Abstract
Glutamate release, particularly in pathologic conditions, may result in cellular swelling. The authors studied the effects of glutamate, N-methyl-d-aspartate (NMDA), and α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) on extracellular pH (pHe), extracellular potassium concentration ([K+]e), and changes in extracellular space (ECS) diffusion parameters (volume fraction α, tortuosity λ) resulting from cellular swelling. In the isolated spinal cord of 4-to 12-day-old rats, the application of glutamate receptor agonists induced an increase in [K+]e, alkaline-acid shifts, a substantial decrease in α, and an increase in λ. After washout of the glutamate receptor agonists, α either returned to or overshot normal values, whereas λ remained elevated. Pretreatment with 20 mmol/L Mg++, MK801, or CNQX blocked the changes in diffusion parameters, [K+]e and pHe evoked by NMDA or AMPA. However, the changes in diffusion parameters also were blocked in Ca2+-free solution, which had no effect on the [K+]e increase or acid shift. The authors conclude that increased glutamate release may produce a large, sustained and [Ca2+]e-dependent decrease in α and increase in λ. Repetitive stimulation and pathologic states resulting in glutamate release therefore may lead to changes in ECS volume and tortuosity, affecting volume transmission and enhancing glutamate neurotoxicity and neuronal damage.
Glutamate is the most common excitatory neurotransmitter in the mammalian central nervous system (CNS). Glutamate synapses are an integral part of neuronal pathways in various areas, taking part, for example, in the initiation and performance of movements (Vezina and Kim, 1999; Walwyn et al., 1998), sensory input (Bradley and Grabauskas, 1998; Thoreson and Witkowsky, 1999), pain modulation (Urban and Gebhart, 1998), and the formation of memory traces during long-term potentiation (LTP) (Kaczmarek et al., 1997; Raymond et al., 2000). The fast and immediate effect of glutamate on the postsynaptic membrane is mediated through α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA)/kainate and N-methyl-d-aspartate (NMDA) subtypes of the glutamate ionotropic receptors (Mayer and Westbrook, 1987). The slow prolonged effect of glutamate results from the activation of metabotropic receptors associated with cyclic adenosine monophosphate (cAMP) formation (Pin and Duvoisin, 1995). However, a number of studies have demonstrated that the release of excitatory amino acids, particularly glutamate, plays a major role in neuronal damage after ischemia and CNS injury (Kimelberg and Ransom, 1986; Benveniste et al, 1989; Choi, 1992).
It has been shown that the release of glutamate in either physiologic or pathologic conditions is accompanied by an increase in extracellular K+ concentration ([K+]e), alkaline-acid changes in extracellular pH (pHe), and an increase in Ca2+ metabolism (Chesler and Kaila, 1992; Deitmer and Munsch, 1994). Glutamate released into the extracellular space (ECS) is quickly removed and transported into the astroglial syncytium by high affinity and high capacity uptake carriers (Attwell, 2000). During pathologic conditions such as ischemia or anoxia, this process may collapse—rather than removing extracellular glutamate to protect neurons, an excessive release of glutamate reaches neurotoxic levels (Benveniste et al., 1989; Choi, 1992; Zorumski and Olney, 1993). It has been suggested that the transmembrane ionic shifts observed after glutamate release are accompanied by water movement from the extra-to the intracellular space, and that activation of both ionotropic and metabotropic glutamate receptors may result in rapid cellular swelling (Kimelberg, 1987; Chan et al., 1990; Koyama et al, 1991; Hannson et al, 1994). The influx of Na+, Cl−, and water, particularly because of the activation of the Na+-K+-2Cl− cotransporter, leads to glutamate-induced astroglial swelling (Kempski et al., 1991; Walz et al., 1993).
Furthermore, glutamate induces astrocyte swelling through the stimulation of a K+ influx dependent on extracellular Ca2+ (Bender and Norenberg, 1994). Tissue acidosis during ischemia or trauma induces glia swelling, which depends on the presence of extracellular Ca2+; alterations in [Ca2+]i do not seem to be involved (Plesnila et al., 1998). Oxygen-glucose deprivation induces substantial neuronal uptake of Ca2+ (Goldberg and Choi, 1993) and an abnormal influx of Ca2+ into myelinated axons in CNS white matter (Waxman et al., 1993). Because cellular swelling leads to an extracellular space volume decrease and to changes in ECS geometry (Voříšek and Syková, 1997; Syková et al., 1999, 2000, the diffusion of transmitters and other neuroactive substances through the ECS will be impaired and glutamate neurotoxicity and neuronal damage enhanced.
In the current study performed on the isolated rat spinal cord, where unlike in tissue culture, the cells are exposed to substances in their natural environment and the ECS is preserved, the authors studied the effects of glutamate receptor agonists on ECS diffusion parameters and compared these with the changes in [K+]e and shifts in pHe. To the authors' knowledge, the absolute values of the ECS volume decrease and changes in ECS geometry related to glutamate-induced cell swelling have not yet been determined. Such studies cannot be performed in tissue culture, and the only available method for obtaining the absolute value of the ECS volume in situ is the real-time iontophoretic method using ion-selective microelectrodes to determine the ECS diffusion parameters (Nicholson and Phillips, 1981; Nicholson and Syková, 1998).
The diffusion of substances released from neurons or glial cells into the ECS is hindered by three factors. First, diffusion in the ECS is constrained by the restricted volume of the tissue available for the diffusing particles, that is, by the extracellular volume fraction α = ECS volume/total tissue volume. Second, tortuosity λ (λ2 =D/ADC, where D is the free diffusion coefficient and ADC is the apparent diffusion coefficient in the brain) represents the restrictions on diffusion in the ECS that are imposed by cellular membranes, neuronal and glial processes, fixed negative and positive surface charges, glycoproteins, and macromolecules of the extracellular matrix. In addition to these two constraints, the diffusion of substances may be affected by nonspecific uptake, k ′, a factor describing the loss of a substance across cell membranes (Nicholson and Phillips, 1981; Nicholson and Syková, 1998). It has been shown that reversible changes in extracellular space volume and tortuosity result from cell swelling under a number of physiologic as well as pathologic conditions (Syková, 1997; Syková et al., 2000; Syková and Chvátal, 2000). Long-term or even permanent changes require other morphologic changes—for example, cell death, changes in cellular processes, astrogliosis, and/or changes in the extracellular matrix (Syková et al., 2000).
MATERIALS AND METHODS
Animal preparation and solutions
The current study was performed on 81 four-to 12-day-old Wistar rat pups. Experiments were performed in accordance with the European Communities Council Directive of 24 November 1986 (86/609/EEC). After decapitation performed under ether anesthesia, spinal cords were isolated in a chamber with cold (8°C) artificial cerebrospinal fluid (aCSF) of the following composition (in mmol/L): NaCl 117.0, KCl 3.0, NaHCO3 35.0, Na2HPO4 1.25, d-glucose 10.0, MgCl2 1.3, CaCl2 1.5. The solution was saturated with 95% O2 and 5% CO2 (pH 7.3). The isolated spinal cord was placed in a chamber of approximately 2 mL in volume, which was continuously perfused with aCSF containing 0.1 mmol/L tetramethylammonium chloride (TMACl) at a rate of 5 to 10 mL/min. The temperature in the chamber was increased to 21°C to 22°C during 1 hour. Ca2+-free solutions were prepared by omitting CaCl2 and adding 1 mmol/L EGTA. The following drugs were dissolved in aCSF and applied to the spinal cord: glutamate, NMDA, AMPA, dizolcilpine malate (MK-801), 6-cyano-7nitro-quinoqalin-2,3dion (CNQX), MgCl2. The osmotic strength of solutions was kept constant at 300 mmol kg−1 and was measured using a vapor pressure osmometer (Vescor, Logan, UT, U.S.A.). All substances were purchased from Sigma (St. Louis, MO, U.S.A.).
