Abstract
Impairment of inhibitory neurotransmission has been reported to occur in widespread, structurally intact brain regions after focal ischemic stroke. These long-lasting alterations contribute to the functional deficit and influence long-term recovery. Inhibitory neurotransmission is primarily mediated by γ-aminobutyric acid (GABA)A receptors assembled of five subunits that allow a variety of adaptive changes. In this study, the regional distribution of five major GABAA-receptor subunits (α1, α2, α3, α5, and γ2) was analyzed immunohistochemically 1, 7, and 30 days after photochemically induced cortical infarcts. When compared with sham-operated controls, a general and regionally differential reduction in immunostaining was found within the cortex, hippocampus, and thalamus of both hemispheres for almost all subunits. Within ipsilateral and contralateral neocortical areas, a specific pattern of changes with a differential decrease of subunits α1, α2, α5, and γ2 and a significant upregulation of subunit α3 was observed in the contralateral cortex homotopic to the infarct. This dysregulation was most prominent at day 7 and still present at day 30. Interestingly, a single application of the noncompetitive N-methyl-D-aspartate–receptor antagonist MK-801 during lesion induction completely blocked these bihemispheric alterations. Cortical spreading depressions induced by topical application of KCl do not change GABAA-receptor subunit expression. As alterations in subtype distribution crucially influence inhibitory function, ischemia-induced modifications in GABAA-receptor subtype expression may be of relevance for functional recovery after stroke.
The functional long-term consequences of clinical stroke are predicted not only by the size and location of the ischemic lesion, but also by the capacity of the brain to compensate and reorganize lost functions. Although recent research has led to a better understanding of the pathophysiologic events taking place during the acute phase of the ischemic injury (Dirnagl et al., 1999; Lee et al., 1999), comparatively little is known about subacute alterations in brain function. Accumulating evidence indicates that primarily uninjured tissue surrounding the ischemic lesion as well as remote ipsilateral and contralateral brain regions play a crucial role in long-term recovery (Eysel, 1997; Liepert et al., 2000b; Nudo et al., 1996, 2001; Witte et al., 2000). Various functional alterations occur within these structurally intact regions that may be both favorable and disadvantageous, compensating for lesion-induced deficits while causing a deterioration of functional impairment or even postlesional epilepsy (So et al., 1996).
Experimental studies indicate widespread and partially persistent alterations in cortical excitability immediately adjacent to the focal ischemic infarct and also in remote ipsilateral and contralateral brain regions. Electrophysiologic recordings using a paired-pulse protocol in brain slice preparations revealed an impairment of functional cortical inhibition (Buchkremer-Ratzmann et al., 1996; Reinecke et al., 1999) accompanied by a reduction of γ-aminobutyric acid (GABA)A-dependent inhibitory function (Neumann-Haefelin et al., 1995). Additional in vivo recordings demonstrated an increased spontaneous activity in the surrounding infarct, and receptor autoradiography revealed widespread reduction in binding of radiolabeled muscimol to GABAA receptors (Qu et al., 1998a; Schiene et al., 1996). Although transient changes of glutamatergic neurotransmission contribute to the hyperexcitability after focal brain infarcts (Qu et al., 1998b; Que et al., 1999), these studies point toward a substantial disturbance of inhibitory function that is primarily mediated by GABAA receptors.
To analyze whether modifications of GABAA-receptor subtype expression contribute to the widespread and long-lasting alterations in GABAergic function, the distribution of five major receptor subunits were immunohistochemically investigated in adult rats 1, 7, and 30 days after focal cortical infarcts using the photothrombosis model (Watson et al., 1985). GABAA receptors are assembled of five subunits deriving from a multitude of subunit subtypes encoded by at least 19 genes (14641–6, β1–3, γ1–3, δ, ε, π, θ, and ρ1–3) (Barnard et al., 1998; Whiting et al., 1999), which allow for numerous combinatorial variants (Olsen et al., 1999). Although only little is known about the specific properties of single subunits, functional studies clearly demonstrate that subunit composition of receptor subtypes crucially influences their electrophysiologic and pharmacological properties (Barnard et al., 1998; Narahashi, 1999). Since these properties are primarily determined by expression of different α-subunit variants (Sieghart et al., 1999), alterations in expression of four α-subunits and one γ-subunit were investigated. Using this approach, bihemispheric regionally differential alterations in GABAA-receptor subunit expression were observed, which could be completely suppressed by pharmacological blockade of N-methyl-D-aspartate (NMDA) receptors during lesion induction.
Parts of this paper have been published previously in abstract form (Redecker et al., 2000a).
MATERIALS AND METHODS
Induction of focal cortical infarcts
The experiments were carried out on a total of 47 male Wistar rats (280 to 320 g; Experimental Animal Laboratory). A total of 27 animals received photothrombotic cortical infarcts, spreading depressions were induced in 7 animals, and 13 animals served as controls. Photothrombotic cortical infarcts were induced using the method initially described by Watson et al. (1985), with only minor modifications. Briefly, animals were anesthetized with enflurane (1.5% to 2.0%) in oxygen–nitrous oxide (30%/70%). Body temperature was kept constant throughout the surgery at 36.5°C. The rat was placed in a stereotactic frame and an optic fiber bundle connected to the cold light source (Schott KL 1500) was stereotactically positioned 4 mm posterior to bregma and 4 mm lateral to the midline. During the first minute of illumination (total time, 20 minutes), Rose Bengal (1.3 mg/100 mg body weight at a concentration of 10 mg/mL in 0.9% NaCl) was injected through a femoral vein catheter. After this surgery, the animals were returned to their cages where they had free access to food and water. Control animals (n = 13) received the same treatment without illumination of the brain. In a group of 20 animals (14 animals with photothrombotic infarcts, 6 sham-operated controls), the NMDA-receptor antagonist MK-801 (2 mg/kg body weight) was applied intravenously 30 minutes before illumination.
Induction of cortical spreading depressions
In a group of seven animals, spreading depressions were induced by topical application of potassium chloride to the cortical surface using a method described elsewhere (Jander et al., 2001; Schroeter et al., 1995). These experiments were performed to determine whether cortical spreading depressions, which occur after ischemic insults (Dietrich et al., 1994), influence the distribution of GABAA-receptor subtypes. After exposing the skull, two burr holes were made over the left hemisphere while leaving the dura intact. The first hole was positioned directly posterior to bregma and 1 or 2 mm from midline, whereas the second was burred 1 mm anterior to lambda with the same distance to midline. A recording electrode filled with artificial cerebrospinal fluid (124 mmol/L NaCl, 5 mmol/L KCl, 2 mmol/L CaCl2, 2 mmol/L MgSO4, 1.25 mol/L NaH2PO4, 10 mmol/L glucose, and 26 mmol/L NaHCO3) was positioned rostrally on the dura and direct current (DC) potentials were monitored using a home-build amplifier and an analog plotter. Spreading depressions were induced by placing a swab soaked with KCl (3 mol/L) on the dura in the occipital burr hole. After recording seven or eight spreading depressions, the swabs were removed (usually approximately 120 minutes after their application) and the burr holes were flushed with saline. During the next 10 to 15 minutes, a maximum of two more DC deflections occurred. The animals were then allowed to recover and returned to their cages.
Immunohistochemistry
At day 1, 7, and 30 after infarct (or spreading depression) induction, the rats were deeply anesthetized with diethylether and perfused through the ascending aorta with 4% paraformaldehyde and 15% saturated picric acid solution in phosphate buffer (0.15 mol/L, pH 7.4) (Fritschy and Mohler, 1995). Brains were removed immediately after the perfusion and post-fixed for 3 hours in the same fixative at 4°C. All samples were then cryoprotected in phosphate-buffered saline containing 30% sucrose for 24 hours and stored at −75°C for further processing. To enhance the detection of synaptic receptor proteins in the subsequent immunohistochemical staining, the brains were processed with a modified antigen-retrieval procedure (Bohlhalter et al., 1996; Fritschy et al., 1998). The brains were incubated overnight at room temperature in 0.1 mol/L sodium citrate buffer (pH 4.5) and irradiated with microwaves (650 W, 135 seconds) in the same buffer. Coronal sections were cut at 50 μm with a freezing microtome and collected in ice-cold 0.1 mol/L phosphate-buffered saline. A series of sections was Nissl-stained with cresyl violet for histologic analysis of the photothrombotic infarcts.
The GABAA-receptor subunits α1, α2, α3, α5, and γ2 were visualized using subunit-specific antisera raised in guinea pigs against synthetic peptides derived from rat subunit cDNA. These subunit specific antisera have been extensively characterized biochemically by Western blotting and immunoprecipitation (Fritschy and Mohler, 1995) and their suitability for immunohistochemistry has been documented in several previous reports (Fritschy et al., 1998; Fritschy and Mohler, 1995; Redecker et al., 2000b).
