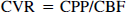Abstract
Little is known of the factors that regulate CBF in sleep. We therefore studied 10 lambs to assess the vasodilatory processes that underlie cerebral autoregulation during sleep. Lambs, instrumented to measure CBF (flow probe on the superior sagittal sinus), sleep state, and cerebral perfusion pressure (CPP), were rapidly made hypotensive by inflating a cuff around the brachiocephalic artery to reduce CPP to 30 mm Hg in each state. During control periods, cerebral vascular resistance (CVR in mm Hg/mL/min) was lower in active sleep (2.8±0.3, mean±SD, P ≤ 0.001) than in wakefulness (3.9±0.6) and quiet sleep (4.3±0.6). The CVR decreased promptly in each state as CPP was lowered. The time (seconds) required for maximal cerebral vasodilation to occur was longer in active sleep (35±11) than in quiet sleep (20±6, P ≤ 0.001) and wakefulness (27±11, P ≤ 0.05). The CVR decreased less in active sleep (0.6±0.3, P ≤ 0.001) than in quiet sleep (1.5±0.3), although the changes in CPP induced with brachiocephalic occlusion were equal in each state. In conclusion, our studies provide the first evidence that the vasoactive mechanisms that underlie autoregulation of the cerebral circulation function during sleep. Moreover, our data reveal that the speed and the magnitude of the vasodilatory reserves available for autoregulation are significantly less in active sleep than in quiet sleep.
Autoregulation of the cerebral circulation is a fundamental protective mechanism that acts to maintain CBF constant by adjusting cerebral vascular resistance (CVR) to changes in arterial pressure (Edvinsson et al., 1993; Paulson et al., 1990). This vasoactive process is crucial for maintaining the integrity of the brain by protecting the neural tissues from ischemic hypoxia during hypotension and from hemorrhage and edema during hypertension (Edvinsson et al., 1993; Paulson et al., 1990). Surprisingly, given the importance of autoregulation, and given that human beings spend one third of their lives sleeping, little is known of the factors that regulate CBF in sleep. Moreover, no study has examined the vascular responses that might compensate for variations in cerebral perfusion pressure (CPP) during sleep.
It is known that CBF changes dramatically with alterations of behavioral state. In all species, the transition from quiet sleep to active sleep is accompanied by an increase in CBF, regardless of the nature of the accompanying blood pressure change, which generally is small and variable between species (Cianci et al., 1991; Franzini, 1992; Grant et al., 1995b; Madsen, 1993; Zoccoli, 1994). In contrast, the change in CBF with the transition from wakefulness to quiet sleep is variable, depending on the species (Franzini, 1992). Just as CBF differs substantially between behavioral states, so, too, do cerebral neural activity, cerebral metabolic rate, and neural control of the peripheral circulation (Cianci et al., 1991; Franzini, 1992; Madsen, 1993), all factors that may impact on the control of the cerebral circulation. Thus, the ability of the cerebral circulation to adapt to circulatory disturbances such as hypotension may differ substantially between behavioral states. Moreover, the increase in CBF that is typical of active sleep results from cerebral vasodilation (Grant et al., 1995b); on this basis, it can be predicted that the vasodilatory reserve of the cerebral circulation, and, therefore, its ability to compensate to acute hypotension, would be limited in this sleep state.
This study was designed to assess the cerebral vascular responses to hypotension in sleep by using a newly developed quantitative technique for continuously measuring CBF (Grant et al., 1995b). Our study used the newborn lamb, an animal that has been widely used in the study of sleep physiology, since it has active and quiet sleep phases comparable with those of the human infant (Baker and Fewell, 1988; Horne et al., 1991; Grant et al., 1995a). This species also has proven to be of value over many years in the study of the cerebral circulation (Purves and James, 1969; Szymonowicz et al., 1990) and of the cerebral circulation in sleep, where it exhibits the stereotyped increase of blood flow during active sleep and the decrease in quiet sleep that is common to humans and most other species (for reviews see Greenberg, 1980; Madsen and Vorstrup, 1991; Franzini, 1992). Specifically, we measured both the rapidity and the magnitude of the vasodilatory response of the cerebral circulation to acute hypotension in wakefulness, quiet sleep, and active sleep to ascertain if the mechanisms that underlie the process of cerebral autoregulation function in sleep as they do in wakefulness.