The authors' previous studies with ion-selective microelectrodes revealed that after changing the pH or potassium concentration of the perfusate upstream of the perfusion chamber, the half-rise time in the spinal dorsal horn (at a depth of 250 to 350 μm from the dorsal surface) is 1 to 2 minutes, including a delay of 30 to 60 seconds before the modified perfusate first reaches the tissue in the chamber. Equilibrium is attained within 5 to 7 minutes of the switch from aCSF to the modified solution (Jendelová and Syková, 1991; Syková, 1992b; Syková et al., 1999).
Measurements of extracellular space diffusion parameters
To measure diffusion parameters in the ECS, the real-time iontophoretic method originally developed by Nicholson and Phillips (1981) and described in the authors' previous studies (Lehmenkühler et al., 1993; Syková et al., 1994, 1999) was used. Briefly, an extracellular marker such as tetramethylammonium ions (TMA+), to which cell membranes are relatively impermeable, was administered into the nervous tissue by iontophoresis. The concentration of TMA+ was measured in the ECS by a TMA+-selective microelectrode (TMA+-ISM) and is inversely proportional to the ECS volume. Double-barreled TMA+-ISMs were prepared by the procedure described by Syková (1992a). The liquid ion-exchanger highly sensitive to TMA+ was Corning 477317. The TMA+-sensitive barrel of the microelectrode was backfilled with 100 mmol/L TMACl, whereas the reference barrel contained 150 mmol/L NaCl. Iontophoretic micropipettes were pulled from theta glass (Clark Electromedical Instruments, Edenbridge, U.K.). Before backfilling with 0.1 mol/L TMA+, the shank of the electrode was bent so that it could be aligned parallel to the TMA+-ISM. To stabilize the intertip distance (100 to 180 μm), both electrodes were glued together with dental cement (Fig. 1). Typical iontophoresis parameters were a +20 nA bias current (continuously applied to maintain a constant transport number) and a +200 nA current step of 60 seconds duration to generate the diffusion curve. TMA+ diffusion curves were captured on a digital oscilloscope (Nicolet 310), transferred to a PC-compatible computer, and analyzed to yield the volume fraction α, tortuosity λ, and nonspecific uptake k ′ using the program VOLTORO (kindly provided by C. Nicholson).
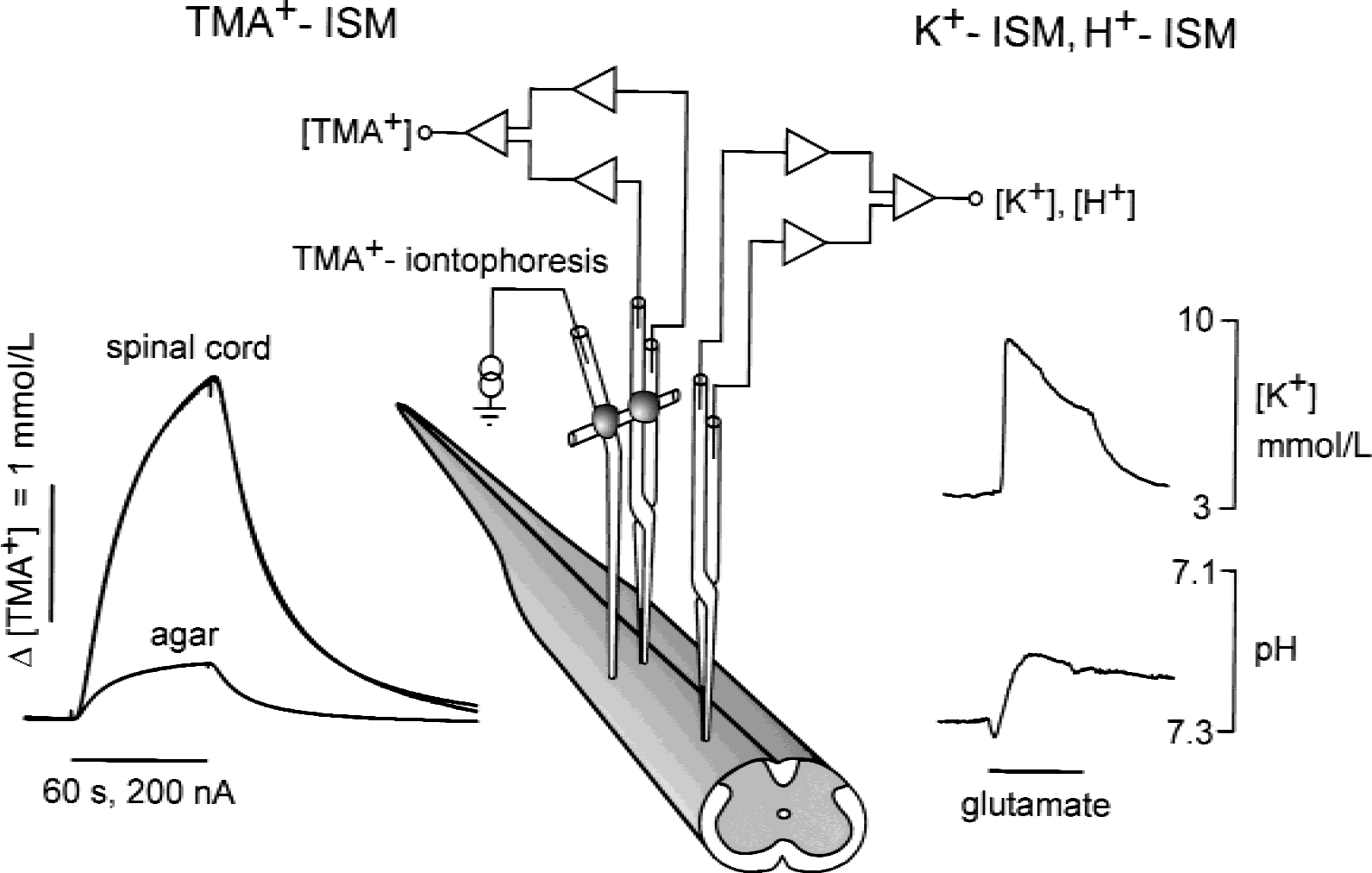
Experimental set-up and representative TMA+-diffusion curves, [K+]e, and pHe recordings obtained in the spinal cord. Note the large difference between the amplitudes of the TMA+-diffusion curves in spinal cord and diluted agar. In agar, where diffusion is free, the diffusion curves recorded by TMA+ selective microelectrodes (TMA+-ISM) are small, whereas in spinal cord the diffusion curves are larger, indicating the constrained diffusion in tissue caused by the smaller space available for diffusion and the presence of various barriers. Concomitantly, changes in either the extracellular concentration of K+ or pH could be recorded by the respective ISM. Examples of K+ increase and an alkaline-acid shift evoked by glutamate are shown.
TMA+ diffusion curves were first recorded in 0.3% agar gel (Special Noble Agar; St. Louis, MO, U.S.A.) dissolved in a solution of 150 mmol/L NaCl, 3 mmol/L KCl, and 1 mmol/L TMACl, in which by definition α = 1 = λ and k ′ = 0 (free-diffusion values). The obtained diffusion curves were analyzed by fitting the data using a nonlinear, curve-fitting simplex algorithm to yield the iontophoretic electrode transport number (n) and free diffusion coefficient for TMA+ (D). The electrode array then was lowered into an isolated spinal cord through a small opening in the dura and pia matter at the dorsal spinal surface to a depth of 250 to 350 μm, and diffusion curves were recorded in dorsal horn gray matter (Fig. 1). Knowing n and D, the ECS diffusion parameters α, λ, and k ′ were calculated from modified diffusion equations (Nicholson and Phillips, 1981; Nicholson and Syková, 1998).
