Abstract
The authors compared the influence of environmental enrichment on intact and lesioned brain, and tested the hypothesis that postischemic exposure to an enriched environment can alter dendritic spine density in pyramidal neurons contralateral to a cortical infarct. The middle cerebral artery was occluded distal to the striatal branches in spontaneously hypertensive rats postoperatively housed either in a standard or in an enriched environment. Intact rats were housed in the same environment. Three weeks later the brains were perfused in situ. The dendritic and spine morphology was studied with three-dimensional confocal laser scanning microscopy after microinjection of Lucifer yellow in pyramidal neurons in layers II/III and V/VI in the somatosensory cortex. In intact rats, the number of dendritic spines was significantly higher in the enriched group than in the standard group in all layers (P < 0.05). Contralateral to the infarct, pyramidal neurons in layers II/III, which have extensive intracortical connections that may play a role in cortical plasticity, had significantly more spines in the enriched group than in the standard group (P < 0.05). No difference was observed in layers V/VI. They conclude that housing rats in an enriched environment significantly increases spine density in superficial cortical layers in intact and lesioned brain, but in deeper layers of intact brain.
Postischemic housing in an enriched environment can significantly improve functional outcome after permanent focal brain ischemia in the rat (Grabowski et al., 1995; Olsson and Johansson, 1995; Johansson and Ohlsson, 1996), even when delayed for 2 weeks after the ischemic insult (Johansson, 1996). Different mechanisms may contribute to the environmental effects in the postischemic brain. Neuronal changes in other parts of the brain have been documented from minutes to months after focal brain lesions (Nicolelis, 1997). Postlesion events may be due to deafferentation, alteration of the balance between excitation and inhibition, changes in membrane excitability, unmasking of preexisting connections, and growth of new connections (Hallett, 1999). The extent to which postischemic functional improvement depends on changes in areas surrounding the infarct in contralateral cortex or in subcortical regions is unclear, and may vary with lesion size and location (Johansson, 2000).
Dendritic spines are the primary postsynaptic targets of excitatory glutamatergic synapses (Eilers and Konnerth, 1997; Boyer et al., 1998; Harris, 1999). Strategically located as bridges between axons and dendrites, dendritic spines were early to be proposed as primary sites of synaptic plasticity (Eccles, 1979; Crick, 1982). The spine cytoskeleton consists of actin filaments, and video recordings from hippocampal neurons expressing actin tagged with fluorescent protein have shown that the shape of spines can change rapidly (Fischer et al., 1998; Matus, 1999). Spines are calcium compartments with a constantly changing ability to compartmentalize calcium (Majewska et al., 2000; Yuste et al., 2000). Highly localized dendritic calcium signals limited to small dendritic segments and even to single dendritic spines have been documented (Svoboda et al., 1997), and are thought to be caused primarily by the entry of extracellular calcium (Kovalchuk et al., 2000).
Considering the central role of dendritic spines in brain function, we hypothesized that they may play a role in brain plasticity after ischemic brain lesions. Housing intact animals in an enriched environment can increase dendritic branching and number of synapses and dendritic spines (Volkmar and Greenough, 1972; Globus et al., 1973; Greenough et al., 1973; Kolb, 1995; Comery et al., 1996), whereas rearing animals in social isolation has the opposite effect (Bryan and Riesen, 1989). Most studies on environmental influence on dendritic morphology have been performed using modifications of the Golgi technique or with electron microscopy of thin sections. Because of the small size, large number, and highly variable shapes of dendritic spines, those techniques are not optimal for identification and quantification of dendritic spines, for which three-dimensional visualization is essential (Harris et al., 1992).
The aims of our study were to test the hypothesis that postischemic housing conditions can influence dendritic morphology and number of spines, and to compare the effect of an enriched environment on intact and lesioned brain. Microinjection of Lucifer yellow into individual neurons combined with confocal microscopy allows a three-dimensional view of dendritic spines and better quantification of spines than standard light microscopy (Belichenko and Dahlström, 1995). With this technique, we have analyzed dendritic and spine morphology of pyramidal cells in layers II/III and V/VI of the somatosensory cortex of intact and postischemic brains.
METHODS
The local Ethical Committee for Animal Experiments approved the experimental protocol.
Intact rats
Adult (3-month-old) male spontaneously hypertensive rats who were previously housed in standard laboratory cages (550 × 350 × 200 mm per four rats) either were kept in the same environment or were transferred to an enriched environment (i.e., 820 × 610 × 450 mm cages with horizontal and inclined boards, equipped with various activity-stimulating items such as a chain, swing board, wooden blocks, or balls; see Olsson and Johansson, 1995). Some changes, such as altering the space between the boards and adding some objects while removing others, were made three times a week.
Rats subjected to middle cerebral artery occlusion
Rats of the same strain and age as intact rats who were preoperatively housed in standard environment were anesthetized with methohexital sodium 50 mg/kg intraperitoneally. Using a microsurgical approach, the right middle cerebral artery was ligated with a 10–0 monofilament nylon thread distal to the origin of the striatal branches (Grabowski et al., 1995). The rats were housed in individual cages for 30 hours postoperatively, then returned either to standard cages or to an enriched environment. Although spontaneously hypertensive rats get more consistent infarcts than normotensive rats (Duverger and MacKenzie, 1988), there is usually some variation in the extension of the infarcts after arterial ligation. Therefore, we chose to have eight rats in these groups to select the four most comparable standard and enriched rats for the detailed spine study.
