Abstract
This study assesses the effect of neuronal voltage-sensitive Ca2+ channel blockers, ω-conotoxin GVIA (CTX), and ω-agatoxin IVA (AgTX) on the vasodilation and release of calcitonin gene-related peptide (CGRP), both of which were induced by either application of capsaicin or acute stepwise hypotension. Changes in pial arterial diameter were determined directly through a closed cranial window. The vasodilation of pial artery induced by either CGRP (0.1 μmol/L) or capsaicin (0.3 μmol/L) was significantly inhibited by CGRP8–37 (0.1 μmol/L) (P < 0.05 and P < 0.05, respectively). The autoregulatory vasodilation to acute stepwise hypotension was severely attenuated by pretreatment with either CTX or AgTX. When the hypotension was kept for 2, 4, and 10 minutes, the releasable CGRP-like immunoreactivity (CGRP-LI) level (vehicle, 13.4 ± 1.5 fmol/mm2/30 min) by 10 μmol/L capsaicin from the isolated pial arteries was significantly reduced in the 4- and 10-minute hypotension groups (11.3 ± 1.2 fmol/mm2/30 min, P < 0.05, and 11.1 ± 1.5 fmol/mm2/30 min, P < 0.05, respectively), but not in 2-min group. Moreover, the CGRP-LI level released by 10 μmol/L capsaicin (13.7 ± 0.9 fmol/mm2/30 min) also was significantly depressed by pretreatment with 1 μmol/L CTX to 10.4 ± 1.0 fmol/mm2/30 min (P < 0.01) and with 0.1 μmol/L AgTX to 8.7 (1.7 fmol/mm2/30 min (P < 0.001), as well as by pretreatment with 10 μmol/L capsaicin (6.0 ± 1.6 fmol/mm2/30 min, P < 0.001). These results suggest that the neuronal N- and P-type voltage-sensitive Ca2+ channels are implicated in the release of CGRP from capsaicin-sensitive perivascular sensory nerves in response to acute hypotension, and that the released CGRP may contribute to the autoregulatory vasodilation in the cerebral microcirculation.
Keywords
Autoregulation of cerebral blood flow is an intrinsic ability of cerebral vessels to maintain constant cerebral perfusion in the face of changes in systemic arterial blood pressure (Lassen, 1959). Pial arterioles are regarded as the primary sites of autoregulatory function because they change their calibers in response to blood pressure changes (Kontos et al., 1978; MacKenzie et al., 1979). We have shown that vasodilation of rat pial arteries in response to transient systemic hypotension is partly mediated by calcitonin gene-related peptide (CGRP), which is released from the perivascular sensory fibers, and this peptide causes an increase in intracellular K+ channel-coupled cyclic AMP formation through activation of CGRP1 receptors (Hong et al., 1994; 1996, 1997). The CGRP is contained in large amounts in the cerebral perivascular sensory nerves arising from the trigeminal ganglia (McCulloch et al., 1986). Either topical application of capsaicin or pretreatment with capsaicin in neonatal rats was reported to deplete CGRP and substance P in the periarterial nerves that supply the brain (Lundberg et al., 1985).
On the other hand, Ca2+ influx has been reported as a key step in excitation-release coupling for neurotransmitter from afferent sensory nerves (Franco-Cereceda et al., 1989), and its influx is governed by diverse voltage-sensitive Ca2+ channels (VSCC). The N-, P- and Q- subtypes of neuronal Ca2+ channels (Zhang et al., 1993; Randall and Tsien, 1995) have been demonstrated as being closely related with the release of neurotransmitters (Llinas et al., 1992; Olivera et al., 1994). However, few researchers have focused their interest on the role of N- and P-type Ca2+ channels in the release of neuropeptides such as CGRP. Thus, with the availability of ω-conotoxin GVIA (CTX), a blocker of neuronal N-type VSCC (McCleskey et al., 1987), and ω-agatoxin IVA (AgTX), a blocker of P-type VSCC in the central synapses (Llinas et al., 1989; Turner et al., 1993), we aimed to explore the relative importance of N- and P-type Ca2+ channels in the release of neurotransmitters from the pial arteries.
This study first determined the effect of CGRP8–37 fragment, CTX, and AgTX on the capsaicin-induced vasodilation of the rat pial artery. If these toxins altered or eliminated the capsaicin-induced vasodilation of the rat pial artery, they also would alter the autoregulatory vasodilation of the pial artery in response to acute stepwise hypotension. Then we examined whether the autoregulatory vasodilation was altered under pretreatment with these ω-toxins as with capsaicin. In line with these studies, lastly, we measured the releasable levels of CGRP-like immunoreactivity (CGRP-LI) from the perivascular beds by application of capsaicin under pretreatment with ω-toxins.
METHODS
Preparation of animals
Male Sprague-Dawley rats (250 to 300 g) were anesthetized with urethane (1 g/kg intraperitoneally) and placed on a heating pad to maintain a constant body temperature. After a tracheostomy was performed, the trachea was cannulated and each rat was mechanically ventilated with room air by a respirator (model 683, Harvard Apparatus, South Natick, MA, U.S.A.) after immobilization with gallamine triethiodide (2 to 5 mg/kg, intravenously). Animals were anesthetized throughout terminal experiments. The depth of anesthesia was examined often by observing the effects of pressure to the paw on heart rate and blood pressure. Catheters were placed in both femoral arteries to measure the systemic arterial blood pressure (Statham P23D pressure transducer, Gould, Cleveland, OH, U.S.A.) and to sample arterial blood. Arterial blood was collected before and after installation of a cranial window for blood gas and pH determination (STAT Profile 3, Nova Biomedicals, Boston, MA, U.S.A.). The mean arterial blood gas and pH values were within normal limits as follows: pH 7.37 ± 0.08, PaCO2 31.3 ± 13.4 mm Hg, and PaO2 98.0 ± 18.4 mm Hg. Rectal temperature was kept constant (37°C) with a heating pad. Procedures and handling of the animals were reviewed and approved by the Institutional Animal Care and Use Committee of Pusan National University.
