-echo images (slice thickness 6 mm with a 0.6-mm interslice gap, repetition-to-echo time [TR/TE] 600/20 milliseconds, matrix 256 × 205, and a field of view [FOV] 220 × 165 mm), dual T2-weighted fast spin-echo images (slice thickness 3 mm without interslice gap, TR/TE 3,000/27/120 milliseconds, matrix 256 × 205, FOV 220 × 220 mm), and fast fluid-attenuated inversion recovery images (3.0/0.0 mm, TR/TE 8,000/100 milliseconds, inversion time 2,000 milliseconds, matrix 256 × 192, FOV 220 × 176 mm) were obtained. All images were performed in the axial plane parallel to the inferior border of the genu and splenium of the corpus callosum. A gradient-echo phase-contrast technique (TR/TE 16/9 milliseconds; flip angle 7.5°; 5-mm slice thickness; FOV 250 × 188 mm; matrix 256 × 154) with a velocity encoding of 100 cm/s was used for nontriggered flow measurements (Bakker et al., 1996). The scan plane was chosen perpendicular to the basilar artery and both internal carotid arteries. Cerebral blood flow was measured 10 minutes before the administration of 14 mg/kg acetazolamide intravenously and repeated at 5, 10, 15, and 20 minutes after the administration of acetazolamide. The same position of the scan plane was used for repeated flow measurements. Blood pressure, peripheral saturation, and heart rate were continuously monitored.
For cerebral blood flow assessment, images were analyzed using the software package FLOW (van der Geest et al., 1998; van Mil et al., 2002). A region of interest was manually drawn around the basilar artery and the left and right internal carotid artery in the phase image by one observer (R. v. d. B.). The flow volume was calculated by integrating the flow velocity values within the contour multiplied by the area. Flow in the basilar artery and the left and right internal carotid artery were added and considered to represent TCBF in milliliters per minute (Buijs et al., 1998). The baseline TCBF and maximum TCBF were respectively defined TCBF before and the maximum TCBF obtained after the administration of acetazolamide. Cerebrovascular reactivity was defined as: cerebrovascular reactivity = (maximum TCBF – baseline TCBF)/baseline TCBF × 100%.
White matter hyperintensities and lacunar infarcts rating
WMH were defined as white matter areas with increased signal intensity on intermediate, T2, and fluid-attenuated inversion recovery weighted images. WMH volume (in milliliters) measurements were performed on fast spin-echo dual images by one observer, using a locally developed semiautomated segmentation software that combines knowledge-based fuzzy clustering and region-growing techniques. The software computes an additional T2/proton density angle image to distinguish the lesions from cerebrospinal fluid. Volume of WMH was corrected for total brain volume by dividing the individual volume of WMH by intracranial volume and expressed in percent. The whole postprocessing procedure yielded an intraclass correlation coefficient of >0.99 (95% CI: 0.96–1.0) in an analysis of data sets from 10 patients examined twice by the same observer who performed the automated segmentation.
In addition, the number of lacunar infarcts, defined as parenchymal defects not extending to the cortical gray matter with a signal intensity following that of cerebrospinal fluid on all pulse sequences, irrespective of size, were counted on hardcopies by an experienced neuroradiologist. In the basal ganglia, areas that were isointense to cerebrospinal fluid on all pulse sequences and located in the lower one third of the corpus striatum were excluded in an effort to differentiate lacunar infarcts from normal dilated perivascular spaces (Jungreis et al., 1988; Heier et al., 1989).
Statistical analysis
Differences between NOTCH3 mutation carriers and nonmutated persons in volume of WMH, number of lacunar infarcts, and flow parameters were performed with analysis of variance. Increase of TCBF after acetazolamide was analyzed with paired t-test and the effect of age on volume of WMH and baseline TCBF with linear regression analysis. In a second phase of the analysis, the mutation carriers were separated into groups with minimal volume of WMH and with moderate or severe volume of WMH, to assess the nature of the correlation between WMH and flow changes. Analysis of variance was performed with age and sex as covariates. A P value of <0.05 was considered significant. The statistical package SPSS-10 (SPSS, Inc., Chicago, IL, U.S.A.) was used for data analysis.