Perfusion and fixation
Three weeks after placing the rats in different environments, rats were anesthetized with sodium pentobarbital 100 mg intraperitoneally and their brains were perfused in situ with 0.9% NaCl for 1 minute, followed by 4% paraformaldehyde solution in 0.1 mol/L phosphate buffer (pH 7.4) for 10 minutes.
Tissue specimens and Lucifer yellow microinjection
Coronal blocks (bregma −2.8 mm) (Paxinos and Watson, 1982) were taken from both sides of the sensorimotor cortex in intact brains and from the cortex contralateral to the infarct in the infarcted brain. The tissue was sectioned serially perpendicular to the pial surface with an Oxford vibratome at 200 μm, and stored in 0.1 mol/L phosphate buffer at 4°C until microinjection of Lucifer yellow. Some of the sections were stained by the Nissl method to observe layers and area borders. Vibratome sections were immersed in phosphate buffer and held in place with a Millipore filter, with a window over the area to be injected to allow glass pipette access. The brain slices were placed under a fluorescence microscope, where neuronal cell bodies could be observed because of the presence of lipofuscin. In the fluorescence microscope, which was equipped with a 10X objective, single pyramidal cells in layers II, III, V, and VI of the somatosensory cortex were selected, impaled with a glass pipette, and injected iontophoretically with 6% Lucifer yellow. The procedure was performed in three to four serial slices of each specimen, and 15 to 20 neurons were injected with the tracer in each section.
Morphologic analysis
The dendritic and spine geometry was studied with three-dimensional confocal laser scanning microscopy. Dendritic spines were identified along the dendrites on confocal images, and represented as number of spines per 100-μm length of dendrite. Because the distribution of dendritic spines is not equal along the dendrites, the calculation always started from the same position: 30 to 60 μm from the cell body in basal and oblique apical dendrites, and 50 to 75 μm from the cell body in apical dendrites, and followed to the distal end of the dendrite (70–120 μm).
Statistical analysis
The two intact groups and the two lesioned groups of rats were compared separately. Although the in vivo part of the study was done at the same time for all four groups, it was not technically possible to inject and analyze the large amount of neurons studied for more than two groups at a time. The statistical analysis was performed with nonparametric analysis of variance (StatView software, version 5.0; SAS Institute, Inc., Cary, NC, U.S.A.). A P value < 0.05 was considered significant.
RESULTS
Each experimental group consisted of four rats. The data are based on examination of 344 pyramidal neurons in the somatosensory cortex. Each neuron was treated as an independent observation, and the exact number of neurons in each layer and each group is specified in Figs. 2 and 6.
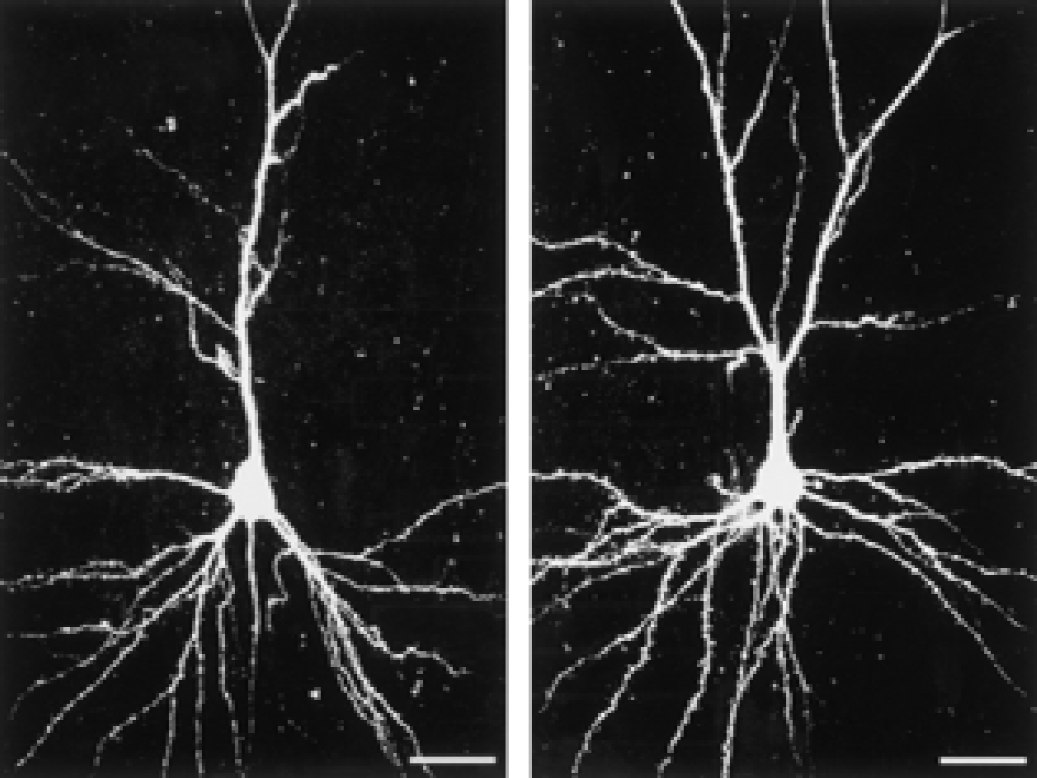
Dendritic morphology of pyramidal neurons in layer III of the somatosensory cortex in a rat housed in standard (left) and enriched (right) environments, as viewed in confocal imaging after microinjection of Lucifer yellow into the neurons. Bar = 25 μm.