Measurement of vessel diameter
Pial microvessels were visualized through an implanted closed cranial window with a slight modification of the methods described by Levasseur and colleagues (1975) and Morii and associates (1986). Briefly, the head was fixed in prone position with a stereotaxic apparatus (Stoelting, Wood Dale, IL, U.S.A.), and a square-shaped (5–5 mm) craniotomy was made over the right parietal cortex. The dura was carefully resected, and the burr hole was covered with warmed mineral oil during the operation. Pial precapillary microvessels (35 to 50 μm) were visualized through the implanted cranial window, where prewarmed artificial CSF (37°C) saturated with a gaseous mixture of 95% O2 and 5% CO2 was constantly perfused at 0.3 mL/min. Cerebral microvessels were allowed to equilibrate for 60 minutes after installation of the cranial window. The image of pial vessels was captured with a CCD video camera (VDC 3900, Sanyo, Japan) through a stereomicroscope (model SMZ-2T, Nikon, Japan) and fed to a television monitor for direct observation, and the caliber was measured using a width analyzer (C3161, Hamamatsu Photonics, Hamamatsu, Japan). The composition (in millimolar amounts) of the artificial CSF was as follows: 132 NaCl, 2.9 KCl, 1.4 MgCl2, 24.6 NaHCO3, 1.2 CaCl2, 6.7 urea, and 3.7 D-glucose (pH 7.4). The intracranial pressure was maintained at a constant of 5 to 6 mm Hg throughout the experiment by adjusting the height of the free end of the plastic tubing, which was connected to the outlet of the window. In each rat, only one arteriole was observed under the window.
Measurement of calcitonin gene-related peptide-like immunoreactivity
Two experiments were performed. In the first, after operation the systemic arterial blood pressure of the anesthetized rats was reduced to lower than 50% of the initial blood pressure by bleeding of the blood into the reservoir, and the reduced state was kept for 2, 4, and 10 minutes; the pial arteries then were quickly isolated. In the second experiment, the intact pial arteries were isolated from the anesthetized rats without bleeding procedure. The animals were anesthetized throughout the procedures. The isolated pial arteries were equilibrated in the aerated Krebs buffer solution containing (in millimolar amounts) 130 NaCl, 4.7 KCl, 1.6 CaCl2, 1.18 NaH2PO4, 14.9 NaHCO3, and 5.5 D-glucose for 1 hour at 37°C. Thereafter, the strips were preincubated in the Ca2+-free Krebs buffer solution containing 0.1 mmol/L EGTA for 30 minutes, and thereafter incubated again in the Krebs solution in the presence of a vehicle (Krebs buffer solution), CTX, AgTX, and capsaicin (10 μmol/L of each) for 30 minutes each. Additionally, 10 μmol/L capsaicin was applied to facilitate the release of CGRP from the arteries. The reaction was stopped by adding the assay buffer solution (a 50-mmol/L sodium phosphate buffer containing 80 mmol/L NaCl, 0.1% bovine serum albumin, 0.05% NaN3, and 0.1% Triton X-100, 25 mmol/L Na2-ethylenediamine tetra-acetic acid (Na2EDTA), and 500 kallikrein-inhibiting units/mL Trasylol; pH 7.4). The strips then were removed for measurement of the surface area of the pial artery. The CGRP-LI content in the buffer solution was determined by radioimmunoassay method as described by Fujimori and others (1989). Briefly, the samples were preincubated with rabbit anti-human CGRP serum at 4°C for 10 to 12 hours. The reaction mixture then was incubated with (2-[125I]iodohistidyl10) CGRP (human) for an additional 24 to 36 hours at 4°C. The incubation buffer for the radioimmunoassay consisted of 50 mmol/L sodium phosphate buffer (pH 7.4) containing 80 mmol/L NaCl, 25 mmol/L Na2EDTA, 0.05% NaN3, and 500 kallikrein-inhibiting units/mL Trasylol.
The antibody-bound antigen was separated from free antigens by incubation with the goat anti-rabbit IgG serum and normal rabbit serum for 2 hours at room temperature. After addition of the radioimmunoassay buffer, the samples were centrifuged and the radioactivity in the pellets was counted using a gamma counter (Wizard 1470, Wallac, Finland).
After terminating the incubation, the microvessel sample was removed on the slide glass and stained with 2.5 mmol/L methylene blue. The magnified surface areas of the vessels were measured with planimeter (type KP-21, Koizumi, Japan). The released CGRP-LI was expressed as fmol/mm2/30 min.
Protocol of in vivo experiments
First, to examine the vasodilating responses of the resting pial arteries to CGRP and capsaicin, the cortical surface was suffused with artificial CSF containing either CGRP (0.1 μmol/L) or capsaicin (0.3 μmol/L).
Second, two groups of adult rats (12 to 14 weeks old) were used for capsaicin treatment. One group was treated with capsaicin (3 μg/kg) intracisternally 5 hours before the experiment under urethane anesthesia; they were maintained in the anesthetized state. The other group comprised the age-matched adult rats neonatally treated with capsaicin to destroy small unmyelinated sensory fibers (Jancso et al., 1977). Rat pups were injected with capsaicin subcutaneously (50 mg/kg in a volume of 0.1 mL) over the lower back within the first 48 hours after birth without anesthesia. After the injection, each neonate was placed in a small chamber for up to 1 hour into which flowed a fine mist of a 0.25% solution of isoproterenol through a nebulizer.
Third, the inhibitory effects of CGRP8–37 (0.1 μmol/L), CTX (1 μmol/L), and AgTX (0.1 μmol/L) were observed for the CGRP- and capsaicin-induced vasodilation. The inhibitors were previously applied from 30 minutes before suffusion of either CGRP or capsaicin.
Lastly, we initially examined the normal autoregulatory function of the pial artery to a stepwise hypotension and its reverse under suffusion with an artificial CSF-containing vehicle. Afterward, we retested the autoregulatory responses under pretreatment with ω-toxins. Stepwise hypotension was induced by controlled withdrawal of blood into the reservoir. The decreased blood pressure at each step was maintained for approximately for 2 minutes, and vessel diameter was measured during the last minute. The toxins were applied 30 minutes before inducing hypotension. Alterations in pial arterial diameter and blood pressure were expressed as the percent change of the baseline diameter and blood pressure.
Drugs
Calcitonin gene-related peptide (human), CGRP8–37 (human alpha) fragment, rabbit anti-human CGRP serum, CTX, and AgTX were purchased from Peninsula Laboratories, Inc. (Belmont, CA, U.S.A.). The (2-[125I]iodohistidyl10) CGRP was purchased from Amersham International plc (Buckinghamshire, U.K.). CGRP, CTX, and AgTX were dissolved in 0.1% bovine serum albumin, and CGRP8–37 was dissolved in 1% acetic acid to make a stock solution of 0.1 mmol/L. Capsaicin (Sigma Chemical Co., St. Louis, MO, U.S.A.) was dissolved in the mixture of Tween 80:ethanol:normal saline (1:1:8 v/v).
Statistical analysis
Results are expressed as means ± SD. Student's t test was performed for the differences between the results of vehicle and toxin-treated groups, and between the slopes of linear regression lines, which were plotted with the changes in pial arterial diameter as a function of changes in mean arterial blood pressure. The difference in time relation of arteriolar diameter between groups was analyzed by analysis of variance with repeated measures followed by Tukey's multiple comparisons test. A value of P < 0.05 was accepted as statistically significant.