Measurement of [K+]e and pHe
Extracellular K+ concentrations were recorded by means of double-barreled, K+-selective microelectrodes prepared similarly as TMA+-ISM (Fig. 1); however, the K+-sensitive barrel of the microelectrode was backfilled with 0.5 mol/L KCl (Syková, 1992a). The microelectrodes were calibrated in solutions containing 3, 4, 5, 6, 8, or 10 mmol/L KCl in 150 mmol/L NaCl. A similar procedure was used to prepare double-barreled, pH-sensitive microelectrodes (Chvátal et al., 1988; Syková, 1992a). The pH-sensitive channel contained liquid Hydrogen Ion Ionofore II-Cocktail A (Fluka, Prague, Czech Republic) and was backfilled with a buffer adjusted to pH 7.4. Electrode sensitivity was tested in solutions with a pH of 7.0, 7.2, 7.4, 7.6, 7.8 and 8.0, which contained as a background 150 mmol/L NaCl and 3 mmol/L KCl.
Immunohistochemistry and densitometry
Astrogliosis and morphologic changes were studied in 21 isolated spinal cords that were incubated in aCSF only, or in aCSF containing glutamate (10−3 mol/L, 10−2 mol/L), NMDA (5 × 10−5 mol/L, 10−4 mol/L), or AMPA (10−6 mol/L, 10−5 mol/L). Spinal cords were fixed with 4% paraformaldehyde in 0.1 mol/L phosphate-buffered saline (PBS; pH 7.5) for 4 hours, trimmed, then immersed in PBS containing 30% sucrose. Frozen transverse sections (40 μm) were cut through the lumbar part of the spinal cord. Selected sections were labeled with cresyl violet using Nissl staining. Astrocytes were identified using monoclonal antibodies to glial fibrillary acidic protein (GFAP; Boehringer-Mannheim, Mannheim, Germany), which were diluted to 0.4 μg/mL in PBS containing 1% bovine serum albumin (BSA, Sigma, St. Louis, MO, U.S.A.) and 0.2% Triton X-100. After overnight incubation in the primary antibodies at 4°C, the floating sections were washed and processed using biotinylated anti-mouse secondary antibodies and the peroxidase-labeled, avidin-biotin complex method (Vectastain Elite; Vector Laboratories, Burlingame, CA, U.S.A.). Immune complexes were visualized using 0.05% 3,3′-diaminobenzidine tetrachloride (Sigma) in 0.05 mol/L Tris buffer (pH 7.6) and 0.02% H2O2.
Transversal sections of spinal cord, stained for GFAP, were analyzed using an image analysis system (KS 400; Carl Zeiss Jena, Germany). Optical densities (OD) were measured in different regions of interest (ROIs) in the dorsal horns and in a reference region with low immunoreactivity (REF). Relative optical densities (ROD) were calculated as ROD = [(ODROI/ODREF) − 1] × 100. At least nine ROIs were analyzed in each measurement.
Statistical analysis of the difference between groups was performed using a one-way analysis of variance test. P < 0.05 was considered statistically significant. Data are expressed as mean ± SD.
RESULTS
Changes in pHe, [K+]e, and ECS diffusion parameters evoked by glutamate, NMDA, or AMPA
The control values of pHe, [K+]e, and the ECS diffusion parameters α, λ, and k ′ were first measured in dorsal horn gray matter at a depth of approximately 250 μm from the dorsal surface in isolated spinal cords superfused with aCSF (see mean values in Table 1). These values remained stable during superfusion of spinal cords with aCSF for 2 to 3 hours. Changes in pHe and [K+]e evoked by a 5-or 20-minute application of an agonist at variable concentrations were dependent on the concentration of glutamate, NMDA, or AMPA (Table 1). Glutamate at a concentration of 10−4 mol/L evoked a small increase in [K+]e of approximately 0.2 mmol/L, which was accompanied only by an alkaline shift whereas no acid shift was observed. Glutamate at a concentration of 10−3 mol/L or 10−2 mol/L evoked an initial alkaline shift that was accompanied by a [K+]e increase of approximately 4 or 12 mmol/L, respectively, and followed by an acid shift (Fig. 2, Table 1).
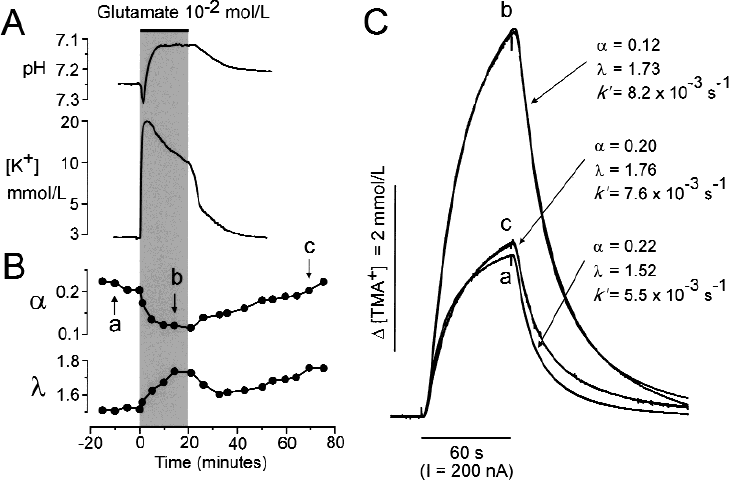
A typical effect of glutamate on pHe, [K+]e, and extracellular space diffusion parameters in the spinal cord of a 5-day-old rat.
The mean values of [K+]e′ pHe′ α γand k′ evoked by the application of glutamate, NMDA, or AMPA in the dorsal horn of the isolated rat spinal cord at postnatal days 4 to 12

The values of [K+]e tortuosity l, and nonspecific uptake k′ (×10−3 s−1) represent either the stable values in aCSF or the values after a 20-minute application of glutamate, NMDA, or AMPA (mean ± SD; n is the number of animals). Statistical significance was evaluated as the difference between controls (aCSF) and the values during application.
P < 0.05. aCSF, artificial cerebrospinal fluid; GLU, glutamate; NMDA, N-methyl-D-aspartate; AMPA, α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid.
NMDA or AMPA, starting at a concentration of 10−5 mol/L or 10−7 mol/L, respectively, also evoked an initial alkaline shift and a concomitant small increase in [K+]e (Table 1). The initial alkaline shift was followed by an acid shift. The application of NMDA at 5 × 10−5 mol/L or 10−4 mol/L or of AMPA at 10−6 mol/L or 10−5 mol/L evoked more pronounced changes in pHe and [K+]e (Figs. 3 and 4, Table 1). It is evident that the larger the concentration of glutamate receptor agonist, the greater the increase in [K+]e and the more pronounced the acid shift. At the highest agonist concentrations used, [K+]e reached 15 to 25 mmol/L.
