Abstract
The two structurally related peptides, vasoactive intestinal polypeptide (VIP) and pituitary adenylate cyclase activating polypeptide (PACAP), are present in cerebral vascular nerve fibers. Biologic actions of VIP are exerted through two receptors, VPAC1 and VPAC2, having similar binding affinity for both VIP and PACAP. In the current study, the authors have developed a specific antibody against the rVPAC1 receptor to examine the localization of rVPAC1 immunoreactivity in cerebral arteries and arterioles of the rat by immunohistochemistry using fluorescence confocal microscopy. Specificity of the antiserum was ensured by immunoblotting and immunocytochemistry of cells transfected with cDNA encoding the different PACAP-VIP receptor subtypes. The rVPAC1 receptor immunoreactivity was localized to the plasmalemma of circularly orientated smooth muscle cells on superficial cerebral arteries and arterioles taken from the basal surface of the brain. By double immunostaining VIP immunoreactive nerve fibers and, to a lesser extent, those containing PACAP were shown to have intimate contact with the receptor protein. Vasoactive intestinal polypeptide and PACAP containing cerebrovascular nerve fibers were found in separate nerve populations with different distribution pattern and density. In brain sections processes of cortical VIP-, but not PACAP-, containing neurons seemed to innervate the rVPAC1 receptor of pial arterioles on the brain surface. The current findings provide the neuroanatomical substrate for a role of VIP and maybe PACAP in the regulation of cerebral blood flow.
Vasoactive intestinal polypeptide (VIP) is a 28 amino acid pleiotropic peptide with a widespread occurrence in neurons of the central and peripheral nervous system (Fahrenkrug, 1993). A rich supply of nerve fibers containing VIP has been described around cerebral blood vessels in a number of species including mouse, gerbil, rat, cat, dog, pig, cow, monkey, and human (Edvinsson et al., 1980; Edvinsson and McCulloch, 1985; Gibbins et al., 1984; Hara et al., 1985,1989; Larsson et al., 1976; Lee et al., 1984; Matsuyama et al., 1983; Miao and Lee, 1990; Suzuki et al., 1988; Uemura et al., 1988; Zhang et al., 1991). In rat and dog, the nerve cell bodies giving rise to the VIP immunoreactive fibers of the large cerebral arteries are contained in the ipsilateral sphenopalatine and otic ganglia and in the internal carotic miniganglion (Hara et al., 1989; Suzuki et al., 1988; Uemura et al., 1988).
Pituitary adenylate cyclase activating polypeptide (PACAP), recently identified as a member of the VIP family of neuropeptides, exists in two biologically active forms, PACAP-38 and PACAP-27, both derived from the same precursor. Like VIP, PACAP is widely expressed in the nervous system having a broad spectrum of biologic functions including neurotransmitter and neuroprotective actions (Arimura, 1998). Nerve fibers containing PACAP have been demonstrated in the adventitia and adventitia media border of cat cerebral arteries (Uddman et al., 1993), and PACAP and VIP can dilate cerebral blood vessels suggesting a role in the regulation of cerebral circulation (Dacey et al., 1988; Edvinsson and McCulloch, 1985; Jansen-Olesen et al., 1994; Larsson et al., 1976; McCulloch and Edvinsson, 1980; Seki et al., 1995; Uddman et al., 1993; Wei et al., 1980). Because PACAP-27, PACAP-38, and VIP result in a similar concentration-dependent vasorelaxation, and a VIP receptor antagonist was able to inhibit the VIP and PACAP-38 provoked increase in vertebral artery blood flow, it is possible that the peptides are working through a common receptor (Anzai et al., 1995; Seki et al., 1995). Vasoactive intestinal polypeptide and the two alternative processing forms of PACAP are recognized by 3 G-protein coupled receptors with a large extracellular N-terminal domain and 7 transmembrane helices: the PACAP (or PAC1) receptor that has a much higher affinity for the 2 forms of PACAP than for VIP, and the VIP1 (or VPAC1) and VIP2 or (VPAC2) receptors that have a similar binding affinity for VIP and PACAP (Harmar et al., 1998).
In the current study, the authors have developed an antiserum against the N-terminus of the rVPAC1 receptor and used it for studying rat cerebral blood vessels by immunohistochemistry. The localization of the rVPAC1 receptor immunoreactivity and the relationship between the receptor protein and VIP or PACAP immunoreactive nerve fibers are described.
MATERIALS AND METHODS
rVPAC1 receptor antibody preparation
Polyclonal antibodies were generated against the rVPAC1 receptor N-terminal fusion protein in random bred white Danish rabbits. Each animal received 1 mg of the receptor fusion protein per immunization emulsified in double volume Freunds adjuvans.