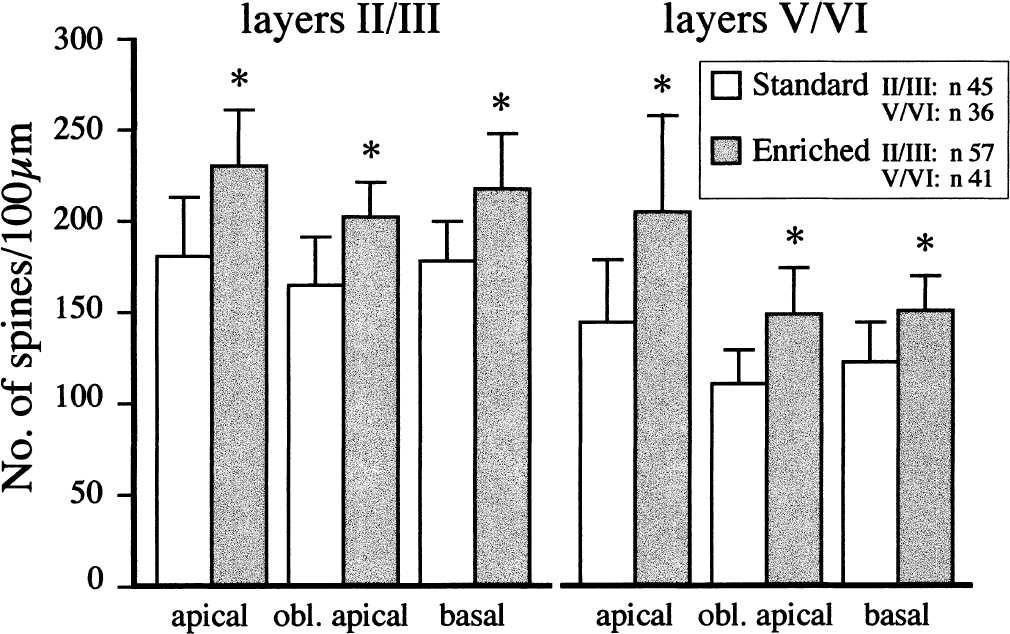
Dendritic spine density presented as spines per 100-μm length of dendrite on apical, oblique (obl) apical, and basal dendrites of pyramidal neurons in a somatosensory cortex of adult intact rats housed in standard environment or transferred to an enriched environment for 3 weeks. Data are mean ± SD. * P < 0.05 for differences between standard and enriched rats.
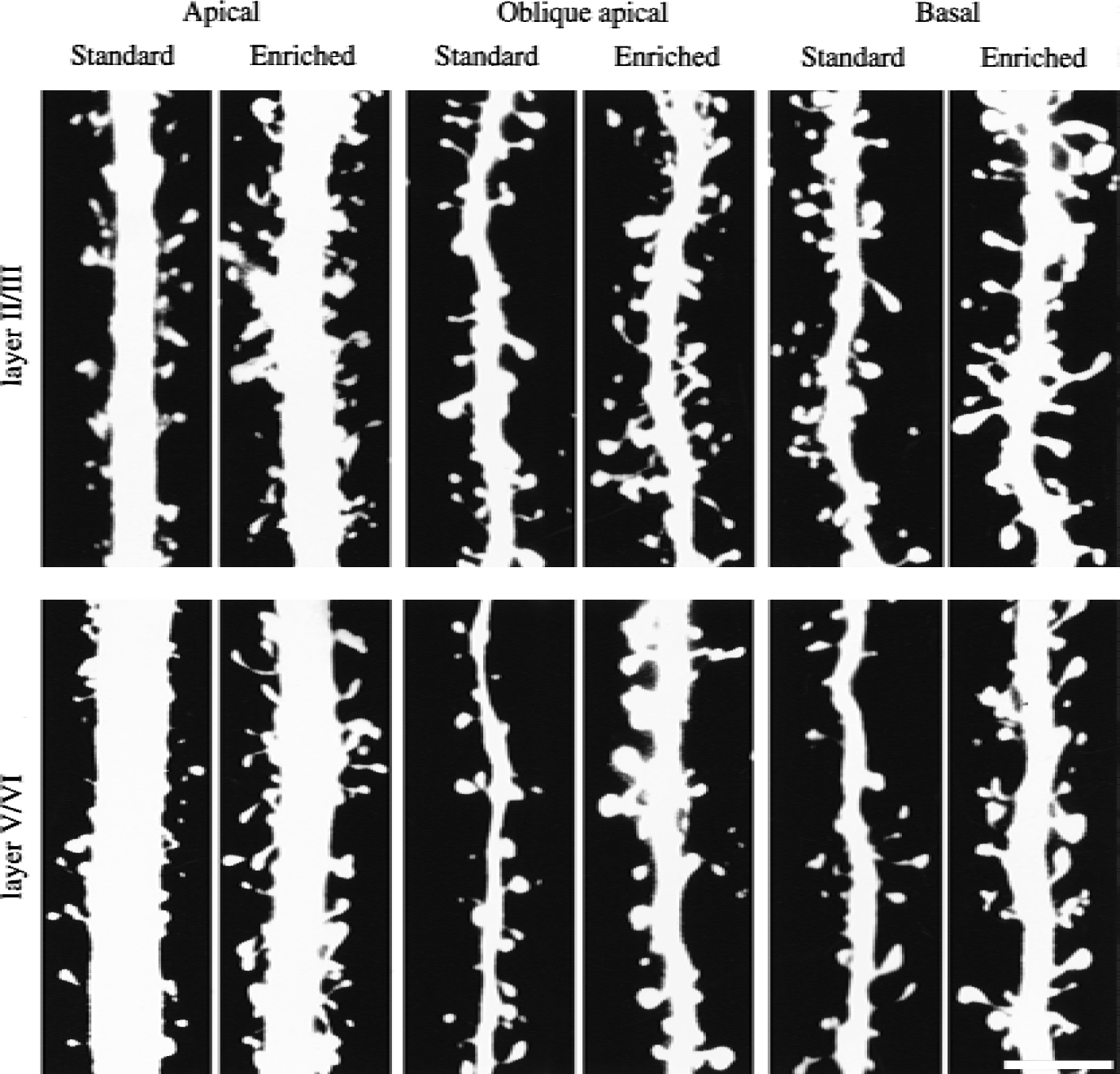
Confocal imaging of dendritic spines in somatosensory cortex in adult rats housed in standard environment or exposed