Abstract
Astrocytes play a pivotal role in cerebral glutamate homeostasis. After 90 minutes of middle cerebral artery occlusion in the rat, the changes induced in neuronal and astrocytic metabolism and in the neuronal—astrocytic interactions were studied by combining in vivo injection of [1-13C]glucose and [1,2-13C]acetate with ex vivo 13C nuclear magnetic resonance spectroscopy and HPLC analysis of amino acids of the lateral caudoputamen and lower parietal cortex, representing the putative ischemic core, and the upper frontoparietal cortex, corresponding to the putative penumbra. In the putative ischemic core, evidence of compromised de novo glutamate synthesis located specifically in the glutamatergic neurons was detected, and a larger proportion of glutamate was derived from astrocytic glutamine. In the same region, pyruvate carboxylase activity, representing the anaplerotic pathway in the brain and exclusively located in astrocytes, was abolished. However, astrocytic glutamate uptake and conversion to glutamine took place, and cycling of intermediates in the astrocytic tricarboxylic acid cycle was elevated. In the putative penumbra, glutamate synthesis was improved compared with the ischemic core, the difference appeared to be brought on by better neuronal de novo glutamate synthesis, combined with normal levels of glutamate formed from astrocytic glutamine. In both ischemic regions, γ-aminobutyric acid synthesis directly from glucose was reduced to about half, indicating impaired pyruvate dehydrogenase activity; still, γ-aminobutyric acid reuptake and cycling was increased. The results obtained in the current study demonstrate that by combining in vivo injection of [1-13C]glucose and [1,2-13C]acetate with ex vivo 13C nuclear magnetic resonance spectroscopy, specific metabolic alterations in small regions within the rat brain suffering a focal ischemic lesion can be studied.
Keywords
The neuron—its pathophysiology and pharmacology—has been the focus of cerebral ischemia research. Elevated extracellular glutamate (Glu) levels resulting in excessive and prolonged stimulation of Glu receptors is considered to initiate a cascade of detrimental biochemical events, ultimately leading to neuronal death from ischemia. Under normal conditions, astrocytes are intimately involved in the brain's Glu homeostasis (Hertz, 1979). Glu released as a neurotransmitter is taken up primarily into astrocytes (Erecinska, 1987) and converted to glutamine (Gln) by the glia specific enzyme glutamine synthetase (enzyme commission 6.3.1.2). Glutamine synthetase is predominantly astrocytic (Norenberg and Martinez-Hernandez, 1979) but also can be found in oligodendrocytes (Tansey et al., 1991). However, Gln—from astrocytic tricarboxylic acid (TCA) cycle activity or synthesized from Glu taken up from the extracellular space—leaves the astrocytes and is taken into the neuronal terminals to replenish the Glu pool and the neuronal TCA cycle. The neuronal TCA cycle lacks an anaplerotic pathway and is therefore continuously drained from neurotransmitter production and release, consequently rendering the neurons dependent on metabolic support from astrocytes (Shank et al., 1985; Kaufmann and Driscoll, 1993). In the CNS, pyruvate carboxylase (PC; E.C. 6.4.1.1) is the principal anaplerotic enzyme (Patel, 1974) and is localized in astrocytes (Shank et al., 1985).
The effects ischemia exerts on neuronal—astrocytic interactions still remain to be elucidated. Morphologic studies reveal early and progressive structural alterations in response to ischemia in both neurons and astrocytes. Based on differences in the temporal sequence and the magnitude of structural alterations, neurons are considered to be more vulnerable to ischemia than astrocytes (Garcia et al., 1977). In vitro observations of neuronal and astrocytic metabolism under ischemic conditions are conflicting. Some evidence indicates astrocytic metabolism to be more severely affected by hypoxia than neuronal metabolism (Sonnewald et al., 1994). Yet the presence of astrocytes has been shown to improve neuronal survival after exposure to anoxia (Vibulsreth et al., 1987). And finally, some experimental findings connect astrocytic dysfunction per se to neuronal death under ischemic conditions (Largo et al., 1995).
This study explores the metabolic perturbation in the putative ischemic core and penumbra after 90 minutes of middle cerebral artery occlusion (MCAO) in the rat, with emphasis on astrocytic function and the possible consequences that alterations in the interaction between astrocytes and neurons have for neuronal survival. In vivo infusion of 13C metabolites combined with ex vivo 13C nuclear magnetic resonance spectroscopy (NMRS) of distinct anatomical regions in the CNS subjected to in vivo focal injury has not been undertaken before our pilot study of the ischemic core in rats with MCAO (Sonnewald et al., 1996).
MATERIALS AND METHODS
All experimental procedures were approved by the Norwegian Committee for Animal Experiments.
Animal surgery and sample preparation
Six male Wistar rats (Møllegaard Breeding Center, Copenhagen, Denmark) weighing 320 to 340 g were fasted overnight. Anesthesia was induced with 3.5% isoflurane in 70%/30% N2/O2. During surgery, the animals were spontaneously breathing, anesthetized with 2% isoflurane in 70%/30% N2/O2 delivered through a close-fitting snout mask. The temperature was maintained at 37°C using a feedback-controlled heating blanket connected to a rectal temperature probe. The rats underwent MCAO using a modified intraluminal filament technique. Briefly, the right external carotid artery is ligated, and through the proximal end a nylon filament with a diameter of 0.27 mm and a rounded tip is introduced. The filament is pushed into the internal carotid artery and advanced 19 to 20 mm, thereby blocking the origin of the middle cerebral artery. A catheter was introduced into the right femoral vein, externalized at the tail radix, and taped in place. All wounds were sprayed with lidocaine (10-mg doses) before suturing. After surgery, the animals were returned to individual cages and allowed to recover from anesthesia. Seventy-five minutes after MCAO, the fully awakened animals received an intravenous infusion of a 0.3 mmol/L solution (1 mL/100 g rat) of sodium-[1,2-13C]acetate and [1-13C]glucose (Cambridge Isotope Laboratories, Woburn, MA, U.S.A.) dissolved in water. The infusion was given over 2 minutes. Fifteen minutes later (ie, 90 minutes after MCAO), the animals were decapitated and the heads snap-frozen in liquid nitrogen. The brains were removed under intermittent nitrogen irrigation. From the brains, a 3-mm coronal slice extending caudally from the chiasma opticum was cut using a brain tissue matrix (RBM-40000, Activational Systems, Warren, MI, U.S.A.). The lateral caudoputamen and lower parietal cortex weighing 0.07 ± 0.02 g (mean ± SD), and the upper frontoparietal cortex weighing 0.03 g ± 0.01 g, were sampled from both hemispheres and stored at −80°C. The lateral caudoputamen and lower parietal cortex correspond to the putative ischemic core, whereas the upper frontoparietal cortex represents the putative penumbra in the hemisphere ipsilateral to the MCAO. The brain samples were homogenized in 1 mL 70% ethanol and centrifuged at 4000 g for 5 minutes. This procedure was repeated twice, with the supernatant retained every time. The supernatant then was lyophilized and stored at −20°C. Before 13C NMRS, the samples were redissolved in 0.5 mL 99.9% D2O (Fluorochem Limited, Old Glossop, U.K.) containing 0.1% dioxane (C4H8O2, Fluka Chemiæe AG, Buchs, Switzerland) as a chemical shift and quantification reference. Blood was collected from the severed neck vessels and immediately centrifuged, and the plasma was retained and frozen at −80°C. One milliliter of 7% perchloric acid (w/v) was added to 1 mL plasma and centrifuged at 4000 g for 10 minutes. The supernatants were neutralized with KOH before lyophilization. Plasma samples were redissolved in 0.5 mL D2O containing 0.1% dioxane before 1H and 13C NMRS. Before spectroscopy, pH was adjusted to 6.5 to 7.5 in all samples.
13C nuclear magnetic resonance spectroscopy
Proton-decoupled 125.5-MHz 13C NMR spectra were obtained on a Bruker DRX-500 spectrometer. The spectra were accumulated using a 35° pulse angle, with 25-kHz spectral width and 64-K data points. The acquisition time was 1.3 seconds per scan, plus 5.0 seconds for relaxation delay. The number of scans was typically 600 for blood and 8000 for the brain samples. To avoid nuclear overhauser effects, some spectra underwent broadband decoupling during acquisition only. The respective nuclear overhauser effect factors then were applied to the spectra obtained from corresponding samples. Correction for T1, was not necessary with a 5.0-second relaxation delay. Typical 13C spectra obtained from the lateral caudoputamen and lower parietal cortex and from the upper frontoparietal cortex are shown in Figs. 1 and 2.