The 363 bp SacII/BbsI fragment of the rVPAC1 receptor complementary deoxyribonucleic acid (cDNA) containing the N-terminal was subcloned as a blunt end fragment in the BamHI site of pGEX2T (Amersham Pharmacia Biotech, Allerød, Denmark). The fusion protein product, with glutathione S-transferase followed by the rVPAC1 receptor N-terminal, was termed VNG270. Fusion protein production in UT5600 E. coli cells, grown in Luria-Bertani broth (10 g bacto-tryptone/L, 5 g bacto-yeast extract/L, and 5 g sodium chloride/L), was initiated by 0.5 mmol/L Isopropyl beta D-thiogalactopyranoside and incubated at 15°C overnight. Cells (7 g) were pelleted at 8,000 g for 10 minutes and resuspended in 320 mL 40 mmol/L 2-amino-2-(hydroxymethyl)-1,3-propandiol (Tris), 20 mmol/L hydrochloric acid (HCl), 10 mmol/L dithiothreitol (DTT), 1 μmol/L leupeptin, 1 mmol/L phenylmethylsulfonylflorid and sonicated (150 W) for 10 minutes in an ice bath. The homogenate was centrifuged at 30,000 g for 10 minutes at 4°C. Sodium phosphate was added to the supernatant to a final concentration of 50 mmol/L and pH 7.0 followed by the addition of 2 mL glutathione Sepharose (Amersham) and incubated for 2 hours at 4°C. The glutathione Sepharose was pelleted by centrifugation for 10 minutes at 500 g at 4°C. The pellet was washed with 3 × 40 mL 20 mmol/L 2-(bis(2-hydroxyethyl)imino)-2- (hydroxymethyl)-1,3-propanediol (Bis-Tris), 10 mmol/L HCl, 10 mmol/L DTT, and 4 mL 50% glycerol was added to the pellet and stored at −20°C. Glutathione Sepharose, with VNG270 attached, was washed twice with 20 mmol/L Bis-Tris, 10 mmol/L HCl, 10 mmol/L DTT, and VNG270 was detached from the Sepharose by incubation with 50 mmol/L Tris, 15 mmol/L acetic acid, 10 mmol/L glutathione, 5 mmol/L DTT for 30 minutes at room temperature. The suspension was centrifuged for 2 minutes at 500 g at 20°C and iodoacetamide was added to the supernatant to a final concentration of 30 mmol/L and incubated for 30 minutes at room temperature.
Antiserum, code No. 4G28, was selected, and the antibody specificity was determined by using Chinese hamster ovary cells (CHO) stably transfected with rVPAC1 and human embryonic kidney cells 293 (HEK 293) transiently transfected with rPAC1 and rVPAC2 receptor.
Purification of rVPAC1 receptor N-terminal
Sodium dodecyl sulfate (SDS) with a final concentration of 0.01% was added to the purified product, VNG270, used for immunization and incubated overnight at room temperature with 0.1 g/L thrombin. Acetonitrile was added to a final concentration of 32% and pH was adjusted to pH 2 with trifluoroacetic acid, and the digest was separated at 50°C by C-4 reverse phase HPLC on a 4.6 × 250 mm column using a linear gradient from 32% to 45% acetonitrile in 0.1% trifluoroacetic acid at 1 mL/min for 60 minutes. Sodium dodecyl sulfate polyacrylamide gel electrophoresis was used to identify the peak containing the rVPAC1 receptor N-terminal, and amino acid analysis verified the expected content of amino acids.
Immunoblot analysis
Confluent monolayers of transfected cells were washed with phosphate buffered saline (PBS) and detached from their plastic support using a cell scraper. Cells were solubilized in 20 mL 25 mmol/L N-2-hydroxyethylpiperazine-N′-2-ethane-sulfonic acid (HEPES), 2.5 mmol/L CaCl2, 1.0 mmol/L MgCl2, 50 mg/L bacitracin, pH 7.4 per plate and disrupted using a polytron for 30 seconds. The homogenate was spun for 20 minutes at 30,000 g at 4°C. The resulting pellet was resuspended in 15 mL 25 mmol/L HEPES, 2.5 mmol/L CaCl2, 1.0 mmol/L MgCl2, 50 mg/L bacitracin, pH 7.4 per plate. The preparation was aliquoted and stored at −80°C.
Membrane protein samples were electrophoresed on 10% Tricine SDS-polyacrylamide gels according to the methods of Schägger and von Jagow (1987). After electrophoresis the proteins were transferred to polyvinylidendifluorid (PVDF) membranes (NOVEX, San Diego, CA, U.S.A.). The PVDF membranes were incubated in Tris buffer saline (TBS; 20 mmol/L TrisCl, 140 mmol/L NaCl, pH 7.4) containing 10% Tween-20 overnight at room temperature followed by an incubation with 0.01 mg/mL avidin and the antireceptor antiserum diluted to 1:2,000 in TBS containing 0.1% Tween-20 (TBS-T). After two quick washes with TBS the washing was extended with the addition of 0.01 mg/mL biotin for 3 × 10 minutes. Hereafter the PVDF membranes were incubated with biotinylated donkey anti-rabbit IgG (Jackson ImmunoResearch Laboratories, West Grove, PA, U.S.A.) diluted to 1:20,000 in TBS-T for 30 minutes at room temperature. After two quick and three 10-minute washes with TBS-T, the PVDF membranes were incubated with StreptABComplex/HRP (Dako A/S, Copenhagen, Denmark) diluted to 1:40 in TBS-T for 30 minutes at room temperature. After two quick and three 10-minute washes with TBS-T, the PVDF membranes were incubated with enhanced chemiluminescence reagents according to the manufacturer's instructions (Amersham) and apposed to Hyperfilm (Amersham) and developed for 5 seconds to 20 minutes.
Preabsorption of rVPAC1 receptor antiserum
Glutathione Sepharose with VNG270 attached, 0.5 mL, was applied on a poly-prep chromatography column, 0.8 × 4 cm (Bio-Rad Laboratories, Hercules, U.S.A.), and washed with 3 × 5 mL 20 mmol/L sodium phosphate buffer, 1 mmol/L DTT, pH 7.0. One mL 20 mmol/L sodium phosphate buffer, 10 mmol/L iodoacetamide, pH 7.0 were applied on the column and washed with 5 mL 20 mmol/L sodium phosphate buffer, pH 7.0. One mL 4G28 antiserum was applied on the column and the run through was used as preabsorbed antiserum.
Immunocytochemical characterization of the rVPAC1 receptor antiserum
The transfected cells were grown in eight-chamber glass slides washed with PBS and fixed for 1 hour in 2% paraformaldehyde, 0.2% picric acid in 0.1 mol/L sodium phosphate buffer, pH 7.2. After washing and drying the cells were immunostained with anti-rVPAC1 receptor antiserum (code No. 4G28) in a dilution of 1:640 using a biotinylated donkey anti-rabbit secondary antiserum (711-065-152 Jackson ImmunoResearch, diluted 1:1,000) and the avidin-biotin complex/streptavidin horseradish peroxidase complex as previously described (Fahrenkrug and Hannibal, 1998).