Free-floating sections were washed three times in Tris-buffer (Tris saline, pH 7.4, 0.05% Triton X-100) and incubated at 4°C overnight in primary antibody solution diluted in Tris-buffer containing 2% normal goat serum. The following dilutions of the antisera were used: GABAA-receptor subunit α1, 1:20,000; subunit α2, 1:2,000; subunit α3, 1:2,000; subunit α5, 1:4,000; and subunit γ2, 1:3,000. Sections were then washed three times in Tris buffer and incubated in biotinylated secondary antibody solution (Jackson Immunoresearch, West Grove, PA, U.S.A.) diluted 1:300 in Tris buffer containing 2% normal goat serum for 30 minutes at room temperature. After additional washing, sections were transferred to the avidin-peroxidase solution (Vectastain Elite Kit, Vector Laboratories, Burlingame, CA, U.S.A.) for 25 minutes, washed, and processed using diaminobenzidine hydrochloride (DAB; Sigma-Aldrich, Munich, Germany) as chromogen. Sections were mounted onto gelatincoated slides, air-dried, dehydrated with ascending series of ethanol, cleared, and coverslipped with toluene (Entellan; Merck, Darmstadt, Germany).
Changes in the regional and laminar distribution of GABAA-receptor subunits were analyzed by light microscopy. For semiquantitative image analysis, sections were digitized with a charge-coupled device camera and processed with an imaging program (NIH Image; Research Service Branch, NIH, Bethesda, MD, U.S.A.). Measures of relative optical density (ROD) of GABAA-receptor subunit staining were performed on one section per animal and per antibody. The following cytoarchitectonic brain regions were evaluated on both hemispheres according to the atlas of Zilles (1992): the frontal (motor) cortex (Fr), the hindlimb representation cortex (HL), the primary and secondary somatosensory cortex (Par1, Par2), the hippocampal formation, and the thalamus. Delineations of the cytoarchitectonic cortical areas were defined using adjacent cresyl-violet–stained sections. For background correction, the signals obtained in the corpus callosum were subtracted. An independent investigator who was blinded to the data performed all ROD measurements. Optical densities are given as mean ± SD unless stated otherwise. The RODs of different GABAA-receptor subunits in ipsilateral and contralateral brain regions in the different experimental groups were compared with the appropriate ROD measurements of sham-operated controls. To assess statistical significance, an unpaired t-test was performed followed by the multiple test procedure of Holm (α = 0.1), which is a refinement of the Bonferroni method (Holm, 1979). For display, images were contrast enhanced and color coded on a 256-level scale.
RESULTS
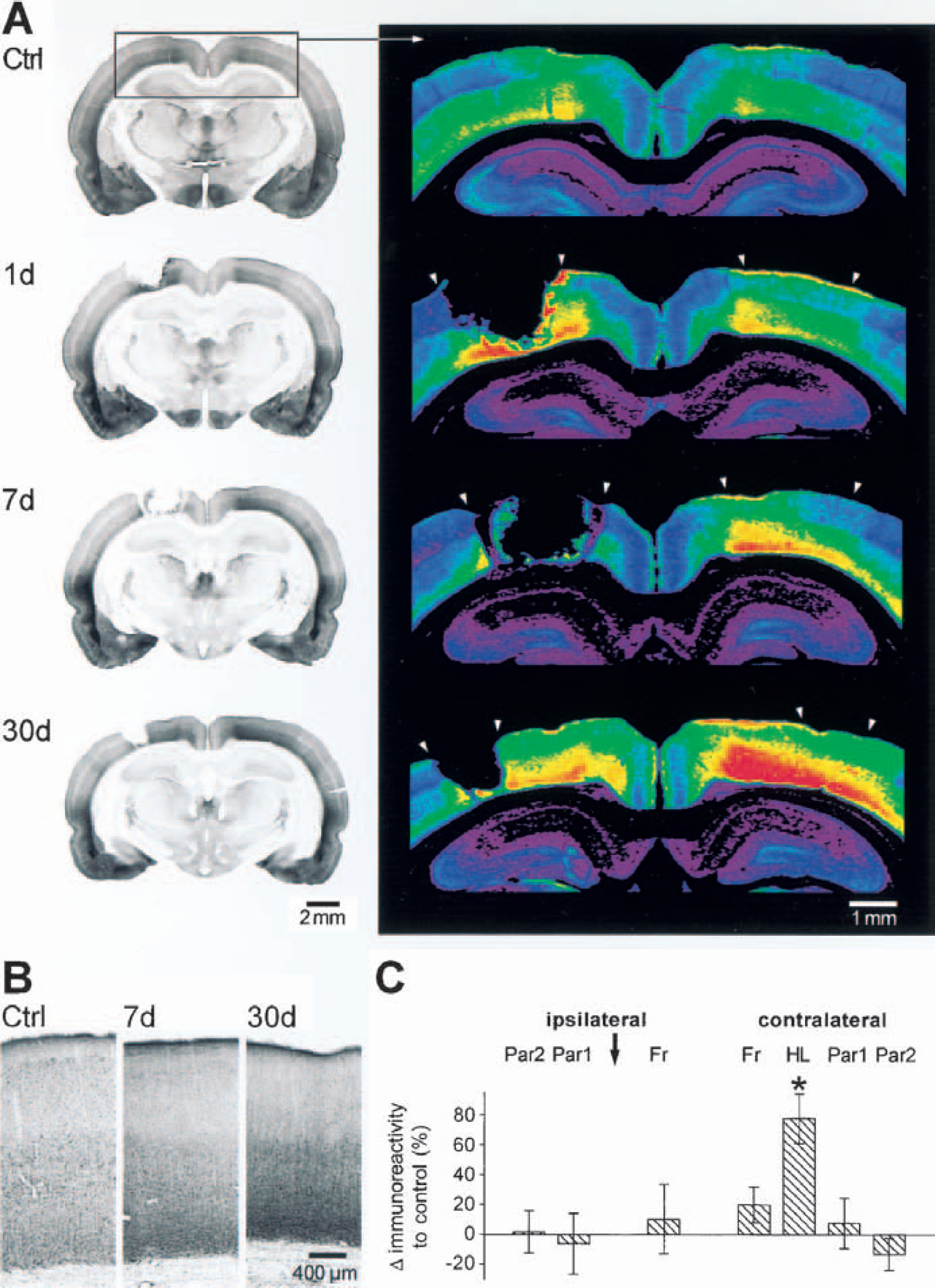
Morphology of the photothrombotic infarcts and distribution of GABAA-receptor subunits in controls
All animals in the experimental subgroups had typical cortical infarcts in the HL area, involving in some cases medial parts of Par1 (Zilles, 1992) (Fig. 1). The photothrombotic infarcts affected all cortical layers, whereas the subcortical white matter appeared to be intact in the majority of animals. The mediolateral diameter of the lesions, as measured on immunohistochemical sections taken at the center of the ischemic lesion, was 2.3 mm (± 0.7 mm, n = 13). Within the first month after the photothrombotic infarct, the diameter of the lesions decreased from 2.9 ± 0.4 mm (n = 4) at day 1 and 2.5 ± 0.3 mm (n = 4) at day 7 after lesion induction to 1.6 ± 0.4 mm (n = 5, −44.8%) at day 30. This decrease in lesion diameter was mainly due to scar formation processes as well as to a shift of surrounding tissue toward the lesion center. No structural abnormalities were found in sections collected from sham-operated animals (n = 7).
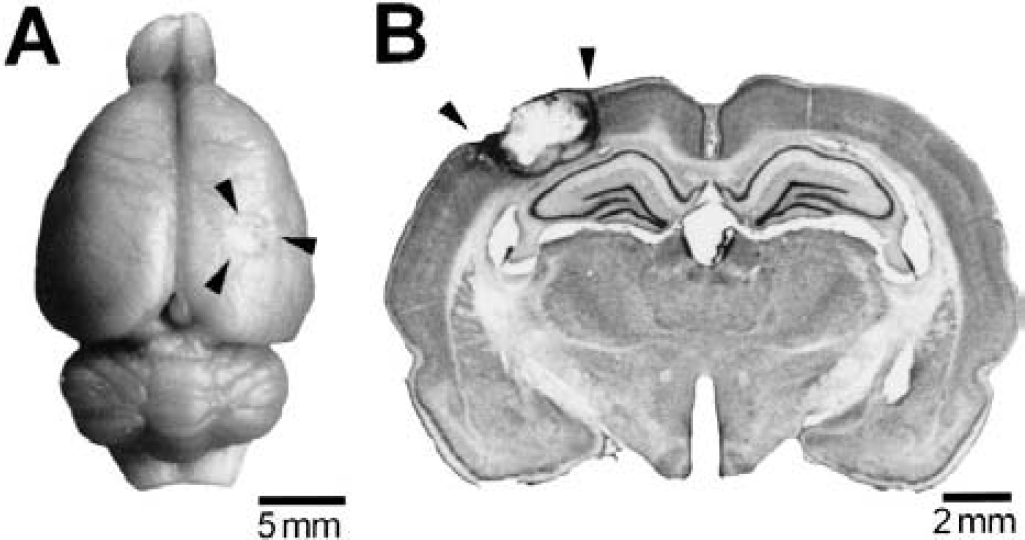
Morphology of photothrombotically induced focal cortical infarcts.
