Abstract
The binding of [11C]diprenorphine to µ, κ, and Δ subsites in cortical and subcortical structures was measured by positron emission tomography in vivo in six patients before and after surgical relief of trigeminal neuralgia pain. The volume of distribution of [11C]diprenorphine binding was significantly increased after thermocoagulation of the relevant trigeminal division in the following areas: prefrontal, insular, perigenual, mid-cingulate and inferior parietal cortices, basal ganglia, and thalamus bilaterally. In addition to the pain relief associated with the surgical procedure, there also was an improvement in anxiety and depression scores. In the context of other studies, these changes in binding most likely resulted from the change in the pain state. The results suggest an increased occupancy by endogenous opioid peptides during trigeminal pain but cannot exclude coexistent down-regulation of binding sites.
Trigeminal neuralgia (TN) is clinically well defined, yet little is known about the central representation of the pain experience. Other types of neuropathic pain have been studied with positron emission tomography (PET). The ongoing neuropathic pain of mononeuropathy studied in patients before and after acute nerve block was found to be represented by increased regional cerebral blood flow (rCBF) in bilateral insula, inferior frontal and prefrontal cortices, and ipsilateral anterior cingulate cortex. There were decreases in rCBF in the thalamus ipsilateral to the pain (Hsieh et al., 1995). Acute allodynic pain in patients with lateral-medullary (Wallenberg) infarcts was found to be represented by increased rCBF contralateral to the pain in the insula, prefrontal, motor, SI and SII cortices, and lateral thalamus, with ipsilateral responses in the inferior parietal cortices. Interestingly, there were decreases in rCBF in the anterior cingulate cortex (Peyron et al., 1998). Increases and decreases in rCBF cannot be equated directly with net inhibition or excitation. However, the decreases in thalamic rCBF observed both in the chronic neuropathic pain study of Hsieh and colleagues (1995) and during chronic cancer pain suggest differences in the modulation of the rCBF responses between acute and chronic pain (Jones, 1998). The decreases in rCBF with acute allodynic pain (Peyron et al., 1998) in the context of increases in rCBF in anterior cingulate cortex during experimental pain in all normal volunteer PET studies (Derbyshire et al., 1998) suggest substantial modulation of anterior cingulate function in this type of central deafferentation pain. So far, all PET studies of ongoing or tonic clinical pain have demonstrated alterations in rCBF within components (thalamus, insula, prefrontal and anterior cingulate cortices) of the “pain matrix” that have high concentrations of opioid receptor binding (Jones et al., 1994b). These all are subcortical or cortical components of the “medial pain system” (Jones, 1998), which is associated with processing of affective and higher cognitive aspects of nociception such as attention and aversive conditioning. No changes in rCBF in SI or SII have been associated with chronic pain. These cortical areas are the principal cortical projections of the “lateral pain system” and are associated more with aspects of localization and intensity coding (Treed et al., 1999). Notice that both primary occipital and somatosensory cortices are distinguished from other cortical areas by their low opioid receptor binding (Jones et al., 1991c). The common clinical observation that synthetic opiates reduce the unpleasantness of the pain that still can be localized makes some sense of this physiologic and pharmacologic division between the medial and lateral pain systems.
One candidate class of compounds that may have a role in modulating nociceptive responses within some of the components of the medial pain system are the endogenous opioid peptides. Since these were discovered (Hughes, 1975), their precise function in humans has remained elusive, although there is reasonable evidence for their involvement in the modulation of nociceptive processing in animals (Hollt et al., 1985). This is partly because of difficulties of access and partly because of difficulties with the use and interpretation of the effects of opiate antagonists such as naloxone. Techniques to measure availability of brain in vivo opioid receptor binding in human subjects have been developed using the opioid antagonist [11C]diprenorphine and PET, which provide the possibility of measuring changes in occupancy of opioid receptors by endogenous opioid peptides (Jones et al., 1994b).
The other advantage of [11C]diprenorphine is that it has been previously shown that there is no correlation between rCBF and K1/k2 or k3/k4 in normal volunteers (Jones et al., 1994).