RESULTS
Study population
Of 63 CADASIL family members, 41 subjects had a mutation in the NOTCH3 gene and 22 did not. One person had an incomplete MRI examination because of claustrophobia and was therefore excluded from the analysis. The mean age of the remaining 40 NOTCH3 mutation carriers was 45 years ± 10 years. Thirty-three patients of this group were symptomatic, with symptoms ranging from one transient ischemic attack to multiple strokes and cognitive decline. Seven mutation carriers were asymptomatic. The 22 nonmutation carriers had a mean age of 39 ± 12 years and were all asymptomatic (Table 1). Twenty-three mutation carriers (6 with minimal and 17 with moderate and severe WMH) and 15 nonmutated carriers consented to administration of acetazolamide.
Baseline characteristics of the CADASIL population

CADASIL, cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy.
Comparison between NOTCH3 mutation carriers and nonmutation carriers
The total volume of WMH, number of lacunar infarcts, and flow parameters are given in Table 2. The mutation carriers had a significantly higher volume of WMH and a higher number of lacunar infarcts, whereas baseline TCBF was significantly lower. In both groups, the average TCBF increased significantly after the administration of acetazolamide (P < 0.001). Because the increase in TCBF on administration of acetazolamide was proportional to baseline TCBF, no significant difference in cerebrovascular reactivity was found.
Differences between NOTCH3 nonmutation and NOTCH3 mutation carriers in WMH volume, number of lacunar infarcts, and flow parameters

P values by analysis of variance. For mean CVR, n = 38 (see Methods).
WMH, white matter hyperintensities; bTCBF, baseline total cerebral blood flow; CVR, cerebrovascular reactivity.
The changes in volume of WMH according to age are graphically represented in Fig. 1. This figure shows that there is an increase in volume of WMH in mutation carriers with age; volume of WMH in nonmutated carriers did not increase.
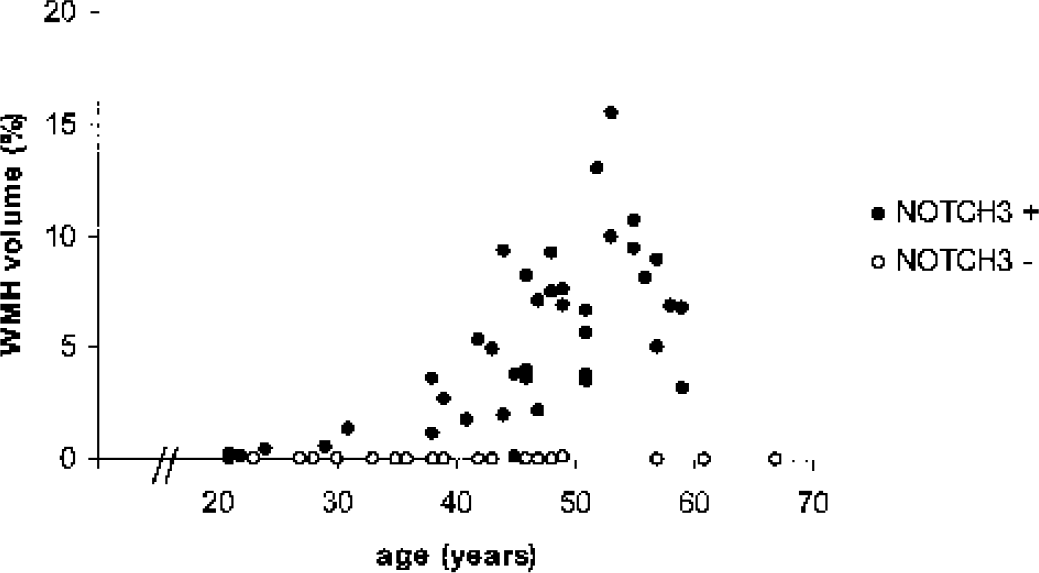
Graph shows white matter hyperintensities (WMH) volume according to age in NOTCH3 nonmutation and mutation carriers. WMH increase with age in NOTCH3 mutation carriers but not in NOTCH3 nonmutation carriers.
