Abstract
Helospectin I and II and helodermin are nonamidated, vasoactive intestinal peptide (VIP)-like peptides, isolated from the salivary gland venom of the lizards Heloderma suspectum and Heloderma horridum. Helospectin I has 38 amino acid residues and differs from helospectin II in that it has an additional serine residue at the C-terminus. Numerous nerve fibers containing helospectin-like immunoreactivity (LI) and a few fibers containing helodermin-LI were present in the adventitia and at the adventitia-media border of cat cerebral arteries. In the sphenopalatine ganglion, numerous nerve cell bodies containing helospectin-LI were seen. Double immunostaining revealed that helospectin-LI nerve cell bodies coexisted with VIP-containing cell bodies. Radioimmunoassay showed high levels of helospectin-LI in extracts of cerebral vessels from the circle of Willis (27.4 pg/mg [wt/wt]). Helospectin I and II and helodermin (10−10 to 10−6 mol/L) produced concentration-dependent relaxations of feline middle cerebral arteries amounting to 50% to 80% of precontraction induced by U46619. The maximum effects and the potency were similar to that of VIP. Neither of these peptides elicited endothelium-dependent relaxations. Intracerebral microinjection of helospectin and helodermin produced a moderate concentration-dependent increase of the cerebral blood flow of α-chloralose anesthetized cats. The maximum increase (21 ± 5%) was observed after the injection of 5 μg helodermin, whereas 16 ± 7% was seen with helospectin I and 19 ± 5% with helospectin II. The results suggest that helospectin/helodermin-like peptides co-localize with VIP in perivascular nerve fibers originating in the sphenopalatine ganglion. They seem to have strong and potent vasodilator effects.
Keywords
Helodermin and the helospectins belong to the family of vasoactive intestinal peptide (VIP)-related peptides, among which are pituitary adenylate cyclase activating peptide (PACAP) (Miyata et al., 1989, 1990), glucagon, secretin, and peptide histidine isoleucine. Helospectin I and II are two closely related peptides first isolated from the venom of the lizard Heloderma horridum (Parker et al., 1984; Vandermeers et al., 1984). Helodermin is an amidated peptide of 35 amino acid residues originally isolated from the salivary gland venom of the lizard Heloderma suspectum (Hoshino et al., 1984; Robberecht et al., 1984; Vandermeers et al., 1984). Helospectin I consists of 38 and helospectin II of 37 amino acid residues. Helospectin II is identical to helospectin I except for the lack of serine-38 in position 38. These peptides are homologous with helodermin; helospectin I differs from helodermin in four positions only. There is evidence that helospectin- and helodermin-like peptides are distributed in mammalian endocrine-like cells (Bjartell et al., 1989; Grunditz et al., 1989; Luts et al., 1991) and in neurons in the central and peripheral nervous system (Kivipelto et al., 1992; Sundler et al., 1988; Grimes et al., 1995).
In the current study, the distribution of helospectin-like immunoreactivity in the sphenopalatine ganglion and around feline cerebral arteries has been examined. Moreover, in view of their VIP-like effects on rat blood vessels (Grundemar and Högestätt 1990; Cardell et al., 1993), the effects of helospectin and helodermin on feline cerebral blood vessels have been examined in vitro and compared with that of VIP. The effects of local cortical injection of helospectin and helodermin on cerebral blood flow have been examined in vivo.