Previous studies (Jones et al., 1994a) have shown substantial changes in cerebral 11C-diprenorphine binding in relation to chronic inflammatory pain states in patients with rheumatoid arthritis. The latter are consistent with changes in opioid peptide concentrations in the brain of arthritic animals (Panerai et al., 1987). We have used [11 C] diprenorphine and PET (Jones et al., 1987) in six patients with TN to evaluate whether similar changes in opioid receptor binding occur during the chronic neurogenic pain characteristic of this condition. The hypothesis was that reduced [11 C]diprenorphine binding would occur during ongoing pain as a result of increased occupancy by endogenous opioid peptides, and that this would occur mainly within the components of the medial pain system.
METHODS
Six male patients (mean age 61.5 ± 9.5 years; range 46 to 72 years) with TN were studied. Each had a typical clinical presentation of TN with a mean length of history of 15 ±8 years (range 3 to 26 years). Five subjects were right sided and one was left sided. All patients were affected by pain in the following trigeminal nerve divisions: the maxillary division (three patients), maxillary and mandibular divisions (two patients), or just the mandibular division (one patient). Routine diagnostic magnetic resonance (MR) imaging brain scans had been performed. Results of these studies were normal in five patients, whereas an incidental subfrontal meningioma was found in one. All six patients were taking carbamazepine as their only medication with a mean dose of 800 ± 300 mg (range 400 to 1300 mg), which was kept constant throughout the duration of the study, spanning both presurgical and postsurgical scans. Three patients had no previous radiofrequency thermocoagulation (RFTC) treatments, whereas the others had undergone one, two, or three previous RFTC treatments.
Two PET studies were performed on each patient: one before RFTC treatment (“in pain”), and one within 3 weeks after treatment (“pain free”). The RFTC treatments were considered technically successful in all cases, producing analgesia in the appropriate trigeminal divisions while maintaining light touch sensibility.
Measures of pain, anxiety, and depression were performed before each scan: namely, the Visual Analogue score of pain within the previous 24 hours, and the Hospital Anxiety and Depression scale of anxiety and depression.
11C-Diprenorphine PET studies were performed on the CTI 953B PET scanner (CTI, Inc., Knoxville, TN, U.S.A.) at the MRC Cyclotron Unit. This scanner has retractable interplane septa and thus the ability to acquire all lines of coincidence between all planes (Spinks et al., 1992). The inplane resolution of the scanner in three-dimensional mode is 8.5 × 8.5 × 4.3 mm (full width-half maximum). After intravenous injection of 11C-diprenorphine (specific activity range 7000 to 3800 mBq/µmol), a dynamic scan over 90 minutes was performed consisting of 17 time frames (6 × 180 seconds, 3 × 240 seconds, 4 × 300 seconds, 4 × 600 seconds). For attenuation correction, a transmission scan was taken using three external line sources of germanium-68. To obtain the 11C-diprenorphine input function, continuous radioactivity monitoring of the subjects' arterial blood, with intermittent sampling to measure unchanged 11C-diprenorphine and metabolites, was carried out (Jones et al., 1987).
The PET images then were reconstructed on a Supercard SCl computer (CSPI, Billerica, MA, U.S.A.) and pixel-by-pixel functional images obtained of the [11C]diprenorphine binding equivalent to total volume of distribution (Vdtott) using a spectral analysis technique (Cunningham and Jones, 1993; Tadokoro et al., 1993). The use of Vdtot as a measure of specific binding of [11C]diprenorphine is discussed in detail by Jones and associates (1994b). It is directly proportional to the binding potential and has been shown to be independent of changes in CBF.
Correction of intrasubject head movement during each study was carried out using the Automated Image Registration program (Woods et al., 1992), which normalized each scan to the first image acquired. Results from preoperative and postoperative scans were normalized to the standardized Talairach Atlas (Talairach and Tournoux, 1988; Friston et al., 1991) and superimposed on the standardized MR space. The data from the preoperative and postoperative studies were pooled into two normalized preoperative and postoperative images. The single patient with left-sided pain was mirror-transformed before group analysis.