to an enriched environment for 3 weeks. Pyramidal neurons in the enriched group had more dendritic spines than the standard group in layers II/III (upper panel) and V/VI (lower panel), and the individual spine morphology was more variable. Scale bar = 5 μm.
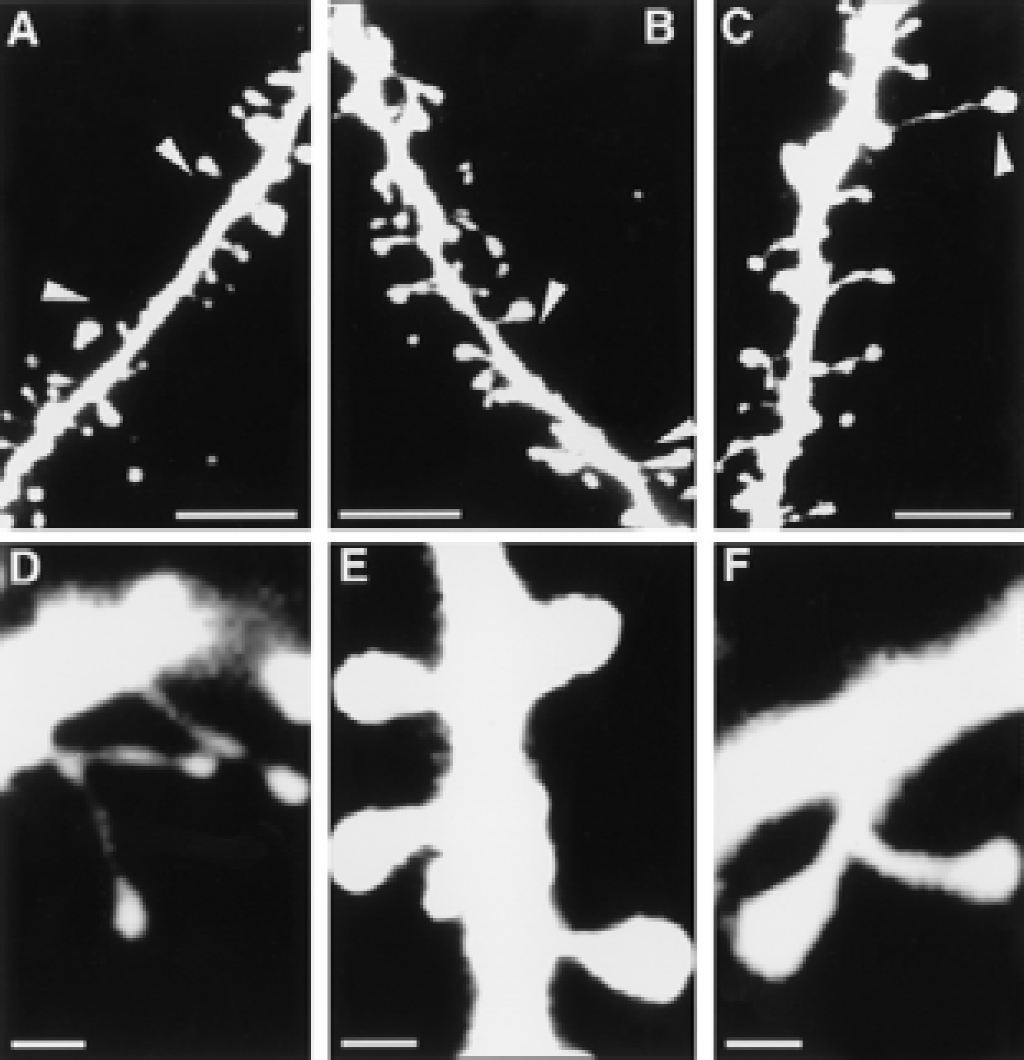
Morphologic features of dendritic spines in the somatosensory cortex of adult rats housed in standard or enriched environments. “Naked” sections of dendrites (arrowheads) in rats housed in standard environment
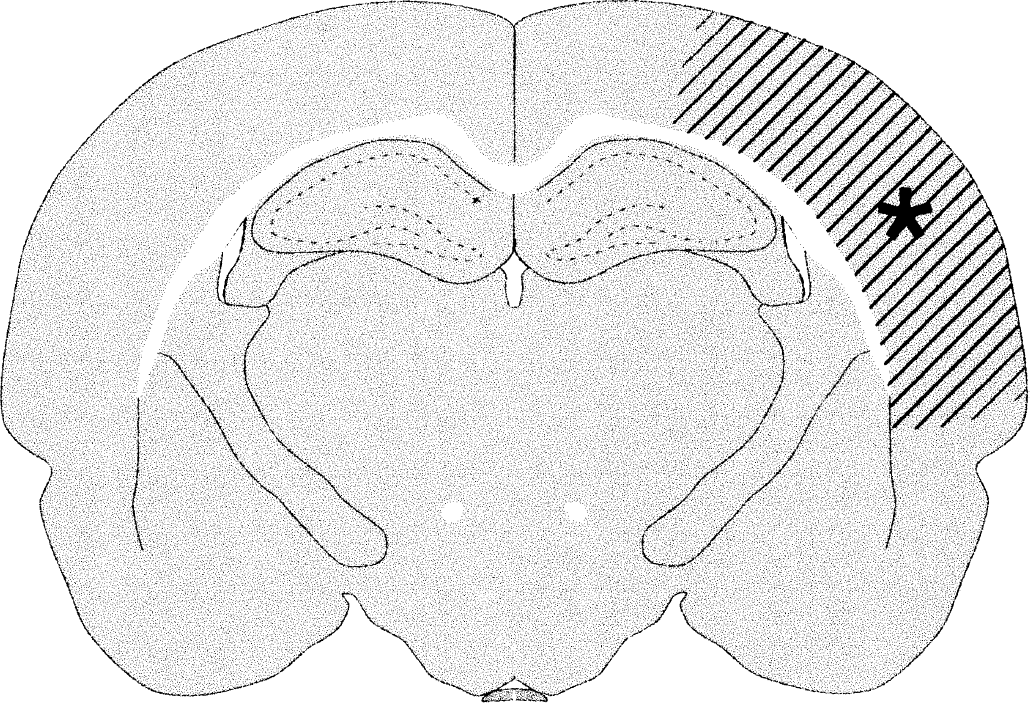