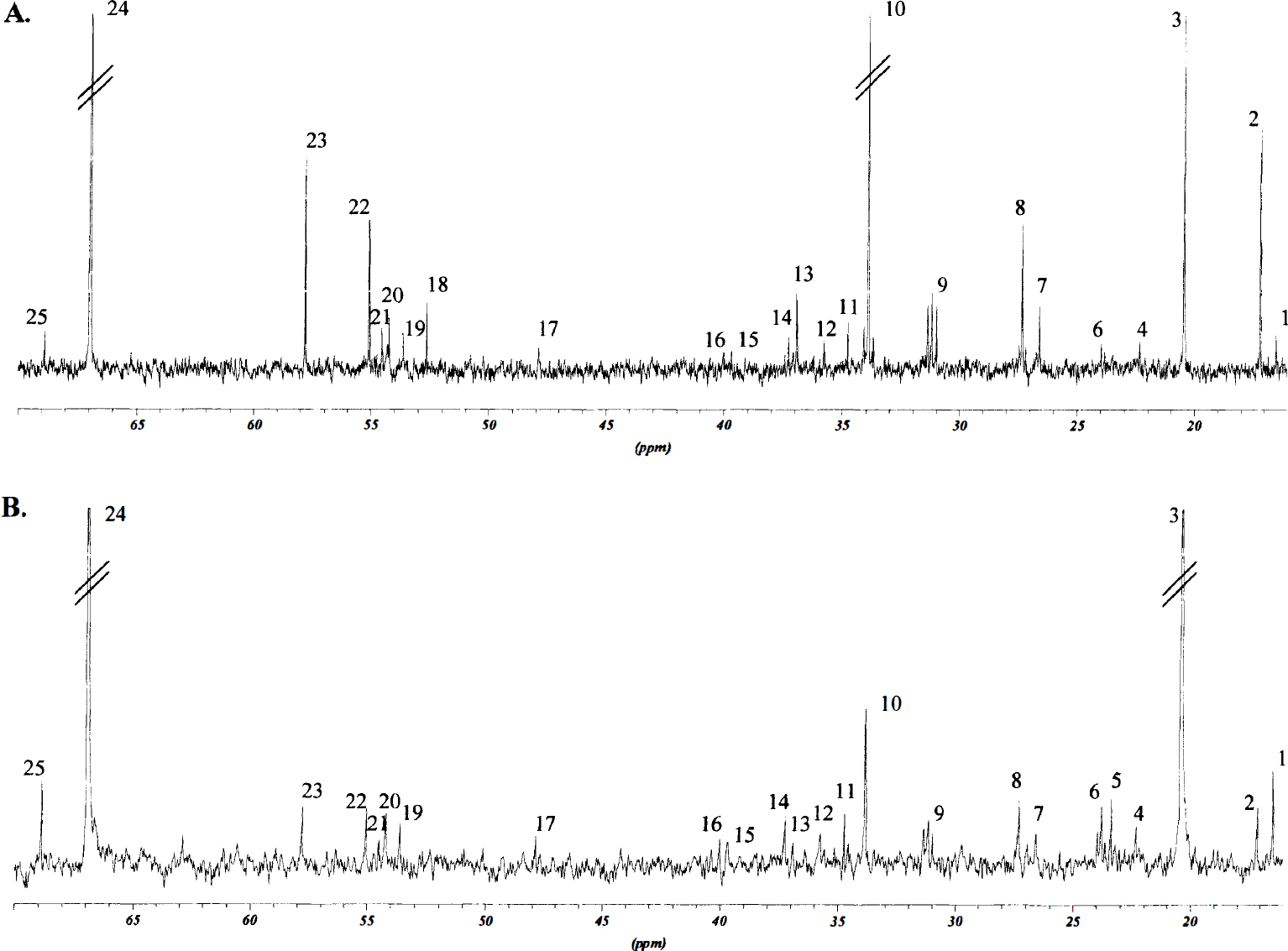
Typical 13C nuclear magnetic resonance (NMR) spectra from the lateral caudoputamen and lower parietal cortex in rats subjected to 90 minutes of middle cerebral artery occlusion (MCAO). The upper spectrum
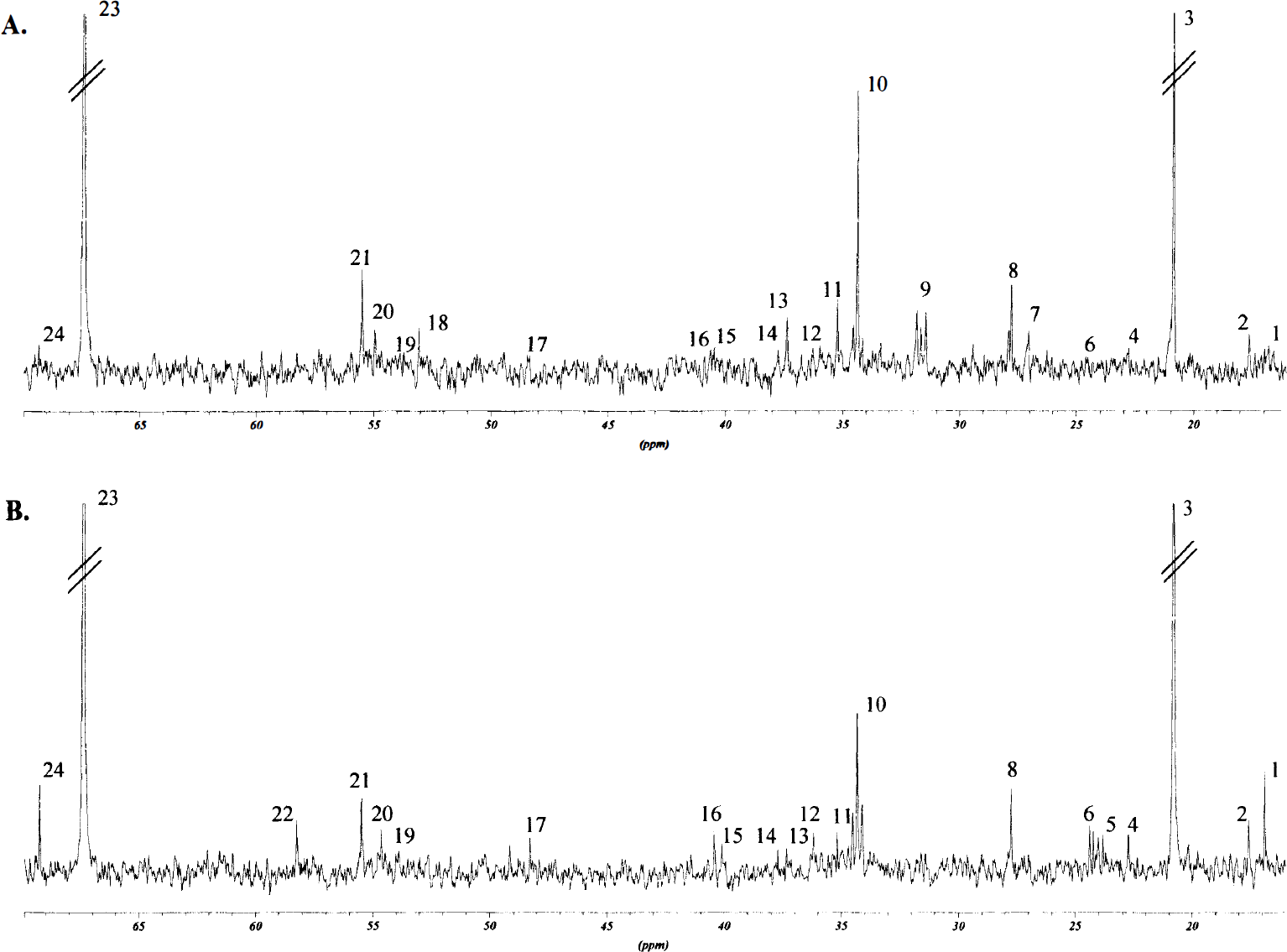
Typical 13C NMR spectra from the upper frontoparietal cortex in rats subjected to 90 minutes of MCAO. The upper spectrum
1H nuclear magnetic resonance spectroscopy
Five hundred megahertz 1H NMR spectra of the plasma samples were obtained on a Bruker DRX-500 spectrometer using a 35° pulse angle, with 7.5-kHz spectral width and 16-K data points. The acquisition time was 1.1 seconds per scan, and an additional 15.0-second relaxation delay was used. The number of scans typically was 200.