In Fig. 2, changes in baseline TCBF versus age are shown. As shown by the parallel regression lines through the NOTCH3 mutation carriers and the NOTCH3 nonmutation carriers, the TCBF was lower at any age in the NOTCH3 mutation carriers (mean difference 124 mL/min, P = 0.001).
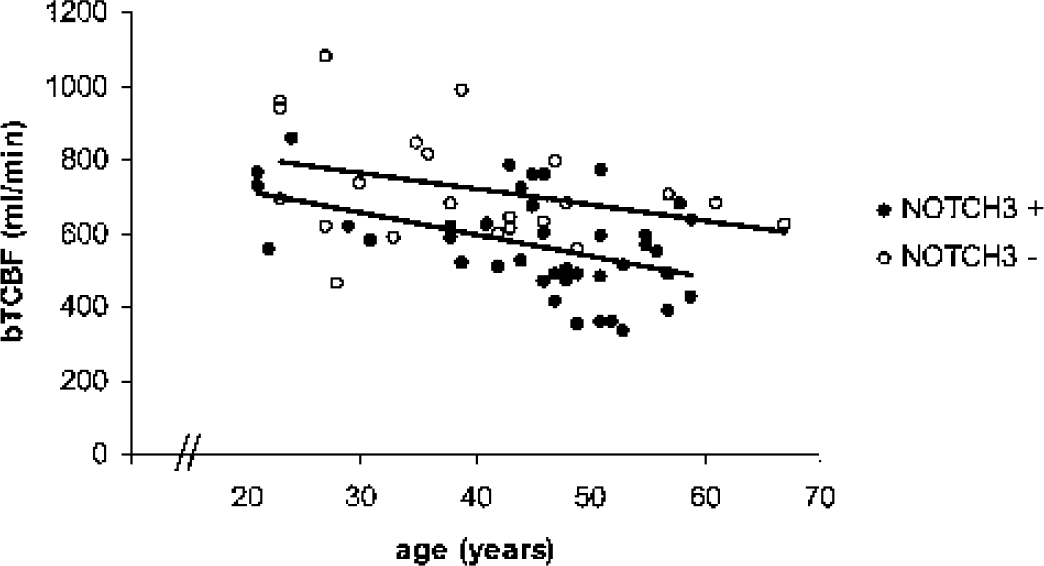
Baseline total cerebral blood flow (bTCBF) in NOTCH3 mutation and nonmutation carriers. bTCBF is significantly lower in NOTCH3 mutation carriers compared with NOTCH3 nonmutation carriers (mean difference 124 mL/min).
Comparison between NOTCH3 mutation carriers with minimal and moderate–severe WMH
Figure 1 shows that there were 8 NOTCH3 mutation carriers with minimal and 32 NOTCH3 mutation carriers with moderate or severe WMH. The cutoff value of volume of WMH was 1.5%. To study the correlation between low TCBF and WMH in patients with a NOTCH3 mutation, we used this cutoff value to separate the NOTCH3 mutation carriers in those with minimal WMH and those with moderate or severe WMH (Table 3). The highest baseline TCBF was present in the NOTCH3 nonmutation carriers, followed by the NOTCH3 mutation carriers with minimal and NOTCH3 mutation carriers with moderate or severe WMH (analysis of variance, P < 0.001). After adjustment for age and adjustment for age and sex, the difference in corrected baseline TCBF values remained significant. Post hoc analysis showed that the difference in baseline TCBF between NOTCH3 mutation carriers with and without moderate or severe WMH was not significant.