METHODS
Ten newborn lambs (Merino/Border-Leicester cross) were separated from their ewes within 24 hours of birth and housed within a plexiglass cage. The lambs were taught to feed from a nipple connected to a continuous supply of lamb milk replacer (Veanavite Pty. Ltd., Shepparton, Australia) and gained weight normally. Once feeding independently, each lamb was prepared for a series of chronic studies. All surgical and experimental procedures were performed in accordance with the guidelines of the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes established by the National Health and Medical Research Council of Australia, the Commonwealth Scientific and Industrial Research Organization, and the Australian Agriculture Council, and were approved by both the Monash University and Monash Medical Centre Committees on Ethics in Animal Experimentation.
Surgical preparation
Each lamb was anesthetized (1.5% halothane, 50% O2, balance N2O), intubated, and then ventilated. Using sterile surgical techniques, each lamb underwent instrumentation to record CBF as previously described (Grant et al., 1995b). In brief, a 2 times 2 cm section of the skull overlying the intersection of the lambdoid and sagittal sutures was removed to access the superior sagittal sinus. A transit time ultrasonic flow probe (2 mm diameter, Transonic Systems, Inc., Ithaca, NY, U.S.A.) was carefully positioned around the superior sagittal sinus, taking care not to damage neural or vascular tissue. The probe then was covered with dental acrylic to form a rigid cap, which stabilized the probe and replaced the section of skull that had been removed. This technique provides a simple, quantitative, and beat-by-beat measurement of CBF. The technique uses two ultrasonic transducers within a perivascular probe, with the transducers passing ultrasound alternately with and against the flow of blood. The transit time of the ultrasound beam varies with blood flow velocity, and the difference between the integrals of the upstream and downstream ultrasound transit time is a measure of volume flow. The technique has been validated for use on the sagittal sinus of the lamb (Grant et al., 1995b). Ultrasonic flow probe measurements were linearly related to venous outflow measured directly by blood collection, and to arterial inflow determined using radioactive microspheres. Retrograde dye injection revealed that the measurement represents blood flow from 35% of the total brain mass, principally the frontal and anterior parietal lobes. Postmortem examination confirmed the correct positioning of the probe and the patency of the sagittal sinus.
We also positioned saline-filled catheters (nonocclusive, 0.86-mm inside diameter, 1.52-mm outside diameter) in the carotid artery for blood pressure monitoring and for blood sampling. A catheter (1.57-mm inside diameter, 2.41-mm outside diameter) also was positioned under the dura to record intracranial pressure. To determine behavioral states of wakefulness, quiet sleep, and active sleep, we implanted pairs of Teflon-coated stainless-steel wires on the parietal cortex (electrocorticogram [ECOG]), at the inner and outer canthus of the left eye (electrooculogram), and in the dorsal musculature of the neck (nuchal electromyogram [EMGn]) (Grant et al., 1995a). Wakefulness was defined as a period where the lamb was lying down, when the ECOG displayed a pattern of low voltage and high frequency, and when eye movements and EMGn tone were present (Fig. 1). In quiet sleep, the ECOG displayed a pattern of high voltage and low frequency, eye movements were absent, and EMGn tone was reduced when compared with the awake state (Fig. 1). In contrast, during active sleep, the ECOG displayed a pattern of low voltage and high frequency, rapid eye movements were present, and EMGn tone was absent (Fig. 1).
Physiologic recording from a sleeping lamb illustrating that CBF increases on entering active sleep from quiet sleep. The increase in CBF corresponds to a decrease in cerebral vascular resistance, since cerebral perfusion pressure (CPP), on average, did not change. EMGn, electromyogram of the nuchal (neck) muscles; EOG, electrooculogram; ECOG, electrocorticogram.
Finally, we positioned an inflatable silicon rubber cuff around the common brachiocephalic artery through a left thoracotomy in the second intercostal space. Inflation of this cuff rapidly reduced arterial pressure distal to the cuff, and thus reduced CPP (carotid arterial pressure minus intracranial pressure).
Conditions of study
After a minimum of 72 hours postoperative recovery, the lambs were studied on 2 to 4 separate days. During these studies, the lamb's cage was partitioned to prevent the lamb from turning around, while still allowing freedom to move forward and backward, and to stand up and lie down. Food was available to the lambs throughout the study, and room temperature was maintained between 22° and 25°C.