High performance liquid chromatography analysis
Fifty microliters of the brain extracts were retained after 13C NMRS, lyophilized, and redissolved in water before analysis of total amino acid content with HPLC (Specta System Gradient Pump, Freemont, CA, U.S.A.) and fluorescence detection (Shimaduz RF 530, Tokyo, Japan), after derivatization with ophthaldialdehyde (Sigma Chemical, St. Louis, MO, U.S.A.) using a Waters Nova Pack C18 column, 3.9 mm × 15 cm (Waters, Milford, MA, U.S.A.).
Data analysis
All peaks were integrated, and the amounts of 13C or 1H in the resonance of a particular metabolite were quantified from the integrals of the peak area using dioxane as an internal standard (Badar-Goffer et al., 1990). Isotopomers with two consecutive 13C atoms, called homonuclear spin-spin couplings, were analyzed as described by Cerdan and colleagues (Cerdan et al., 1990). In the plasma samples, the percent enrichment of 13C in glucose, acetate, lactate, and hydroxybutyrate was calculated from the 1H spectra (Badar-Goffer et al., 1990).
Injection of [1-13C]glucose and [1,2-13C]acetate permits simultaneous monitoring of neuronal and astrocytic TCA cycle activity, as well as neuronal—astrocytic interactions in the same animal (McLean et al., 1993; Taylor et al., 1996; Hassel et al., 1997). Because of expression of different enzymes in astrocytes and neurons (Fig. 3), a 13C label originating from either [1-13C]glucose or [1,2-13C]acetate is incorporated into different molecular positions in the amino acids (Fig. 4), reflecting neuronal or astrocytic metabolic activity, respectively. The distribution and the amount of 13C in the different molecular positions can be identified and quantified with 13C NMRS, and from this, information on relative changes in the metabolic pathways leading to Glu, Gln, and γ-aminobutyric acid (GABA) synthesis can be derived (Taylor et al., 1996; Hassel et al., 1997).
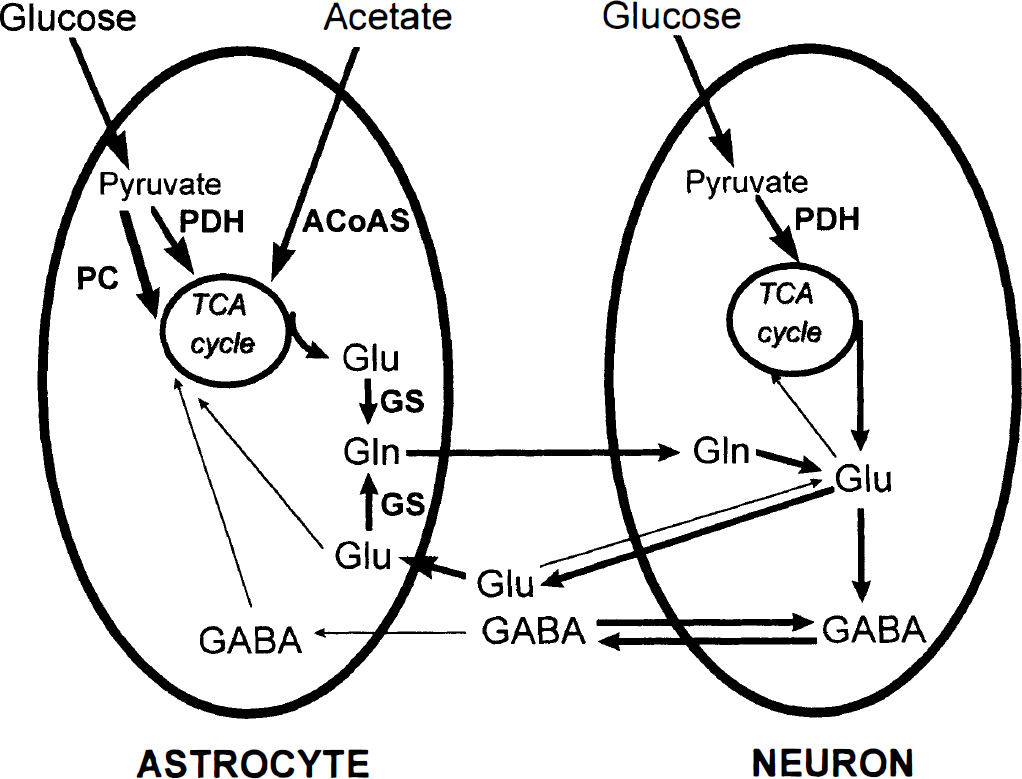
Schematic representation of the neuronal and the astrocytic compartments in CNS. For simplicity, the depicted neuron is a nonexisting pluripotent neuron, using both Glu and GABA as neurotransmitters. (See text for further details.) PC, pyruvate carboxylase; PDH, pyruvate dehydrogenase; ACoAS, acetyl coenzyme A synthetase; GS, glutamine synthetase; TCA cycle, tricarboxylic acid cycle; Glu, glutamate; Gln, glutamine; GABA, γ-aminobutyric acid.
Pyruvate carboxylase versus pyruvate dehydrogenase activity. The contribution from the anaplerotic versus the oxidative pathway in the formation of Glu, Gln, and GABA can be expressed as the ratio of PC activity versus pyruvate dehydrogenase (PDH; E.C. 1.2.4.1) activity (Taylor et al., 1996). This ratio also is an approximation for astrocytic support of neuronal neurotransmitter production. [1-13C]glucose oxidation yields labeling of the C-4 position in Glu and Gln, and the C-2 position in GABA (Fig. 4). Carboxylation, on the other hand, enriches the Glu and Gln C-2 position and the GABA C-4 position (Fig. 4). If the 13C label remains in the TCA cycle for subsequent turns, new isotopomers will arise. In the second turn, the original labeling of the Glu and Gln C-4 position will be equally distributed between the C-3 and the C-2 positions (Fig. 4) because of scrambling in the symmetrical succinate step. Thus, labeling of the Glu and Gln C-2 position represents either 13C incorporated through PC, or 13C originating from PDH activity, which has remained in the TCA cycle for more than one turn. Hence, only C-2 labeling in excess of C-3 labeling is considered resulting from PC activity. For Glu and Gln the PC/PDH ratio may be expressed as (C-2 – C-3)/C-4, and for GABA (C-4 – C-3)/C-2.
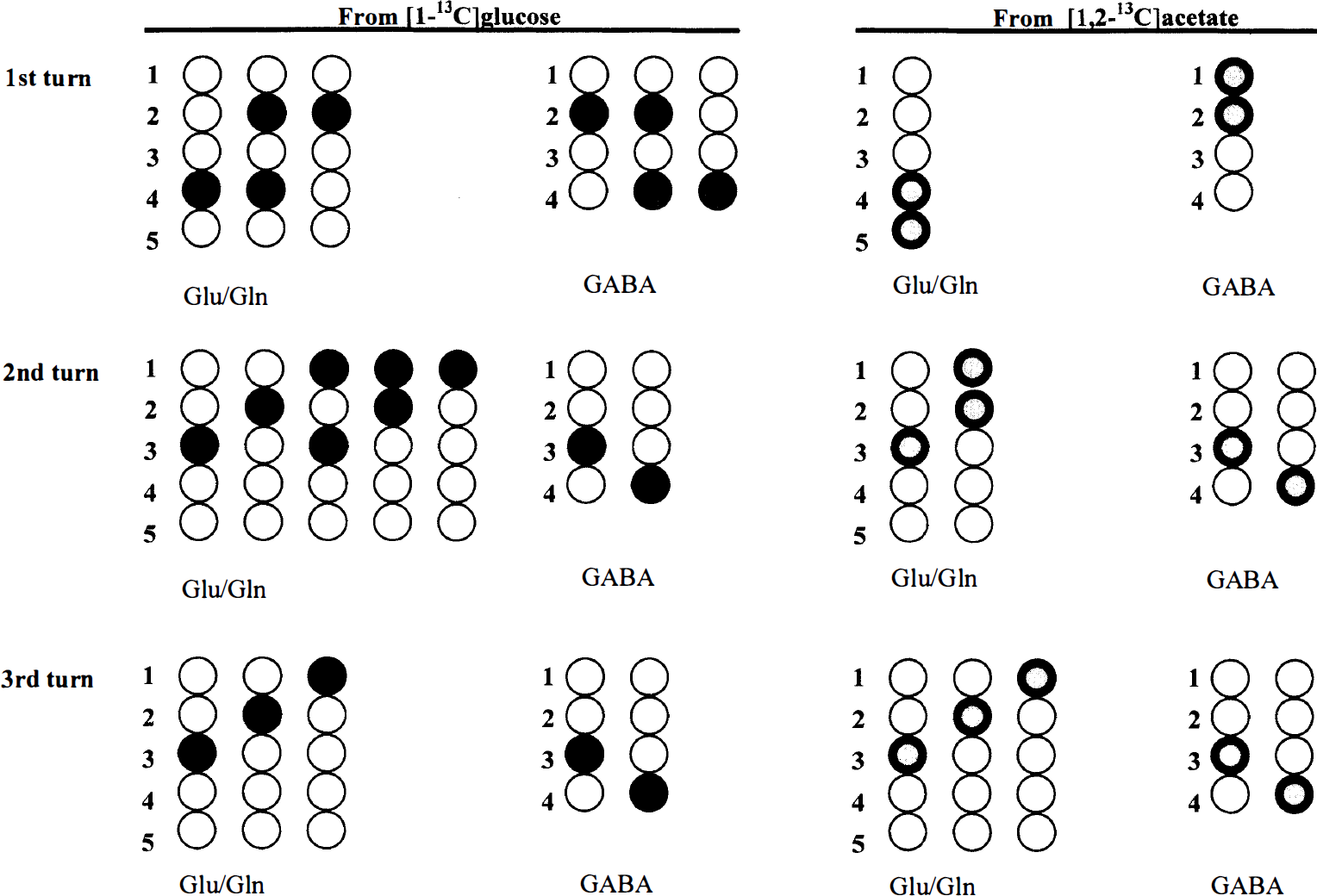
Schematic representation of 13C labeling originating from either [1-13C]glucose or [1,2-13C]acetate in Glu, Gln, and GABA. First turn refers to Glu, Gln, or GABA synthesized during the first passage of the 13C atom in the TCA cycle. The 13C atom from [1-13C]glucose can enter the TCA cycle through • pyruvate dehydrogenase (PDH) or ● pyruvate carboxylase (PC), and the 13C atoms from [1,2-13C]acetate via Acetyl coenzyme A synthetase. (See text for further details.)