The extension of the infarct at the level the neurons were injected with Lucifer yellow on the contralateral intact side.
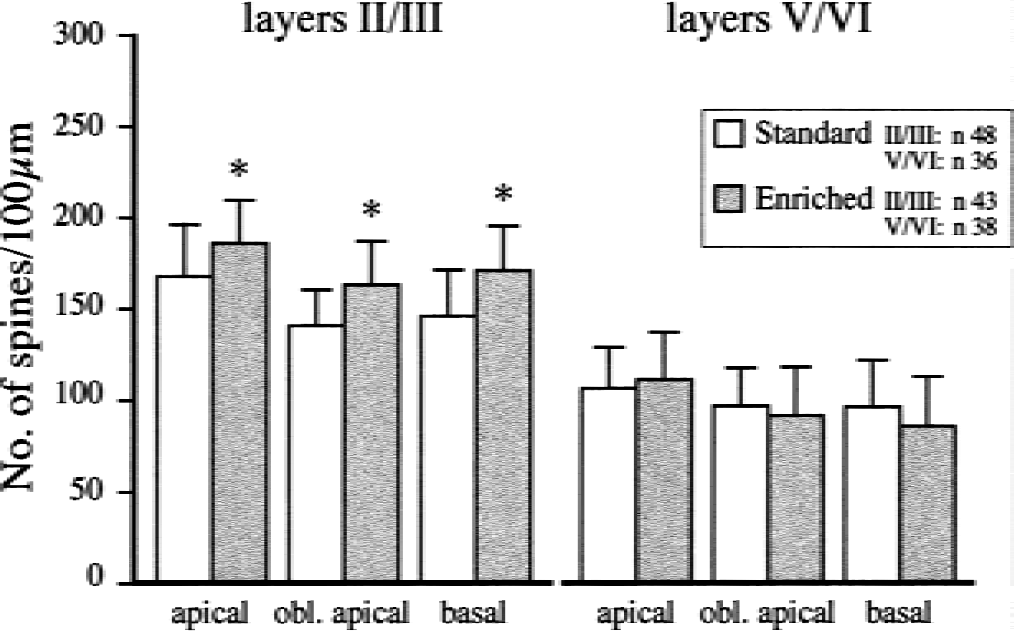
Dendritic spine density presented as spines per 100-μm length of dendrite on apical, oblique (obl) apical, and basal dendrites of pyramidal neurons in somatosensory cortex 3 weeks after ligation of the middle cerebral artery in rats housed in standard environment or transferred to an enriched environment for 3 weeks. Data are mean ± SD * P < 0.05 for differences between the standard and enriched rats.