Animal preparation
Male Wistar rats weighing 180 to 200 g and housed under standard laboratory conditions in a 12-hour light—dark cycle with free access to food and water were used. On the day of fixation the animals were anesthetized with tribromoethanol (20 mg/100 g, BW) and perfused through the heart with a room temperature solution of saline 0.9% to which heparin (15,000, IU/L) was added 75 to 100 mL over 3 minutes. The perfusion was followed by 2% paraformaldehyde, 0.2% picric acid in 0.1 mol/L sodium phosphate buffer, pH 7.2, 300 mL over 15 minutes. In 10 rats, brains were rapidly removed from the skull and the major extracerebral arteries on the base of the skull were removed by careful dissection under a stereomicroscope. The vessels were postfixed in the same fixative for 4 hours at 4°C, and subsequently the vessel preparations were washed in PBS overnight. Fixed brains from 6 additional rats were postfixed for 24 hours at 4°C and subsequently equilibrated in PBS 0.05 mol/L, pH 7.4 containing 30% sucrose for 48 hours and 4°C and then sectioned coronally at 40 μm in a cryostat. The resulting sections were collected as free floating sections in PBS.
Immunohistochemistry
A previously characterized mouse monoclonal PACAP antibody (Code No. MabJHH1, diluted 1:5) directed against an epitope of the amino acid sequence 6–16 of PACAP recognizing both PACAP-38 and PACAP-27 was used for PACAP immunostaining (Hannibal et al., 1995). The monoclonal mouse anti-VIP antibody (Code No. 55, diluted 1:500) was kindly donated by John H. Walsh (Wong et al., 1996). The rabbit anti-VIP antiserum (Code No. 291E, diluted 1:1,000) has been characterized previously (Fahrenkrug et al., 1995). As controls, sections were routinely incubated with antibodies preabsorbed with the respective antigen (20 μg/mL) that abolished all staining. The rVPAC1 receptor antibody was used in a dilution of 1:500. Whole mounts of cerebral arteries and arterioles and free floating brain sections were processed for single or double antigen fluorescence immunohistochemistry as previously described (Fahrenkrug and Hannibal, 1998). Briefly, the whole mounts or sections were incubated with combinations of primary antibodies for 12 to 18 hours at 4°C. After washing for 3 × 10 minutes in 0.25% bovine serum albumin + 0.1% Triton X-100 (PBS-BT) the whole mounts or sections were incubated with a combination of Cy2 conjugated donkey anti-rabbit (diluted 1:100 for visualization of VIP and rVPAC1 receptor), or Texas Red® conjugated goat anti-mouse antisera (diluted 1:200 for visualization of VIP), or both, and biotinylated donkey anti-mouse antiserum (diluted 1:600 for visualization of PACAP). All secondary antisera were from Jackson Immunoresearch Laboratories. After washing for 3 × 5 minutes in PBS-BT and incubation for 30 minutes at room temperature in ABC-Streptavidin horse radish peroxidase complex (diluted 1:150; Vector, Burlingame, CA, U.S.A.), the sections or whole mounts were incubated in biotinylated tyramide (diluted 1:50 in application buffer; Tyramide System Amplification, DuPont NEN, Boston, MA, U.S.A.). After washing for 3 × 15 minutes in PBS-BT, the material was incubated for 30 minutes in streptavidin Texas Red conjugated complex (diluted 1:200; Amersham) followed by 3 × 10 minutes washes in PBS-BT and mounting in glycerol/PBS 1:1). For absorption control the anti-rVPAC1 receptor antiserum was incubated with 2 × 10−5 mol/L N-terminal receptor fusion protein for 24 hours at room temperature before immunohistochemistry. In addition, control experiments with omission of anti-rVPAC1 receptor antibody from the first incubation did not detect any staining imputable to the second antibody.
Whole mounts and brain sections were examined in a Leitz DMRB fluorescence microscope equipped with standard filter settings (13, code No. 513 808, excitation BP 450–490 nm, and N2.1, code No. 513 803, excitation PB 515–560 nm) and a red—green filter (code No. 513 803) allowing simultaneous visualization of red and green fluorescence. Photomicrographs were obtained using a Leica DC 200 camera and software (Leica, Herlev, Denmark), or a confocal laser scanning Zeiss LSM 510 microscope, or both. Sections obtained by the confocal microscope were 1.5 to 5.5-μm thick. Image editing software (Adobe Photoshop and Adobe Illustrator, Adobe, San José, CA, U.S.A.) was used to combine the obtained images into plates, and figures were printed on a Tektronix (Wilsonville, OR, U.S.A.) Phase 450 dye sublimation printer.