Acetate versus glucose utilization. The proportion of metabolism in astrocytes versus neurons is reflected in the acetate-versus-glucose utilization ratio (Taylor et al., 1996). Glucose is the predominant substrate for the neuronal TCA cycle (Künnecke et al., 1993; Hassel et al., 1997), entering solely through PDH, and therefore represented by 13C label in the Glu and Gln C-4, and the GABA C-2 positions. The astrocytic compartment is characterized by its unique ability to use acetate as a substrate for the TCA cycle (Van den Berg, 1973; Hassel et al., 1997). Incorporation of [1,2-13C]acetate yields label in the Glu and Gln C-4,5, and the GABA C-1,2 positions (Fig. 4). For Glu and Gln, the acetate-glucose utilization ratio may be expressed as C-4,5/C-4, and for GABA C-1,2/C-1.
13C cycling. An indication on how swiftly TCA cycle intermediates leave the TCA cycle can be obtained from the ratio of isotopomers arising from the second turn of the TCA cycle compared with isotopomers originating from the first (Hassel et al., 1995). For Glu and Gln, the 13C cycling ratio may be expressed as C-3/(C-4 + C-4,5), and for GABA C-3/(C-2 + C-1,2).
Statistical analysis
All values are given as mean ± SD. Samples from the hemispheres contralateral and ipsilateral to the MCAO were compared using paired Student's t test. Comparisons between plasma samples analyzed with 13C and 1H NMRS were performed with paired Student's t test. P < 0.05 was considered significant.
RESULTS
13C and 1H nuclear magnetic resonance spectroscopy of plasma samples
The plasma glucose and acetate levels calculated from the 13C and the 1H spectra were in good agreement (Table 1). The total amount of plasma glucose was high, but only glucose enriched in the C-1 position was detected in significant amounts. Both glucose C-1 and acetate C-1 and C-2 were over 50% enriched with 13C. There was no 13C enrichment in glucose C-1,2 or C-5,6, and glucose C-2,3 and 4,5 was only slightly labeled (approximately 1%). From the 1H spectra, the amount of single-labeled acetate was estimated to have constituted 4.9% ± 1.6% of the total acetate. There was some 13C enrichment in lactate and hydroxybutyrate, but no 13C label was detected in the plasma amino acids.
Total amounts of glucose, acetate, lactate, and hydroxybutyrate in plasma calculated from 13C and 1H NMR spectra
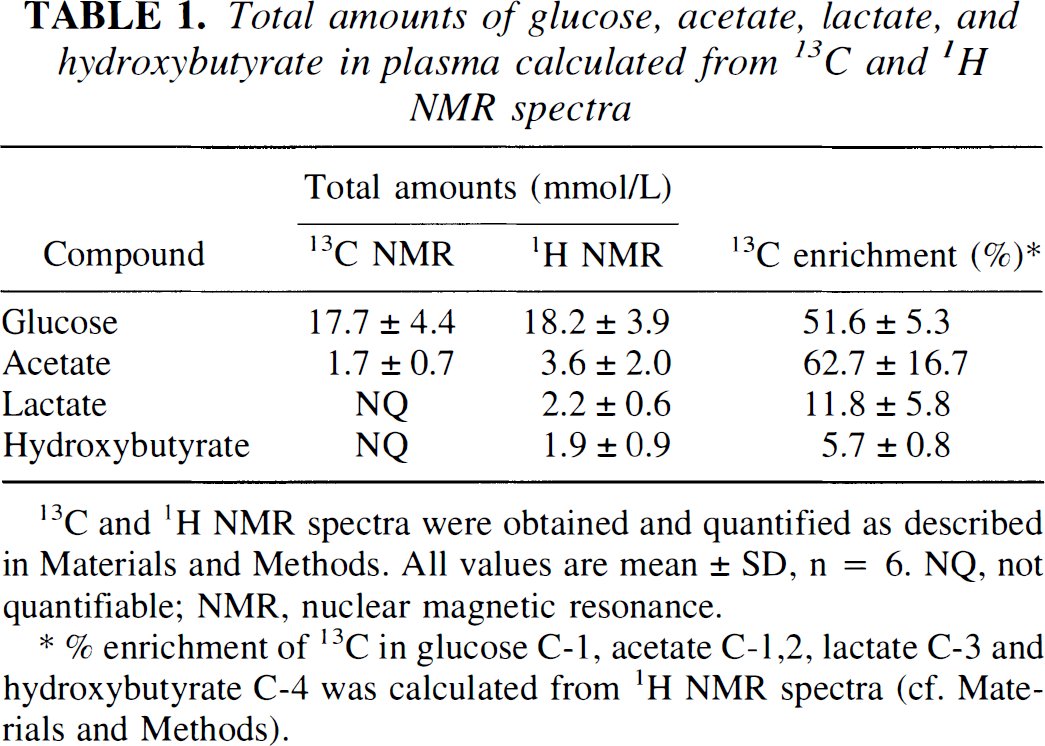
13C and 1H NMR spectra were obtained and quantified as described in Materials and Methods. All values are mean ± SD, n = 6. NQ, not quantifiable; NMR, nuclear magnetic resonance.
% enrichment of 13C in glucose C-1, acetate C-1,2, lactate C-3 and hydroxybutyrate C-4 was calculated from 1H NMR spectra (cf. Materials and Methods).
High performance liquid chromatography analysis of total amounts of amino acids in brain samples
In the lateral caudoputamen and lower parietal cortex ipsilateral to the MCAO, the total amount of Glu and aspartate had fallen significantly compared with the contralateral side (Table 2). The Gln content remained at a level comparable with the nonischemic hemisphere. A significant increase in the alanine concentration was observed, and a trend toward increased amounts of GABA was detected.