METHODS
Immunohistochemistry
Pial vessels at the base of the brain, sphenopalatine ganglia, trigeminal ganglia, and superior cervical ganglia were collected from five adult cats (body weight 3 to 4 kg). The specimens were fixed by immersion in an ice cold mixture of 2% formaldehyde and 0.2% picric acid in 0.1 mol/L phosphate buffer (pH 7.2) overnight and thoroughly rinsed in Tyrode solution containing 10% sucrose. The specimens then were frozen on dry ice and sectioned at 10-μm thickness in a cryostat. Other vessels and cortical pial membranes were fixed as free floating tissues overnight and processed as described earlier. They then were briefly rinsed in phosphate buffer and stretched on chrome-alum subbed microscope slides as whole mounts. Cryostat sections and whole mounts were processed for the immunocytochemical demonstration of helodermin, helospectin, and VIP. The helodermin antiserum (code no. 8706, Eurodiagnostica, Malmö, Sweden) was raised in rabbit against synthetic helodermin and used in a dilution of 1:160, and the helospectin antiserum (code no. 8843, Eurodiagnostica) was raised in a rabbit against helospectin I and used in dilution of 1:320. To demonstrate VIP, we used two different antisera. One antiserum (code no. 7852, Eurodiagnostica) was raised in a rabbit and used in a dilution of 1:640. The other (code no. 8701, Eurodiagnostica) was raised in a guinea pig and used in a dilution of 1:640. The latter antiserum was used together with the helospectin antiserum for double immunostaining (see later) to examine possible coexistence of the two immunoreactants. The sections were exposed to the peptide antiserum for 24 hours at 4°C in a moist chamber. The site of the antigen-antibody reaction was revealed by application of fluorescein isothiocyanate-labeled IgG antibodies (Dakopatts, Copenhagen, Denmark) in a dilution of 1:320 for 1 hour at room temperature. Control sections were exposed to primary antiserum that had been preabsorbed with excess amounts of each of the antigen (10 μg of synthetic peptide per milliliter of diluted antiserum). The helospectin I antiserum recognized helospectin II and helodermin, but not VIP, peptide histidine isoleucine, PACAP, secretin, or glucagon (Kivipelto et al., 1992). The helodermin antiserum did not cross-react with VIP, peptide histidine isoleucine, PACAP, glucagon, or secretin (Luts et al., 1991; Kivipelto et al., 1992). The VIP antisera previously have been characterized at the immunocytochemical level (Grunditz et al., 1986; Luts and Sundler 1989). They were found not to cross-react with helodermin, helospectin I or II, or PACAP-27 or PACAP-38. Since cross-reaction with other peptides or proteins containing amino acid sequences recognized by the antisera cannot be excluded, it is appropriate to refer to the immunoreactive material as helospectin-like and VIP-like, respectively. For simultaneous double immunostaining, the guinea pig VIP antiserum was demonstrated using a second antibody labeled with fluorescein isothiocyanate, whereas the (rabbit) helospectin I antiserum was demonstrated using a second antibody labeled with tetramethyl rhodamine.
Radioimmunoassay
The cerebral blood vessels from the circle of Willis of six cats were harvested and extracted by boiling in 0.9% NaCl for 10 minutes followed by homogenization (Polytron, 1 to 2 minutes). The homogenate was centrifuged at 3000 g for 10 minutes at +4°C. The sediment was re-extracted by boiling in 0.5 mol/L acetic acid for 15 minutes, followed by homogenization and centrifugation as described earlier. The pooled supernatant was freeze-dried and dissolved in 0.05 mol/L phosphate buffer, pH 7.5. The helospectin-like peptide was measured by using antiserum no. 8709 (Eurodiagnostica). The radioimmunoassay has been described in detail previously (Kivipelto et al., 1992).
Response of isolated cerebral artery segments
Seven adult cats of either sex, weighing 2 to 4 kg, were exsanguinated under pentobarbitone anesthesia (phenobarbital [Nembutal] 30 mg/kg intraperitoneally). The brains were removed, and the middle cerebral arteries then were dissected free and placed in a cold buffer solution (4°C) aerated with 5% CO2 in O2. The composition of the buffer solution was (in millimolar amounts): NaCl 119, KCl 4.6, CaCl2 1.5, MgCl2 1.2, NaHCO3 15, NaH2PO4 1.2, and glucose 11.0, pH 7.4. Vessel segments 2 to 3 mm long were mounted between two L-shaped metal prongs in a 2.5-mL mantled tissue bath at 37°C to record circular vasomotor activity. Isometric tension was measured with Grass force displacement transducers and recorded on a Grass polygraph (Grass Instruments, Quincy, MA, U.S.A.). Circular segments were given a passive load of 3 mN and were allowed to attain a steady level of tension and to stabilize for 90 minutes before testing with an elevated (60 mmol/L) potassium buffer solution obtained by an equimolar substitution of NaCl for KCl, resulting in a contraction amounting to 10.2 ± 1.2 mN. When the vessels were examined for relaxation, the vascular segments were given an active tone (4.4 ± 0.7 mN) by adding U 46619 (3 × 10−7 mol/L). This resulted in a contraction that was stable for at least 30 minutes, during which the peptides were tested in a cumulative manner. The endothelium was not damaged by these procedures, as evidenced by intact relaxation to the cumulative administration of acetylcholine (Furchgott, 1984).
In some experiments, the endothelium was removed by perfusing with a buffer solution containing 0.1% Triton X-100 for 10 seconds (Hamel et al., 1987). Verification of the absence of endothelium was always checked by the lack of dilatory response to acetylcholine (Furchgott and Zawadzki 1980).
Analysis of cerebral blood flow
Eight cats were anesthetized with α-chloralose (60 mg/kg intraperitoneally), paralyzed (gallamine triethiodide, 6 mg/kg intravenously), ventilated with a mixture of 60% air-40% O2, and placed in a stereotaxic device (David Kopf Instruments, CA, U.S.A.). End-expiratory CO2 was continuously monitored and adjusted by altering the stroke volume of a ventilator (Harvard Instruments, Cambridge, MA, U.S.A.), and the fractional concentration of O2 in inspired gas was continuously observed (DATEX Instruments, Finland). Arterial blood gases were measured during the study. Polyethylene catheters were placed in the femoral artery and vein bilaterally to measure blood pressure (using a Statham pressure transducer) and administer drugs or fluids. The level of anesthesia was monitored by observing cardiovascular responses to nociceptive stimuli, and supplemental doses (10 mg/kg intravenously of α-chloralose) were administered.