RESULTS
Immunoblot analysis
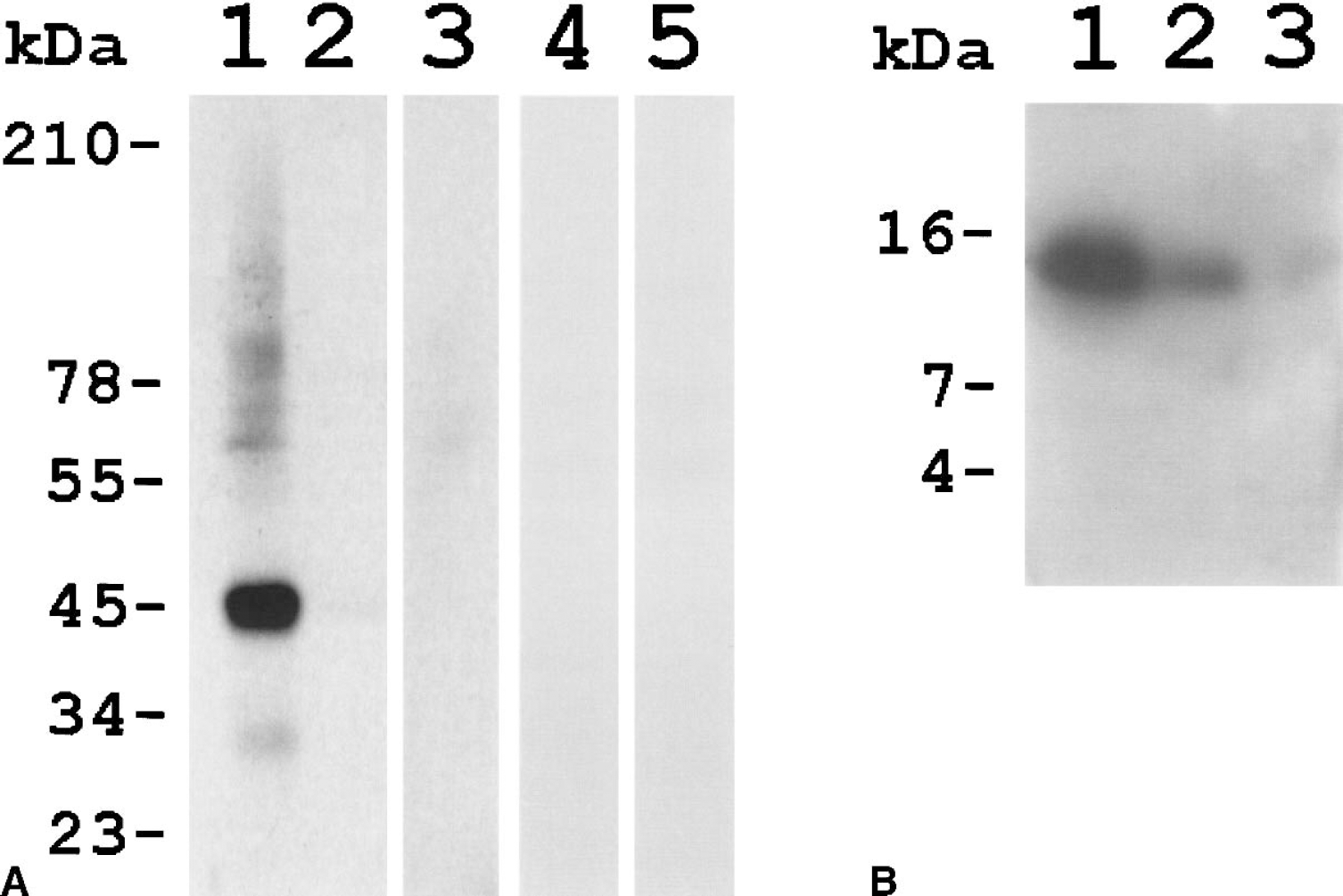
The antiserum 4G28 reacted strongly with a band of 45 kDa in CHO cells stably transfected with a rVPAC1 receptor expression plasmid, whereas the immunoreactivity was completely abolished after preabsorption of the antiserum with the rVPAC1 receptor N-terminal fusion protein (Fig. 1A). The antiserum did not react with the parental CHO cells that do not express the rVPAC1 receptor nor with membrane preparations from cells transfected with the rVPAC2 or rPAC1 receptor subtypes (Fig. 1A). The antiserum detected the purified rVPAC1 receptor N-terminal expressed in E. coli as a band of 14 kDa, and it was estimated that the antiserum was able to detect the protein above 3 pmol (Fig. 1B).
Visualization of the rVPAC1 receptor in transfected cells
The CHO cells stably transfected with the cDNA encoding the rVPAC1 receptor were immunostained with the 4G28 antiserum (Fig. 2A), whereas no immunoreaction was detected in nontransfected CHO cells (Fig. 2B) or cells transfected with the cDNA encoding the other receptor subtypes (data not shown).