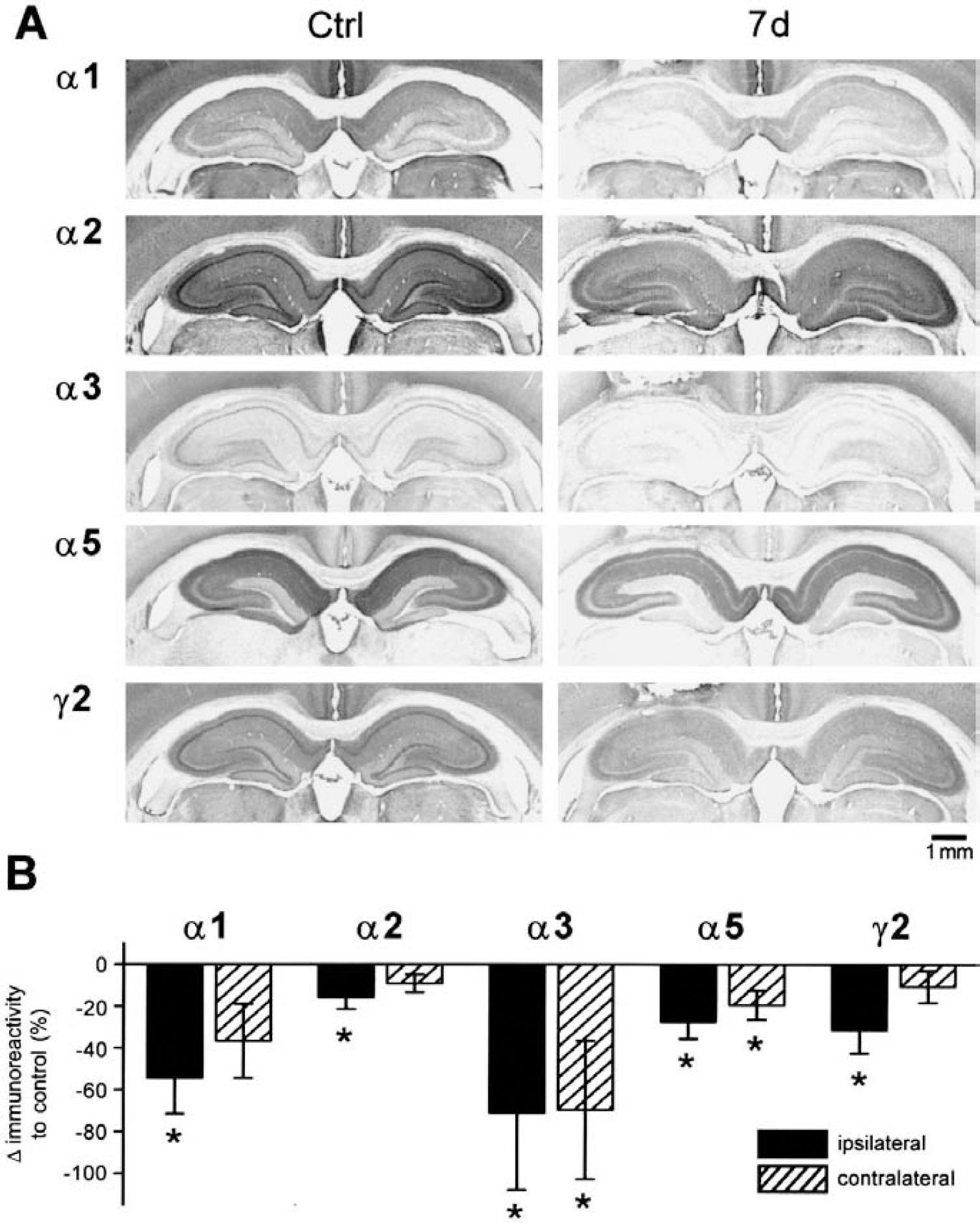
The expression of the GABAA-receptor subunits a1, α2, α3, α5, and γ2 was identical in sham-operated animals at day 1 (n = 3) and day 7 (n = 4) after the surgery and showed the same distribution pattern as described previously for nontreated animals (Fritschy and Mohler, 1995) (Fig. 2). Therefore, both groups of animals were merged and used as controls for the subsequent experimental groups. Subunits α1 and γ2 were abundantly expressed within the cortex in all layers, with particularly intense staining in layers III-IV, whereas subunit α2, α3 and α5 were primarily confined to certain cortical layers. Subunit α2 predominated in superficial layers, where subunits α3 and α5 were almost absent. Subunit α3 and on a lower level subunit α5 were only slightly expressed in deep cortical layers. In contrast to the cerebral cortex, the hippocampal formation showed a different GABAA-receptor subunit expression with particularly strong immunoreactivity of subunits α2 and α5, and, to a lesser extent, of γ2. Subunit a1 revealed moderate staining within this brain region, whereas subunit α3 was almost absent.
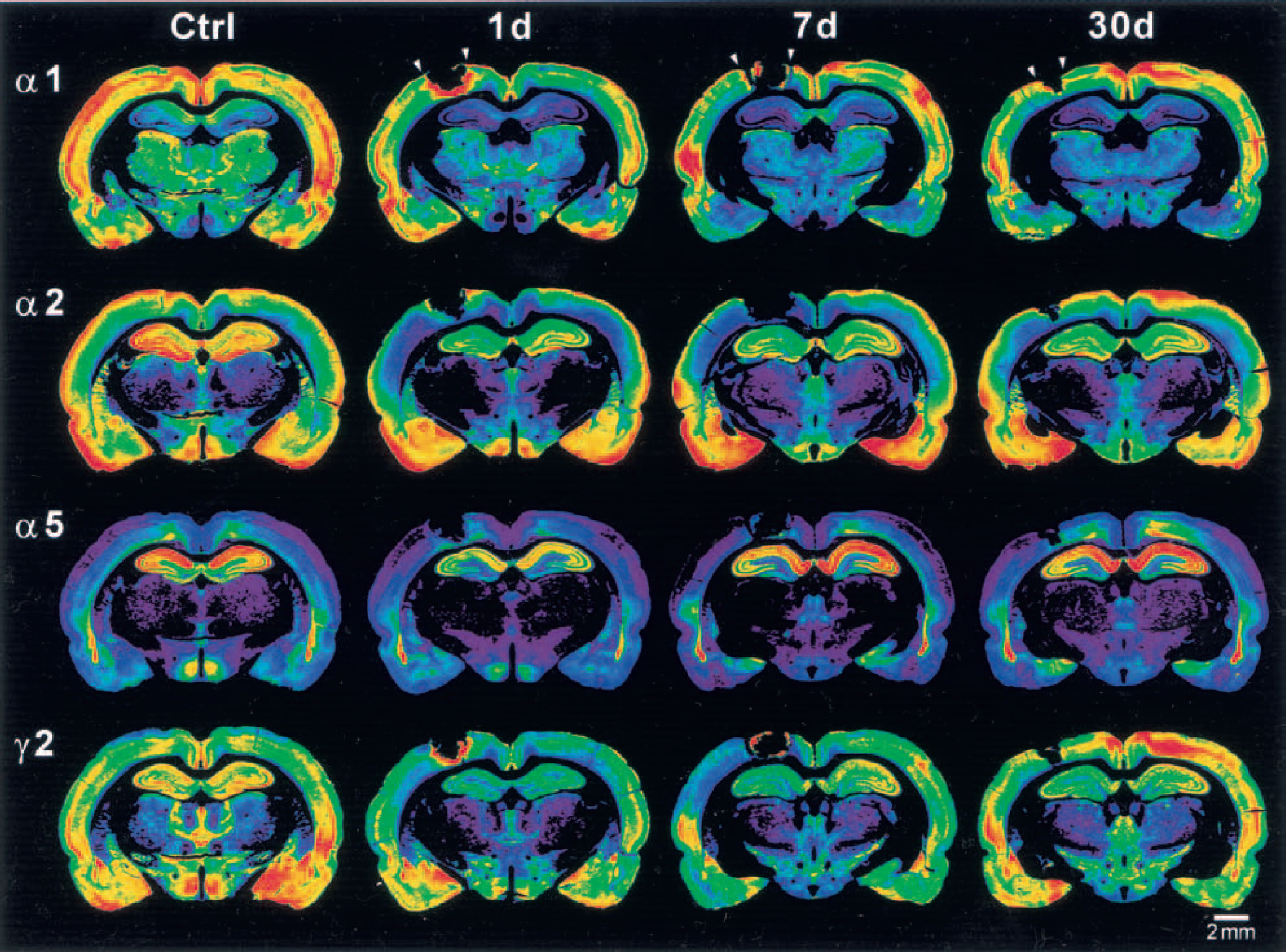
Distribution of GABAA-receptor subunits α1, α2, α5, and γ2 in sham-operated controls as well as in rats with photothrombotic infarcts 1, 7, and 30 days after the lesion. Color-coded images from immunohistochemically processed coronal sections through the infarct. For each subunit, the optical density of the immunoreactivity product was color-coded using a standard 256-level scale ranging from black for background to violet, blue, green, yellow, and red for the most intense signals. Sham-operated rats show the typical distribution pattern of subunits α1, α2, α5, and γ2 with symmetric intensities on both hemispheres. Animals with photothrombotic infarcts (arrowheads) display bihemispheric regionally differential reductions in immunostaining for all these subunits involving different cortical areas, the hippocampal formation as well as thalamic nuclei. The downregulation was emphasized on the ipsilateral hemisphere, was most prominent at day 1 and 7, and was still present at day 30. GABA, γ-aminobutyric acid.