RESULTS
Under control conditions, mean arterial blood pressure was similar in the various groups of animals (124.3 ± 12.9 mm Hg, n = 18). The resting pial arterial diameter was 39.6 ± 5.4 μm, which remained constant throughout the experiment unless there was a bleeding or an infusion of blood. When the cortical surface was suffused with artificial CSF containing either CGRP (0.1 μmol/L) or capsaicin (0.3 μmol/L), each caused a sustained vasodilation of the resting pial arteries for more than 5 minutes (Fig. 1). In response to CGRP and capsaicin, the maximum diameters of the pial arteries under cranial window increased by 119.0 ± 31.6% (n = 7) and 89.3 ± 26.2% (n = 9), respectively.
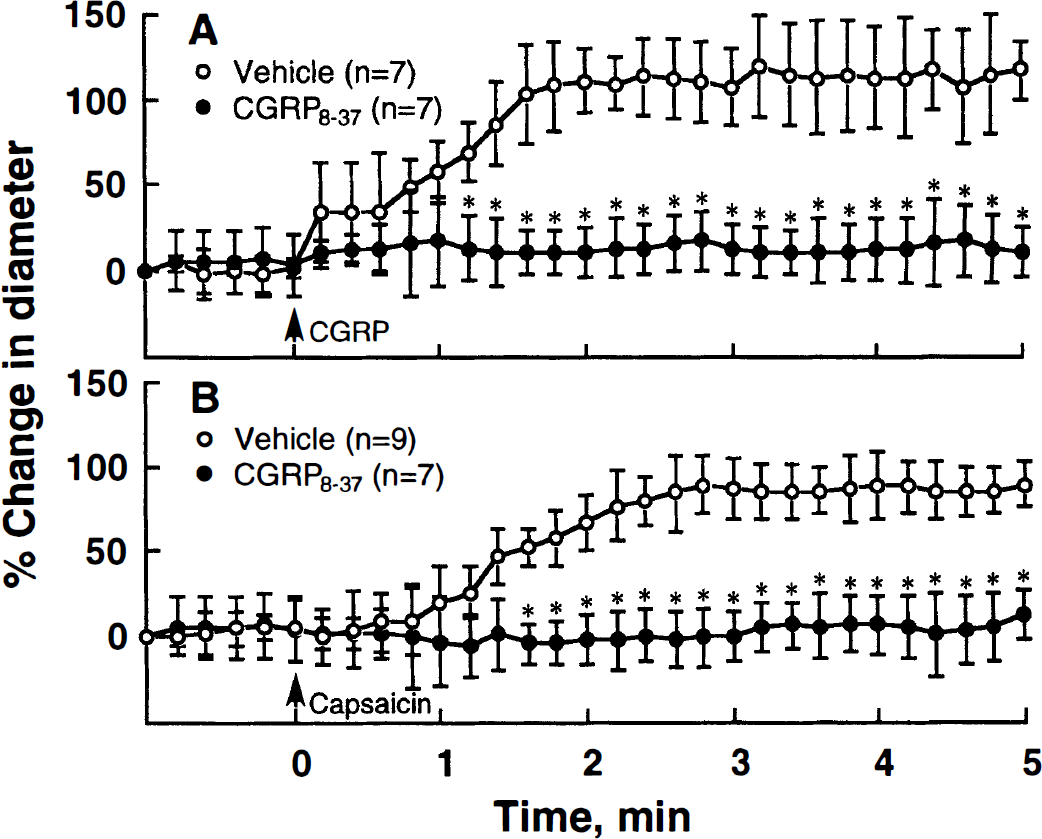
Graphs showing the percent changes in the diameter of the pial arteries in response to calcitonin gene-related peptide (CGRP)
Effect of CGRP8–37 on capsaicin-induced vasodilation
Under previous suffusion with artificial CSF containing the 0.1 μmol/L CGRP8–37 fragment, the capsaicin-induced vasodilation was significantly inhibited (P < 0.05), as was the CGRP-induced vasodilation (P < 0.05) (Fig. 1). When the cortical surface was suffused with normal artificial CSF for 30 minutes, the diameter almost returned to the baseline level. Thereafter, reapplication of CGRP again evoked a similar magnitude of vasodilation. However, in the case of capsaicin, the vessel showed almost complete desensitization on reapplication of the same concentration of capsaicin.
Effect of ω-toxins on capsaicin-induced vasodilation
Capsaicin-induced vasodilation was significantly inhibited under suffusion with either 1 μmol/L CTX (P < 0.05) or 0.1 μmol/L AgTX (P < 0.05) (Fig. 2). Suffusion with artificial CSF containing 1 μmol/L CTX at 30 minutes before and during experiment showed little change in the resting pial arterial diameter; however, AgTX at 0.1 μmol/L caused a marginal increase in baseline diameter by 14.6 ± 10.1% (Table 1).