Total amino acid concentrations (µmol/g tissue) in brain samples from rats subjected to 90 minutes of MCAO
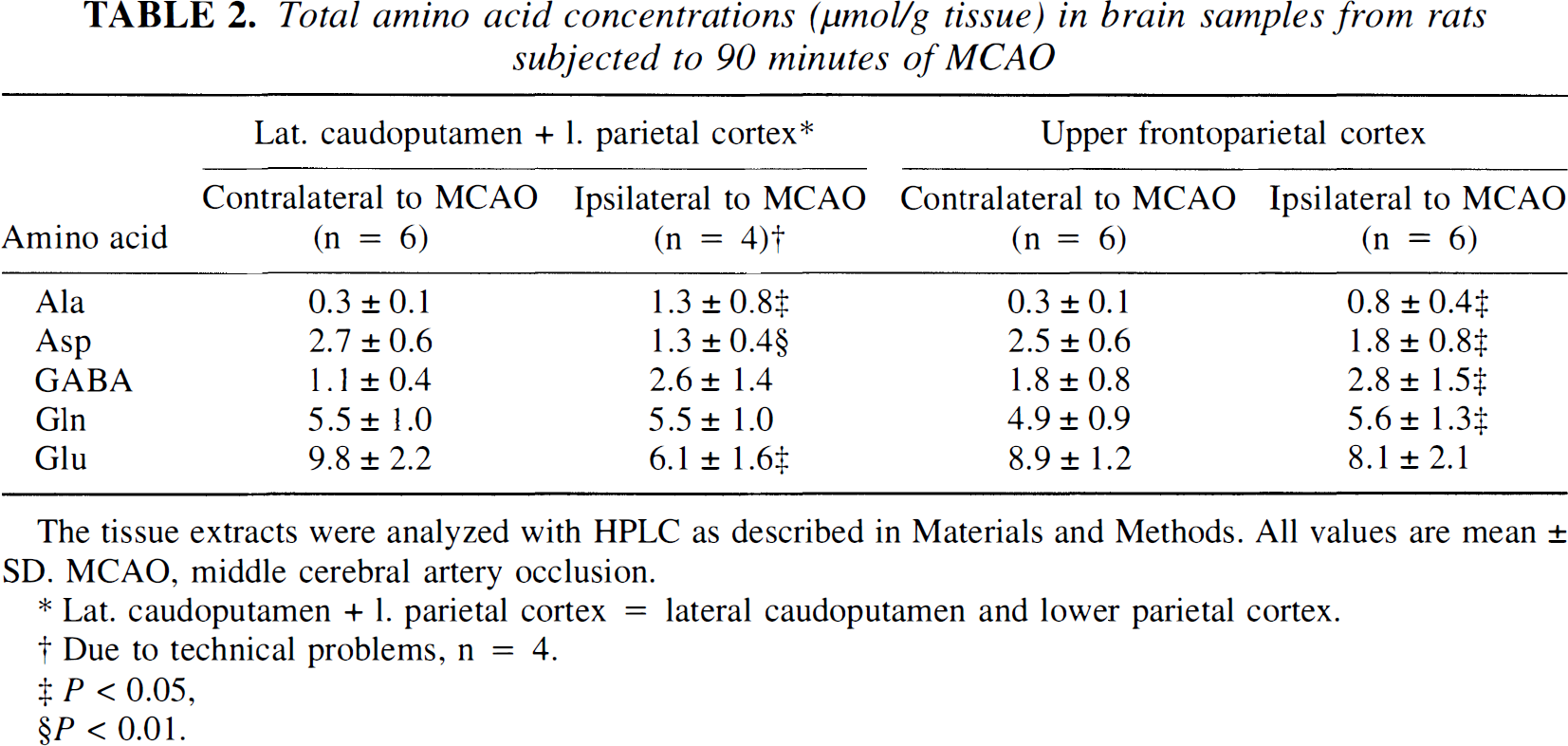
The tissue extracts were analyzed with HPLC as described in Materials and Methods. All values are mean ± SD. MCAO, middle cerebral artery occlusion.
Lat. caudoputamen + 1. parietal cortex = lateral caudoputamen and lower parietal cortex.
Due to technical problems, n = 4.
P < 0.05.
P < 0.01.
In the upper frontoparietal cortex ipsilateral to the MCAO, the Glu content equaled the amount found in the contralateral hemisphere. For aspartate, a significant reduction in the total amount was detectable, whereas statistically significant increased concentrations of Gln, alanine, and GABA were found.
13C nuclear magnetic resonance spectroscopy of brain extracts
[1-13C]glucose, [1,2-13C]acetate, [2-13C]- and [3-13C]lactate. The amount of [1-13C]glucose detected in the samples from the areas contralateral and ipsilateral to the MCAO were not statistically different, although a trend toward reduced glucose content was found in the lateral caudoputamen and lower parietal cortex ipsilateral to the MCAO (Table 3). Unmetabolized [1,2-13C]acetate was not detected in any samples from the hemispheres contralateral to the MCAO, but was present in all samples ipsilateral to the MCAO (Fig. 1 and 2). In lactate only, singlets were detected in the C-2 and C-3 peaks (Fig. 1 and 2). In the ischemic lateral caudoputamen and lower parietal cortex, the total amount of 13C in both lactate C-2 and C-3 were significantly increased, whereas in the upper frontoparietal cortex ipsilateral to the MCAO, only the amount of label in lactate C-3 was significantly elevated.
Total amounts of 13C (10−8mol/g tissue) in glucose, acetate, and lactate in brain samples from rats subjected to 90 minutes of MCAO
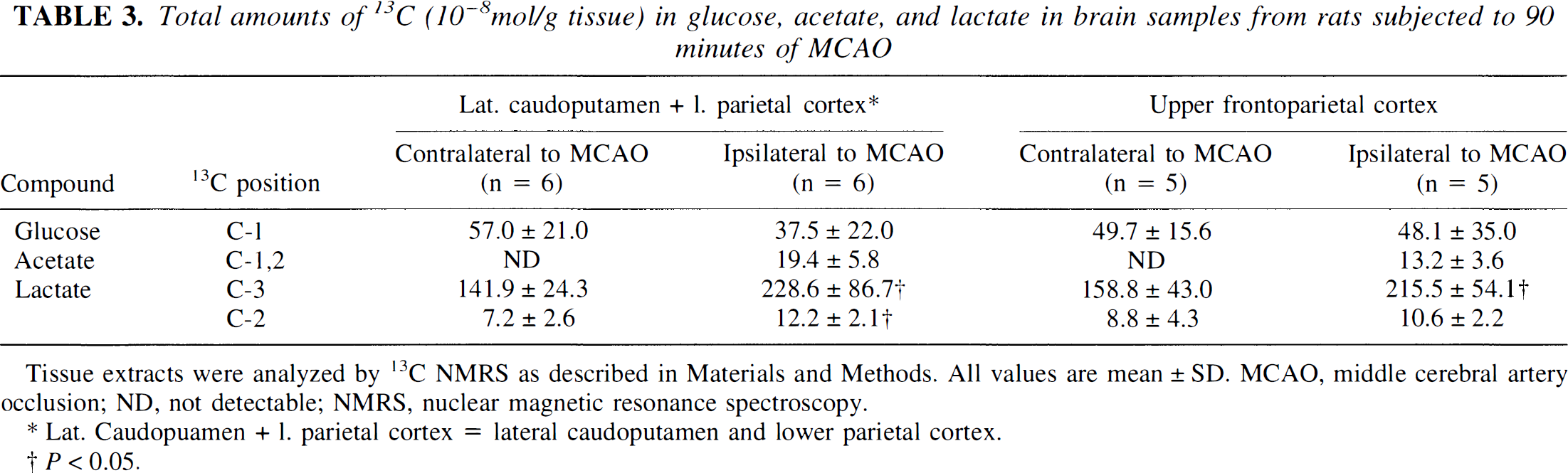
Tissue extracts were analyzed by 13C NMRS as described in Materials and Methods, All values are mean ± SD, MCAO, middle cerebral artery occlusion; ND, not detectable; NMRS, nuclear magnetic resonance spectroscopy.
Lat. Caudopuamen + 1. parietal cortex = lateral caudoputamen and lower parietal cortex.
P < 0.05.
Labeling patterns in glutamate, glutamine, and γ-aminobutyric acid. In the lateral caudoputamen and lower parietal cortex ipsilateral to the MCAO, the amount of labeled Glu C-4 was significantly reduced to about one fifth (Table 4). Statistically significant reductions also were seen in the total amounts of 13C in Glu C-3, C-2, and C-3,4, as well as in Glu C-4,5. Also, in the upper frontoparietal cortex ipsilateral to the MCAO, the amount of 13C label in Glu C-4, C-3, C-2, and C-3,4 was decreased significantly. Still, the total amounts of 13C were clearly higher in these isotopomers in the upper frontoparietal cortex ipsilateral to the MCAO compared with the ischemic lateral caudoputamen and lower parietal cortex. The amount of 13C label in Glu C-4,5 in the ischemic upper frontoparietal cortex was at a level comparable with the value found in the corresponding region in the contralateral hemisphere.