Widespread reduction of GABAA-receptor subunits α1, α2, α5, and γ2 in ipsilateral and contralateral neocortex after focal cortical infarcts
After focal ischemic infarcts, the distribution of GABAA-receptor subunits was different in widespread ipsilateral and contralateral brain regions compared with sham-operated controls in all animals (Fig. 2, Table 1). Although the extent of these changes varied to some degree in each group of animals, a consistent pattern of alterations was observed in the surrounding and remote ipsilateral and contralateral neocortical areas (Fig. 2). Owing to the free-floating immunohistochemical procedure, the center of the ischemic lesion was missing in the majority of sections taken at the level of the lesions and only a small rim of necrotic tissue adjacent to the structurally intact surrounding cortex was preserved. Within this small necrotic rim, a strong immunoreactivity was found for subunits α1 and γ2 at day 1 and less prominent immunoreactivity was found at day 7 after the infarct (Fig. 2). Because of the necrotic tissue and the lack of specific staining in high-resolution microscopic analysis, however, this strong immunoreactivity was likely due to unspecific reactions. Therefore, the present study focused on alterations in GABAA-receptor subunit expression in structurally intact brain regions.
Densities of immunoreactivity of different GABAA-receptor subunits after focal cortical infarcts
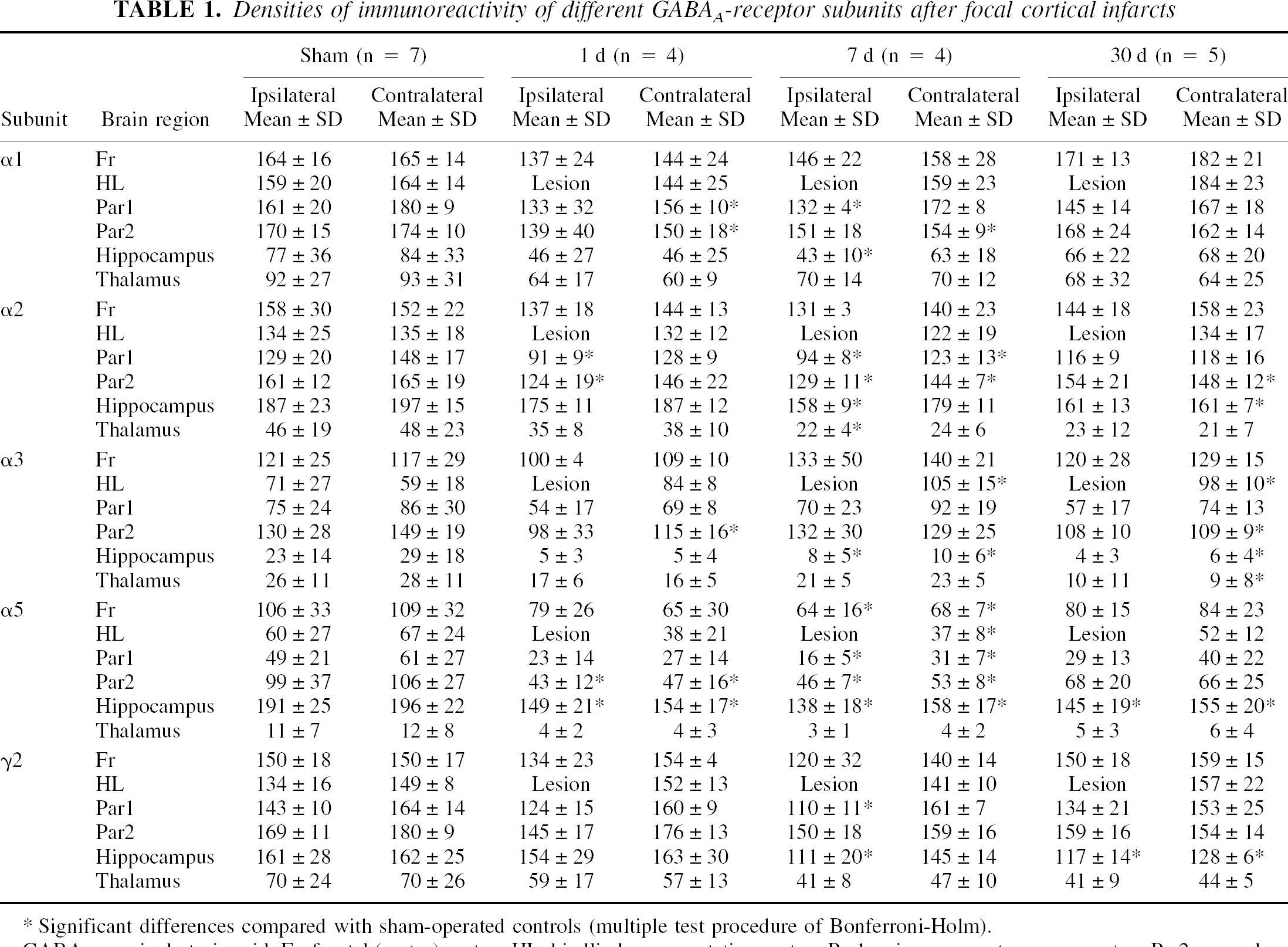
Significant differences compared with sham-operated controls (multiple test procedure of Bonferroni-Holm).
GABA, γ-aminobutyric acid; Fr, frontal (motor) cortex; HL, hindlimb representation cortex; Par1, primary somatosensory cortex; Par2, secondary somatosensory cortex.