Flow parameters in NOTCH3 nonmutation carriers and the NOTCH3 mutation carriers with minimal WMH and moderate and severe WMH
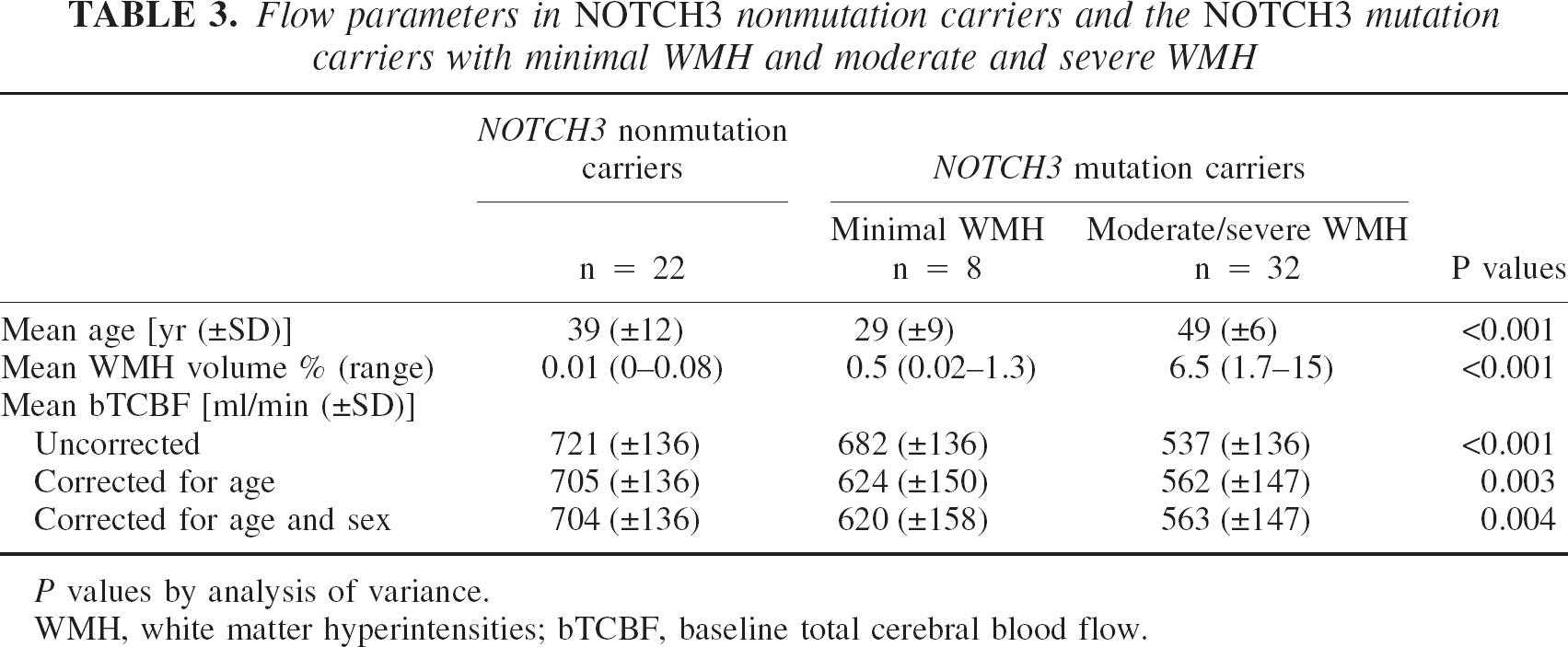
P values by analysis of variance.
WMH, white matter hyperintensities; bTCBF, baseline total cerebral blood flow.
DISCUSSION
In CADASIL patients, two types of lesions affecting small cerebral arteries are observed that might affect cerebral hemodynamics. First, the arteriolar lumen is narrowed as a consequence of fibrous thickening of the arterial wall. This phenomenon presumably reduces baseline blood flow. Second, vascular smooth muscle cells are destroyed, which might impair the vascular dilatory response to hypoxia (Chabriat et al., 2000; Pfefferkorn et al., 2001). In this study, we found a decrease in baseline TCBF in NOTCH3 mutation carriers as compared with NOTCH3 nonmutation carriers, whereas the capacity of the arterial wall to dilate, measured by acetazolamide response, appeared normal in NOTCH3 mutation carriers. These observations suggest that narrowing of the arterial lumen has a more profound effect on cerebral hemodynamics in CADASIL patients than dysfunction of vascular smooth muscle cells.