Statistical differences between the preoperative and postoperative groups were analyzed. This included pixel-based analysis of covariance (ANCOVA correction) to eliminate the effect of random global differences in [11 C]diprenorphine binding between groups (Friston et al., 1990). The statistical differences between the preoperative and postoperative groups were displayed as a statistical parametric map (Friston and Frackowiak, 1991).
RESULTS
Results from the measures of pain, anxiety, and depression performed before each scan were as follows. Between studies, the pain Visual Analogue score (which ranges from 0 [pain free] to 100 [maximal pain possible pain]) changed from a mean of 45 (range 12 to 73) to 0, whereas the scores on the Hospital Anxiety and Depression scale (a score greater than 9 on the depression subscale indicates possible depression, and a score of lion the anxiety subscale indicates possibly significant anxiety) altered from a mean of 8.5 (anxiety) and 8.5 (depression) to 4 and 3.5, respectively. Statistical analysis on such small numbers was not considered appropriate. All patients thus were rendered pain free with a reduction in associated anxiety and depression. All patients experienced pain on the day of their scan, but none of them experienced pain during the scanning period.
Selection of areas with bilateral increases in cortical [11C]diprenorphine binding with pain relief
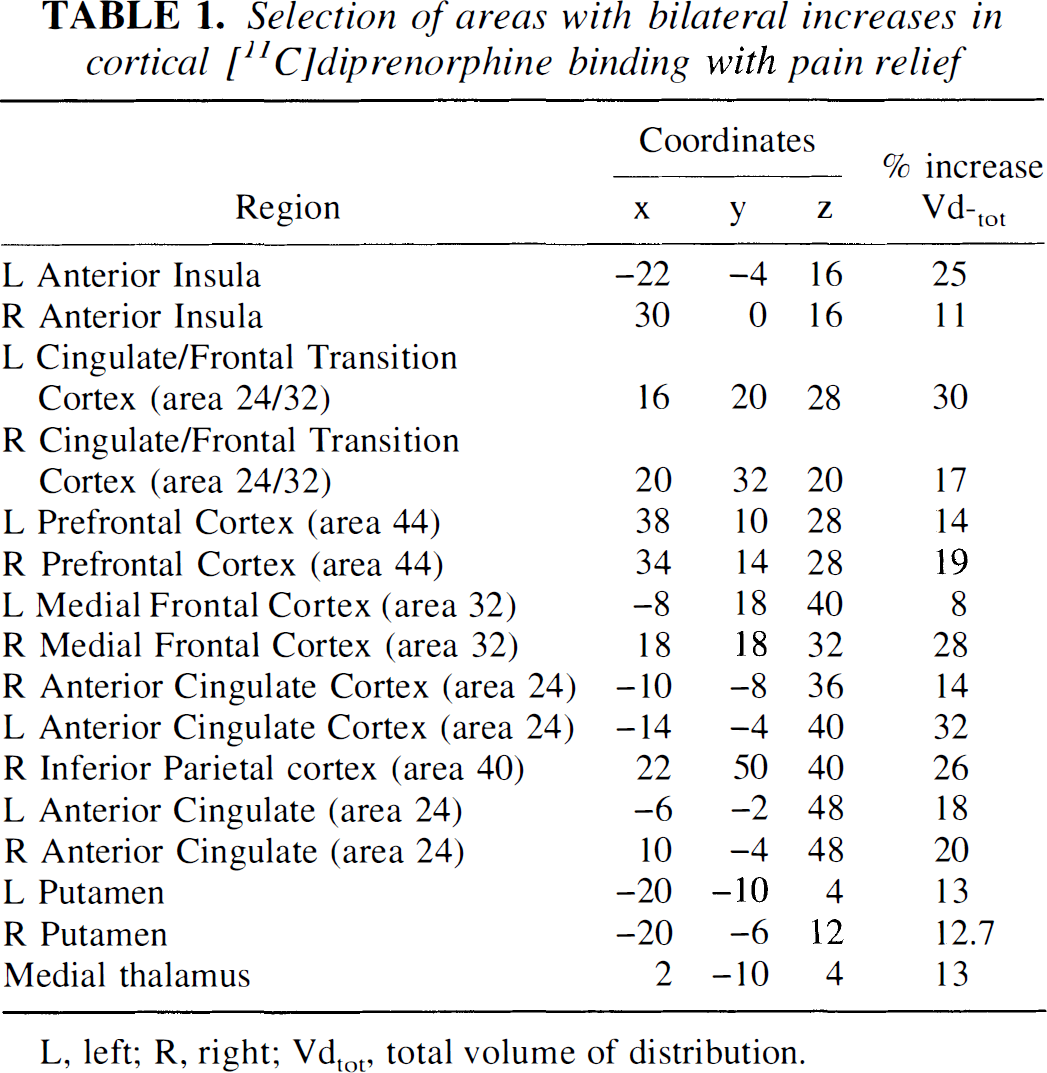
L, left; R, right; Ydtot total volume of distribution.