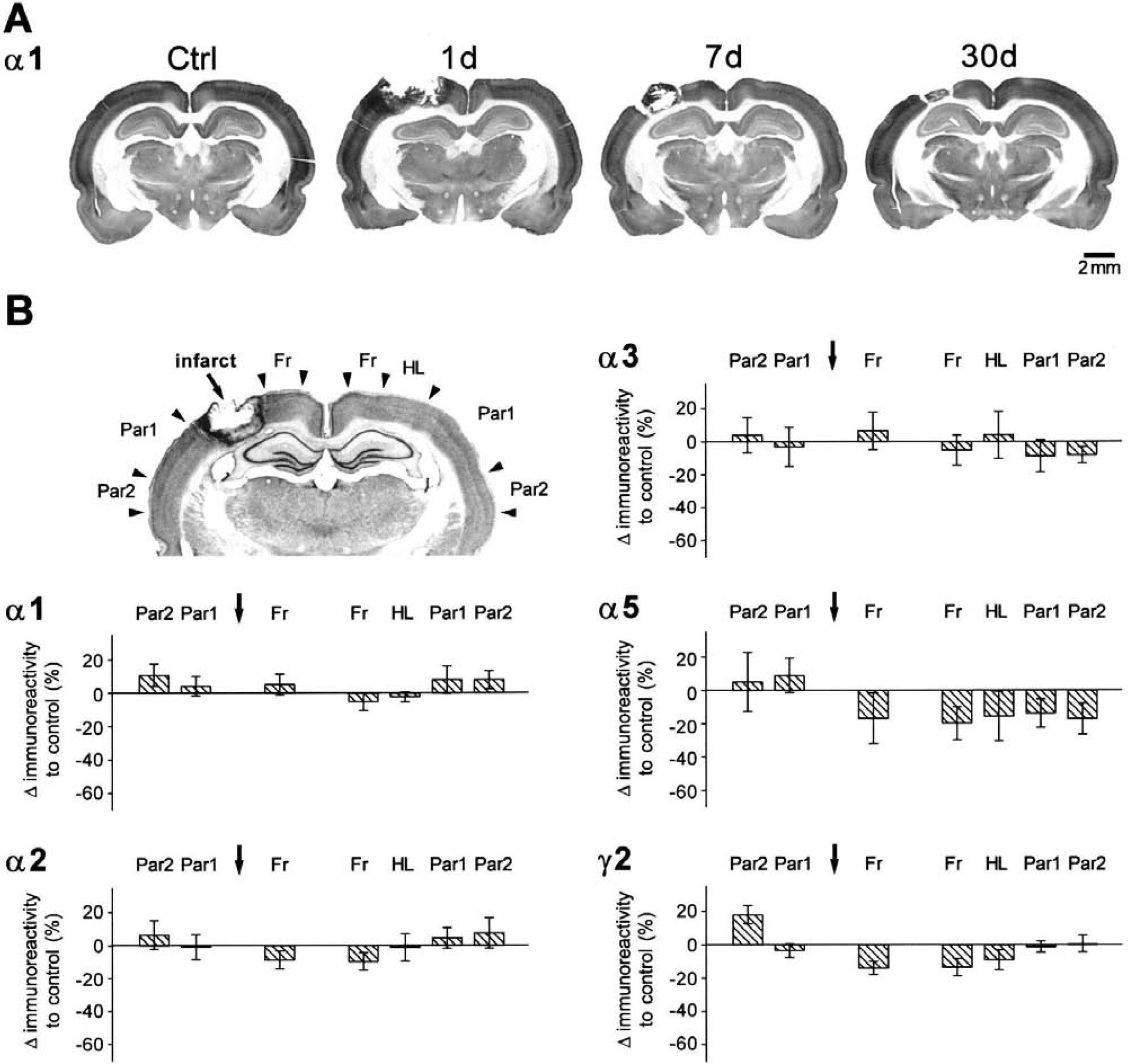
Within the ipsilateral neocortex surrounding the photothrombotic infarcts, qualitative and semiquantitative evaluation revealed a remarkable decrease in immunoreactivity of subunits α1, α2, α5, and γ2 (Fig. 2, Table 1). This downregulation occurred during the first day after the infarct, was most prominent at day 7, and was still present at day 30 (Fig. 2, Table 1). The mean values of optical density measurements for the different groups and brain areas are displayed in Table 1. In the following text, the relative differences of optical densities as well as the corresponding P values are given for the 7-day group. Subunits α1 and γ2, which were both abundantly expressed within the cortex of control animals, showed a marked reduction in immunoreactivity in the surrounding lesion as well as in remote ipsilateral cortical areas. This reduction was most prominent at day 7 in Par1 (α1: −18.0%, P < 0.05; γ2: −23.3%, P < 0.05), but also in Par2 (αd: −13.7%, P = ns; γ2: −10.9%, P = ns) and the Fr (α1: −10.8%, P = ns; γ2: −20.0%, P = ns) showed a slight decrease in staining of these receptor subunits at this time point. In addition, immunoreactivity for subunit α2 and also for subunit α5, which is only expressed on a low level in deep cortical layers of controls, showed a decreased staining in surrounding and remote ipsilateral cortical areas at day 7 after the infarct. In accordance with the findings for subunits α1 and γ2, subunit α2 was also reduced within Par1 (−27.0%, P < 0.05) and Par2 (−20.0%, P < 0.05). Within the Fr, a decreased expression of subunit α2 (−17.0%, P = ns) was also found in most of the animals with photothrombotic infarcts. Staining for subunit α5 revealed a strong decrease in all neocortical areas at day 7 (Fr: −43.7%, P < 0.05; Par1: −68.5%, P < 0.05; Par2: −53.7%, P < 0.05).
The downregulation of GABAA-receptor subunits α1, α2, α5, and γ2 involved the contralateral neocortex too (Fig. 2). The time course of these alterations were similar to the ipsilateral cortex, showing the most prominent reduction at day 7 after the infarct. This decrease in immunoreactivity, however, varied to some degree. Subunit α5 showed a clear reduction in all contralateral neocortical areas (Fr: −37.4%, P < 0.05; HL: −45.3%, P < 0.05; Par1: −54.9%, P < 0.05; Par2: −49.4%, P < 0.05), whereas subunits α1, α2, and γ2 were not consistently reduced. Interestingly, the staining of these subunits was only slightly, but not significantly, reduced in the contralateral brain areas located homotopic to the photothrombotic infarct (Fr α1: −4.3%, P = ns; Fr α2: −7.9%, P = ns; Fr γ2: −6.2%, P = ns; HL α1: −3.4%, P = ns; HL α2: −9.7%, P = ns; HL γ2: −5.3%, P = ns), whereas a remarkable decrease was found in more lateral contralateral brain regions within Par1 for subunit α2 (−19.0%, P < 0.05) and within Par2 for subunits α1 (−11.4%, P < 0.05), α2 (−15.3%, P < 0.05), and γ2(−11.4%, P = ns) (Figs. 2 and 3, Table 1).
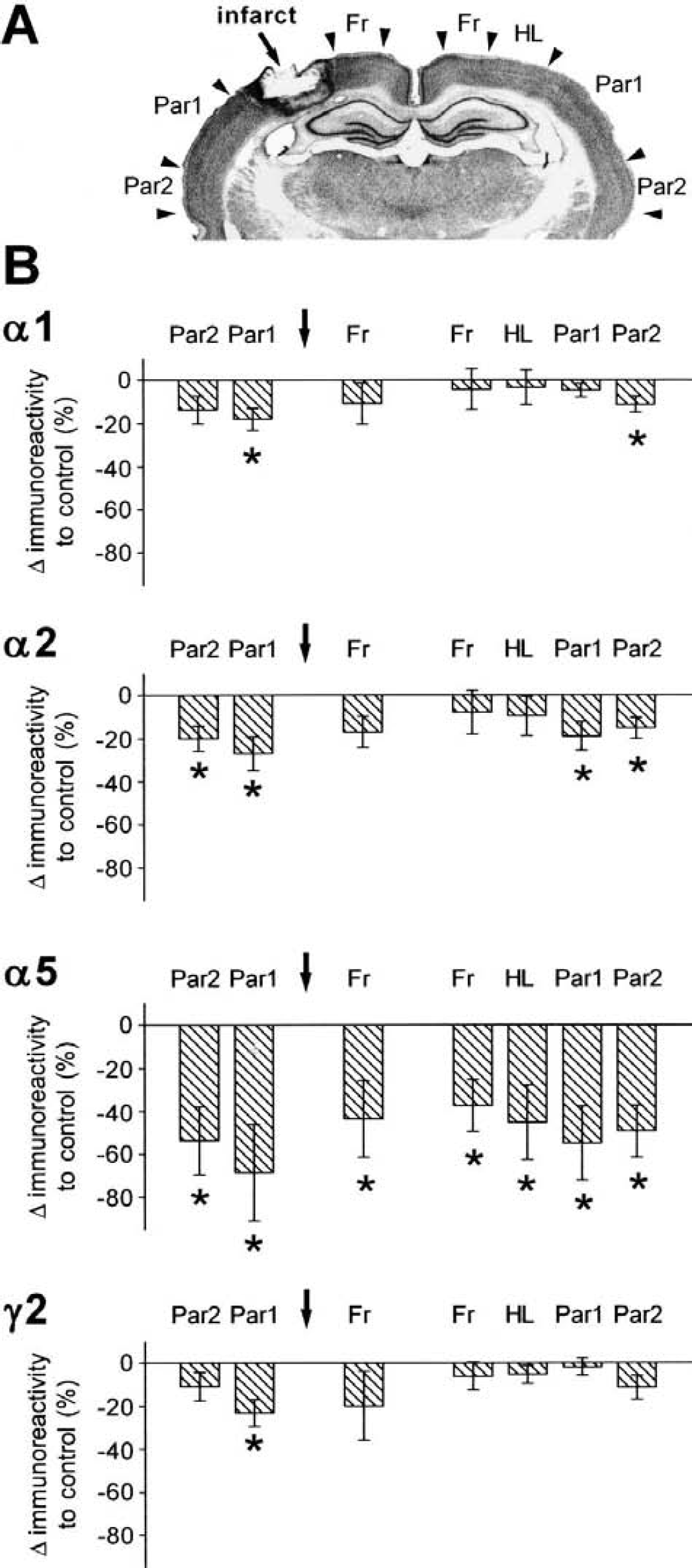
Semiquantitative analysis of regional alterations in immunoreactivity of GABAA-receptor subunits α1, α2, α5, and γ2 at day 7 after the photothrombotic infarct.