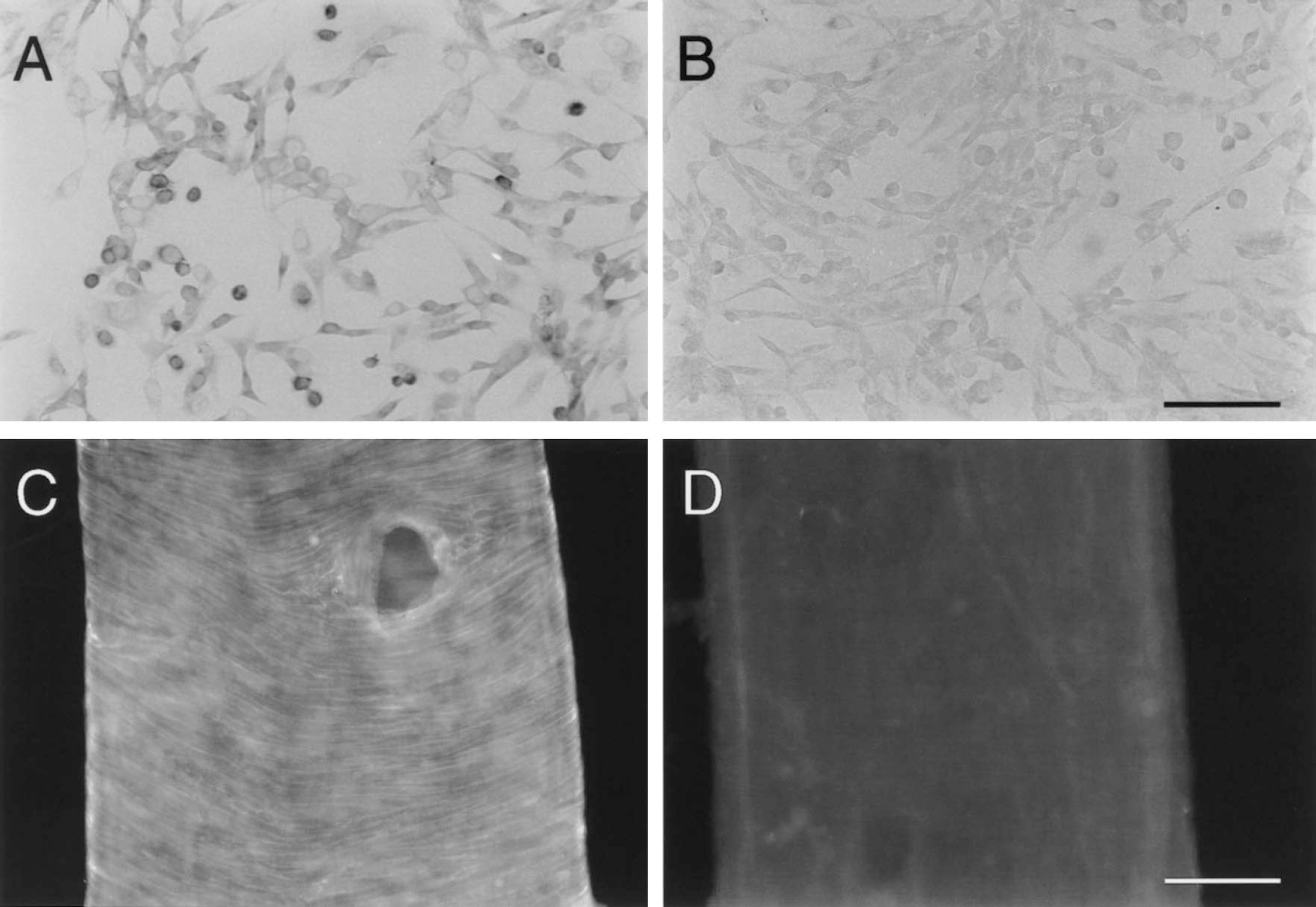
Immunocytochemical staining of the rVPAC1 receptor in Chinese hamster ovary (CHO) cells using antiserum 4G28. Specific staining is seen in the cells stably transfected with cDNA encoding the rVPAC1 receptor
Visualization of the rVPAC1 receptor in cerebral arteries and arterioles
Whole mount preparation of the basal cerebral arteries showed a distinct pattern of immunostaining that was absent after preabsorption of the rVPAC1 receptor antibody with the N-terminal receptor protein (Fig. 2C and 2D). The rVPAC1 receptor immunoreactivity was observed in smooth muscle cells both in superficial cerebral arteries and in arterioles taken from the basal surface of the brain. The strongly immunostained circularly orientated smooth muscles cells were seen in the basilar artery (Fig. 3A and 3B), the arteries of the circle of Willis, the middle cerebral artery, and the anterior and posterior cerebral arteries and their branches (Figs. 3G, 4A, and 4C). In the confocal microscope the rVPAC1 receptor immunoreactivity was exclusively detected on the plasmalemma of the vascular smooth muscle cells (Fig. 3A, 3B, and 3E) and no immunostaining of the endothelial cell layer was observed.
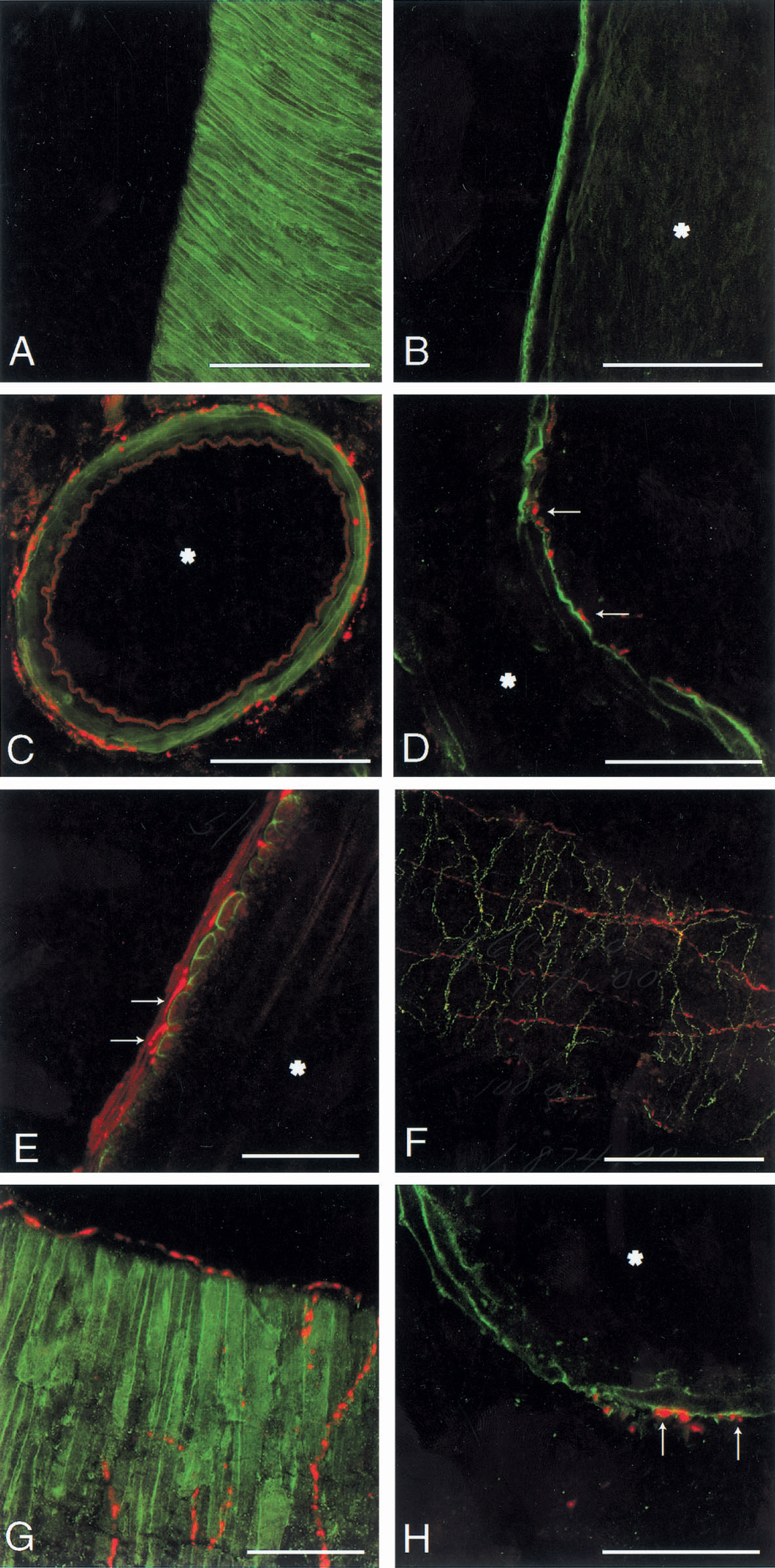
Immunofluorescence photomicrographs of arteries and arterioles isolated from the basal surface of rat brain. rVPAC1 staining along the surface of circularly orientated smooth muscle cells in a large