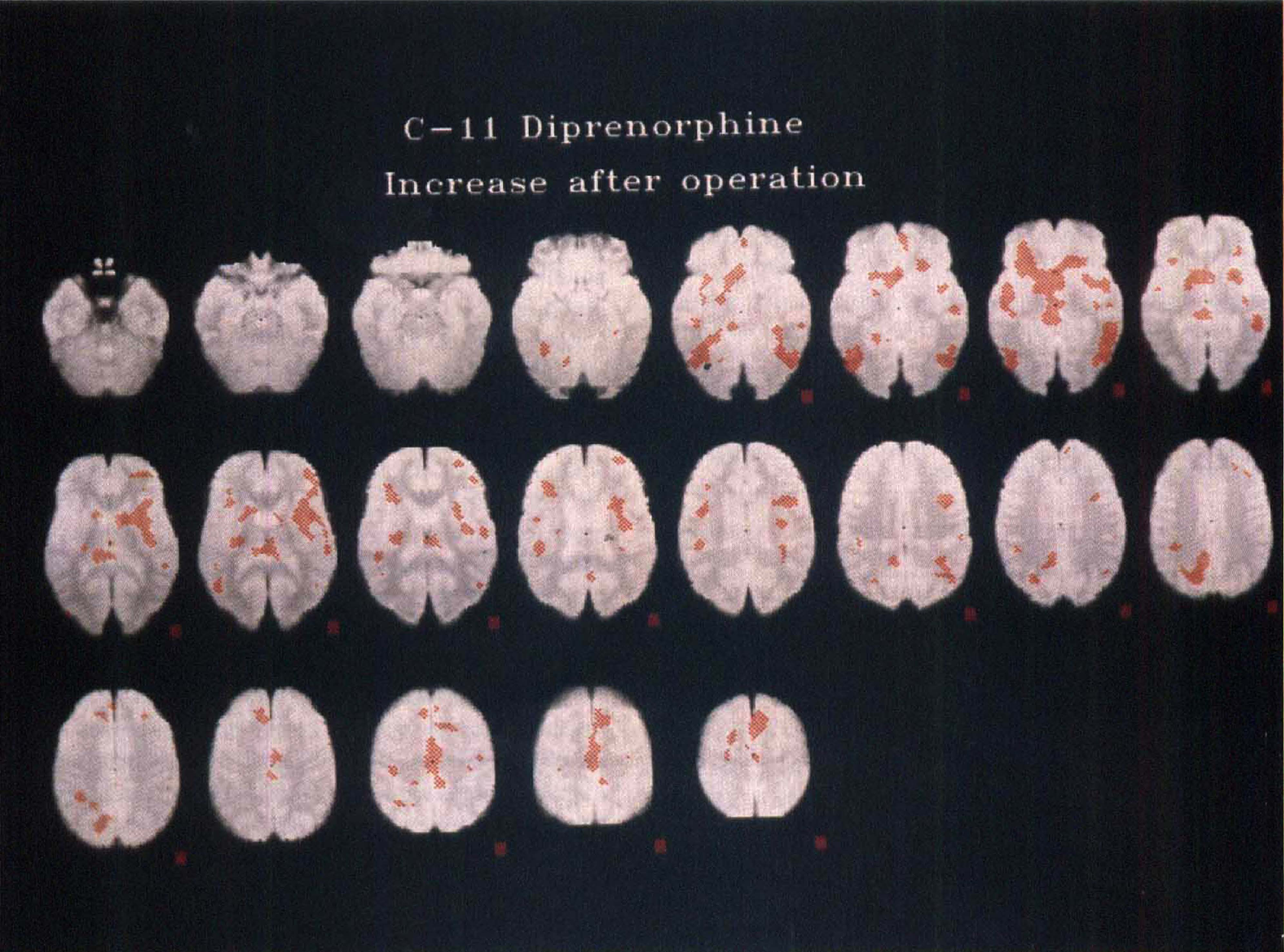
Statistical parametric map of significant increases in [11C]diprenorphine binding (denoted by red areas) as a result of surgical pain relief in six patients with trigeminal neuralgia coregistered in standardized MR/Talairach space. Right is the right side of the brain; top left is the bottom of the brain; bottom right, the top, with slices taken at 4-mm intervals. Plane five of the top row is 0 mm relative to the line joining the anterior and posterior commissures (AC-PC line).
DISCUSSION
Focal brain increases in diprenorphine binding when pain free are most likely to be caused by a decreased occupation of opioid receptors (µ Δ, and κ) by endogenous opioid peptides, resulting in increased available sites for [11C]diprenorphine binding, up-regulation of total binding sites, or both. This technique cannot distinguish between these possibilities. Two high and low specific activity scans in each state might go some way to achieve this (Sadzot et al., 1991) but would have been unlikely to be acceptable to the patients. This entire procedure requires' that patients lie still in the scanner for a minimum of 2 hours. Changes in affinity state as an explanation for these results are less likely, since these have not been observed in the brain during experimental pain (Yonehara et al., 1983). The posttreatment scans were performed within 3 weeks of being rendered pain free and a mean of II days after the operation. It is unlikely that this response represents a nonspecific reaction Selection of areas with bilateral increases in cortical to the stress of the operation or altered stress associated with the actual scan, since previous studies in normal volunteers and patients suggest reproducible binding parameters for both pulse-chase and tracer-alone protocols (Jones et al., 1994b). Stress in animals has been shown to produce reductions in mainly subcortical in vivo 3H]diprenorphine binding (Seeger et