Mean arterial blood pressure and baseline diameter of pial arteries before and after various treatments
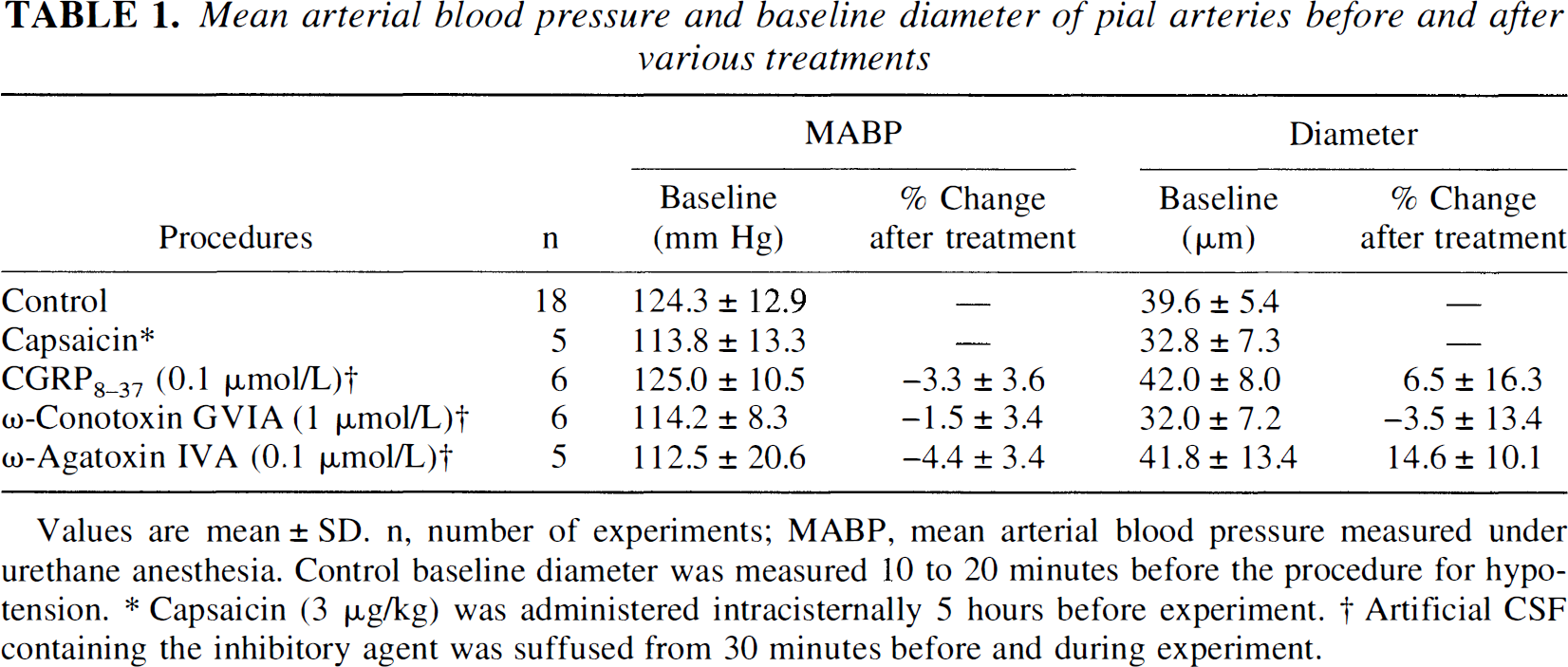
Values are mean ± SD. n, number of experiments; MABP, mean arterial blood pressure measured under urethane anesthesia. Control baseline diameter was measured 10 to 20 minutes before the procedure for hypotension.
Capsaicin (3 μg/kg) was administered intracisternally 5 hours before experiment.
Artificial CSF containing the inhibitory agent was suffused from 30 minutes before and during experiment.
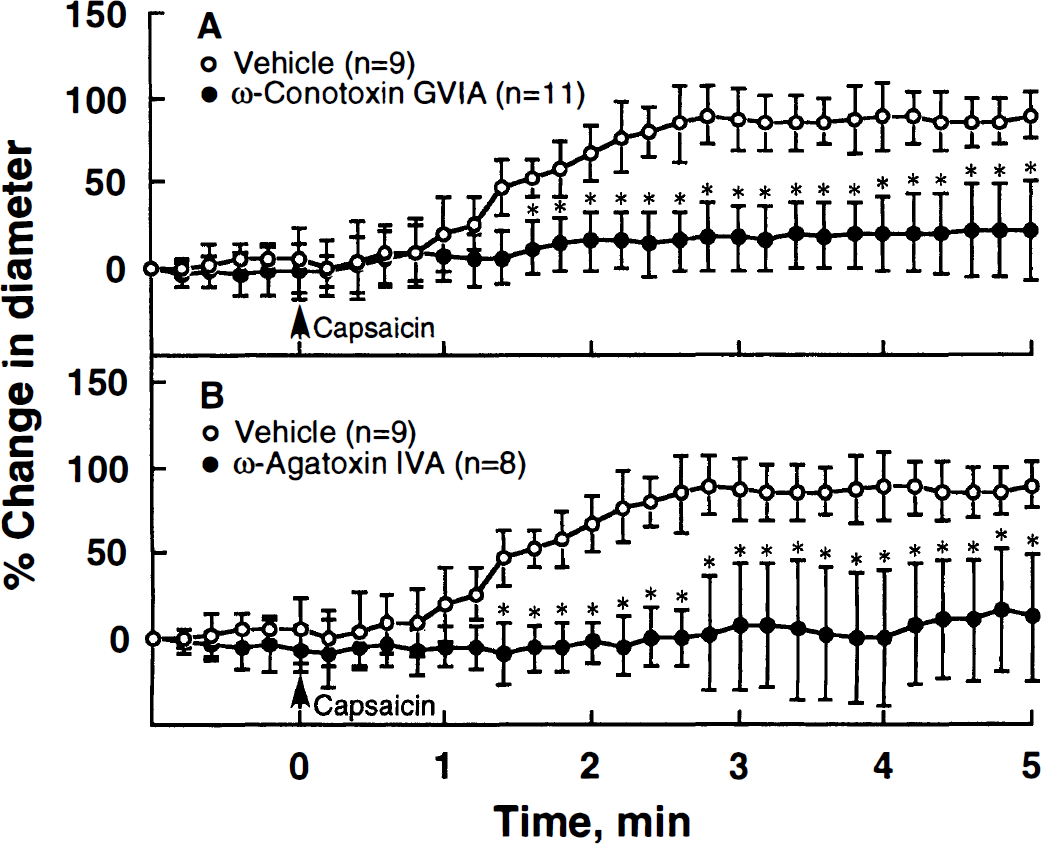
Graphs showing the percent changes in the diameter of pial arteries in response to capsaicin (0.3 μmol/L, open circle) under pretreatment (closed circle) with ω-conotoxin GVIA (1 μmol/L)
Alteration in autoregulation by capsaicin
The alteration in autoregulatory vasodilation in response to a stepwise hypotension was observed after capsaicin pretreatment (Fig. 3). Changes in pial arterial diameter were plotted as a function of changes in mean arterial blood pressure. In the vehicle group, the pial arterial diameter increased during stepwise hypotension and quickly decreased on recovery of the arterial pressure. In this experiment, two methods for capsaicin treatment were employed: one was the topical intracisternal treatment with capsaicin 5 hours before experiment (Fig. 3B), and the other was treatment with capsaicin in neonatal age (Fig. 3C). In the rats either topically treated or systemically treated during neonatal age, the autoregulatory vasodilator and vasoconstrictor responses of the pial arteries were markedly attenuated or almost eliminated. The slopes for vasodilation phase (vehicle group, −1.78 ± 1.32) were markedly increased to −0.26 ± 0.86 (P < 0.05) for the topically treated group and −0.02 ± 0.34 (P < 0.01) for the neonatally treated group (Table 2).