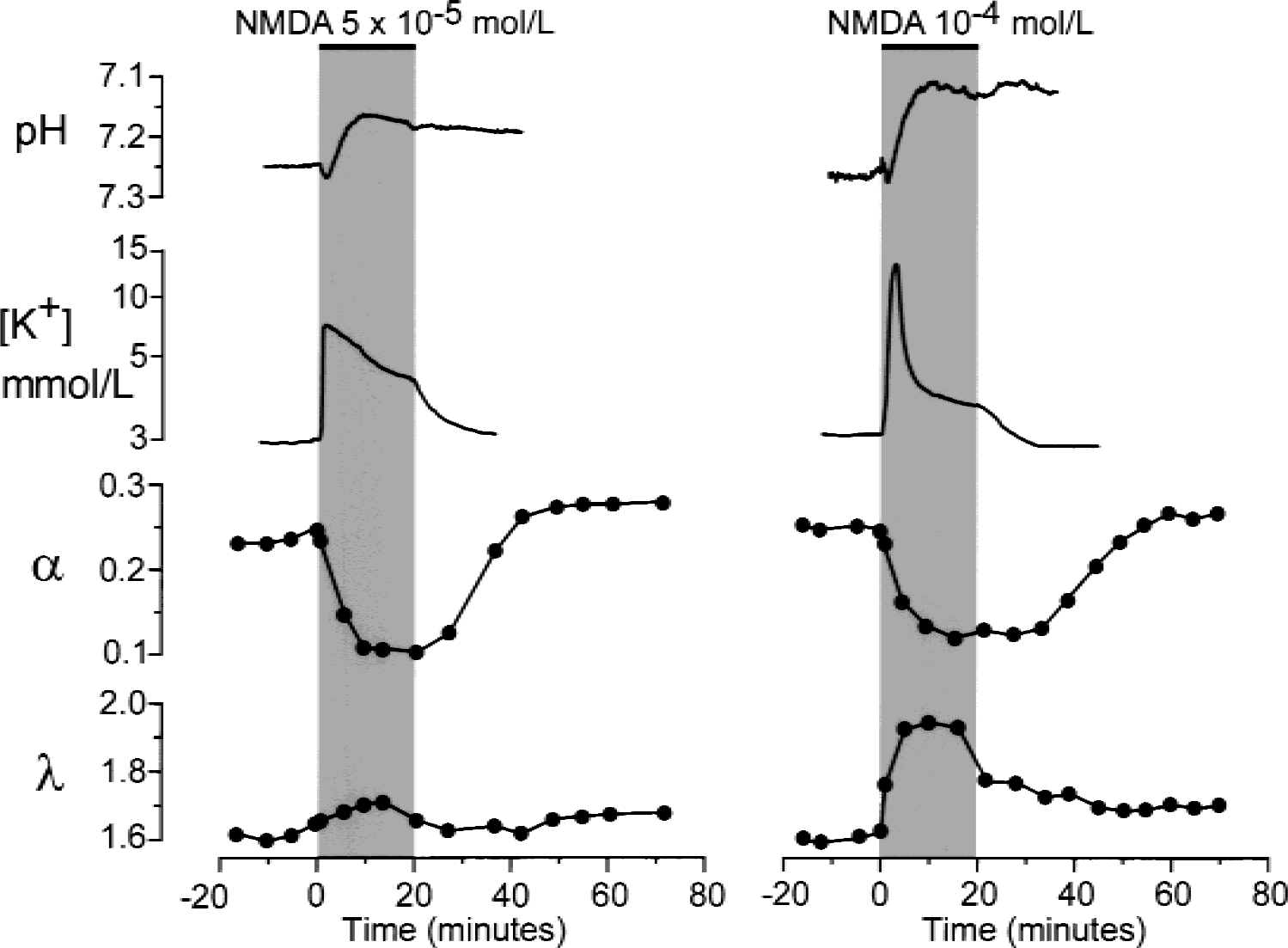
Typical changes in pHe, [K+]e, and extracellular space diffusion parameters evoked by N-methyl-d-aspartate (NMDA). NMDA at concentrations of 5 × 10−5 mol/L (left) or 10−4 mol/L (right) was applied for 20 minutes. Recordings and plots represent changes in [K+]e, pHe, volume fraction α, and tortuosity λ before, during, and after the application of NMDA. Data shown on the left were obtained in a 5-day-old rat; data shown on the right were obtained in a 10-day-old rat.
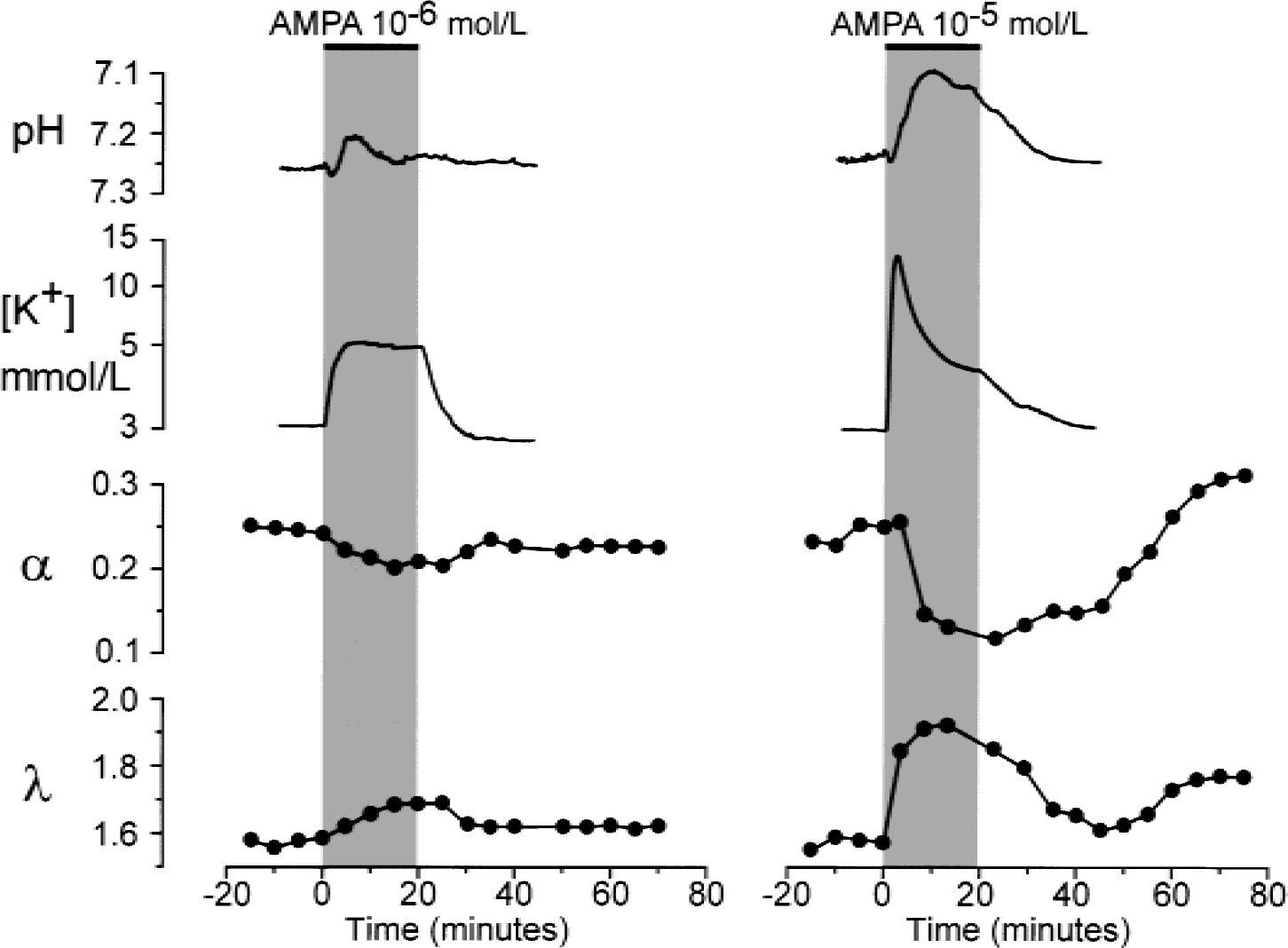
Typical changes in pHe, [K+]e, and extracellular space diffusion parameters evoked by AMPA. AMPA at concentrations of 10−6 mol/L (left) or 10−5 mol/L (right) was applied for 20 minutes. Recordings and plots represent changes in [K+]e, pHe, volume fraction α, and tortuosity λ before, during, and after the application of AMPA. Data shown on the left were obtained in a 5-day-old rat; data shown on the right were obtained in a 10-day-old rat.
The ECS diffusion parameters were measured in the same area of the isolated rat spinal cord as were [K+]e and pHe. Twenty to 30 minutes before the application of a glutamate receptor agonist, 4 to 6 diffusion curves were obtained in the isolated spinal cord superfused with standard aCSF (Fig. 2), and control ECS diffusion parameter values were obtained for α, λ, and k ′. These control values did not differ from those described previously in spinal cord in vivo (Svoboda and Syková, 1991; Šimonová et al., 1996). Glutamate at 10−4 mol/L had no effect on the ECS diffusion parameters, whereas at 10−3 mol/L or 10−2 mol/L it induced a decrease in α of approximately 20% or 60%, respectively, an increase in λ, and no significant change in k ′ (Fig. 2, Table 1).