al., 1984) rather than increases. Although it is possible that relative changes in rCBF may have a small effect on the Vdtot of [11C]diprenorphine binding, this is unlikely to be substantial. It has been previously demonstrated that there is no correlation between rCBF and K1/k2 or k3/K4 in normal volunteers (Jones et al., 1987), and it is therefore unlikely that the situation would be different in this group of patients.
Anxiety, depression, and pain scores all improved after RFTC treatments with complete resolution of the pain. It is unlikely that the changes in [11C]diprenorphine binding are purely a result of the change in mood state, since the current evidence suggests that an increase rather than a decrease in binding should occur during depression. Substantial up-regulation of frontal and anterior cingulate opiate receptor binding, which may be compensatory for reduced opioid peptide release, has been well documented in suicide victims (Gross-Isseroff et al., 1990). Preliminary PET results have confirmed a similar finding in patients with depression (Mayberg et al., 1991). If this were a significant factor in our studies, this would produce the opposite effect of that observed. It is therefore likely that the major psychological component resulting in the change in [11C]diprenorphine binding is the change in the pain state. As far as we are aware, this is the first direct evidence for activation of the endogenous opioid system during neuropathic pain. Most of the areas where significant changes in binding occurred also have been demonstrated to be implicated in acute (Jones et al., 1991a; Talbot et al., 1991; Jones et al., 1992; Casey et al., 1994) and tonic experimental pain processing (Derbyshire and Jones, 1998; Di Pierro et al., 1994). Most of these structures apart from dorsolateral prefrontal cortex also have been shown to be involved in the processing of chronic neuropathic pain (Hsieh et al., 1995; Cesaro et al., 1991).