We connected the flow probe to the flow meter (model T101, Ultrasonic Blood Flow Meter, Transonic Systems), which, along with the electrodes, was connected to a signal conditioner (Cyberamp 380, Axon Instruments, Inc., Foster City, CA, U.S.A.). Electrophysiologic signals were filtered with the signal conditioner (0.3 to 80 Hz, 0.3 to 80 Hz, and 30 to 80 Hz for ECOG, electrooculogram, and EMGn, respectively). Vascular and intracranial catheters were connected to calibrated strain-gauge manometers (Cobe CDX III, Cobe Laboratories, Lake-wood, CO, U.S.A.), and all pressures were referenced to the midthoracic level. The pressure and flow signals were low-pass filtered at 100 Hz. Physiologic signals were recorded on a thermal chart recorder (model 7758A, Hewlett Packard, Waltham, MA, U.S.A.) and simultaneously stored on computer (486 DX/50) at a sampling rate of 200 Hz, using an analog-digital converting board (ADAC 4801A, ADAC Corp., Woburn, MA, U.S.A.) and acquisition software (CVSOFT Data Acquisition and Analysis Software, Odessa Computer Systems, Ltd., Calgary, Canada).
Protocol
To assess the vasodilatory mechanisms on which cerebral autoregulation depends, we rapidly reduced CPP (Fig. 2) by briefly inflating the silicon rubber cuff around the brachiocephalic artery. Inflations were maintained for 50 seconds during uninterrupted, well-defined epochs of wakefulness, quiet sleep, and active sleep. These brief inflations were repeated over a period of 2 to 4 days until four inflations were completed in each behavioral state in each lamb. The 10-second period preceding the inflation was the control period. Cuff inflations were adjusted to reduce CPP to approximately 30 mm Hg and to maintain it at this level. Data from the four inflations were averaged to provide a single result for each lamb in each state. The lambs tolerated the hypotension without arousal or distress, showed no lasting effects from the test procedure, and grew and behaved normally throughout the study. Release of the brachiocephalic cuff produced a brief period of hypertension that was accompanied by arousal from sleep (Home et al., 1991); we therefore did not analyze data recorded after release of the cuff. Inflations were repeated until four inflations that were not associated with behavioral state transitions were completed in each lamb. To achieve this and to obtain comparable levels of blood pressure in each state, an average of two to three attempts were required for each successful inflation.