Laser Doppler flowmetry
The principles of operation have been well described by Stern and colleagues (Stern et al., 1977), and the application of the technique in this type of study also has been recently described (Goadsby, 1991). Briefly, infrared light from a laser diode with a wavelength of 780 nm is directed to the surface of the brain using a fiber optic cable. The cable contains two further fibers that detect backscatter from a tissue area of approximately 1 mm3. Analysis of the Doppler shift caused by that portion of light scattered by the red blood cells gives the velocity of flow for those cells, whereas the proportion of light backscattered is directly related to the volume of moving red cells in the brain sampled. A flow index thus can be derived by multiplying these signals and represents cerebral cortical blood flow (CBFLDF).
In these experiments, the BPM 403A (TSI Inc., MN, U.S.A.) instrument was used with a pencil probe (P-433, TSI) mounted in a stereotaxic manipulator (David Kopf Instruments) and the signals for flow, velocity, and volume continuously monitored. Biparietal craniotomies were carried out with a low-speed dental drill that was cooled with saline to prevent underlying thermal injury, and the dura was left intact. Cerebrovascular reactivity to a brief period of hypercapnia (6% to 8%) was tested before collecting data. Only data points collected from animals responsive to hypercapnia are included in this analysis. The blood pressure, heart rate, end-expiratory CO2, inspiratory O2, and laser Doppler volume, flow, and velocity signals were passed to a signal conditioning device and then to an analogue-to-digital converter (LabMaster DMA, Scientific Solutions, OH, U.S.A.) in an 80386/80387-based microcomputer. Data was monitored by averaging over a respiratory pump cycle. All data were stored on disk for later analysis and plotting.
Local cortical injection
Solutions of either vehicle (phosphate-buffered saline), helospectin I or II, or helodermin were injected directly onto the cortex in volumes of 1 μL. The injections were made from a glass micropipette driven by a pressure injecting system that was constructed in the laboratory. The volume was delivered over 5 minutes under the area of probe sampling with the glass pipette inclined so that the dura could be incised for its passage at a distance from the recording site. Cortical vascular reactivity was tested after pipette insertion and before the injections were made. Helospectin I or II and helodermin each were injected in doses of 0.05, 0.5, and 5 μg, and the vehicle was injected in the same volume.
Drugs
The following drugs were used: acetylcholine (Sigma Chemical Co., St. Louis, MO, U.S.A.), atropine sulphate (Sigma), cimetidine hydrochloride (S.K.F., U.K.), helodermin (Peninsula, Merseyside, U.K.), helospectin I and II (Peninsula), propranolol (Zeneca, Macclesfield, U.K.), Spantide (Ferring AB, Malmo, Sweden), U46619 (9,11-dideoxy-11α, 9α-epoxymethano-prostaglandinF2α; Sigma), and VIP (Peninsula, U.S.A.). All drugs were diluted to appropriate concentrations with 0.9% NaCl containing 1 mmol/L of ascorbic acid. Helodermin and helospectin I and II were dissolved and further diluted in saline containing bovine serum albumin (1%) and used in the experiments within 30 minutes to avoid possible breakdown).
Statistics
Data throughout the text are expressed as mean ± SD. The CBF data were converted to the percentage change from previous baseline level for the calculations (Kaiser et al., 1989). Local cortical injection data were tested using analysis of variance followed by Scheffe's method to test specific differences between groups (Wallenstein et al., 1980). Data obtained by studying vasomotor responses in vitro were tested using Kruskal Wallis test, followed by Mann-Whitney U test to test specific differences between groups.
RESULTS
Immunohistochemistry
A moderate supply of helospectin-like immunoreactive fibers and a few fibers containing helodermin-like immunoreactivity were regularly encountered in the adventitia and at the adventitia-media border of all cerebral arteries examined (Fig. 1). Control sections were devoid of such an immunoreactivity. Few nerve fibers were observed in material from the basilar and the vertebral arteries. The small cerebral arteries and arterioles on the cortical surface received immunoreactive fibers, although to a lesser extent than the major cerebral arteries at the base of the brain. In the sphenopalatine ganglion, numerous nerve cell bodies of the total cell population displayed helospectin-like immunoreactivity (Fig. 1). A smaller amount of cell bodies displayed helodermin-like immunoreactivity. Helospectin- or helodermin-containing nerve cell bodies were not seen in the trigeminal or superior cervical ganglia.