Changes in slopes of linear regression lines during hypotension and reverse of blood pressure
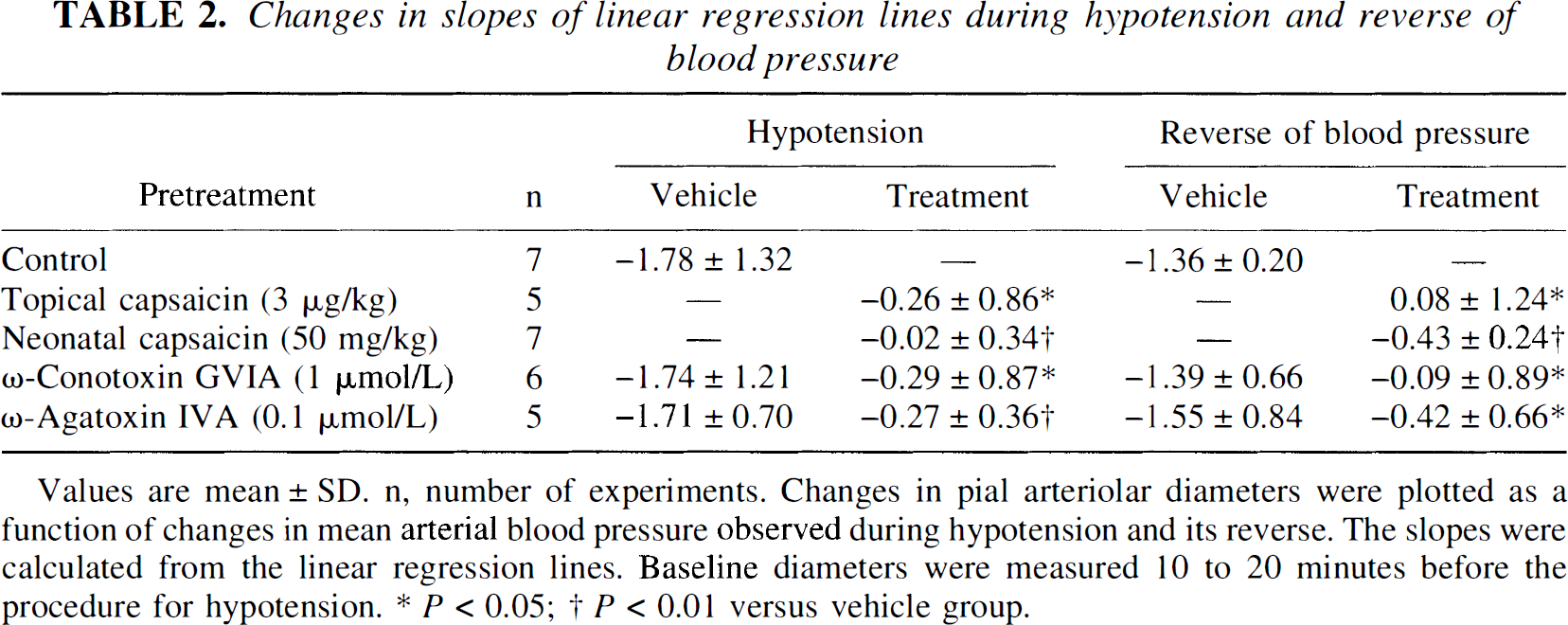
Values are mean ± SD. n. number of experiments. Changes in pial arteriolar diameters were plotted as a function of changes in mean arterial blood pressure observed during hypotension and its reverse. The slopes were calculated from the linear regression lines. Baseline diameters were measured 10 to 20 minutes before the procedure for hypotension.
P < 0.05;
P < 0.01 versus vehicle group.
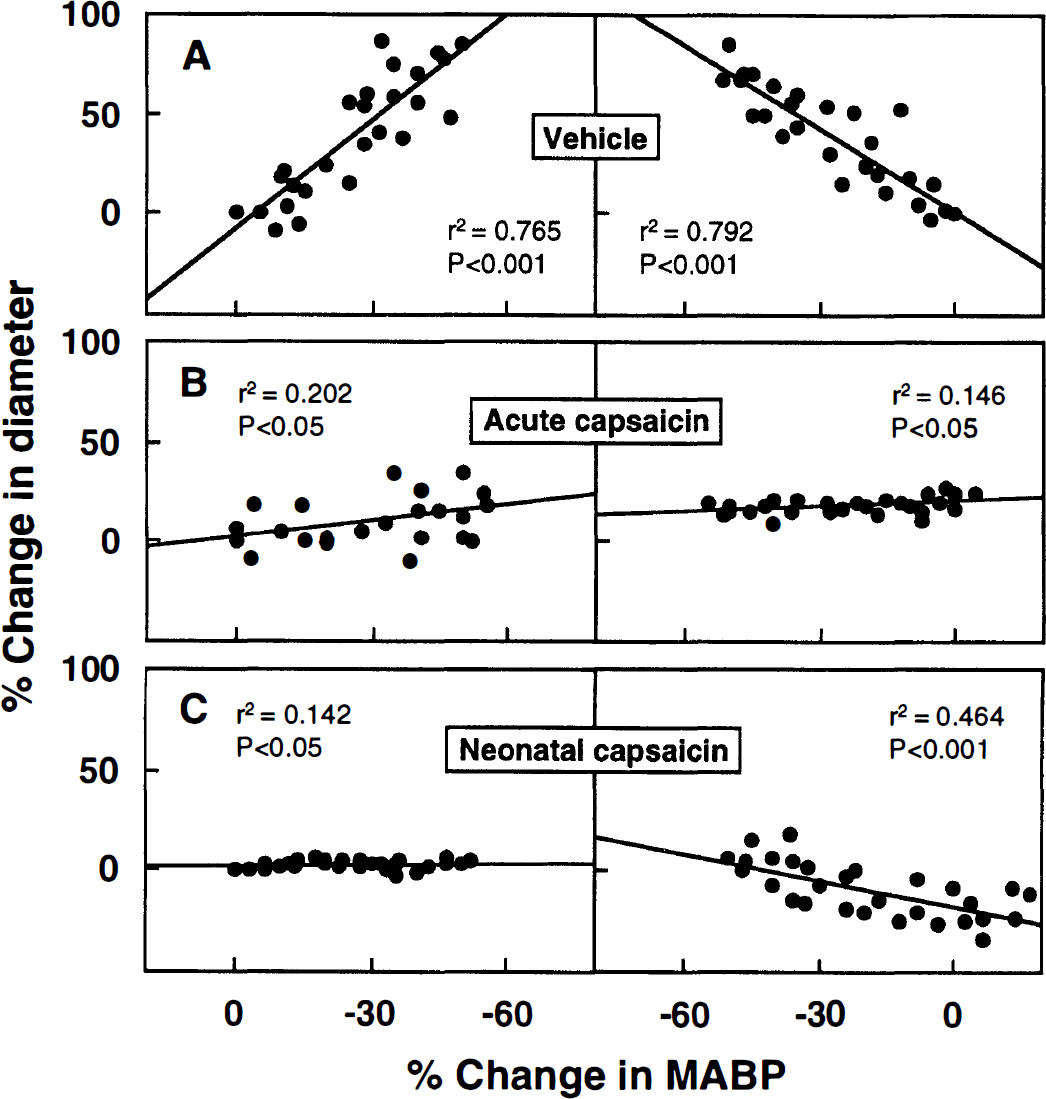
Graphs showing the alterations in the autoregulatory vasodilator and vasoconstrictor responses after capsaicin treatment in comparison with vehicle group
Effect of ω-toxins on the autoregulation
Under suffusion of cortical surface with artificial CSF containing 1 μmol/L CTX at 30 minutes before and during the stepwise bleeding, the slope of regression line for vasodilation phase significantly increased from −1.74 ± 1.21 (vehicle) to −0.29 ± 0.87 (P < 0.05), and the slope for vasoconstriction phase changed from −1.39 ± 0.66 (vehicle) to −0.09 ± 0.89 (P < 0.05) (Fig. 4, Table 2).