Although opiate receptors are widespread and have several different functions in the brain, previous in vivo PET studies using diprenorphine suggest that there are more discrete areas such as prefrontal cortex and anterior cingulate where endogenous opiates may have a discrete function in relation to chronic inflammatory pain (Jones et al., 1994a). Also notice that the synthetic mu-agonist fentanyl in analgesic doses also causes significant increases in rCBF in bilateral prefrontal, anterior cingulate, and caudate nuclei (Firestone et al., 1996). Preliminary data suggest that morphine-induced analgesia for cancer pain is associated with increased rCBF in frontal, anterior cingulate, and insula cortices (Jones et al., 1991b). From our own preliminary results, it appears that the main endogenous opiate responses are within the subcortical and cortical projections of the so-called medial pain system. So far, no changes in opioid receptor binding have been observed in the cortical projections of the lateral pain system to the primary or secondary somatosensory cortices during either inflammatory or neuropathic pain, the former having relatively low levels of opioid receptor binding (Jones et al., 1991c). Notice that the main differences in patterns of cortical activation in different types of chronic pain also have been in the anterior cingulate and prefrontal cortical (Derbyshire et al., 1994; Jones et al., 1997) responses.
The cortical projections of the medial pain system appear to be mainly concerned with the affective, attentional, and intensity coding components of nociceptive processing, whereas the somatosensory cortex is more robustly activated when there is a prominent localization element to the stimulus (Jones, 1998). This has been demonstrated by manipulating the affective components of a nociceptive stimulus while maintaining the intensity rating constant (Rainville et al., 1997) using hypnosis and PET. Using a hypothesis-driven region of interest approach to the analysis, rCBF changes in the anterior cingulate cortex, were clearly correlated with changes in affective response with an absence of correlation in SI. It is therefore not surprising that the main changes in the endogenous opioid system should be found within the projections of the medial pain system.
Although early pain activation studies emphasize the lateralization of cerebral pain responses (Casey et al., 1994; Talbot et al., 1991), subsequent studies with a more sensitive scanner in three-dimensional mode of acquisition provide evidence for bilateral activation of anterior cingulate, insula, and prefrontal cortices and thalamus (Vogt et al., 1996).
After surgical pain relief of trigeminal neuralgic pain, there are significant increases in [11C]diprenorphine binding within some of the cortical and subcortical components of the pain matrix, consistent with the concept of decreased occupancy by endogenous opioid peptides when pain free. It is not within the scope of this experiment to determine the physiologic consequences of the changes in [11C]diprenorphine binding. However, in animals with experimentally induced acute prolonged pain, increased pain behavior has been demonstrated after intracerebroventricular anti-β-endorphin antibody (Porro et al., 1999). Interestingly, this was accompanied by increased metabolic activity in several structures including the thalamus, caudate nucleus, parietal, ingulate, and frontal and insula cortices. This experiment neatly avoided some of the inherent problems associated with naloxone, including peripheral and central agonist and antagonist effects at different doses and the short cerebral half-life. The new generation of enkephalinase inhibitors have been used to elicit the opposite behavioral effects to anti-β-endorphin antibody (Chipkin et al., 1988; Ruiz-Gayo et al., 1992). The substantially increased bioavailability of these compounds provides the potential for human use. The exciting challenge for the future is to establish whether the substantial changes in the endogenous opioid system during trigeminal neuralgic and other types of pain might be amenable to controlled pharmacologic manipulation to further increase opioid receptor occupancy within the medial pain system. The spare opioid binding capacity makes this system amenable to such an approach. Positron emission tomography has been suggested as the technique of choice to establish the optimal dosing regimes for such compounds to achieve specific levels of opiate receptor occupancy and their physiologic effects in humans.