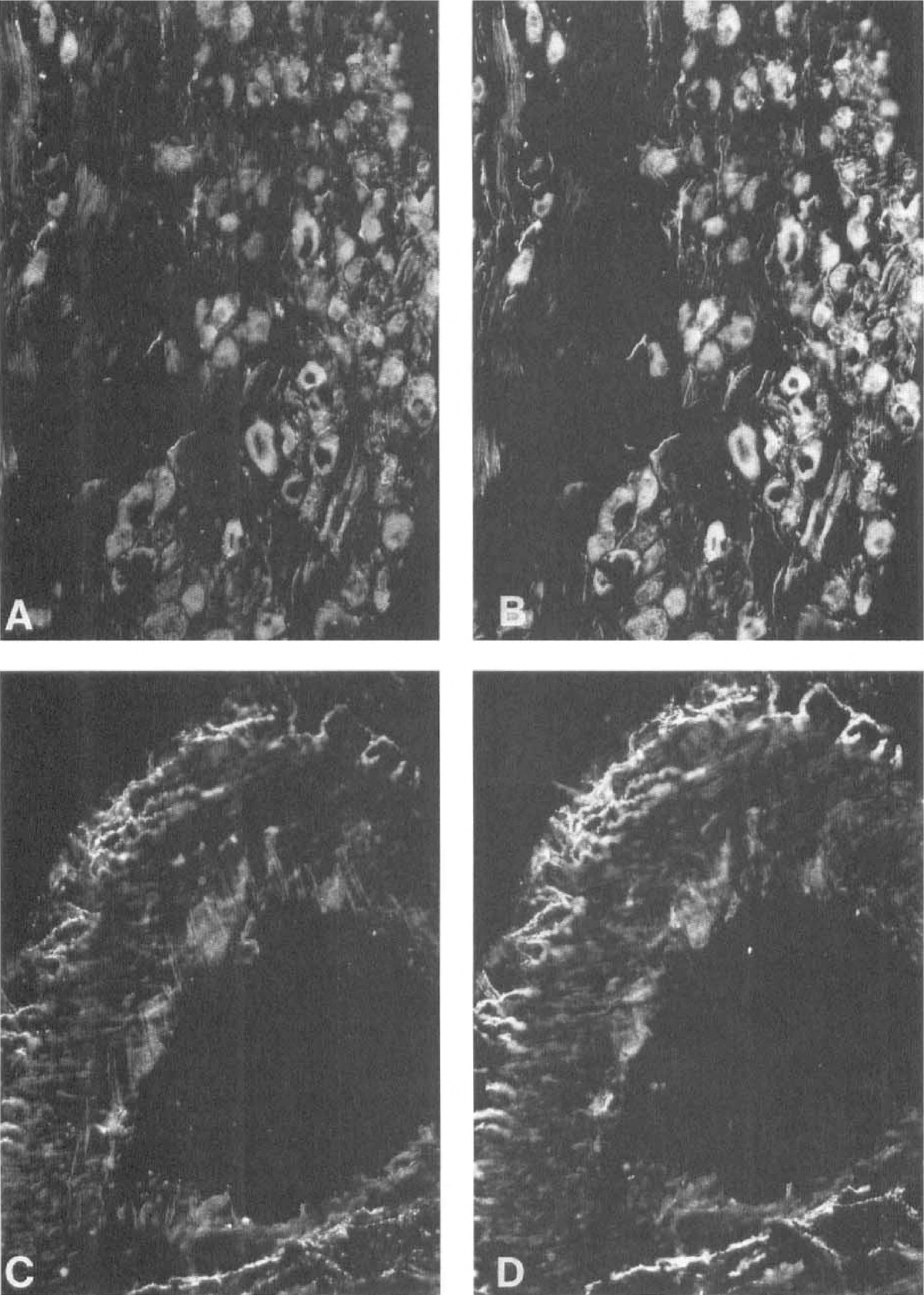
Sphenopalatine ganglion
Double immunostaining revealed that all of the helospectin-like immunoreactive nerve cell bodies in the sphenopalatine ganglion contained VIP. Several VIP-immunoreactive nerve cell bodies did not contain helospectin-like immunoreactivity. Around cerebral arteries, the helospectin-like immunoreactive nerve fibers seemed to coexist with VIP (Fig. 1).
Radioimmunoassay
The entire circle of Willis was collected and pooled from six cats. Using the radioimmunoassay, helospectin-like immunoreactivity was 27.4 pg/mg (wt/wt).