The application of NMDA at 5 × 10−5 mol/L led to a decrease in α of approximately 50% and to an increase in λ (Fig. 3, Table 1). AMPA induced a decrease in α and an increase in λ starting at a concentration of 10−6 mol/L (Fig. 4). At these NMDA or AMPA concentrations, α and λ returned to control values during washout (Figs. 3 and 4). However, higher concentrations of NMDA or AMPA (10−4 mol/L or 10−5 mol/L, respectively) induced an even greater decrease in α (by approximately 60%) and an increase in λ, comparable with the values observed in 10−2 mol/L glutamate. One hour after washout of the agonist, α had “overshot” the control values, whereas λ either remained elevated or increased even further (Figs. 2, 3, and 4). These results suggest that some long-term or permanent diffusion barriers are formed by the release of glutamate, for example, due to astrogliosis (Fig. 8) (Roitbak and Syková, 1999; Syková et al., 1999).
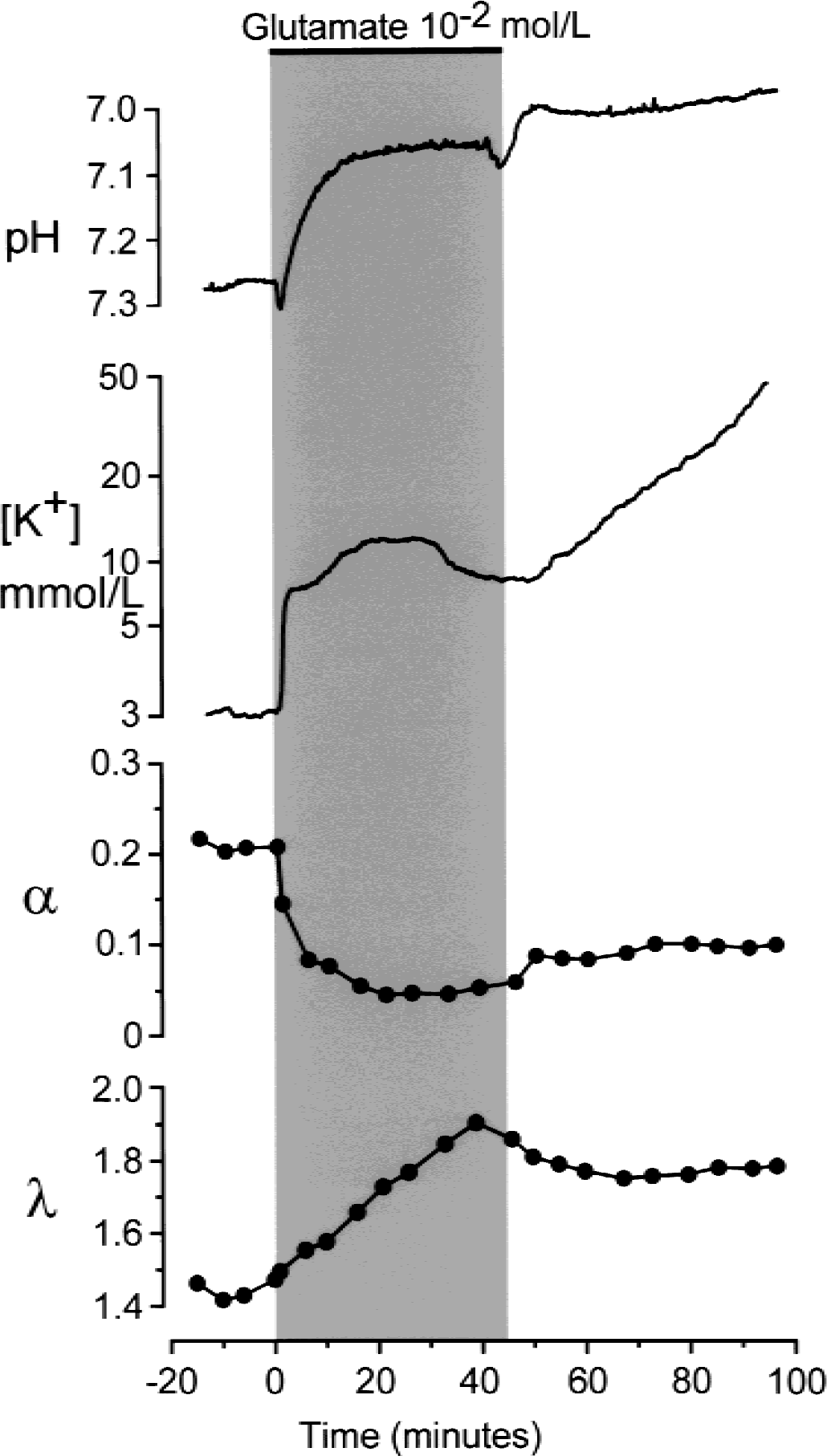
Effect of a 45-minute application of glutamate (10−2 mol/L) on changes in [K+]e, pHe, and extracellular space diffusion parameters. Long-lasting superfusion of spinal cord with a high concentration of glutamate evoked an increase in [K+]e up to 50 mmol/L, a small initial alkaline shift followed by a large and irreversible acid shift and permanent changes in α and λ indicating glutamate neurotoxicity and neuronal damage.
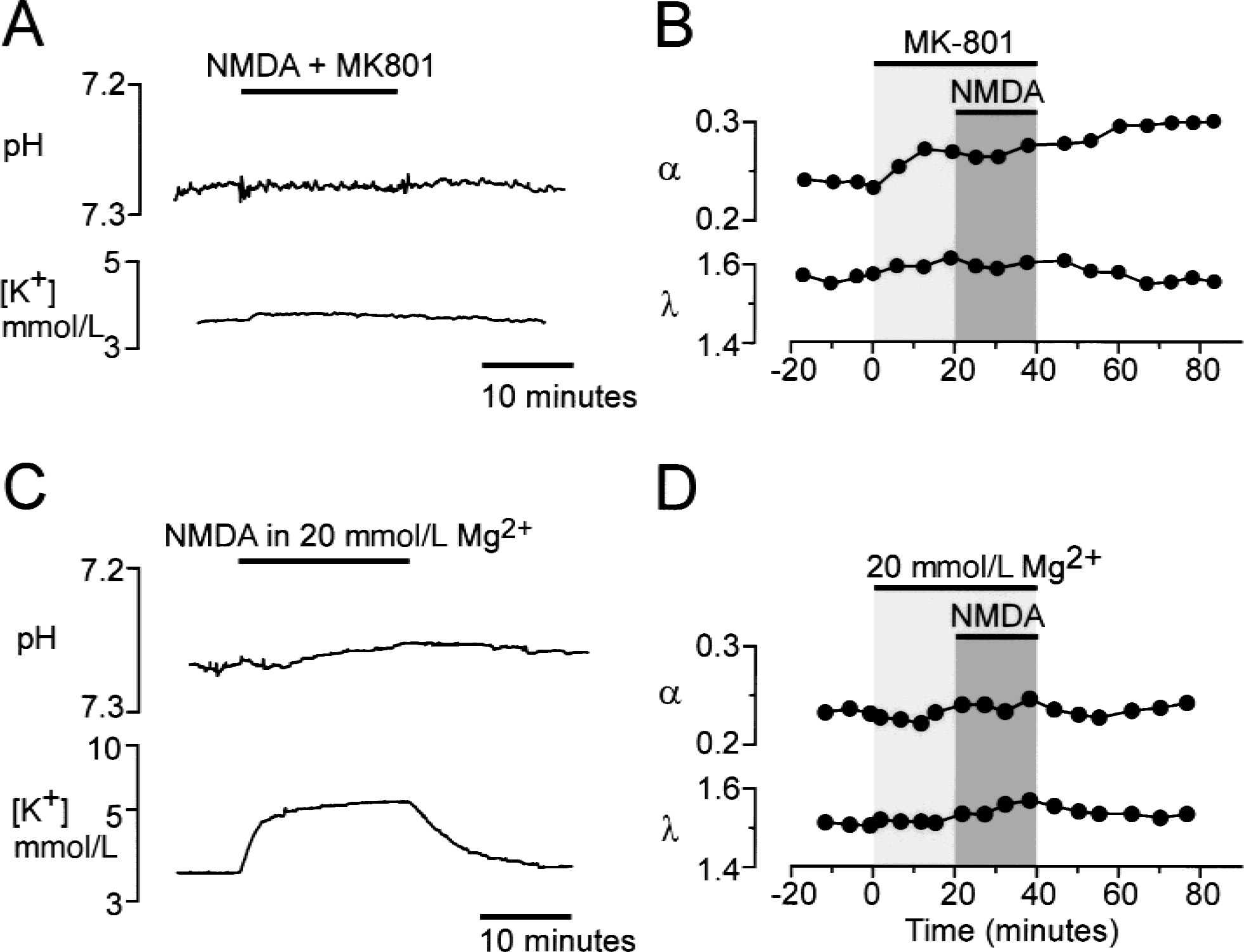
The effects of the competitive N-methyl-d-aspartate (NMDA) receptor antagonist MK-801 and 20 mmol/L Mg2+ on changes in pHe, [K+]e, and extracellular space (ECS) diffusion parameters evoked by the application of NMDA.