Intact rats
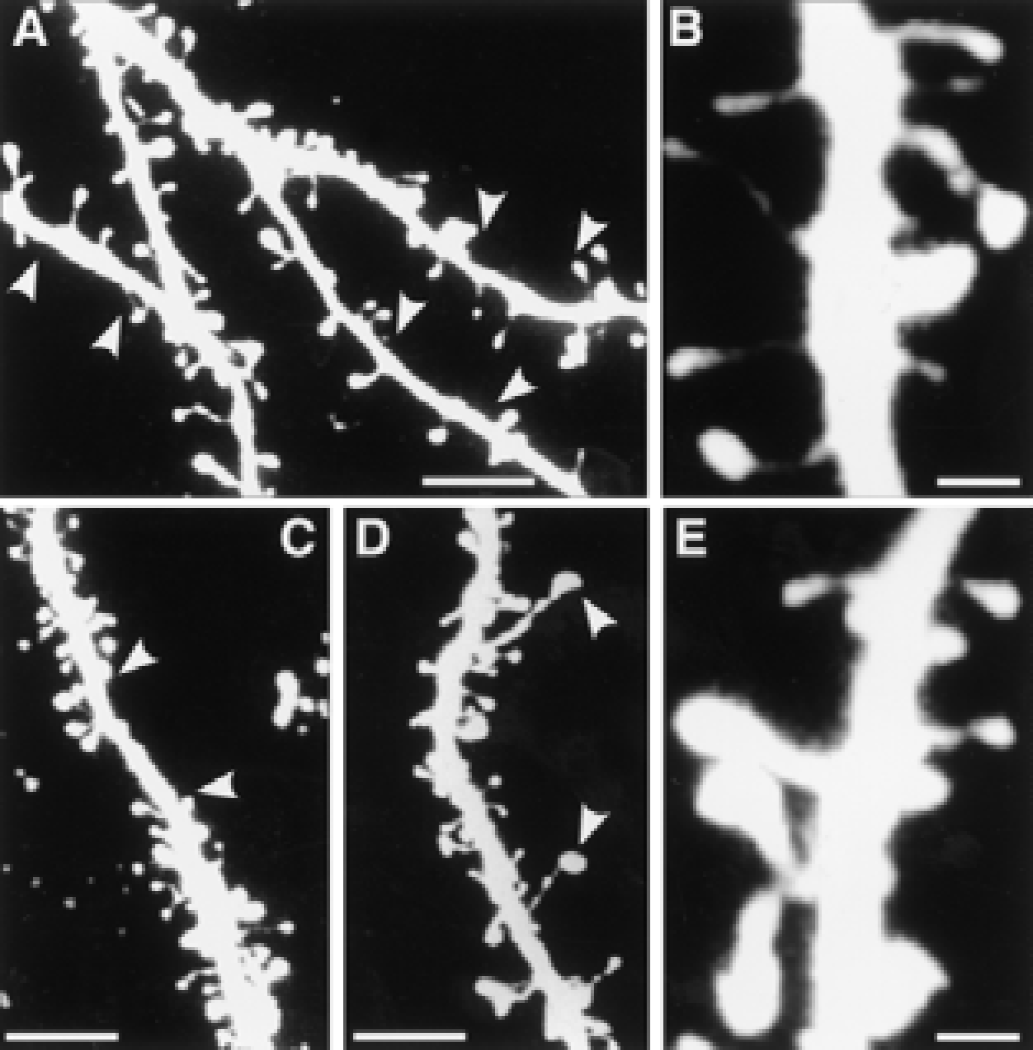
Although we did not quantify dendritic branching, it was evident that dendrites from rats housed in an enriched environment had more branches than dendrites from rats housed in standard environments (Fig. 1). The number of spines per 100-μm length of dendrite was significantly larger in enriched rats than in standard rats (Fig. 2), with the largest difference in apical dendrites (25% in layers II/III, 40% in layers V/VI). In rats housed in standard environments, sections of the dendrites occasionally lacked spines; the length of such “naked” places could be up to 7 to 10 μm (Fig. 3, Figs. 4A and 4B). Some spines appeared “inactive,” with small head areas (0.1–0.2 μm2) and thin necks (Fig. 4C). In rats housed in enriched environments, the individual spine morphology varied more than in rats housed in standard environments (Fig. 3). Dendritic spines with long necks (Fig. 4D), large spine head area (≦ 0.8–1.1 μm2) and neck diameter (Fig. 4E), and more branched spines (Fig. 4F) were frequently observed.