An alternative explanation for the observed reduced baseline TCBF in NOTCH3 mutation carriers could also be raised. NOTCH3 encodes for a transmembrane protein with a receptor and cell signal transduction function. Besides NOTCH3, there are three other known mammalian NOTCH proteins (Gridley, 1997). Evidence from several studies has indicated a role for Notch signaling during vascular development (Gridley, 2001). For example, an activated form of NOTCH4 in the embryonic vasculature causes abnormal vessel structure and patterning (Uyttendaele et al., 2001). Possibly, mutations in NOTCH3 may also lead to an abnormal development of the cerebral vasculature in CADASIL. The observation that flow abnormalities were present in all age groups in this study could thus be explained by such an innate structural abnormality of cerebral blood vessels, and not necessarily acquired structural abnormalities.
Using MRI bolus tracking and acetazolamide in CADASIL patients, Chabriat et al. (2000) found a reduction in both baseline perfusion and cerebrovascular reactivity within WMH. Similarly, Mellies et al. (1998) observed a reduction in gray matter perfusion that correlated with the amount of WMH, using single-photon emission computed tomography. These observations, however, have not made it clear whether structural brain changes are secondary to impaired flow, or whether impaired flow is a consequence of brain damage.
Several observations in our study provide insight into the nature of the correlation between WMH and lacunar infarcts on the one hand, and cerebral flow disturbance on the other. In this study, we assessed TCBF based on phase-contrast MRI. This method provides robust quantitative data on the total amount of blood flowing to the brain (Buijs et al., 1998; Spilt et al., 2002a). Using phase-contrast MRI, we found a significantly diminished baseline TCBF at any age, even in young NOTCH3 mutation carriers with minimal structural lesions (Fig. 2). It is unlikely that the cerebral blood flow to the brain is significantly reduced by a flow reduction in these small lesions, which only represent a very small percentage of the brain volume. A more realistic explanation is that the observed TCBF reduction in these patients is the result of more widespread flow impairment, which affects not just the areas of WMH and lacunar infarcts. Furthermore, in all CADASIL patients with minimal WMH and minimal lacunar infarcts, irrespective of age, baseline TCBF was observed to be abnormal. Both observations suggest that a reduction in baseline flow precedes, and is responsible for, the occurrence of WMH and lacunar infarcts in CADASIL.
In this study, we did not find evidence for a decreased cerebrovascular reactivity (CVR) in CADASIL patients. This finding differs from observations in other studies. Several explanations for this seeming contradiction can be put forward. First, the CADASIL patients who participated in our study were younger than those in the other studies. A reduced CVR might be more pronounced in an older patient population. Second, in our study a different technique for flow quantification was used as compared with previous studies. In these other studies, single-photon emission computed tomography, positron emission tomography, MRI bolus tracking, and transcranial Doppler ultrasound were used to estimate changes in cerebral blood flow (Chabriat et al., 1995b; Mellies et al., 1998; Chabriat et al., 2000; Pfefferkorn et al., 2001). With the exception of transcranial Doppler, these techniques detect flow changes in brain parenchyma, not changes in the supplying vessels of the brain. In a recent article, poor correlations were found between estimates of CVR based on MRI bolus tracking and those based on phase-contrast MRI, which was attributed to the two methods reflecting different aspects of the CVR (Spilt et al., 2002b). However, both transcranial Doppler and phase-contrast MRI enable the measurement of blood flow velocity in extraparenchymal vessels. Still others showed poor correlation between flow data resulting from these two methods, which was attributed to a lack of reproducibility of transcranial Doppler measurements because of operator dependency (Seitz et al., 2001). In this study, we decided to use phase-contrast MRI because this technique has the advantage of being operator independent and involving straightforward flow volume quantification as demonstrated by Bakker et al. (1996) and Spilt et al. (2002a). In addition, phase-contrast MRI has the advantage of providing, during one examination, both flow data and morphologic data, which is not possible with other techniques.
In conclusion, our findings suggest that flow impairment gives rise to the development of WMH and lacunar infarcts in CADASIL patients. Furthermore, in CADASIL patients with minor WMH, the TCBF is reduced in rest, whereas the cerebral vascular dilatory capacity is preserved. This novel observation is relevant because it may imply that, by increasing the cerebral blood flow through a challenge of the remaining vasodilatory capacity, vasodilatory agents might limit the development of WMH and lacunar infarcts in CADASIL patients. Further studies are needed to determine the efficacy of pharmacologic improvement of cerebral blood flow in CADASIL patients.