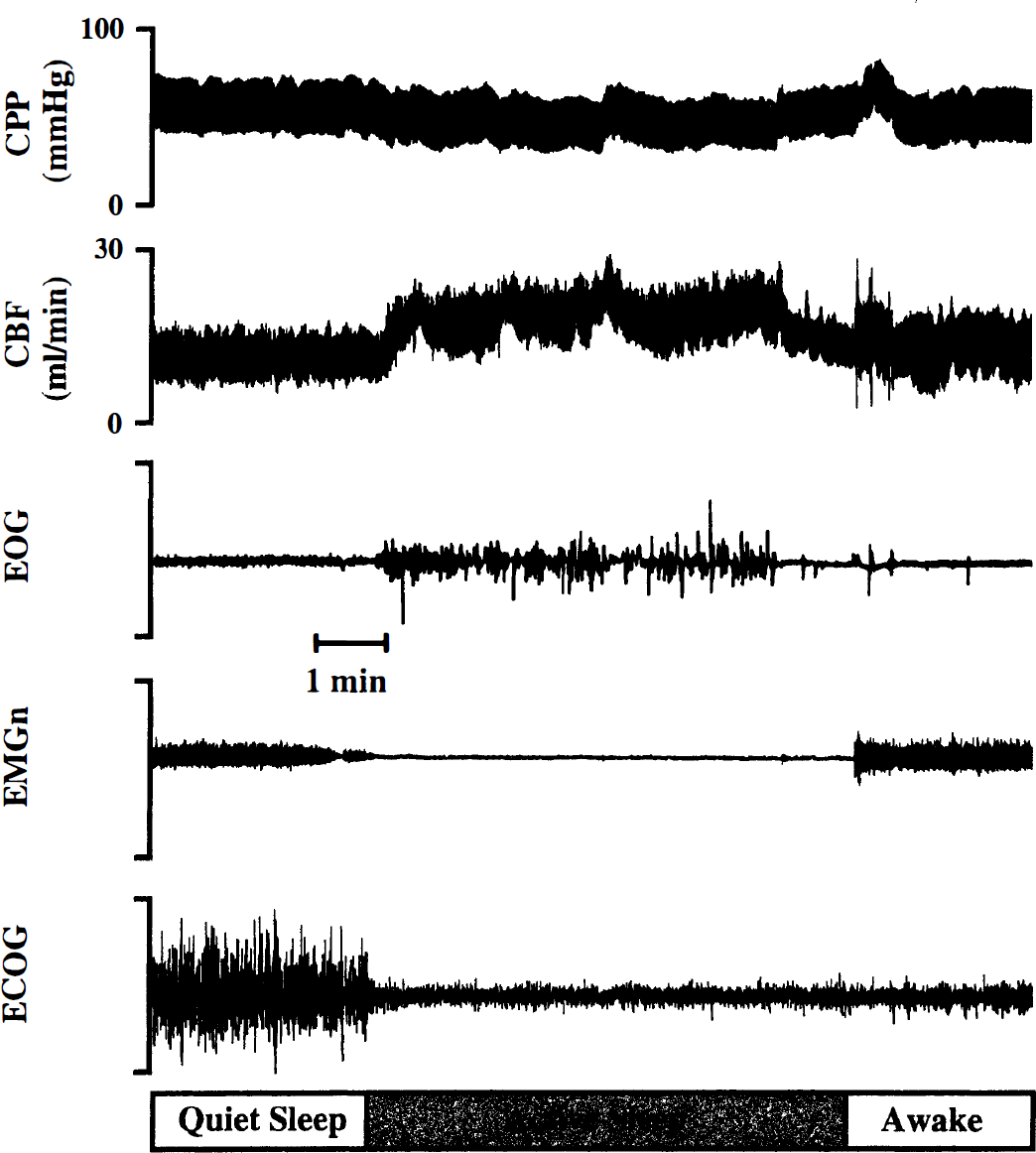
Occluding the brachiocephalic artery (time zero) rapidly decreased cerebral perfusion pressure (CPP) in each behavioral state (AS, active sleep; QS, quiet sleep; W, wakefulness). The CPP fell to a minimum value within 5 to 10 seconds (P ≤ 0.05) and remained unchanged throughout the remainder of the occlusion period. The decreases in CPP were accompanied by significant decreases in CBF (Fig. 3) and cerebral vascular resistance (Fig. 4). Values represent mean±SD.
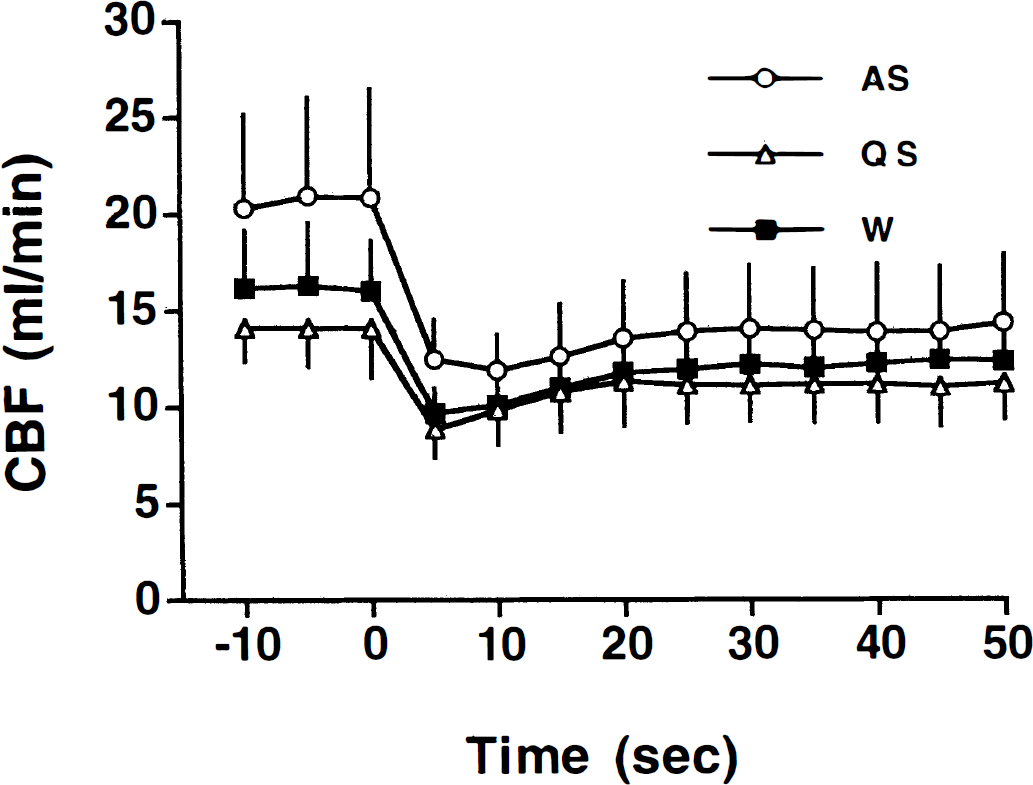
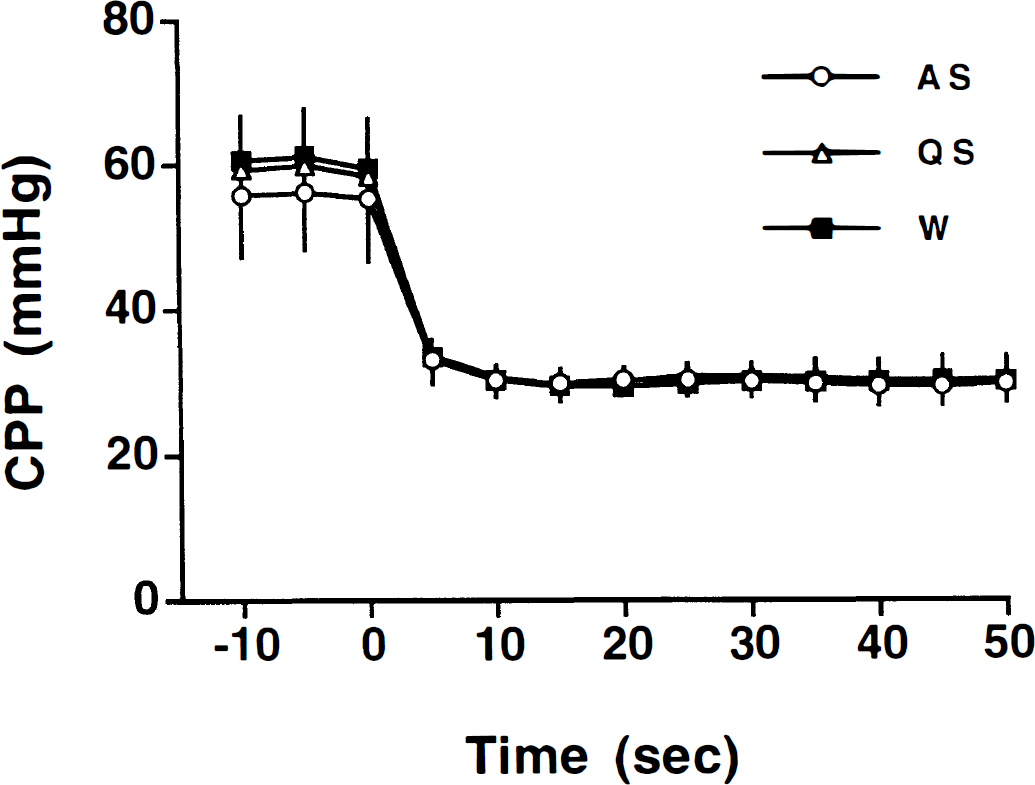
Cerebral blood flow decreased promptly in response to the onset of hypotension (time zero) in each behavioral state. In quiet sleep (QS) and wakefulness (W), CBF partially recovered (within 15 seconds of the onset of hypotension) to a level significantly greater than the minimum value (P ≤ 0.001), although still significantly less than the control value (P ≤ 0.001). In active sleep (AS), the trend for a partial recovery in CBF during hypotension did not reach statistical significance (P ≤ 0.05) until much later (50 seconds). Values represent mean±SD.