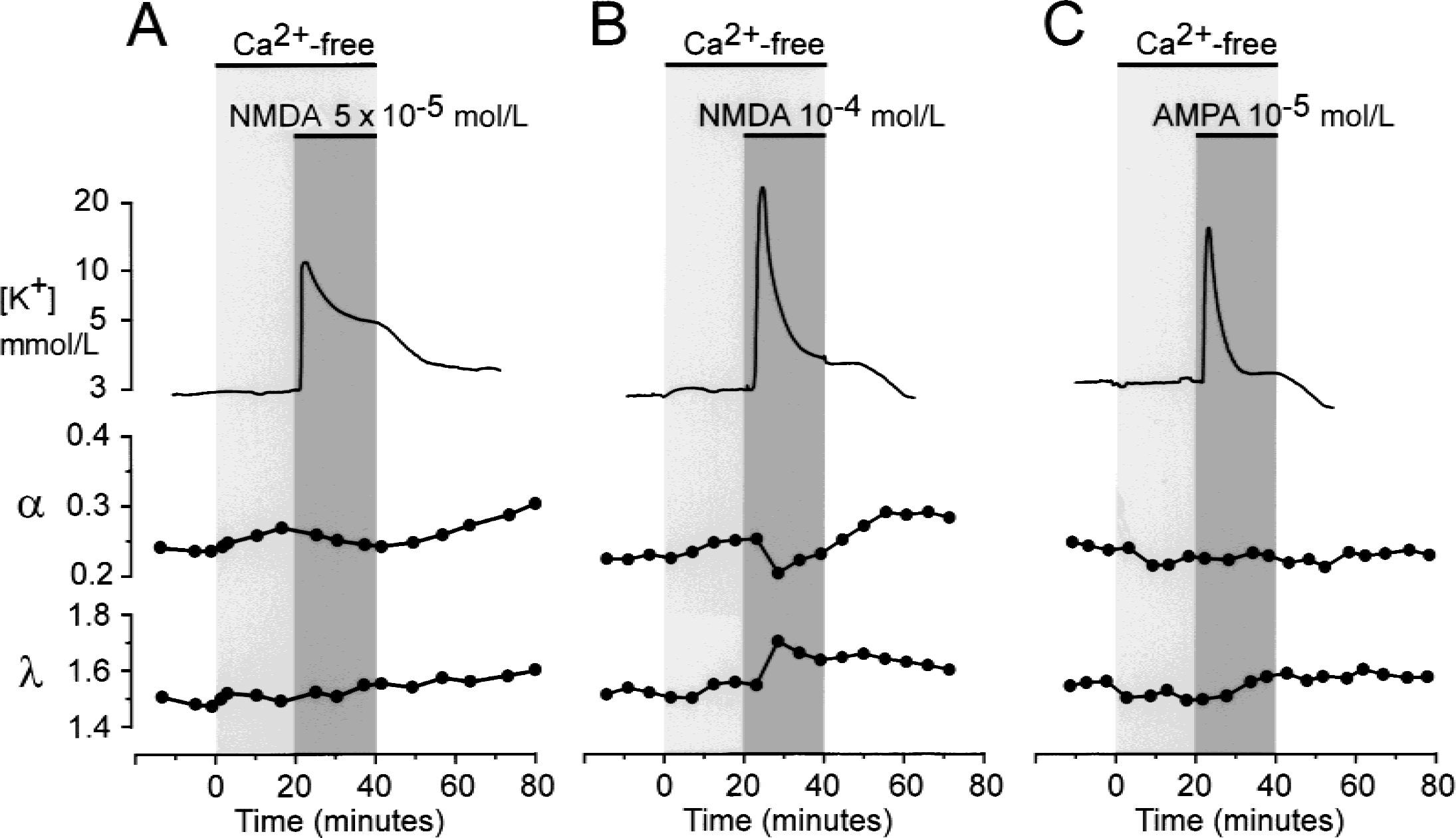
Effect of Ca2+-free solution on changes in [K+]e and extracellular diffusion parameters evoked by the application of N-methyl-d-aspartate (NMDA)
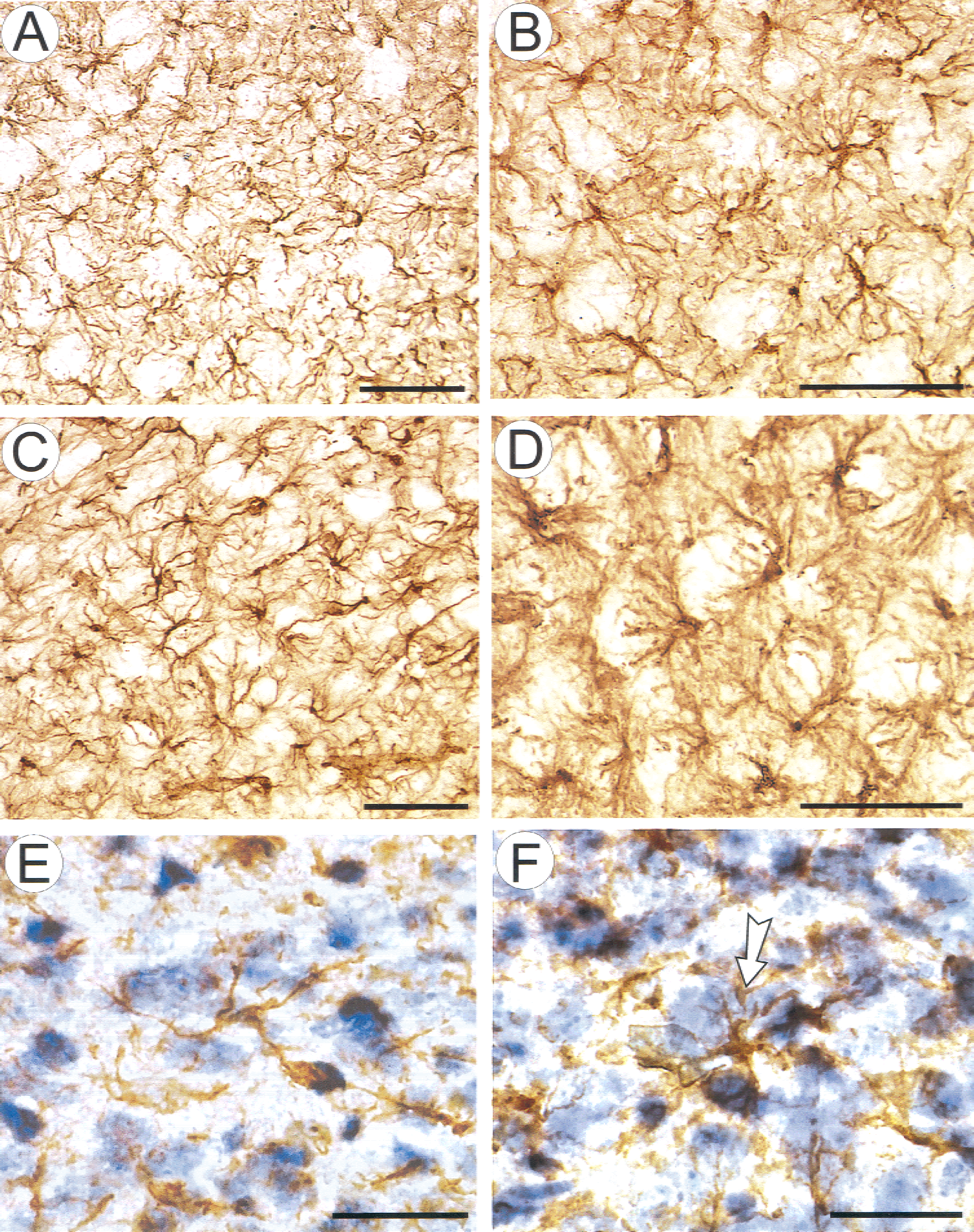
Glial fibrillary acidic protein (GFAP) and cresyl violet staining in the dorsal spinal horn of a 12-day-old rat.