Responses of isolated middle cerebral arteries
Under resting conditions (i.e., when the arteries had reached a steady level of tension of approximately 3 mN), helodermin and helospectin caused no or a minimal relaxation of middle cerebral arteries. In vessel segments precontracted with U46619 (3 × 10−7 mol/L), administration of helodermin or helospectin I or II (10−11 to 10−6 mol/L) resulted in a concentration-dependent relaxation of the cerebral artery segments (Fig. 2). The negative logarithm of IC50, pIC50 (concentrations eliciting half maximum response) were within a narrow spectrum for helodermin and helospectin: 7.65 to 8.08 mol/L (Table 1). The maximum relaxation obtained in vessels with active tension induced by U46619 was 47% to 79% of precontraction (Table 1). The presence of atropine (10−6 mol/L), cimetidine (2 × 10−6 mol/L), Spantide (10−6 mol/L), or propranolol (10−6 mol/L) in the tissue bath did not alter the relaxation of middle cerebral arteries induced by the administration of helodermin or helospectin. The responses induced by helodermin and helospectin I and II did not differ significantly from that induced by VIP (Table 1).
Relaxation induced by VIP, helodermin, helospectin I, and helospectin II on feline middle cerebral artery
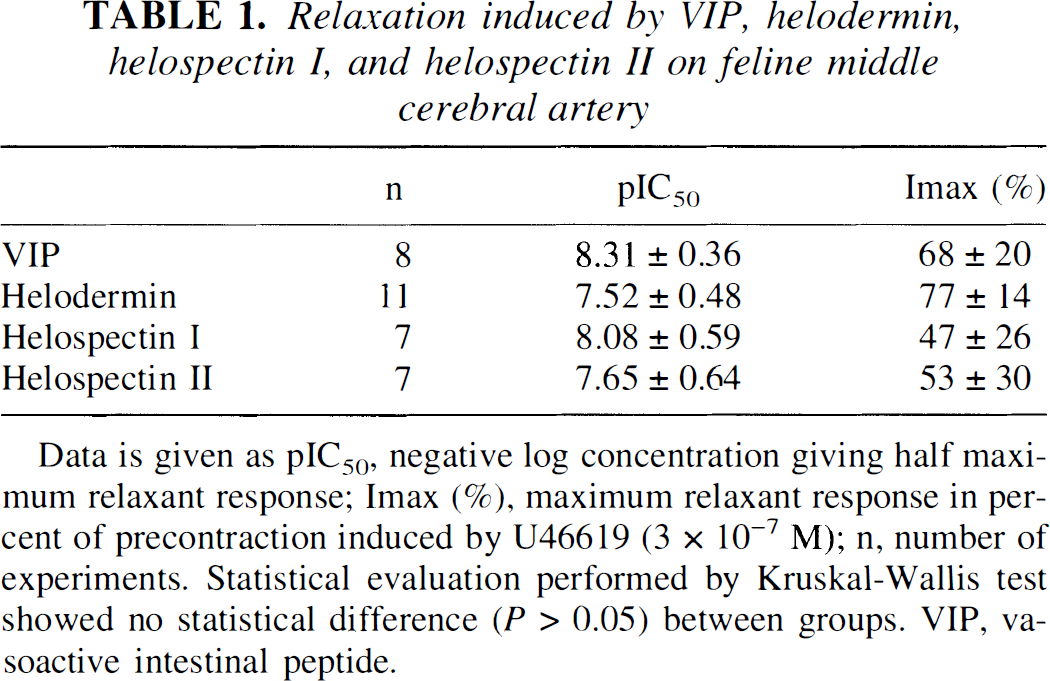
Data is given as pIC50, negative log concentration giving half maximum relaxant response; Imax (%), maximum relaxant response in percent of precontraction induced by U46619 (3 × 10−7 M); n, number of experiments. Statistical evaluation performed by Kruskal-Wallis test showed no statistical difference (P > 0.05) between groups. VIP, vasoactive intestinal peptide.