Postischemic rats
General description.
Figure 5 shows the extension of the infarct at the level where the neurons were studied contralateral to the lesion. Microinjection of the tracer Lucifer yellow was more difficult to perform in these brains than in the intact brains. Injections could be performed as well as in intact animals in part of the neurons; however, in others it was difficult to observe the neurons on the confocal microscope because of the high degree of tracer leakage. In layers II/III, the leakage was less frequent in rats postoperatively housed in an enriched environment, and in these layers the dendrites were also more branched in the enriched group. No apparent difference in dendritic branching was observed in layers V/VI. In the standard-lesioned groups, the neuronal morphology varied more than in intact rats housed in the same environment, particularly in layers II/III, and exhibited fewer basal and oblique apical dendrites. Some dendrites had changed orientation (e.g., basal dendrites going to the “apical” part of the pyramidal neuron, and oblique apical dendrites going in the basal direction) (data not shown).
Spine density.
In layers II/III, rats postoperatively housed in an enriched environment had more dendritic spines than rats housed in standard conditions (P < 0.05, Fig. 6). The variation of spine morphology in apical, oblique apical, and basal dendrites was large in standard rats (Fig. 7, upper part). More “naked” areas (Fig. 8A) and more apparently “inactive” spines were observed (Fig. 8B). In the enriched group, the neurons and spines varied less than in the standard group (Fig. 7, lower panel). However, naked areas could be observed in the enriched group (Fig. 8C). Dendritic spines with long necks (≦ 4.0–4.3 μm) were frequently noted (Fig. 8D). Spine clusters were observed and many spines appeared to be in an active stage with double heads and big head areas (0.9–1.2 μm2, Fig. 8E). No group difference in spine morphology was noted in layers V/VI, and the number of spines appeared to be lower than in intact rats in both lesioned groups (Figs. 2 and 6).

Confocal imaging of dendritic spines on apical, oblique apical, and basal dendrites on pyramidal neurons in layers II/III in the somatosensory cortex contralateral to an infarct induced by ligation of the middle cerebral artery 3 weeks earlier. The rats were postoperatively housed in standard or enriched environments. Note the large heterogeneity of the number of dendritic spines in rats housed in standard environment (upper panel) compared with the more homogenous appearance and average higher number of spines in enriched rats (lower panel). Scale bar = 5 μm.
Detailed morphologic features of dendritic spines on pyramidal neurons in layers II/III contralateral to an infarct induced by a ligation of the middle cerebral artery. “Naked” areas (arrowheads) were frequently seen on the dendrites in rats housed in standard environments
DISCUSSION
Our data show that housing intact rats in an enriched environment significantly increases dendritic spines on pyramidal cells in all cortical layers. Postischemic housing in enriched environment influences spine density in layers II/III but not in layers V/VI in the contralateral hemisphere. Spine density has been shown to be a more sensitive measure of environmental influences than dendritic branching (Bryan and Risen, 1989). Behavioral tests have been reported to increase brain-derived neurotrophic factor gene expression in rats housed in enriched environments, but not in rats housed in isolation (Falkenberg et al., 1992). Therefore, the rats were not tested to avoid any possible confounding effect on spine density. However, the dendritic spines were quantified 3 weeks after the middle cerebral artery occlusion, at which time the difference in functional outcome between animals in standard and enriched environment is well established (Ohlsson and Johansson, 1995; Johansson and Ohlsson, 1996; Risedal et al., 1999).
A striking observation in the intact rats was the frequent presence of “naked” parts of dendrites in the standard group but not in the enriched group. In addition to an increase in spine number, the enriched environment changed spine morphology. More “active” spines with enlarged heads and more double-headed spines were observed. Increased density of multiple-head dendritic spines has been observed on medium-sized spiny neurons of the striatum in rats reared in a complex environment (Comery et al., 1996). Perforated synapses on double-headed dendritic spines have been proposed as possible structural substrate of synaptic plasticity (Geinisman et al., 1989). The existence of two modes of morphologic plasticity in dendritic spines have been suggested: one in which the formation of new spines is initiated by long-term potentiation-like stimulation operating through N-methyl- d -aspartate receptors; and a second in which spine morphology at established synapses is stabilized by AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid) receptor activation (Fisher et al., 2000). The correlation between spine morphology and function is not clear. However, the increase in postsynaptic size and synaptophysin immunoreactivity in connection with long-term potentiation might be an indirect support for the relevance of change in spine morphology (Desmond and Levy, 1988; Bozdagi et al., 2000).
Leakage of Lucifer yellow, which indicates a change in membrane permeability in the cortex contralateral to the infarct, was frequently observed in layers V/VI in both groups but was more common in the standard group than in the enriched group in layers II/III. Clearly, neurophysiologic data are needed to explain this observation, particularly why it was more common in the standard-lesioned group. We speculate that this finding might be related to the long-term cellular dysfunction and change in excitability described in the ipsilateral and contralateral hemisphere after focal brain ischemia (Mittmann et al., 1998; Qu et al., 1998).