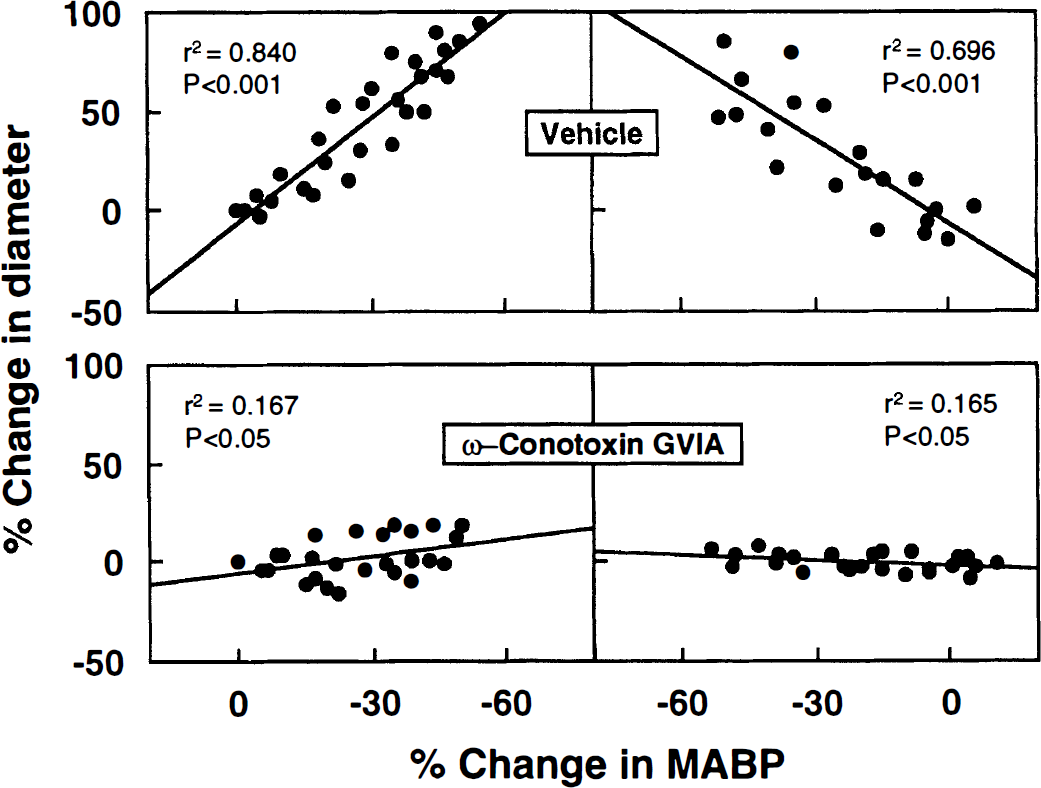
Graphs showing alterations in the autoregulatory responses under pretreatment with ω-conotoxin GVIA. After the autoregulation was confirmed, the cortical surface was suffused with artificial CSF containing ω-conotoxin GVIA (1 μmol/L) from 30 minutes before and during stepwise hypotension. ω-Conotoxin GVIA significantly attenuated the autoregulatory vasodilation. A significant difference (P < 0.05) was shown between the blood pressure-diameter relation of vehicle-treated and ω-conotoxin-treated groups by two-way repeated measures analysis of variance.
Likewise, under application of 0.1 μmol/L AgTX, the autoregulatory vasodilation and vasoconstriction responses were significantly attenuated, as evidenced by the increased slopes of regression lines (vasodilation phase from −1.71 ± 0.35 to −0.27 ± 0.36, P < 0.01; vasoconstriction phase from −1.55 ± 0.84 to −0.42 ± 0.66, P < 0.05) (Fig. 5, Table 2).
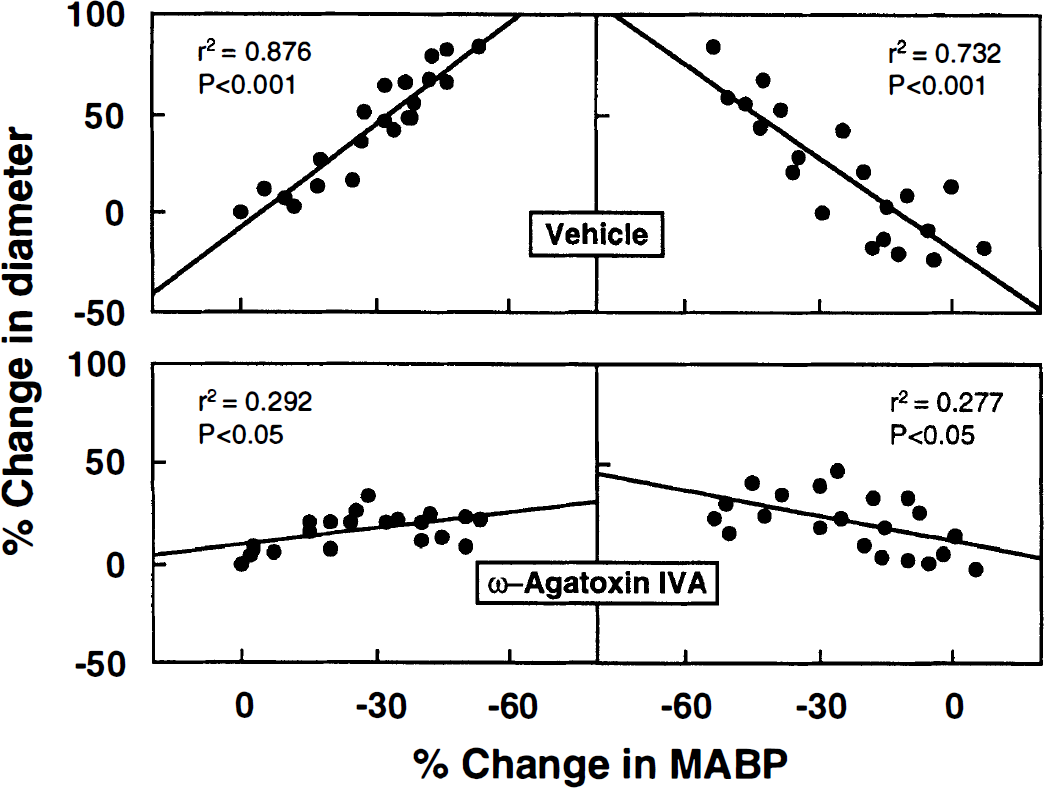
Graphs showing alterations in autoregulatory responses under pretreatment with ω-agatoxin IVA. Under suffusion with ω-agatoxin IVA, the autoregulatory responses determined were severely attenuated. A significant difference (P < 0.05) was identified between the blood pressure-diameter relation of vehicle-treated and ω-agatoxin-treated groups by two-way repeated measures analysis of variance.