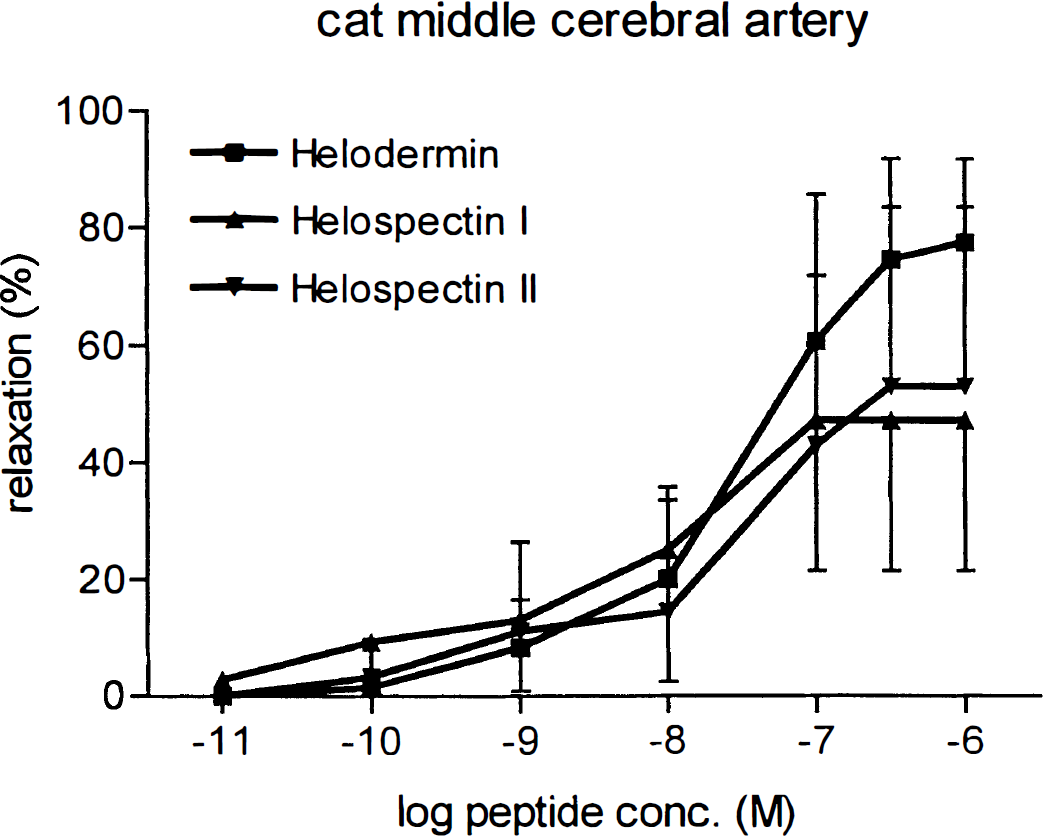
Concentration-dependent relaxation by helodermin, helospectin I, and helospectin II in cat cerebral arteries precontracted with U46619. Values represent mean ± SD; number of experiments = 8.
Endothelium removal
The removal of the endothelium was verified by the absence of relaxant response to acetylcholine in U46619 precontracted arteries (Fig. 3). Cumulative application of helodermin, helospectin I, or helospectin II to endothelium denuded vessel segments caused relaxation, which was similar in magnitude to that seen in vessels with intact endothelium (exemplified with helodermin in Fig. 4).
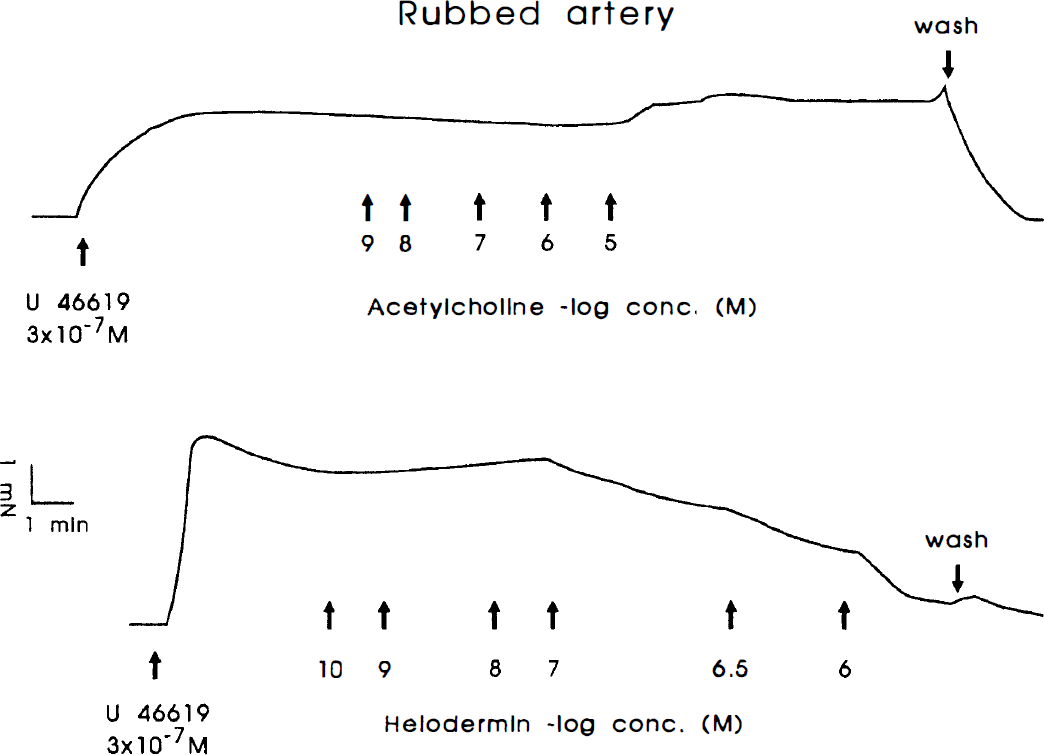
Responses to acetylcholine and helodermin of U46619-precontracted triton X-100-treated vessel segments. The upper panel shows the absence of acetylcholine induced relaxation, whereas helodermin on the same segment induced a strong relaxation.