Rats postoperatively housed in an enriched environment had significantly more dendritic spines in layers II/III than rats housed in standard cages. The most likely explanation for this finding would be that the difference between the infarcted standard and enriched groups was due to more intracortical connections in the enriched group. Pyramidal neurons in layers II/III have abundant horizontal connections with ipsilateral cortical regions, and synaptic plasticity in cortical horizontal connections has been proposed to underlie cortical plasticity and cortical map reorganization (Hess et al., 1996). The environmental effect on the number and morphology of dendritic spines in layers II/III is consistent with the better functional outcome observed in rats postoperatively housed in an enriched environment, but does not prove a causal connection. To what extent changes in the contralateral cortex are essential for functional recovery after cortical infarcts remains to be elucidated.
Many studies have shown that brain lesions can induce neuronal changes in the ipsilateral and contralateral hemisphere that may vary with the type and size of lesion and have a different time course (Raisman and Field, 1973; Wolff et al., 1981, Jones and Schallert, 1992, 1994; Stroemer et al., 1995; Jones et al., 1996; 1999; Napieralski et al., 1996; Cheng et al., 1997; Nicolelis, 1997; Shimada et al., 1997; Schallert and Kozlowski, 1998).
Dendritic spines degenerate after deafferentation, but sprouting from axon terminals can reinnervate dendrites. A progressive and transient loss of dendritic spines has been observed in the striatum after unilateral cortical transection lesions in the adult mouse. A 30% loss of unlesioned control values were observed 3 days after lesion formation, 45.5% 10 days postlesion, and a recovery to 80% of control levels 20 days after postlesion. The primary course of recovery of spine density was axonal sprouting of survival homologous afferent fibers from the contralateral cortex (Cheng et al., 1997).
Sectioning of corpus callosum at birth results in a permanent reduction of branches of oblique apical dendrites in layer-V pyramidal neurons (Globus and Scheibel, 1967). Using a modified Golgi-Cox method and laser scanning microscopy, the number of spines on apical dendrites of pyramidal neurons in layers III and V of the motor area was studied in mice 1, 6, 10, and 12 days after callosotomy (Shimada et al., 1997). A transient increase was observed, with a maximum at 6 to 10 days and a return to a normal-like level at day 12 after callosotomy. No later periods were studied. The authors concluded that dendritic spines might be formed superfluously on the deafferented dendritic portions in the initial period after callosotomy, and that some of the newly formed dendritic spines might disappear if failing to make synaptic contact with axon terminals sprouting form nearby axons.
Cortical electrolytic lesions restricted to the sensorimotor cortex representing the forelimb area have been reported to be associated with an increase in dendritic branching in layer-V pyramidal cells in the contralateral cortex, reaching a maximum approximately 18 days after the lesion. Immobilizing of the intact forelimb prevented both functional recovery and the increase in dendritic branching, which were therefore thought to be due to overuse of the intact forelimb (Jones and Schallert, 1992, 1994). Two studies of cortical aspiration lesions or less extensive electrolytic lesions did not reveal any evidence for use-dependent neuronal growth in the intact hemisphere 18 days after the lesion (Forgie et al., 1996; Prusky and Whishow, 1996). In a later electron microscopic study in the same experimental model as used in Jones and Schallert (1992, 1994) a tendency for reduced synaptic density was observed in the contralateral cortex at 18 days postlesion, but a significant increase was observed 30 days after the lesion (Jones et al., 1996). Motor-skills training significantly enhanced synapse number per neuron in layer V in the motor cortex opposite the damage 28 days after the lesion in the same experimental model (Jones et al., 1999).
Our aim was to evaluate if an enriched postischemic environment could alter spine density 3 weeks after the infarct when the difference in outcome is well established (Ohlsson and Johansson, 1995; Johansson and Ohlsson, 1996; Risedal et al., 1999). In light of the studies on synaptic density published by Jones et al. (1996, 1999), it is possible that we would have obtained different results for layer-V pyramidal neurons at later periods. However, other important factors, including type and size of the cortical lesion and the area investigated (Schallert and Kozlowski, 1998; Chu and Jones, 1999; Zeng et al., 2000), differed from our study factors. Furthermore, we do not know if neuronal plasticity induced by a combined physical and social activation in enriched environments has the same time course as those induced by specific reach training. According to the figures presented in the study on motor-skill training (Jones et al., 1999), the behavioral effects were established within 3 weeks. The electrolytic cortical lesion model data suggest that dendritic growth and functional improvement precede an increase in synaptic density, which is an interesting observation (Jones and Schallert, 1992, 1994; Schallert and Kozlowski, 1998; Jones et al., 1999).
The postischemic environment is an important factor for functional outcome that may also be of clinical significance (Johansson, 2000). We do not suggest that an enriched environment should be a substitute for specific training. However, postischemic housing in an enriched environment can enhance the functional effect of specific rehabilitation methods, which has been shown to be the case for neocortical transplantation (Mattsson et al., 1997; Zeng et al., 2000) and skilled -reach training (Biernaskie and Corbett, 2001).
Footnotes
Acknowledgments:
The authors thank Anna-Lena Ohlsson and Bengt Mattsson for their skillful technical assistance.