Examination of free floating brain sections confirmed the strong VIP receptor immunoreactivity found in isolated arteries and arterioles (Fig. 3C, 3D, and 3H). Based on visualization the intensity of immunostaining seemed to be similar in all arteries and arterioles.
Double immunolabeling
A well-developed plexus of perivascular VIP containing nerve fibers was observed in the wall of all major cerebral arteries and their branches (Figs. 3E, 3F, and 4E). Pituitary adenylate cyclase activating polypeptide immunoreactive nerve fibers were much less abundant, and the density of PACAP positive nerve fibers decreased as the diameter of the arterioles became smaller (Figs. 3F, 3G, 4B, and 4D). Although VIP formed a dense network of varicose nerve fibers mainly circularly orientated, the PACAP containing nerve fibers ran primarily along the long axis of the vessels. By double immunostaining, the two peptides were generally found not to be colocalized in the same nerve fibers (Fig. 3F). In free floating sections delicate varicose PACAP or VIP containing nerve fibers were encountered in the adventitia media border of the arteries (Fig. 3C). Confocal microscopical analysis at high magnification of free floating sections and whole mount arterial preparations after double immunostaining with the receptor antibody disclosed that PACAP and VIP immunoreactive nerve fibers had intimate contact with the receptor protein on the surface of the vascular smooth muscle cells (Fig. 3D, 3E, and 3H).