Total amounts of 13C (10−8mol/g tissue) in Glu, Gln, and GABA in brain samples from rats subjected to 90 minutes of MCAO
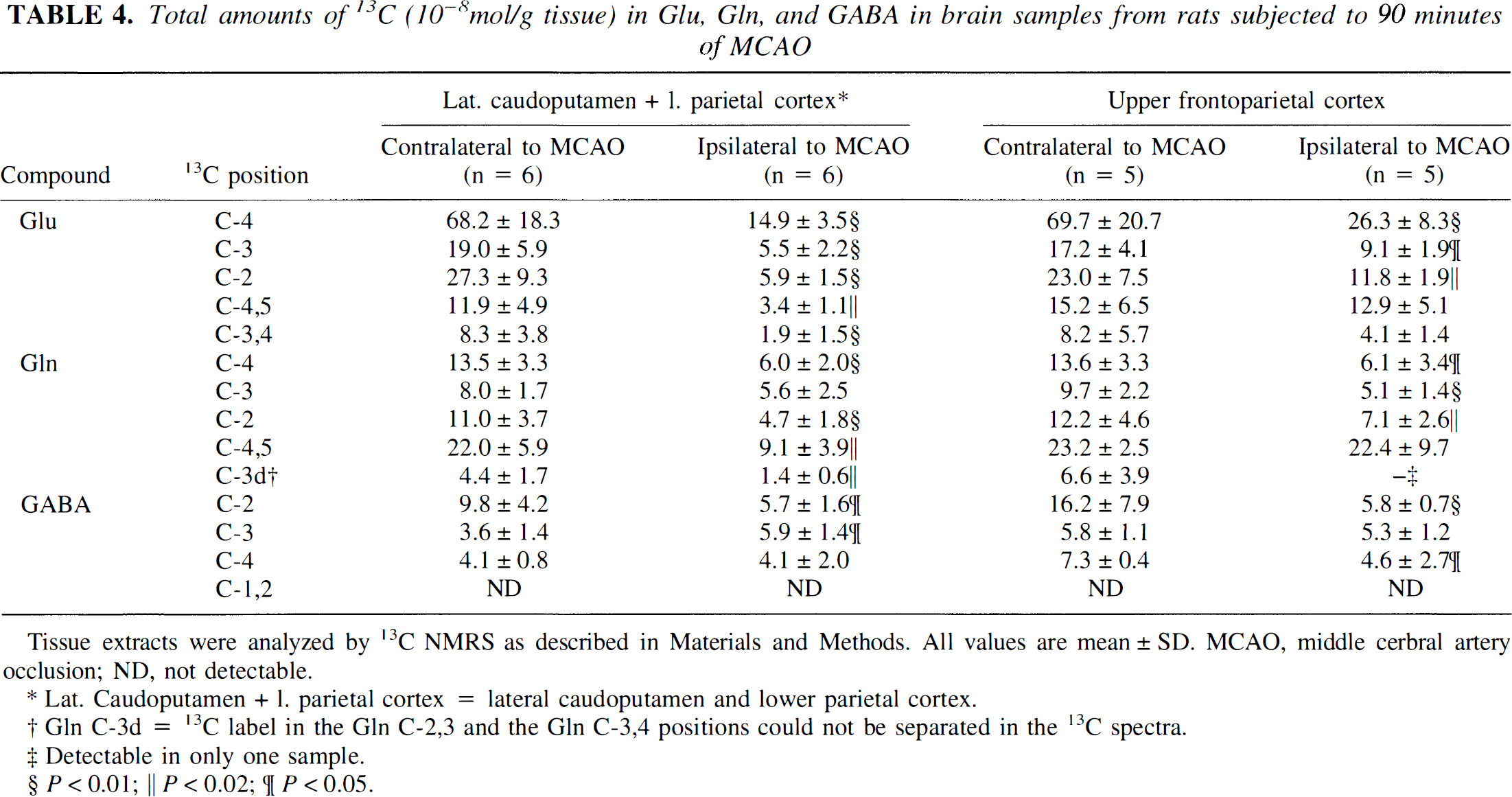
Tissue extracts were analyzed by 13C NMRS as described in Materials and Methods. All values are mean ±SD. MCAO, middle cerbral artery occlusion; ND, not detectable.
Lat. Caudoputamen + 1. parietal cortex = lateral caudoputamen and lower parietal cortex.
Gln C-3d = 13C label in the Gln C-2,3 and the Gln C-3,4 positions could not be separated in the 13C spectra.
Detectable in only one sample.
P < 0.01;
P > 0.02;
P < 0.05.
In the lateral caudoputamen and lower parietal cortex ipsilateral to the MCAO, the amount of GIn C-4,5 was significantly reduced by 50%. Also, Gln labeled in C-4 had fallen significantly, as had the amount of 13C in Gln C-2 and C-3 doublet (double labeling in Gln C-3, ie, 13C label in the Gin C-3,4 or Gln C-2,3 positions, could not be separated in the 13C NMR spectra). Only in Gln C-3 was the amount of label not significantly reduced compared with the corresponding area in the contralateral hemisphere. In the upper frontoparietal cortex ipsilateral to the MCAO, the total amounts of 13C in Gln C-4,5 were at nonischemic levels, but the amounts of 13C in Gln C-4, C-3, C-2, and C-3 doublet were significantly decreased, and were similar to the amounts found in the ischemic lateral caudoputamen and lower parietal cortex.
In the lateral caudoputamen and lower parietal cortex, as well as in the upper frontoparietal cortex ipsilateral to the MCAO, significantly decreased amounts of 13C were detected in GABA C-2, and instead, a near-equal distribution of 13C label between the three GABA isotopomers was observed. There was no label incorporation from [1,2-13C]acetate detectable in GABA in either ischemic or nonischemic samples.
Pyruvate carboxylase versus pyruvate dehydrogenase activity. In the ischemic lateral caudoputamen and lower parietal cortex, no PC activity was detected in Glu, Gln, or GABA (Table 5). In the upper frontoparietal cortex ipsilateral to the MCAO, the PC—PDH ratio was significantly reduced in Glu only.
Ratios for PC versus PDH activity, acetate versus glucose utilization, and 13C cycling in Glu, Gin, and GABA in brain samples from rats subjected to 90 minutes of MCAO
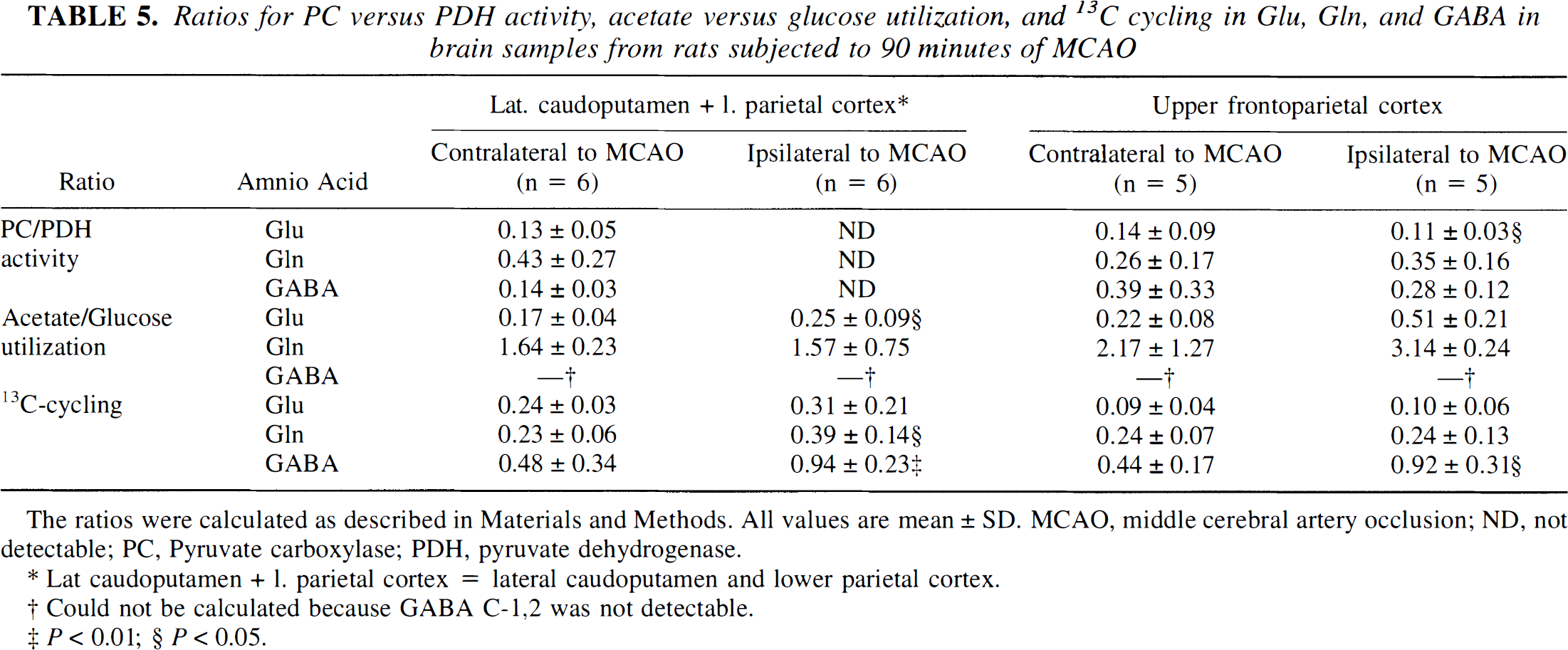
The ratios were calculated as described in Materials and Methods. All values are mean ± SD. MCAO, middle cerebral artery occlusion; ND, not detectable; PC, Pyruvate carboxylase; PDH, pyruvate dehydrogenase.