Increased expression of GABAA-receptor subunit α3 in the contralateral neocortex homotopic to the cortical infarct
In contrast to subunits α1, α2, α5, and γ2, which showed a differentially intense reduction in immunoreactivity in ipsilateral and contralateral cortical areas, an increase in expression of subunit α3 was found in the contralateral neocortex homotopic to the photothrombotic infarct (Fig. 4). Within this area of increased subunit expression, the typical laminar distribution pattern was intact and high magnification did not reveal any qualitative change in immunoreactivity, which is indicated by strong neuropil staining. This upregulation was most prominent in the contralateral HL at day 7 after the infarct (77.7%, P < 0.05). This increase in subunit expression was not consistently found at day 30, but was still present on a high level in two animals (n = 5; mean = 66.3%, P < 0.05; Fig. 4), whereas in others the expression pattern partially normalized. The upregulation also partly involved the Fr (20.1%, P = ns), but was not detected in additional cortical areas. Subunits α1 and γ2 were also upregulated in contralateral areas Fr and HL at day 30 after the infarct in some animals (Table 1, Fig. 2), although not to a significant level.
Distribution of GABAA-receptor subunit α3 in sham-operated controls as well as in rats with photothrombotic infarcts 1, 7, and 30 days after the lesion.
Reduction in GABAA-receptor subunit expression within the hippocampal formation and the thalamus after focal cortical infarcts
Alterations in distribution of GABAA-receptor subunits in animals with focal cortical infarcts also involved the hippocampal formation and thalamic nuclei of both hemispheres (Figs. 2 and 5). Qualitative and semiquantitative evaluation of the regional subunit distribution within the hippocampal formation revealed a reduction of immunoreactivity for all receptor subunits, which was more pronounced on the ipsilateral hippocampus. This downregulation was most prominent within the dentate gyrus and the CA1 region on both hemispheres. The time course of these alterations slightly differed from that described for the cerebral cortex. Although the decrease was maximal at day 7 after the infarct, a strong decrease was directly observed at day 1 and was still present at day 30 (Fig. 2, Table 1). Subunits α1 and α3 were strikingly decreased within the ipsilateral hippocampus at day 7 (α1: −54.3%, P < 0.05; α3: −70.8%, P < 0.05), and subunits α2 (−15.8%, P < 0.05), α5 (−27.5%, P < 0.05) and γ2 (−31.2%, P < 0.05) showed a strong downregulation. Since subunit α3 plays only an inferior role within the hippocampal formation, however, the prominent proportional reduction of this subunit may be of minor functional importance. Within the contralateral hippocampus a similar pattern of downregulation was observed (α1: −36.6%, P = ns; α2: −9.1%, P = ns; α3: −69.5%, P < 0.05; α5: −19.5%, P < 0.05; γ2: −10.6%, P = ns).
Alterations in distribution of GABAA-receptor subunits α1, α2, α3, α5, and γ2 within the hippocampal formation after focal cortical infarcts.
Interestingly, a specific decrease in staining of hippocampal interneurons positive for subunit α1 was observed at high magnification in all hippocampal subregions (CA1, CA2, CA3, and the dentate gyrus) of both hemispheres (Fig. 6). At day 1 after the infarct, a distinct reduction in staining intensity of subunit α1 was observed on somata and processes of interneurons within CA1 to CA3 as well as within the dentate gyrus. This reduction in staining of subunit α1 proceeded until day 7 when only contours of interneurons were detected (Fig. 6). At day 30 after the infarct, this loss of subunit α1 on hippocampal interneurons partially recovered and the typical staining pattern reappeared. This pattern of alterations occurred in all animals with focal photothrombotic infarcts and was more prominent on the ipsilateral hemisphere, though it was also present contralaterally.
After focal photothrombotic infarcts, a downregulation of GABAA receptors was also observed within the thalamus (Fig. 2, Table 1) involving all receptor subunits. This reduction in GABAA-receptor subtypes was not restricted to specific nuclei and was only slightly emphasized on the ipsilateral hemisphere. As observed within the hippocampal formation, the time course of these alterations differs from that of the cortical alterations, in that a strong decrease already occurred during the first day and was still present at day 30 after the infarct.
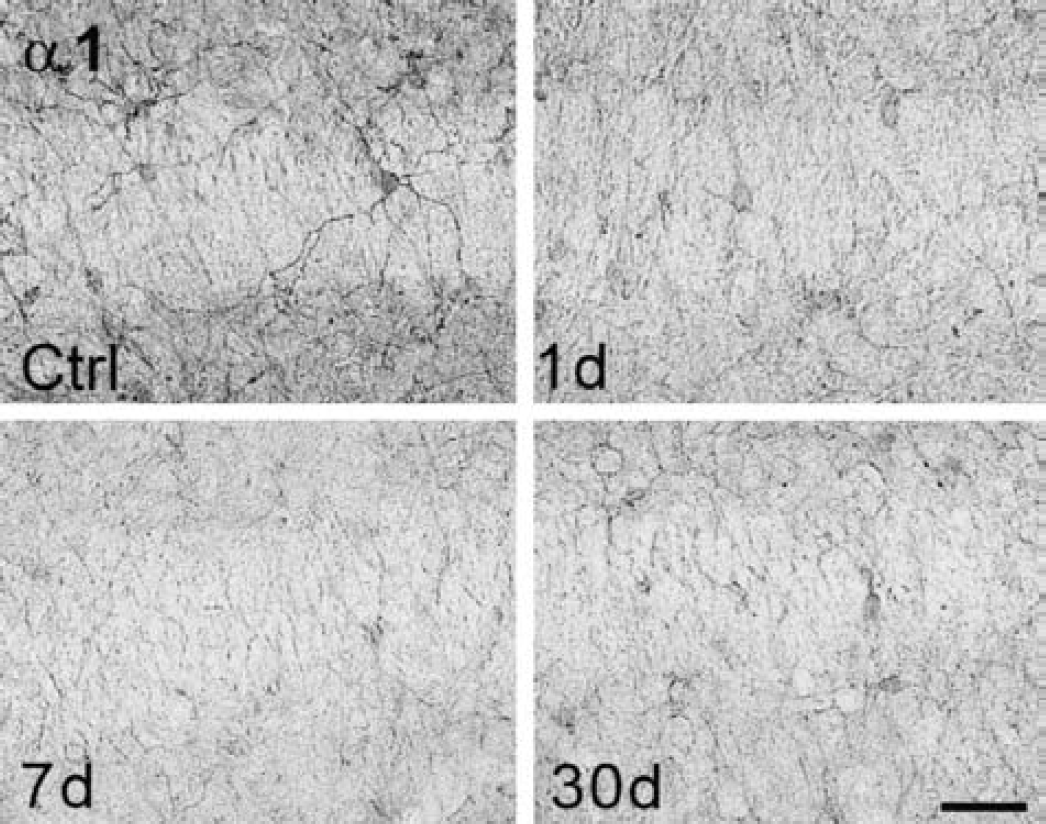
Reduction in GABAA-receptor subunit α1-positive interneurons within CA3 of the ipsilateral hippocampal formation after focal cortical infarcts. One day after the infarct, interneurons within the hippocampal formation lose their crisp cellular staining, at day 7 only contours of subunit α1-positive interneurons were present, and at day 30 this decrease in subunit α1 immunostaining partially recovered. Scale bar, 100 μm. GABA, γ-aminobutyric acid.
Blockade of GABAA-receptor subunit dysregulation after ischemic lesions by application of MK-801
In a further series of experiments, possible underlying mechanisms for this bihemispheric dysregulation of GABAA-receptor subunits after small cortical infarcts were analyzed. The distribution of these alterations in widespread, structurally intact brain regions as well as their early occurrence suggested that acute changes involving large parts of the brain network contributed to this long-lasting dysregulation of GABAA-receptor subunits. One mechanism affecting brain function soon after focal ischemia is cortical spreading depressions, which travel across the cortex during the first hours after the infarct (Schroeter et al., 1995) and induce long-lasting metabolic changes and multiple molecular cascades (Sharp et al., 2000; Witte et al., 2000). To determine whether postischemic spreading depressions contribute to the dysregulation of GABAA-receptor subunits, the competitive NMDA-receptor antagonist MK-801 was applied during lesion induction. This procedure has been shown to completely block spreading depression (Schroeter et al., 1995).