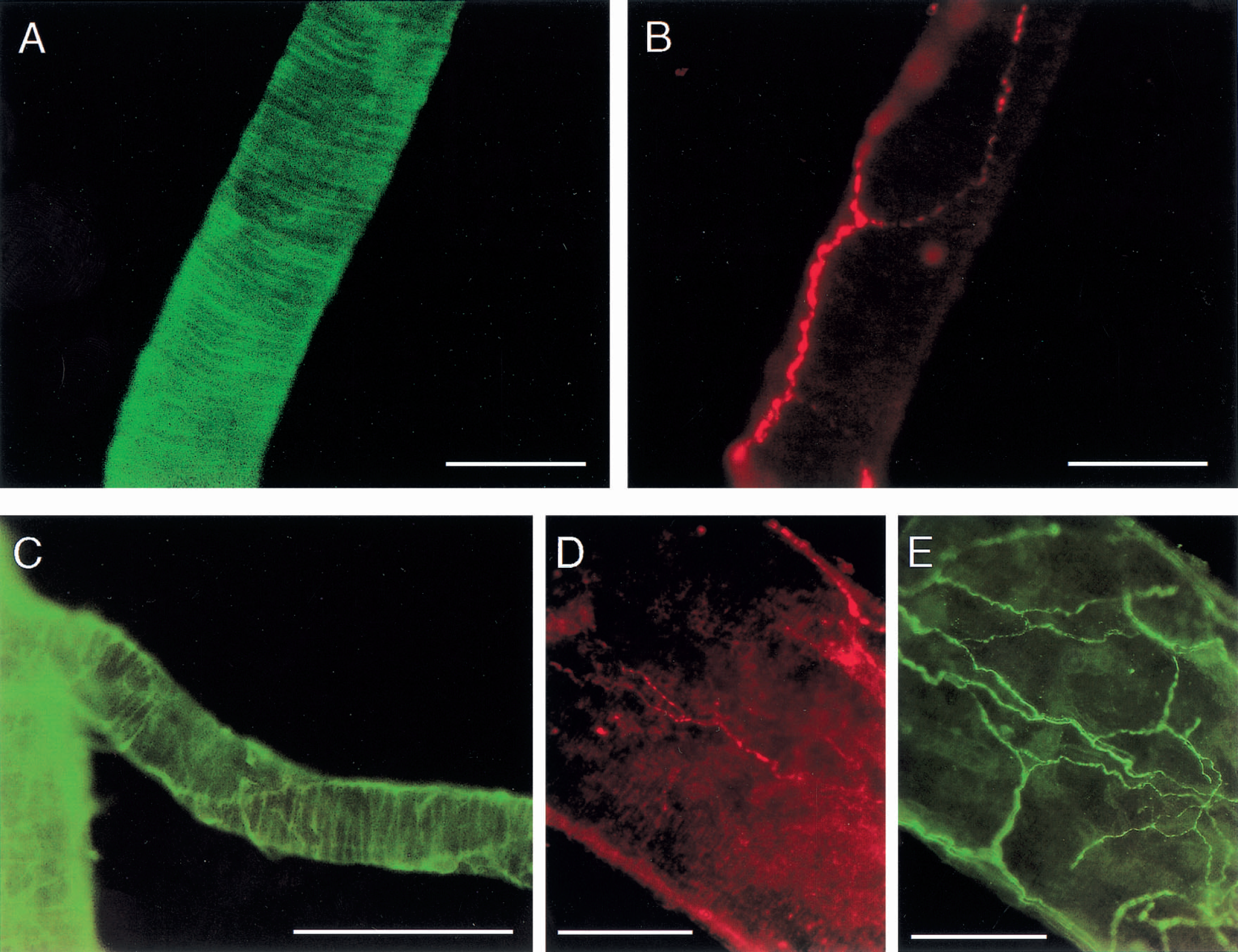
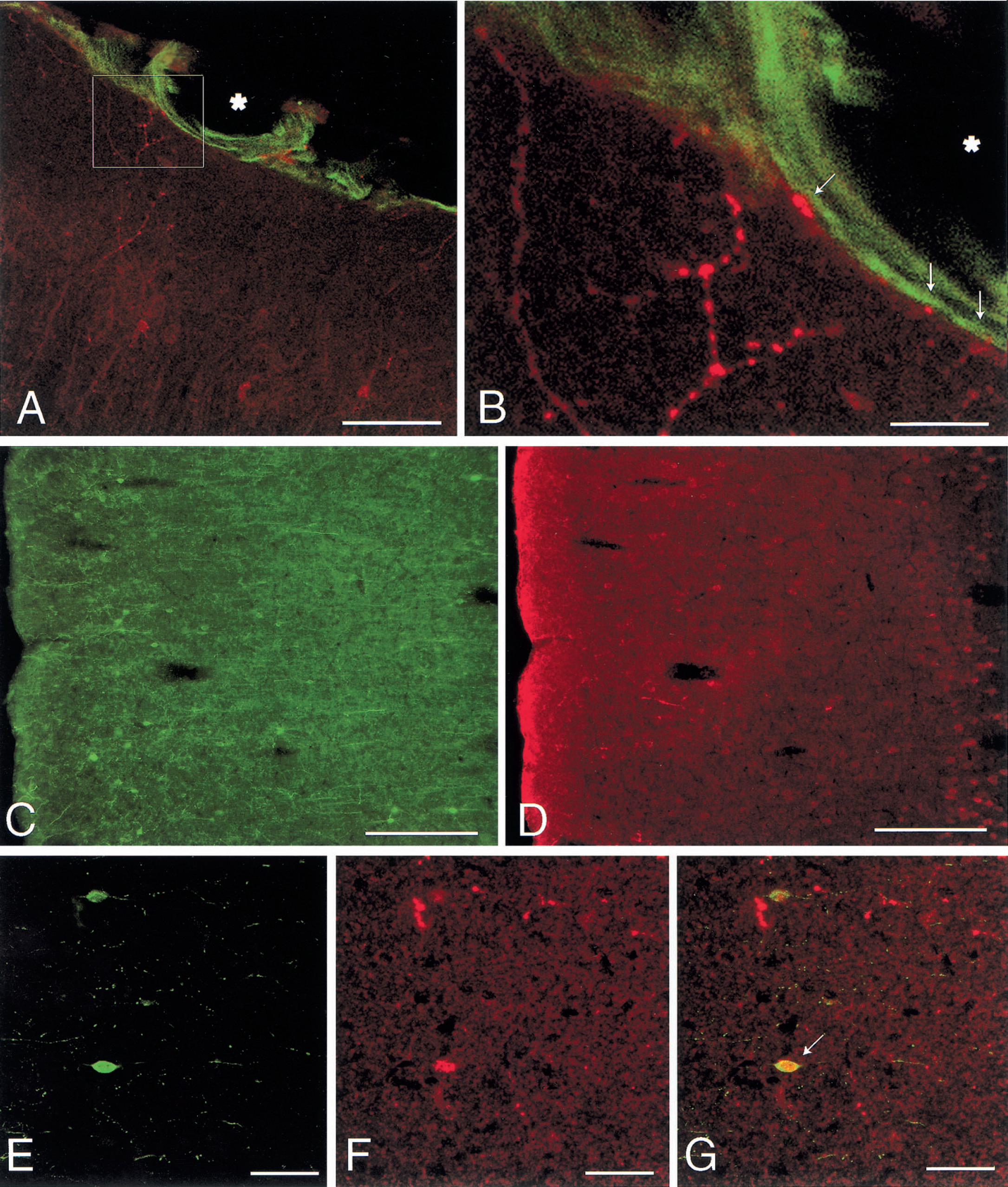
In coronal sections of the cortex processes arising from the superficial poles of VIP containing cell bodies were often found to extend into the pial tissue on the brain surface. By double immunostaining, these processes, which sometimes dichotomized, were shown to innervate the rVPAC1 receptor of the pial arterioles (Fig. 5A and 5B). Pituitary adenylate cyclase activating polypeptide immunoreactive perikarya, however, did not give rise to projections which extended to the pial arterioles. As previously reported VIP immunoreactivity in the parietal cortex was localized in a homogenous population of radially oriented bipolar neurons in cortical layers II-IV and their processes (Fig. 5C) (Lorén et al., 1979; Morrison et al., 1984; Zhang et al., 1991). The distribution pattern of the PACAP immunoreactive cell bodies differed from that of VIP because they were present in all layers (Fig. 5D), although mainly concentrated in the deeper cortical layers. The number of the PACAP immunoreactive nerve cell bodies seemed to outnumber those of VIP (Fig. 5C and 5D). Generally VIP and PACAP were observed to be stored in separate cortical neurons, but occasionally a few cortical perikarya were shown to contain both peptides (Fig. 5E to 5G).