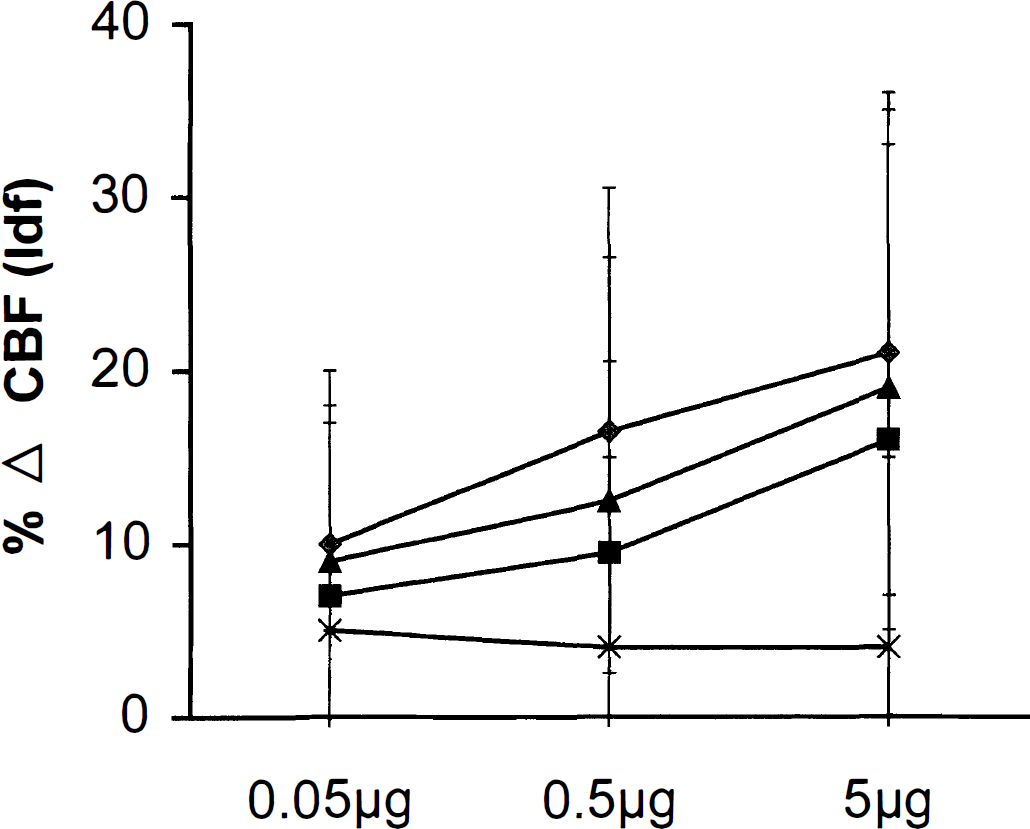
Concentration-response relation for a 1-μL injection of either vehicle, helodermin, or helospectin on CBFLDC. Doses are marked on the abscissae, and the percentage change in CBFLDF on the ordinate, compared with the immediate preinjection control period. There is no effect of vehicle (phosphate-buffered saline), nor does injection of helodermin or helospectin affect flow contralaterally after a single intracortical injection. There is a moderate, consistent concentration-related vasodilator response to helodermin or helospectin injection. (⧫) Helodermin; (▪) helospectin I; (▴) helospectin II; (X) control.
Local cerebral blood flow
The cardiovascular and blood gas parameters for the animals included in the analysis were within the normal physiologic range for the anesthetized cat. Intracerebral injection of 1 μL of phosphate-buffered saline had no effect on CBFLDF with a mean change of 5 ± 11% in the flow signal. In contrast, injections of helospectin or helodermin in the same volume of solution had moderate and consistent vasodilator effects on the CBFLDF, the onset of which was rapid. The CBF was increased for all peptides in a concentration-dependent fashion above the level seen with the vehicle injection (P < 0.01). The mean maximum increase was 21 ± 5% at 5 μg for helodermin, and 16 ± 7% and 19 ± 5% for helospectin I and II, respectively (Fig. 5). There was no significant effect on the contralateral flow signal nor was there a consistent effect on peripheral cardiovascular parameters.