A total of 20 animals (14 animals with photothrombotic infarcts, 6 sham-operated controls) were treated with intravenous MK-801 (2 mg/kg body weight) 30 minutes before illumination or sham surgery. Interestingly, MK-801 failed to reduce the size of the infarcts in these animals, as it has been reported to do in several models of middle cerebral artery occlusion (Dirnagl et al., 1990; Iijima et al., 1992; Ozyurt et al., 1988; Park et al., 1988a,b). In contrast, the mediolateral diameter of the infarcts tends to be slightly increased in MK-801–treated animals (2.7 ± 0.8 mm, n = 14) compared with untreated lesioned animals (2.5 ± 0.5 mm, n = 13), although this difference was not significant. In accordance with the findings in untreated animals, the size of the lesions decreased within the first month after infarct induction (at day 1: 3.1 ± 0.6 mm; at day 7: 3.4 ± 0.1 mm; at day 30: 1.8 ± 0.2 mm, −45.5%). Since the amount of tissue salvageable by MK-801 depends on the size of the partially ischemic penumbral tissue surrounding the lesion (Yao et al., 1993), which is very thin in the photothrombotic model, MK-801 failed to reduce infarct diameter compared with nontreated animals.
The distribution of GABAA-receptor subunits in sham-operated MK-801–treated animals was identical to that seen in untreated controls and showed typical expression patterns for all subunits as described previously (Fig. 7). Even semiquantitative evaluation of regional optical densities did not reveal any consistent alteration in receptor expression. In animals with photothrombotic infarcts the application of MK-801 completely blocked the dysregulation of GABAA-receptor subunit expression. At day 1 (n = 5), 7 (n = 4), and 30 (n = 5) after the infarct, qualitative and semiquantitative evaluation of immunoreactivity for subunits α1, α2, α3, α5 and γ2 did not reveal any consistent alteration compared with MK-801–treated controls. Although the extent of receptor expression varied to some degree, the pattern of changes in GABAA-receptor subunit expression with widespread reduction of subunits α1, α2, α5, and γ2 in ipsilateral and contralateral regions, with upregulation of subunit α3 in the contralateral cortex homotopic to the lesion and bilateral reduction in immunoreactivity within the hippocampal formation and thalamic nuclei, was not observed in any MK-801–treated animal with a photothrombotic infarct (Fig. 7).
Blockade of dysregulation of GABAA-receptor subunits after focal cortical infarcts by a single application of the noncompetitive NMDA receptor antagonist MK-801 30 minutes before lesion induction.
Absence of GABAA-receptor subunit dysregulation after cortical spreading depressions
The complete blockade of postischemic GABAA-receptor dysregulation by MK-801 observed in these experiments did not determine whether cortical spreading depressions alone can influence the distribution of these receptors. To analyze this question, additional experiments were performed and the distribution of GABAA-receptor subunit expression was examined after spreading depressions elicited by topic application of KCl (3 mol/L). Repetitive cortical spreading depressions were elicited in seven animals and immunohistochemical analysis of GABAA-receptor subunit expression was carried out 1 (n = 2), 7 (n = 3), and 30 days (n = 2) after this procedure. During a recording period of 120 to 150 minutes, 9 or 10 DC deflections of approximately 5 mV (typical for cortical spreading depressions) were detected (Fig. 8). Subsequent histologic investigations with standard cresyl-violet stainings revealed no structural damage in all animals, neither in the area of KCl application nor in remote brain areas. The distribution of GABAA-receptor subunits was also identical with controls in all animals with cortical spreading depressions at day 1, 7, and 30 after the surgery (Fig. 8). Qualitative and semiquantitative evaluation did not reveal any significant difference for all receptor subunits investigated in this study.
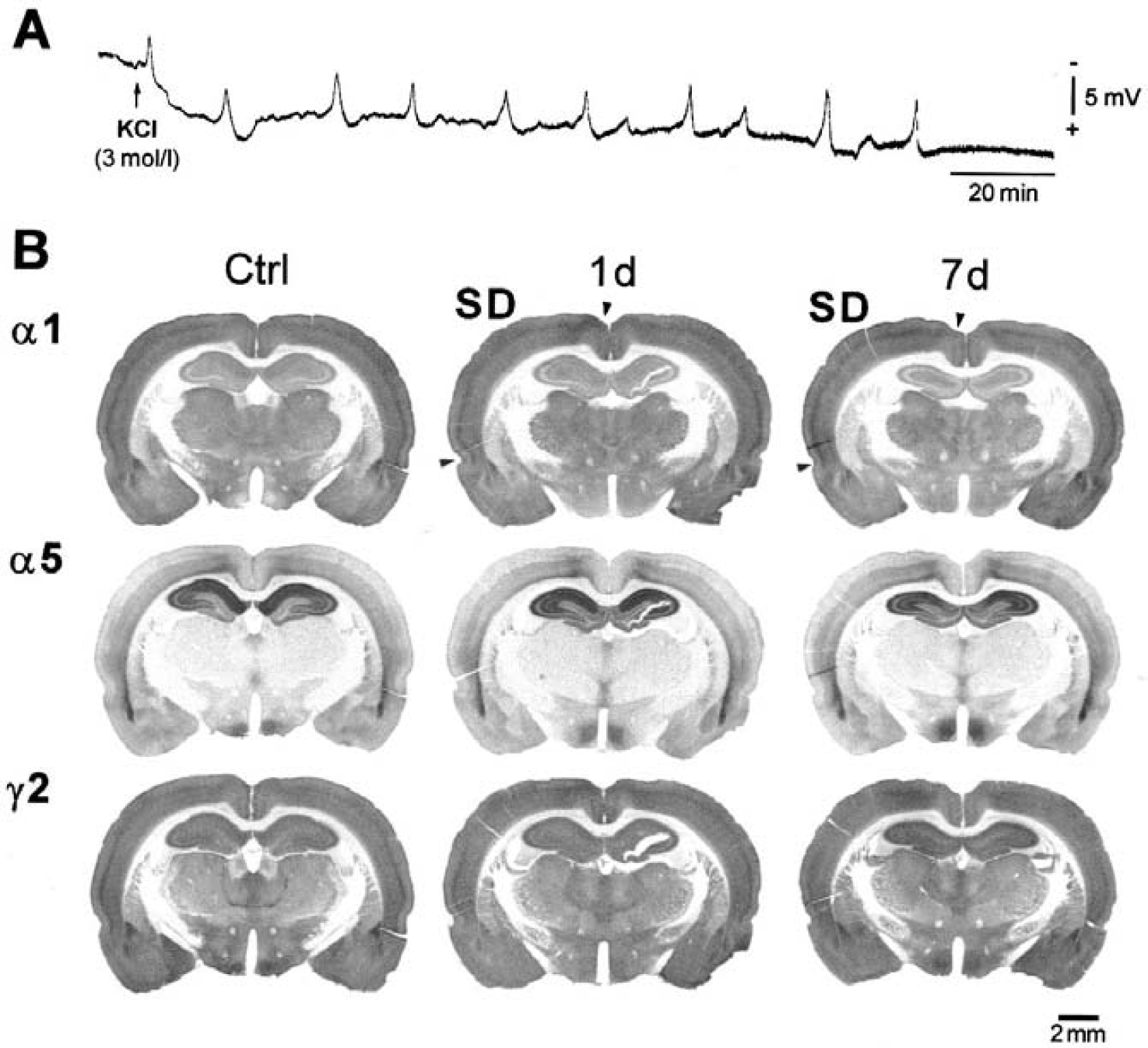
Absence of alterations in GABAA-receptor distribution after cortical spreading depressions induced by topical application of KCl (3 mol/L).
DISCUSSION
The present study clearly demonstrated bihemispheric and long-lasting alterations in GABAA-receptor subunit expression after small photothrombotic infarcts. A general, regionally differential reduction was found within the cortex, hippocampus, and thalamus of both hemispheres for nearly all subunits, indicating an absolute reduction of receptors in these brain regions. The pattern of changes, however, within ipsilateral and contralateral neocortical areas with a differential reduction of subunits α1, α2, α5, and γ2 and a significant upregulation of subunit a3 in the contralateral cortex homotopic to the lesion points toward a considerable modulation of GABAA-receptor subunit composition, which may specifically alter receptor function. The present study further demonstrated that blockade of NMDA receptors by application of MK-801 during lesion induction completely prevented the bihemispheric changes in GABAA-receptor subunit distribution, providing evidence that NMDA-dependent excitatory processes occurring during the first hours of the ischemic infarct mediate these widespread and long-lasting alterations in subtype expression.