Lat caudoputamen + 1. parietal cortex = lateral caudoputamen and lower parietal cortex.
Could not be calculated because GABA C-1,2 was not detectable.
P < 0.01;
P > 0.05.
Acetate versus glucose utilization. A significantly higher proportion of Glu was synthesized from acetate in the lateral caudoputamen and lower parietal cortex ipsilateral to the MCAO (Table 5). In the ischemic upper frontoparietal cortex, a trend toward higher acetate-versus-glucose utilization ratio was seen in both Glu and Gln. Because of the lack of quantifiable amounts of 13C label in GABA C-1,2, the acetate-versus-glucose utilization ratio could not be calculated for GABA.
13C cycling. There were no differences in the Glu 13C cycling ratios between ischemic and non ischemic samples (Table 5). In Gln, the 13C cycling ratio was significantly elevated in the lateral caudoputamen and lower parietal cortex ipsilateral to the MCAO, but not in the ischemic upper frontoparietal cortex. Significantly increased 13C cycling ratios were detected in GABA in both the lateral caudoputamen and lower parietal cortex, and in the upper frontoparietal cortex ipsilateral to the MCAO.
Other metabolites. In the lateral caudoputamen and lower parietal cortex, and in the upper frontoparietal cortex ipsilateral to the MCAO, the amount of 13C label in aspartate was significantly reduced (Table 6). No double-labeled aspartate was detected in the samples from either hemisphere, indicating no 13C incorporation directly from [1,2-13C]acetate. Significantly increased amounts of 13C in alanine were found in the lateral caudoputamen and lower parietal cortex, and in the upper frontoparietal cortex ipsilateral to the MCAO. The amount of 13C in N-acetyl aspartate (NAA) C-2, C-3, and C-6 was not significantly different in the samples from the hemispheres ipsilateral and contralateral to the MCAO. Only the NAA C-6 position was enriched with 13C. If no 13C enrichment had taken place, the total amounts of 13C in the C-2, C-3, and C-6 positions would have been equal; there was, however, more 13C label in C-6 than in C-2 and C-3 positions. No BC label was detected in the NAA C-5 position, indicating no 13C enrichment in NAA from [1,2-13C]acetate. The amount of 13C is normally assumed to be at the natural abundance level in taurine. Still, the total amount of 13C in taurine C-2 was significantly lower in the ischemic lateral caudoputamen and lower parietal cortex compared with the contralateral side. In the upper frontoparietal cortex ipsilateral and contralateral to the MCAO, the amounts of 13C in taurine C-2 were similar.
Total amounts of 13C (10−8mol/g tissue) in Asp, Ala, Tau, and NAA in brain samples from rats subjected to 90 minutes of MCAO
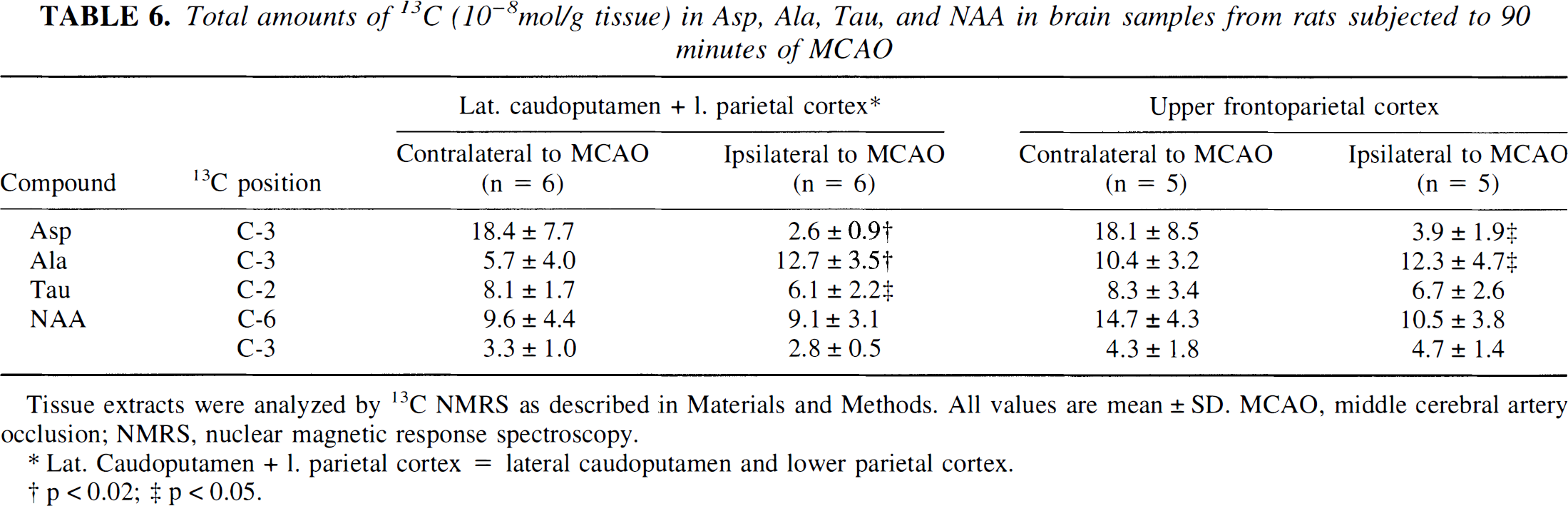
Tissue extracts were analyzed by 13C NMRS as described in Materials and Methods. All values are mean ± SD. MCAO, middle cerebral artery occlusion; NMRS, nuclear magnetic response spectroscopy.
Lat. Caudoputamen + 1. parietal cortex = lateral caudoputamen and lower parietal cortex.
p < 0.02; ‡ p > 0.05.
DISCUSSION
Validation of methods
Metabolite infusion scheme. Cerebral metabolism traditionally has been studied after infusion of metabolite precursors for 45 to 60 minutes to obtain a steady state (Sokoloff et al., 1977; Cerdan et al., 1990), but several inherent characteristics of the ischemic lesion make the steady state approach unfeasible. First, the acute ischemic lesion is continuously evolving, and no circulatory steady state exists; consequently, a conventional steady state cannot be achieved. Furthermore, continuous infusion of glucose for a prolonged time influences the development of the lesion. Postischemic hyperglycemia has been demonstrated to decrease glucose utilization in the ischemic border zone (Nedergaard et al., 1988). Acute hyperglycemia, however, as caused by the rapid glucose injection in the current study, has been shown not to affect the cerebral glucose metabolism (Duckrow and Byran, 1987). Also, systemic administration of large doses of acetate have adverse effects on cerebral function (Israel et al., 1994). Thus, a rapid injection of [1-13C]glucose and [1,2-13C]acetate close to the rats' killing was chosen in the current study. Previous studies indicate a time span of 15 to 30 minutes between injection and decapitation as optimal, since the influx of 13C-labeled acetyl coenzyme A exceeds the efflux of labeled TCA cycle intermediates (Hassel et al., 1995). This was clearly confirmed in the current study, where the isotopomers representing the first turn of the TCA cycle predominated in all samples, except in GABA in the samples from the ischemic hemispheres.
Effects of fasting. To avoid the detrimental effects that elevated preischemic blood glucose has on the evolution of the ischemic lesion, the animals were fasted overnight. The small amount of 13C label detected in glucose C-2,3 and C-4,5 positions (approximately 1%) indicates that some gluconeogenesis from [1,2-13C]acetate had taken place during the 15 minutes from injection to killing, and thus labeling of [1-13C]- and [6-13C]glucose from [1,2-13C]acetate also had occurred to the same extent (approximately 1%) because of the symmetrical succinate step. 13C label from [1,2-13C]acetate incorporated into the glucose C-1 and C-6 positions, however, does not influence the isotopomer analysis or the results. There was no 13C enrichment in glucose C-1,2 and C-5,6, and thus double labeling in Glu and Gln C-4,5 and GABA C-1,2 could not arise from glucose, which would have complicated the isotopomer analysis. Based on the small amount of 13C-enriched lactate present in plasma and the limited access that plasma lactate has to the brain because of poor blood-brain barrier permeability (Pardridge, 1983), the 13C-labeled lactate detected in the brain samples can be regarded as derived from cerebral metabolic activity.