DISCUSSION
The current study shows the existence of helospectin/helodermin-like peptides in nerve fibers around cerebral blood vessels. The identity of the helospectin-like peptide observed around cerebral blood vessels still is unclear. The fact that the antiserum against helospectin recognized helodermin and vice versa prevents a firm conclusion as to whether the immunoreactivity against helospectin constitutes two separate or one single peptide cross-reacting with the antisera. The presence of helospectin-like immunoreactivity in cerebral blood vessels is further strengthened by the fact that we could extract helospectin-like immunoreactivity, which co-eluted with helospectin in the radioimmunoassay from cerebral blood vessels. Helodermin- and helospectin-like immunoreactive neuronal elements have been demonstrated in the rat brain (Kivipelto et al., 1992). In the periphery, immunoreactive helodermin has been demonstrated in peripheral endocrine cells (thyroid C cells, adrenomedullary cells, and endocrine-like cells in the pulmonary parenchyma) of several species (Sundler et al., 1988; Bjartell et al., 1989; Grunditz et al., 1989; Luts et al., 1991). In the gastrointestinal tract, helospectin-like immunoreactive cell bodies can be seen in the myenteric and submucous ganglia, and helospectin-like immunoreactive fibers occur in the muscle layers, submucosa, and mucosa.
Double immunostaining for helospectin and VIP reveals their coexistence in the same population of nerve fibers and cell bodies throughout the gut (Absood et al., 1992). The coexistence of VIP- and helospectin-like immunoreactivity is intriguing, implying that the same neurons produce several homologous peptides (VIP, peptide histidine isoleucine, and helospectin-like peptides). The structural homology of these peptides suggests that they have similar biological activities (Robberecht et al., 1984; Naruse et al., 1986; Vandermeers et al., 1987).
The VIP fibers to the cerebral circulation originate in the sphenopalatine and otic ganglia and in microganglia located along the carotid artery (Suzuki et al., 1988). In the current study, nerve cell bodies containing helospectin-like immunoreactivity were seen in the sphenopalatine ganglion, which confirms and extends similar findings by Grimes and colleagues (1995). It is also likely that nerve fibers containing helospectin/helodermin-like immunoreactivity emanate from the sphenopalatine and otic ganglia. However, definite proof awaits selective lesioning and tracing studies.
Helodermin, helospectins, and VIP induced concentration-dependent relaxations of isolated middle cerebral arteries. The mean effective concentration was comparable with that previously shown for VIP or PACAP (Larsson et al., 1976; Uddman et al., 1993), although the magnitude of the maximum relaxations were slightly lower for helospectin than for helodermin and VIP. Helospectins and helodermin may reduce blood pressure in a dose-dependent manner (Naruse et al., 1986; Konturek et al., 1989; Grundemar and Högestätt, 1990). Helodermin and VIP are equipotent in relaxing rat femoral arteries, whereas helospectins show a lower potency. However, the magnitude of the maximum relaxation is similar (Grundemar and Högestätt, 1990). Thus, there may be regional variability in responses to the VIP family of peptides. Helospectin and helodermin display structural homology with VIP in 15 residues. It is therefore conceivable that VIP, helodermin, and helospectin may act on a common receptor or on a receptor group such as the VIP-PACAP family. The dilatory effects of helodermin and helospectin do not appear to be mediated by histaminergic H2, cholinergic, or β-adrenergic receptors, all of which are known vasodilator systems present in cerebral blood vessels (Edvinsson et al., 1976).
The dilator responses of the middle cerebral arteries to the VIP-like peptides were unaltered by removal of the endothelium, whereas the response to acetylcholine was absent (Edvinsson et al., 1985). Helodermin, helospectins, and PACAP, all members of the family of VIP-related peptides, have the ability to stimulate adenylyl cyclase to form cAMP (Raufman et al., 1982; Robberecht et al., 1984; Vandermeers et al., 1984; Miyata et al., 1989, 1990; Lerner et al., 1994). In addition, helodermin, like VIP, inhibits calcium incorporation in bone (Grunditz et al., 1989). We have previously shown that VIP relaxes cerebral arteries in parallel with activation of adenylyl cyclase (Edvinsson et al., 1985); whether helodermin and helospectin also do this in cerebral vessels remains to be determined. The development of specific VIP/PACAP receptor antagonists will be of considerable value in the search for the functional role for the VIP-like peptides.
Cortical microinjections of helodermin or helospectins caused a moderate but consistent increase in local cerebral perfusion. The magnitude of the flow increase was slightly less than that seen previously for VIP (Jansen-Olesen et al., 1994). Since the parasympathetic innervation involves VIP as a putative transmitter, the colocalization with helospectin-like peptides may add further information on the signal transduction in the parasympathetic control of the brain circulation (Goadsby et al., 1996). The appearance of selective antagonists seems necessary to delineate the role that each of these co-stored peptides might play in parasympathetically mediated flow responses.