In approximately 25% of the experiments (n = 3), the application of high concentrations of glutamate (10−2 mol/L), NMDA (10−4 mol/L), or AMPA (10−5 mol/L) for 45 minutes resulted in additional changes. In these experiments, after 40 to 45 minutes, [K+]e started to increase further, up to 50 to 70 mmol/L, the acid shift also increased further, α remained decreased, and λ increased (Fig. 5). This effect was irreversible even after 2 hours of perfusion with standard aCSF, suggesting a neurotoxic effect of the glutamate receptor agonist and neuronal damage.
Effect of glutamate receptor antagonists, Mg2+ and Ca2+-free solution
The authors studied whether blocking the ionic shifts evoked by glutamate receptor agonists also diminishes the observed changes in ECS diffusion parameters. The specific competitive antagonist of NMDA receptors, MK-801 (10−5 mol/L), and the AMPA/kainate receptor blocker, CNQX (10−5 mol/L), diminished the pHe shifts, [K+]e elevation, and changes in ECS diffusion parameters evoked by glutamate receptor agonists (10−5 and 5 × 10−5 mol/L NMDA or 10−6 mol/L AMPA) (Fig. 6 A to Fig. 6B). K+ and pH changes evoked by glutamate receptor agonists at higher concentrations (10−4 mol/L NMDA and 10−5 mol/L AMPA) were blocked by approximately 40%, whereas changes in α were blocked by approximately 60% (not shown in figure).
The application of an NMDA receptor channel blocker, Mg2+ (20 mmol/L), completely abolished the NMDA-induced alkaline-acid shift and the increase in [K+]e did not exceed 2 mmol/L (Fig. 6 C). NMDA in the presence of 20 mmol/L Mg2+ did not evoke significant changes in the ECS diffusion parameters (Fig. 6D).
The application of Ca2+-free solution (0 Ca2+, 1 mmol/L EGTA) 20 minutes before and during treatment with NMDA or AMPA (Fig. 7) had no significant effect on the NMDA or AMPA-evoked K+ increase or on the alkaline-acid shift. The application of Ca2+-free solution itself did not evoke changes in ECS diffusion parameters, but did completely block both the NMDA-and AMPA-induced decrease in α and the increase in λ (see Fig. 7A and 7 for the application of 5 × 10−5 mol/L NMDA or 10−5 mol/L AMPA). At a concentration of 10−4 mol/L, NMDA evoked only fast and small changes in α and λ, whereas the larger and sustained changes were blocked (Fig. 7B). These results show that sustained cell swelling and ECS shrinkage, which may result in neuronal degeneration, depend on extracellular Ca2+.
Astrogliosis evoked by glutamate, NMDA, or AMPA application results in increased ECS diffusion barriers
It was demonstrated in previous studies that the cell swelling induced by a 45-to 60-minute application of 50 mmol/L K+ or hypoosmotic solution is accompanied by astrogliosis, which results in changes in ECS tortuosity (Syková et al., 1999). Changes in astrocyte morphology and an increase in chondroitin sulphate proteoglycans related to astrogliosis evoked in the rat cortex by a stab wound also resulted in an increase in ECS tortuosity, whereas cell death was reflected by an increase in the ECS volume fraction (Roitbak and Syková, 1999).
In this study, after washing out the glutamate receptor agonists, the authors observed an increase in tortuosity even long after the cells shrank to their original volume (Figs. 2, 3, and 4). Therefore, the authors compared the observed changes in tortuosity evoked by glutamate and its receptor agonists, NMDA and AMPA, with changes in astrocyte morphology and GFAP expression. No distinct increase in GFAP staining was observed after incubation with smaller concentrations of glutamate, NMDA, or AMPA (10−3, 5 × 10−5, and 10−6 mol/L, respectively). Incubation of the spinal cord in 10−2 mol/L glutamate, 10−4 mol/L NMDA, or 10−5 mol/L AMPA for 20 minutes led to an increase in GFAP staining and to changes in glial cell morphology. Glial fibrillary acidic protein staining revealed astrocytes with thicker processes and more dense somata than in spinal cords incubated in normal aCSF (Fig. 8). The relative optical density (ROD) of GFAP staining was quantified using an image analysis system. In spinal cords incubated in aCSF, the mean ROD was 12.3% ± 2.3% (n = 12), whereas in the presence of glutamate receptor agonists, it was significantly greater (P ≤ 0.0005). In glutamate (10−2 mol/L) it increased up to 19.4 ± 2.7 (n = 11), in NMDA (10−4 mol/L) up to 20.6 ± 1.8 (n = 9), and in AMPA (10−5 mol/L) up to 17.9 ± 3.4 (n = 9).
Typically, GFAP-positive processes were found to form “paw”-like nets around large neurons, particularly in the ventral horns (Fig. 8D). Counterstaining with cresyl violet revealed cells, presumably swollen neurons, surrounded by hypertrophied glial processes (Fig. 8F).
The authors conclude that the increase in ECS tortuosity during and after exposure of the rat spinal cord to glutamate receptor agonists correlates with astrogliosis, characterized by the rearrangement and hypertrophy of astrocytic processes.
DISCUSSION
In the authors' experiments, the application of glutamate receptor agonists altered the [K+]e and pHe in the rat spinal cord (see also Jendelová et al., 1994), and the presence of an acid shift was dependent on the amplitude of the [K+]e increase. The application of low concentrations of glutamate or NMDA, which evoked an increase in [K+]e of approximately 2 mmol/L and resulted in only an alkaline shift, was not accompanied by changes in ECS diffusion parameters. Changes in ECS diffusion parameters were found during the application of glutamate, NMDA, or AMPA concentrations that evoked an increase in [K+]e of at least 4 mmol/L and were accompanied by acid shifts (Table 1). An activity-related increase in extracellular K+ concentration and alkaline-acid shifts in extracellular pH have been found to accompany neuronal activity and pathologic states in a variety of animals and brain regions in vivo as well as in situ (Chesler, 1990; Syková, 1992b, 1997). An important role of glial cells in extracellular K+ and pH homeostasis has been demonstrated repeatedly in brain as well as in isolated rat spinal cord, particularly in experiments with incomplete or impaired gliogenesis (Jendelová and Syková, 1991; Syková et al., 1992).