Effects of anesthesia. Anesthesia has multiple effects on the cerebral metabolism. Isoflurane has been shown to uncouple the relation between cerebral blood flow and metabolism and perturb the incorporation of [1-13C]glucose into cerebral amino acids in animals (Shank et al., 1993). To avoid anesthesia, the animals were fully awake when receiving the infusion of labeled substrate and until decapitation. Funnel freezing is considered to be the optimal method for preservation of cerebral metabolites but was not used because it requires anesthesia. Instead, decapitation and immediate immersion of the heads in liquid nitrogen was used, which should not affect the amino acid levels (Siesjö, 1978).
Metabolic changes induced by middle cerebral artery occlusion
Glucose, acetate, and lactate. The trend toward reduced glucose concentration seen in the lateral caudoputamen and lower parietal cortex, but not in the upper frontoparietal cortex ipsilateral to the MCAO, corresponded to earlier findings (Nedergaard et al., 1988). The absence of detectable [1,2-13C]acetate in the nonischemic samples, together with the ubiquitous presence of unmetabolized [1,2-13C]acetate in the samples from the ischemic hemispheres, indicates impaired acetate metabolism, possibly resulting from reduced incorporation of acetate into the astrocytic TCA cycle because of ATP decline, a more general disturbance of astrocyte metabolism, or a combination of the two. Previously, unmetabolized acetate after [1,2-13C]acetate infusion has been detected in brain samples from mice treated with fluoroacetate (Hassel et al., 1997), a selective inhibitor of the glial TCA cycle. Thus, the presence of unmetabolized acetate in all samples from the hemispheres ipsilateral to the MCAO probably implied severely compromised astrocyte function. The increased amounts of 13C label in lactate detected in all samples from the ischemic hemispheres concurred with previous reports, although the total amount of 13C in lactate did not increase to the same extent as the reported total lactate content (Folbergrová et al., 1995). The total amounts of 13C label in lactate in the lateral caudoputamen and lower parietal cortex, and in the upper frontoparietal cortex ipsilateral to the MCAO, were similar, concurring with results obtained measuring total lactate concentrations in the corresponding brain regions 120 minutes after MCAO (Folbergrová et al., 1995).
Glutamate, glutamine, γ-aminobutyric acid, and N-acetyl aspartate synthesis. The 13C labeling patterns in Glu, Gln, and GABA detected in the hemispheres contralateral to the MCAO were consistent with previous reports (Hassel et al., 1995), confirming glucose as the primary substrate in Glu and GABA synthesis, and acetate in Gln. This, again, is in accordance with Glu and GABA being produced in neurons, and Gln in astrocytes.
In the ischemic lateral caudoputamen and lower parietal cortex, the striking fall in total Glu content, combined with greatly reduced total amounts of 13C in all Glu isotopomers, indicates seriously perturbed Glu homeostasis. A marked decline in neuronal Glu synthesized from [1-13C]glucose through PDH was clearly reflected in the reduced amounts of 13C in the Glu C-4 position; at the same time, a significantly increased proportion of Glu originated from acetate, as demonstrated by the increased acetate-versus-glucose utilization ratio in Glu. Together, these results indicate better preservation of Glu formation from astrocytic Gln than Glu synthesis through the glutamatergic TCA cycle in the ischemic lateral caudoputamen and lower parietal cortex. Thus, the significantly reduced Glu content in this region most likely resulted from a metabolic failure in the de novo Glu synthesis in the glutamatergic neurons, perhaps combined with washout of Glu into CSF and blood. Elevated Glu levels have been demonstrated in both plasma and CSF from patients with ischemic stroke (Castillo et al., 1996). Reduced Glu content after ischemia in vivo has previously been reported after 30 minutes of severe incomplete global ischemia followed by recirculation (Erecinska et al., 1984).
In the upper frontoparietal cortex ipsilateral to the MCAO, the total Glu content was at nonischemic levels, and more 13C label was demonstrated in all Glu isotopomers compared with the ischemic lateral caudoputamen and lower parietal cortex. These results, together with the normalization of the acetate-versus-glucose utilization ratio in Glu, indicate better preservation of neuronal Glu synthesis in this region. Also, the transfer of metabolites from astrocytes to neurons for Glu formation was close to normal, as demonstrated by the total amount of 13C in Glu C-4,5 which was at nonischemic levels. Still, the reduced contribution from the anaplerotic pathway in Glu bore evidence of impaired astrocytic support of the glutamatergic neurons.
In the lateral caudoputamen and lower parietal cortex ipsilateral to the MCAO, the reduction in the total amounts of 13C in Gln singlets was comparatively smaller than the reduction in the Glu singlets; also, the acetate-versus-glucose utilization ratio was preserved in Gln, thus indicating preserved astrocytic Glu uptake. Also, previous studies demonstrated the astrocytic Glu uptake to be resistant to ischemia (Torp et al., 1991). The increased Gln 13C cycling ratio detected in the lateral caudoputamen and lower parietal cortex ipsilateral to the MCAO may be attributed to elevated extracellular Glu levels in ischemia causing more of the Glu taken up from the extracellular space to enter the astrocytic TCA cycle before conversion to Gln. It has been shown in vitro that astrocytic Glu metabolism through the TCA cycle increases with increased extracellular Glu concentration (McKenna et al., 1996). It might be hypothesized that increased 13C cycling in Gln is a means to fill up the astrocytic TCA cycle, which could be of significance because of the lack of anaplerotic activity in astrocytes in the putative ischemic core in the lateral caudoputamen and lower parietal cortex ipsilateral to the MCAO. A stop in the anaplerotic pathway, as demonstrated here by the lack of PC activity in Glu, Gln, and GABA, ultimately will have deleterious effects for both astrocytes and neurons and is possibly an indicator of irreversible tissue damage.
The significantly elevated Gln content in the upper frontoparietal cortex ipsilateral to the MCAO concurs with results from Erecinska and colleagues (Erecinska et al., 1984) and is considered to result from increased glutamine synthetase activity in response to acute brain ischemia (Petito et al., 1992). The Gln isotopomer analysis, however, reveals impaired astrocyte function as the amount of DC label in Gln C-4, C-3, and C-2 were similar to the values found in the ischemic lateral caudoputamen and lower parietal cortex, which was in obvious contrast to the increased amounts of 13C label in all the Glu isotopomers in this region. Still, despite evidence of impaired astrocyte function, the astrocytes maintained the transfer of Gln C-4,5 for neuronal Glu production at nonischemic levels, suggesting that support of neuronal metabolism is a priority for astrocytes, even under metabolically challenged conditions.
The total GABA content was significantly elevated in the upper frontoparietal cortex ipsilateral to the MCAO, and a similar trend was seen in the lateral caudoputamen and lower parietal cortex on the same side. Ischemia is known to induce increased GABA levels (Erecinska et al., 1984), possibly through a combination of increased synthesis and decreased breakdown. Reduced amounts of 13C were detected in GABA C-2, indicating that the GABA de novo synthesis directly from glucose was primarily affected in both the lateral caudoputamen and lower parietal cortex, and in the upper frontoparietal cortex ipsilateral to the MCAO. The redistribution seen in the GABA isotopomers in the samples from the ischemic hemispheres implied preserved GABA reuptake. GABA reuptake is mainly neuronal (Erecinska, 1987) and has been suggested to be less sensitive to oxygen deprivation than the Glu reuptake systems (Erecinska et al., 1984), which is in good agreement with the current results. Continued GABAergic TCA cycle activity was demonstrated by the increased GABA 13C cycling ratio. The increased 13C cycling in GABA concurs with the notion that GABAergic neurons are relatively resistant to ischemia (Gonzales et al., 1992).
In the current study, the 13C enrichment into the C-6 position in NAA was similar in samples taken from the hemispheres ipsilateral to the MCAO and contralateral to the MCAO. Since NAA is a neuronal marker (Urenjak et al., 1992), the preserved NAA 13C enrichment indicates neuronal NAA synthesis, even after 90 minutes of MCAO.
CONCLUSION
The results obtained in the current study show that by combining in vivo injection of 13C-labeled metabolite precursors with ex vivo 13C NMRS, specific metabolic alterations in small regions within the rat brain with a focal ischemic lesion can be studied. The temporal evolution of the metabolic disturbances caused by focal ischemia is under investigation and will give more insight into neuronal-astrocytic dysfunction in ischemia.
Footnotes
Acknowledgments
The authors thank Torild Krogstad for performing the HPLC analysis at the Institute of Biotechnology at University of Science and Technology and Professor Anders Johnsson at the Department of Physics, Norwegian University of Science and Technology for valuable discussion.