Effect of ω-toxins on capsaicin-induced calcitonin gene-related peptide release
We determined the time course effect of hypotension on the capsaicin-induced release of CGRP from the pial arteries (Fig. 6). When the reduced state of the systemic blood pressure was kept for 2, 4, and 10 minutes, and the pial arteries then were quickly isolated, CGRP-LI level released by 10 μmol/L capsaicin (13.4 ± 1.5 fmol/mm2/30 min) was little changed in the 2-minute hypotension group (14.8 ± 3.0 fmol/mm2/30 min) but significantly reduced in both the 4-minute (11.3 ± 1.2 fmol/mm2/30 min, P < 0.05) and 10-minute hypotension (11.1 ± 1.5 fmol/mm2/30 min, P < 0.05) groups.
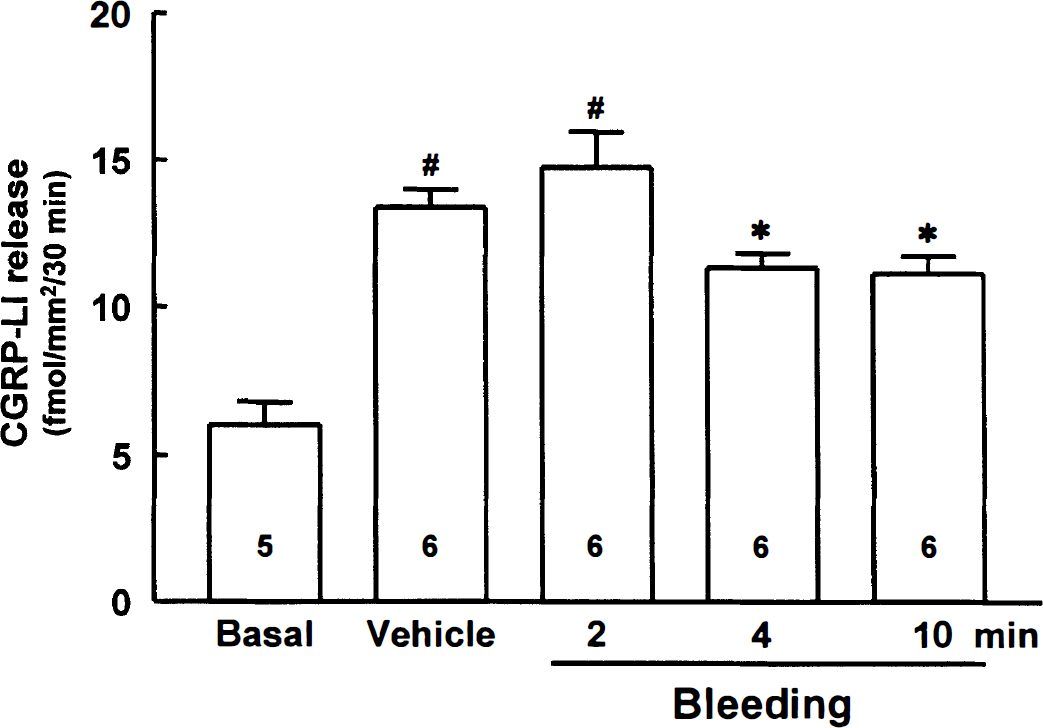
Release of CGRP-like immunoreactivity (CGRP-LI) from the pial arteries isolated from the rats that were subjected to hypotension. The systemic arterial blood pressure of the anesthetized rats was reduced to lower than 50% of the initial blood pressure; the hypotensive stage was kept for 2, 4, and 10 minutes; and then the pial arteries were quickly isolated. All values are means ± SD from the numbers within the columns. Basal represents the released CGRP-LI level in the absence of capsaicin (10 μmol/L). #P < 0.001 versus basal; *P < 0.05 versus vehicle.
Further, effect of CTX and AgTX on the capsaicin-induced release of CGRP-LI from the pial arteries was assessed (Fig. 7). The baseline release (8.2 ± 1.7 fmol/mm2/30 min) was significantly increased by 10 μmol/L capsaicin to 13.7 ± 0.9 fmol/mm2/30 min (P < 0.001), which was significantly depressed either by CTX to 10.4 ± 1.0 fmol/mm2/30 min (P < 0.01) or by AgTX to 8.7 ± 1.7 (P < 0.001). When the arteries were previously treated with 10 μmol/L capsaicin, the releasable CGRP-LI was markedly reduced to 6.0 ± 1.6 fmol/mm2/30 min (P < 0.001).