A number of studies have demonstrated that glutamate receptors are present not only in neurons, but also in glial cells (Hollmann and Heinemann, 1994; Gallo and Russell, 1995; Steinhäuser and Gallo, 1996; Žiak et al., 1998), and that glutamate and its agonists depolarize cells and lead to an increase in [K+]e and to intracellular acidification of both neurons (Endres et al., 1986; Hartley and Dubinsky, 1993) and glial cells (von Blankenfeld and Kettenmann, 1991). The pHe changes in vertebrate and invertebrate preparations induced by the application of glutamate and its agonists are composed of an initial alkaline shift followed by an acidification (Chesler and Rice, 1991; Rose and Deitmer, 1995) and are produced by the activation of various membrane transport mechanisms. It is widely accepted that the initial alkalinization results from neuronal activity and is caused either by direct HCO3− independent proton influx into cells through glutamate receptor-coupled cation channels (Chen and Chesler, 1992; Deitmer and Munsch, 1994) or by the activation of a cellular Ca2+/H+ ATPase subsequent to an increase in intracellular Ca2+ (Schwiening et al., 1993). However, the activity-related extracellular acid shifts are related to Na+/H+ and Na+/Cl−/HCO3−/H+ exchangers located in neurons and glia, and to Na+-HCO3− cotransport specific only for glial cells. Electrogenic Na+-HCO3− cotransport in glial cells is activated during the increase of [K+]e associated with increased neuronal activity and contributes substantially to the formation of an acid shift (Deitmer and Rose, 1996). In addition, the amplitude of the acid shift evoked by electrical stimulation is related to gliogenesis and increases during early postnatal development (Jendelová and Syková, 1991).
The authors' experiments show that glutamate receptor activation, in addition to ionic fluxes, results in cell swelling and a decrease in the ECS volume. It has been suggested by Dietzel et al. (1989) that the accumulation of [K+]e during neuronal activity and neuronal depolarization in response to the application of glutamate receptor agonists is accompanied by neuronal swelling, which may be caused by a NaCl influx into neurons or enhanced intracellular metabolic processes with a subsequent increase in intracellular osmolarity. There is increasing evidence that glutamate application also evokes swelling in astrocytes (Kimelberg, 1987; Chan et al., 1990; Hansson et al., 1994) and that the glutamate-induced swelling of cultured astrocytes is dependent on Cl− and Ca2+ influx (Koyama et al., 1991). In addition to the direct effect of glutamate, glial cells may swell in response to increased [K+]e, which accompanies the activation of glutamate receptors. A number of studies performed on cultured astrocytes have shown that the application of high potassium induces cell swelling (Kimelberg and Frangakis, 1985; Kimelberg and Ransom, 1986). The authors' previous study performed on isolated spinal cord also has shown that both diffusion parameters, ECS volume and tortuosity, change during potassium elevation to 20 to 50 mmol/L as well as during hypotonic stress and that these two variables change independently (Syková et al., 1999). However, changes in the ECS diffusion parameters comparable to those induced by glutamate above a concentration of 10−3 mol/L, NMDA 5 × 10−5 mol/L, or AMPA 10−5 mol/L, require the application of 30 to 50 mmol/L K+ (Syková et al., 1999). K+-induced swelling may result from a net KCl uptake through Na-K-2Cl cotransport and concomitant water influx into astrocytes (Syková et al., 1999). A similar mechanism of astrocytic swelling may also contribute to the changes in ECS volume observed during the application of glutamate receptor agonists, changes that are accompanied by an large increase in [K+]e. In this study, the authors show that cellular swelling in vivo, resulting in ECS volume shrinkage and in an increase of diffusion barriers, occurs at relatively low concentrations of glutamate receptor agonists that result in a relatively small increase in [K+]e. Therefore, it must be assumed that extracellular K+ accumulation cannot be the sole cause of glutamate-induced cell swelling.
The current results are in agreement with the glutamate-induced swelling of cultured astrocytes. This transient swelling apparently also occurs in vivo during physiologic states when the concentration of glutamate may reach relatively high extracellular levels around activated synapses, for example, during repetitive stimulation. The extracellular levels of glutamate reached during ischemia or CNS injury are much greater, that is, in the range used in this study (Benveniste et al., 1989; Xu et al., 1998; Liu et al., 1999). In the current study, glutamate, NMDA, or AMPA application in a Ca2+-free solution had no affect on extracellular K+ accumulation, but the sustained changes in ECS diffusion parameters were blocked. These results indicate that even when neuronal activity in Ca2+-free solution was not blocked, and therefore led to a rise in extracellular K+, glial cells were not able to swell without extracellular Ca2+. It is therefore reasonable to assume that the decrease of ECS volume fraction during the activation of glutamate receptors is predominantly mediated by extracellular Ca2+-dependent swelling of glia. The authors' experiments also show that changes in ECS diffusion parameters were related to the magnitude of the increase in [K+]e evoked by the application of glutamate receptor agonists, which in fact represented the level of neuronal excitation. A more substantial increase in λ and a large decrease in α were evoked by the application of glutamate, NMDA, or AMPA in concentrations that also resulted in an acid shift and a potassium increase greater than 10 mmol/L. In the authors' previous study, the application of 10 or 20 mmol/L K+ induced a minimal decrease in α, whereas λ clearly increased from values of approximately 1.6 to approximately 1.7 or 1.8, respectively (Syková et al., 1999).
After washing out the glutamate receptor agonists the authors found a decrease in α whereas λ remained increased. Therefore it can be speculated that glial cells efficiently regulate their volume; however, astrogliosis results in morphologic changes, for example, increased thickness of fine glial processes, which produce additional diffusion barriers (Syková et al., 1999; Roitbak and Syková, 1999). The current study also indicates that the cellular swelling evoked by low and high concentrations of glutamate receptor agonists has different effects on ECS diffusion parameters. Even though relatively low concentrations led to a transient decrease in ECS volume, only higher concentrations resulted in large and persistent changes in ECS tortuosity. Morphologic analysis showed that this increase in tortuosity was clearly related to astrogliosis (Fig. 8). The current results are in agreement with previous studies, in which the exposure of glial cells to high K+ resulted in increased immunoreactivity for GFAP and astrogliosis (Canady et al., 1990; Del Bigio et al., 1994; Del Cerro et al., 1996; Kraig et al., 1991; Syková et al., 1999). Moreover, the increase in tortuosity also may result from the production of ECM molecules by reactive glial cells (Roitbak and Syková, 1999).
The changes in ECS volume and tortuosity may or may not be fully reversible, depending on the concentration of glutamate in the ECS and on the duration of its action. The current study shows that the duration of cell swelling and ECS shrinkage might be an important factor in neurodegeneration. It was shown that Ca2+-free solution and glutamate receptor antagonists such as MK801 or CNQX reduce the neuronal death induced by hypoxia and ischemia, most likely by reducing excitotoxic injury (Kaku et al., 1993). In the authors' experiments, Ca2+-free solution, MK801 or CNQX, blocked the effect of NMDA and AMPA on ECS diffusion parameters, apparently by blocking cellular swelling. Such a blocking effect of glutamate receptor antagonists may therefore contribute to a decrease of glutamate neurotoxicity.
In more physiologic conditions, glutamate induced modest astrocytic swelling, which affects the size of the ECS volume and tortuosity, slows down diffusion in the ECS, and thus may affect neuronal activity and neuron-glia interaction (Syková, 1997; Zoli et al., 1999). The negative feedback mechanism decreasing neuronal excitability is amplified by cell swelling (Syková, 1997; Chvátal and Syková, 2000). Kullmann and Asztely (1998) have suggested a mechanism of “cross-talk” between distinct synapses caused by the “spillover” of glutamate in the synaptic cleft. Transmitter released from one synapse can lead to significant activation of high-affinity receptors at neighboring synapses. Such cross-talk may be affected by the diffusion parameters of the ECS around the synapses, that is, by the ECS volume and geometry. Because the diffusion properties of the ECS affect the diffusion of substances in nervous tissue (Nicholson and Syková, 1998; Zoli et al., 1999), not only astrogliosis but also more subtle changes in astrocyte morphology can produce transient or persistent changes in extrasynaptic signal transmission.