DISCUSSION
The current study demonstrates for the first time the localization of rVPAC1 receptor immunoreactivity in rat cerebral arteries and arterioles using a polyclonal antibody generated against the N-terminal receptor fusion protein. By Western blot analysis of the expressed receptor N-terminal, the antiserum was found to detect the protein in amounts down to 3 pmol. Through immunoblotting it was also shown that the antibody detected the full length receptor protein expressed in cell lines recognized as a band of the expected 45 kDa, whereas no signal was observed with the related VIP-PACAP receptors. Further support for the specificity of the antibody comes from the immunocytochemical staining of transfected cells and the complete disappearance of rVPAC1 immunoreactivity in cerebral arteries and arterioles after preincubation of the antibody with the rVPAC1 receptor N-terminal fusion protein.
rVPAC1 receptor immunoreactivity was identified in the wall of cortical pial arteries and arterioles and in the major arteries at the base of the brain and their branches. The density and intensity of the receptor immunostaining seemed to be the same in differently sized cerebral vessels. The immunohistochemical receptor localization accords well with a previous autoradiographic study in rat (Amenta et al., 1991). However, in the current study quantitative determination of 125I-VIP binding site density in the different arteries of the circles of Willis demonstrated a higher accumulation of silver grains in the anterior than in the posterior cerebral arteries; furthermore, grain density was higher in pial arteries than in circle of Willis arteries. Autoradiographic studies, however, could reflect binding of radiolabeled VIP to other receptor subtypes than the rVPAC1 receptor.
The rVPAC1 receptor was found to be localized exclusively to the surface cell membrane of the circularly oriented vascular smooth muscle cells and no immunostaining of the endothelial cell layer was observed. This observation accords well with the unaffected ability of VIP to relax cerebral arteries after removal of endothelial cells (Lee et al., 1984). Through double immunostaining a rich supply of VIP immunoreactive nerve fibers were found to have intimate contact to the receptor protein indicating innervation. Vasoactive intestinal polypeptide immunoreactivity has previously been detected in large granular vesicles in nerve terminals and axons in cerebral arterial walls (Lee et al., 1984). Vasoactive intestinal polypeptide released from these autonomic nerve fibers could directly regulate tonus in agreement with the general view on the role of VIP in cerebral vascular control (Edvinsson et al., 1988). Although VIP, agreeing with previous studies, was identified in nerve fibers and dense nerve plexuses of cerebral arterioles and the major arteries and their brances, the number of the longitudinally oriented PACAP containing nerve fibers was much more limited and their density decreased as the vessels became smaller. Some PACAP immunoreactive nerve fibers were, like VIP, found in close apposition to the rVPAC1 receptor protein indicating that this peptide also could have a functional role in the regulation of cerebral blood flow. In accordance with the different distribution pattern of the VIP and PACAP containing nerves, double immunostaining disclosed that the two peptides were localized in separate populations of nerve fibers. This finding disagrees with a previously reported study on cat cerebral blood vessels in which PACAP immunoreactivity was demonstrated in a subpopulation of VIP containing nerve fibers (Uddman et al., 1993). Whether this discrepancy reflects a species difference between rat and cat or could merely be explained by different specificities of the antibodies used remains to be clarified. Although the VIP containing fibers to the cerebral circulation primarily originate in the sphenopalatine and otic ganglia and in microganglia located along the carotic artery (Suzuki et al., 1988), the source of the PACAP containing cerebral vascular fibers remains to be clarified. Pituitary adenylate cyclase activating polypeptide containing cell bodies have been demonstrated in a number of auto nomic ganglia including the two parasympathetic ganglia, the sphenopalatine, and the otic ganglia (Sundler et al., 1996). Pituitary adenylate cyclase activating polypeptide is also present in cell bodies in the trigeminal ganglion, which are sensitive to capsaicin (Moller et al., 1993), suggesting the possibility that PACAP fibers could represent sensory cerebral vascular nerves.
In the current study we also found that intracortical VIP containing processes penetrated the cortical surface and appeared to innervate the rVPAC1 receptor protein on pial arterioles. This innervationlike pattern was also observed after the arterioles had entered the superficial layers of the brain. It is likely that increased activity of the cortical VIP containing neurons causes release, which in turn triggers vasodilation serving as a coupling agent between nervous activity and blood flow (Ingvar and Lassen, 1975). However, processes from the PACAP immunoreactive cortical cells bodies did not project to the surface of the brain and, in general, the distribution pattern of the PACAP and VIP immunoreactive cell bodies in the parietal cortex was quite different suggesting different functional roles.
In conclusion, we have provided direct morphologic evidence that the rVPAC1 receptor is a postsynaptic receptor uniformly present in smooth muscle cells in cerebral arteries and arterioles. The receptor protein was found to be innervated by VIP containing nerve fibers, and to a lesser extent PACAP containing nerve fibers, providing the neuroanatomic substrate for the role of VIP and maybe PACAP in the regulation of cerebral blood flow circulation.
Footnotes
Acknowledgments
The authors gratefully acknowledge the skillful technical assistance of Anita Hansen and Yvonne Søndergaard. The authors thank Dr. Niels Vrang at the Panum Institute for his help in preparing the confocal photomicrographs. The VIP antibody (code MoAb #55) was donated by Dr. J.H. Walsh, CURE, Gastroenteric Biology Center, Department of Medicine, UCLA, Los Angeles, CA, U.S.A.