Topography of alterations in GABAA-receptor subunit distribution
The widespread alterations in GABAA-receptor subunit expression described in this study correspond well to previous receptor autoradiographic studies revealing a decreased binding of the GABA agonist muscimol in bihemispheric brain regions. In accordance with the present study, this decrease in muscimol binding showed the same time course and was more prominent in surrounding and remote ipsilateral areas than in the contralateral cortex (Qu et al., 1998a; Schiene et al., 1996). A decreased binding to GABAA receptors can represent a reduction in density and/or changes in affinity of the receptor. The prominent downregulation of abundantly expressed subunits within the cortex, hippocampus, and thalamus of both hemispheres points toward a general reduction of receptor density, providing further evidence for an involvement of the whole brain network after focal cortical lesions. Since the hippocampal formation as well as thalamic nuclei are both tightly connected to the cortical network via the perforant path and multiple corticothalamic circuits, transsynaptic processes mediate the alterations in receptor expression in structurally intact brain regions.
The present study further demonstrated regionally specific modifications of subunit distribution after focal cortical infarcts. The differential downregulation of subunits α1, α2, α5, and γ2 in neocortical areas of both hemispheres as well as the strong upregulation of subunit α3 within the contralateral cortex homotopic to the lesion point toward specific changes in subunit composition that are likely to modify receptor affinity and function in these brain regions. The findings hereby complement those of a previous study (Neumann-Haefelin et al., 1998) showing a slight downregulation of the GABAA-receptor subunit α1 in the area surrounding the photothrombotic infarct, whereas expression of subunit α2 was unimpaired. Owing to an improvement of the immunohistochemical staining technique with a modified antigen-retrieval procedure (Fritschy et al., 1998), the detection of synaptic GABAA-receptor proteins was enhanced in this study and more widespread alterations could be disclosed. Similar but less extensive alterations have been reported after developmental cortical lesions (Redecker et al., 2000b).
Role of NMDA-dependent excitatory processes
The present study clearly demonstrated that NMDA-dependent excitatory processes crucially influence the expression of GABAA-receptor subunits in widespread brain regions after small photothrombotic infarcts. A single application of the noncompetitive NMDA-receptor antagonist MK-801 30 minutes before infarct induction completely blocked the long-lasting dysregulation of GABAA-receptor subunits in ipsilateral and contralateral brain regions. After focal ischemic brain infarcts, MK-801 has been shown to block periinfarct depolarizations and spreading depressions that travel across the ipsilateral hemisphere during the first hours after the infarct (Dietrich et al., 1994; Mies et al., 1993a; Schroeter et al., 1995) and induce various molecular cascades (Collaco-Moraes et al., 1994; Jander et al., 2001; Miettinen et al., 1997). In addition, electrophysiologic investigations revealed that repetitive spreading depressions in brain-slice preparations cause an impairment of functional GABAergic inhibition (Kruger et al., 1996), providing evidence that spreading depressions possibly alter the ipsilateral GABAA-receptor subunit expression. Surprisingly, application of MK-801 not only blocked GABAA-receptor dysregulation in the ipsilateral hemisphere, but also completely inhibited alterations in subunit distribution on both hemispheres. These results, together with the finding that spreading depressions induced by topical application of KCl in vivo did not change GABAA-receptor subtype expression, clearly demonstrate that spreading depressions do not contribute to the dysregulation of GABAA-receptor subunits after focal brain ischemia.
MK-801 does more than inhibit spreading depressions after focal ischemia. Pretreatment with this drug also influences early NMDA-dependent excitatory processes that occur during lesion induction. Within the ischemic core, neurons deprived of oxygen and glucose release excessive amounts of glutamate, causing an overstimulation of its receptors, a massive calcium influx, and finally cell death (Dugan and Choi, 1994; Lee et al., 1999). Acute ischemic brain injury hereby initiates an early hyperexcitability and hypersynchronous discharges of large populations of excitatory neurons that could spread through the whole brain network. Blockade of NMDA-dependent neurotransmission by MK-801 during lesion induction suppresses this early excitation, which may mediate the postlesional alterations in GABAA-receptor subunit distribution. This hypothesis is supported by an autoradiographic study showing that MK-801 completely abolished a sequential change of local glucose metabolism in the ischemic brain region. Using a model of transient middle cerebral artery occlusion, a shift from excitation-induced hypermetabolism to hypometabolism was observed within the infarct core during ischemia, which was completely blocked by MK-801 (Liu et al., 1997).
Unfortunately, it is unclear whether the dysregulation of GABAA receptors is mediated specifically by NMDA receptors or whether an (over)excitation-induced general decrease in protein synthesis contributes to these alterations. Evidence for an impairment of protein synthesis came from a previous study demonstrating a threefold increase in mRNA levels of subunits α1 and α2 1 week after ischemia in the surrounding infarct (Neumann-Haefelin et al., 1999). This finding, together with the decrease in GABAA-receptor subunit protein, indicates a partial block of translation in the perilesional zone. Inhibition of protein synthesis is well known to occur in association with cerebral ischemia (Hossmann, 1993). This inhibition, however, is usually restricted to the ischemic region or found within the hippocampus after global ischemia (Thilmann et al., 1986). Application of MK-801 reduces this decrease in protein inhibition (Christensen et al., 1996; Mies et al., 1993b) and may therefore contribute to the prevention of ischemia-induced alterations in GABAA-receptor expression after MK-801 treatment. MK-801 not only blocked the general reduction of receptor subunits, however, but also prevented specific changes in subunit distribution in the contralateral cortex.
Functional consequences of alterations in GABAA-receptor subunit distribution
The widespread and long-lasting changes in distribution of GABAA-receptor subunits correlated well with electrophysiologic studies using similar ischemia models (Witte et al., 2000) as well as with clinical investigations demonstrating a modulation of cortical inhibition after stroke (Liepert et al., 2000a,b). Taking into account that the application of the GABAA-receptor antagonist bicuculline at concentrations that are estimated to block no more than 10% to 20% of the GABAA receptors can induce epileptiform activity in vitro (Chagnac Amitai and Connors, 1989), this impairment of GABAergic function may contribute to postlesional epileptogenesis and may disturb information processing within the brain network. GABAergic synapses normally function to restrict cortical activation and thereby contribute to specific input processing (Jones, 1993). Iontophoretic application of the GABAA-receptor antagonists induces a substantial expansion of receptive field size in somatosensory and visual cortical areas (Dykes et al., 1984; Hicks and Dykes, 1983; Kyriazi et al., 1996), as well as a reshaping of motor maps in the motor cortex (Jacobs and Donoghue, 1991). After photothrombotic infarcts, an increase in size of cortical vibrissa representation was also found in the surrounding lesion using metabolic mapping methods (Schiene et al., 1999), indicating an impairment of specific input processing. Decreases in GABAergic function, however, may also facilitate plastic changes and may increase the propensity for long-term potentiation commonly associated with plasticity and learning, as it has been shown in the area surrounding photothrombotic infarcts (Hagemann et al., 1998).
The regionally differential alterations in GABAA-receptor subunit expression probably contribute to these complex changes in inhibitory function after focal ischemia. In particular, the prominent upregulation of subunit α3 in the contralateral cortex homotopic to the lesion points toward a specific change in this brain region. Functionally, extracellular recordings in brain-slice preparations did not reveal any specific alterations in cortical inhibition in this brain area compared with the lesion surround (Buchkremer-Ratzmann et al., 1996), although in vivo recordings within the contralateral homotopic cortex demonstrated layer-specific changes in spontaneous activity that were not observed in the surround of the lesion (Divanach et al., 2001). Interestingly, the slight decrease of subunits α1 and α2 accompanied by a prominent increase of subunit α3 changed the balance of α subunits in favor of subunit α3. Since subunit α3 is abundantly expressed during early postnatal development, even eclipsing the expression of subunit α1 (Laurie et al., 1992), this imbalance may reflect a reestablishment of a developmental GABAA-receptor expression pattern, probably making the cortical network more capable for plastic changes in this brain area.
Footnotes
Acknowledgments:
The authors thank D. Steinhoff, S. Hamm, and A. Sandermann for their excellent technical assistance, Dr. M. Horn (Institute of Medical Statistics and Informatics, Friedrich-Schiller-University Jena) for his help with statistical analysis, and Dr. C. Bruehl for critical comments regarding the manuscript.