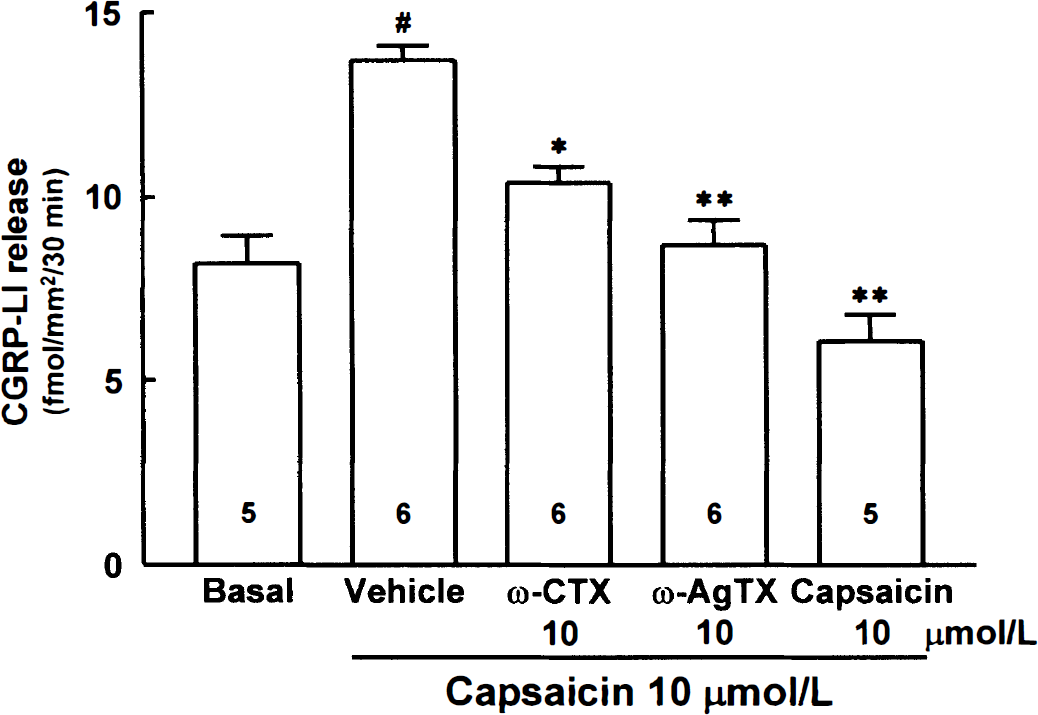
Effects of ω-conotoxin GVIA (ω-CTX), ω-agatoxin IVA (ω-AgTX), and capsaicin on the release of CGRP-like immunoreactivity (CGRP-LI) from the rat pial arteries. All values are means ± SD from the numbers within the columns. Basal represents the released CGRP-LI level in the absence of capsaicin (10 μmol/L). #P < 0.001 versus basal; *P < 0.01 and **P < 0.001 versus vehicle.
DISCUSSION
Two major new findings were identified in the current study. First, the capsaicin-induced vasodilation and the autoregulatory vasodilation of the pial arteries in response to stepwise hypotension were markedly attenuated or almost eliminated under suffusion with artificial CSF containing CTX or AgTX. Second, the capsaicin-induced release of CGRP in the in vitro experiment also was significantly reduced by the ω-toxins.
Calcitonin gene-related peptide is a potent vasodilating peptide that relaxes cerebral arteries in vitro (Edvinsson et al., 1987). The cortical pial arteries and the large cerebral arteries are reportedly invested with CGRP-like immunoreactive nerve fibers originating from trigeminal sensory ganglia (Edvinsson, 1985; McCulloch et al., 1986). Several reports propose that CGRP has a role of neurotransmitter in the nonsympathetic neurogenic vasodilation of the cerebral vascular beds (Bevan and Brayden, 1987; Brian et al., 1996). Our previous results (Hong et al., 1994) showed that the vasodilation of rat pial arteries in response to a stepwise hypotension is partly mediated by CGRP, which is released from the perivascular sensory fibers. This speculation is based on the results that the autoregulatory vasodilation in response to hypotension was attenuated by suffusion with specific CGRP antibody serum or by CGRP receptor desensitization. However, it is not clear whether the mechanisms underlying the autoregulatory vasodilation evoked during stepwise hypotension are linked to the neurogenic control and whether the release of CGRP is coupled to Ca2+ influx governed by neuronal VSCC.
Capsaicin, a pungent algesic substance, has primary effect on the unmyelinated C-fiber that carries sensory signals (Buck and Burks, 1986). Caterina and colleagues (1997) report that capsaicin causes depolarization of the membrane by stimulating the capsaicin receptor, which is a nonselective cation channel structurally related to the members of transient receptor potential family of ion channels. Capsaicin increases the release of neuropeptides, substance P and CGRP from the peripheral nerve endings (Holzer, 1991; Stretton et al., 1992). The CGRP coexists with substance P in the pial arteries (Edvinsson, 1985; Hanko et al., 1985), and both peptides are depleted by application of capsaicin (Lundberg et al., 1985). The finding of our study that the vasodilating action of capsaicin was inhibited by CGRP8–37 fragment, a specific CGRP1 receptor antagonist (Han et al., 1990; Mimeault et al., 1991), indicates that the vasodilation induced by capsaicin was ascribed to the CGRP released from the perivascular sensory nerve, as reported by Lundberg and colleagues (1985). Capsaicin, when given to newborn rats (within 48 hours after birth), is known to cause a permanent and selective degeneration of the peripheral unmyelinated primary sensory neurons in up to 90% (Nagy et al., 1981; Hammond and Ruda, 1989) with diminished CGRP and substance P levels in the neurons (Gamse et al., 1980; Hammond and Ruda, 1989). Our results that the release of CGRP-LI by capsaicin was significantly reduced by long-standing hypotension (4 to 10 minutes) suggest that the releasable amount of CGRP from the perivascular bed was severely exhausted by the hypotensive stress. In line with this speculation, the finding that the autoregulatory vasodilator adjustment in response to hypotension was almost eliminated in the capsaicin-treated rats provides an additional evidence to support the hypothesis that the local sensory implication may dominate in the neurogenic vasodilation of the pial arteries during acute hypotension.
On the other hand, the entry of Ca2+ into the presynaptic nerve endings through VSCC is reportedly essential for the release of neurotransmitters within the nervous system (Mulkey and Zucker, 1991). In our study, the fact that both capsaicin-induced vasodilation and the autoregulatory vasodilation in response to hypotension were inhibited by CTX, a blocker of N-type Ca2+ channels (Olivera et al., 1984), and AgTX, a blocker of P- and Q-type Ca2+ channels (Randall and Tsien, 1995), suggests that both N- and P-type Ca2+ channels are implicated in controlling the Ca2+ influx and CGRP release from the perivascular sensory nerve endings in the pial microvessels.
The fact that the capsaicin-induced CGRP release was significantly inhibited by either CTX or AgTX indicates that the depolarizing action of capsaicin is coupled to Ca2+ influx through the N- and P-type VSCC of the sensory nerve terminals. These speculations are consistent with other reports that multiple Ca2+ channel subtypes coexist to support transmitter release from the presynaptic endings (Miljanich and Ramachandran, 1995).
Taken together, it appears that neuropeptide-containing capsaicin-sensitive sensory nerves exert a potential contribution to the vasodilator phase of the cerebral autoregulation in response to acute stepwise hypotension. Considering the inhibitory effects of CGRP8–37, CTX, AgTX, and capsaicin on the cerebral autoregulatory vasodilatory responses to acute hypotension and capsaicin-induced CGRP release, it is likely that the release of CGRP from capsaicin-sensitive perivascular sensory nerves prominently contribute to the autoregulatory vasodilator hemodynamics in response to a transient hypotension.