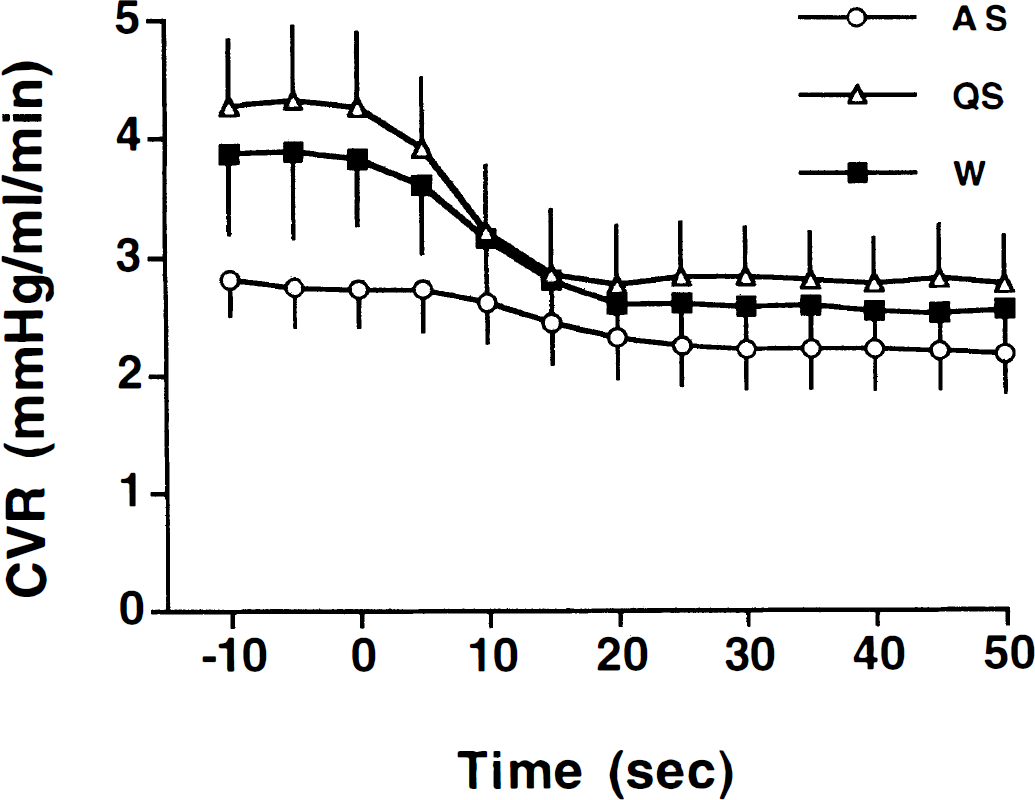
In quiet sleep (QS) and wakefulness (W), cerebral vascular resistance (CVR) decreased promptly during hypotension (within 5 seconds, P ≤ 0.05) and continued to decrease statistically for only 10 to 15 seconds after the onset of hypotension. In active sleep (AS), the fall in CVR was delayed and did not begin until 10 to 15 seconds after the onset of hypotension. Moreover, in active sleep, CVR continued to decrease statistically much longer into the period of hypotension (20 to 25 seconds). Values represent mean±SD.
Data analysis and statistics
The CVR was calculated during control and inflation periods from the second-by-second averages of CPP and CBF:
The time required for maximum vasodilation was determined for each inflation in each lamb. We compared the average CBF, CPP, and CVR recorded from each lamb in the control and inflation periods using an analysis of variance for repeated measures. An analysis of variance also was used to compare the vasodilation times in each of the behavioral states. A Student-Newman-Keuls test was used to isolate differences that were detected by the analysis of variance. A probability (P) of ≤ 0.05 was considered significant. Data are presented as mean±SD.
RESULTS
Cerebral blood flow and CVR differed significantly according to behavioral state under control conditions (Fig. 1, Table 1). In active sleep, control values of CBF were significantly higher (P ≤ 0.001) and CVR (P ≤ 0.001) were significantly lower than in both quiet sleep and wakefulness. In addition, control values of CBF also were significantly higher (P ≤ 0.05) and CVR significantly lower (P ≤ 0.05) in wakefulness than in quiet sleep. Cerebral perfusion pressures did not differ in any behavioral state in the control periods (Table 1), although the variability usually was greater in active sleep (Fig. 1).
Behavioral state effects during control conditions and induced hypotension
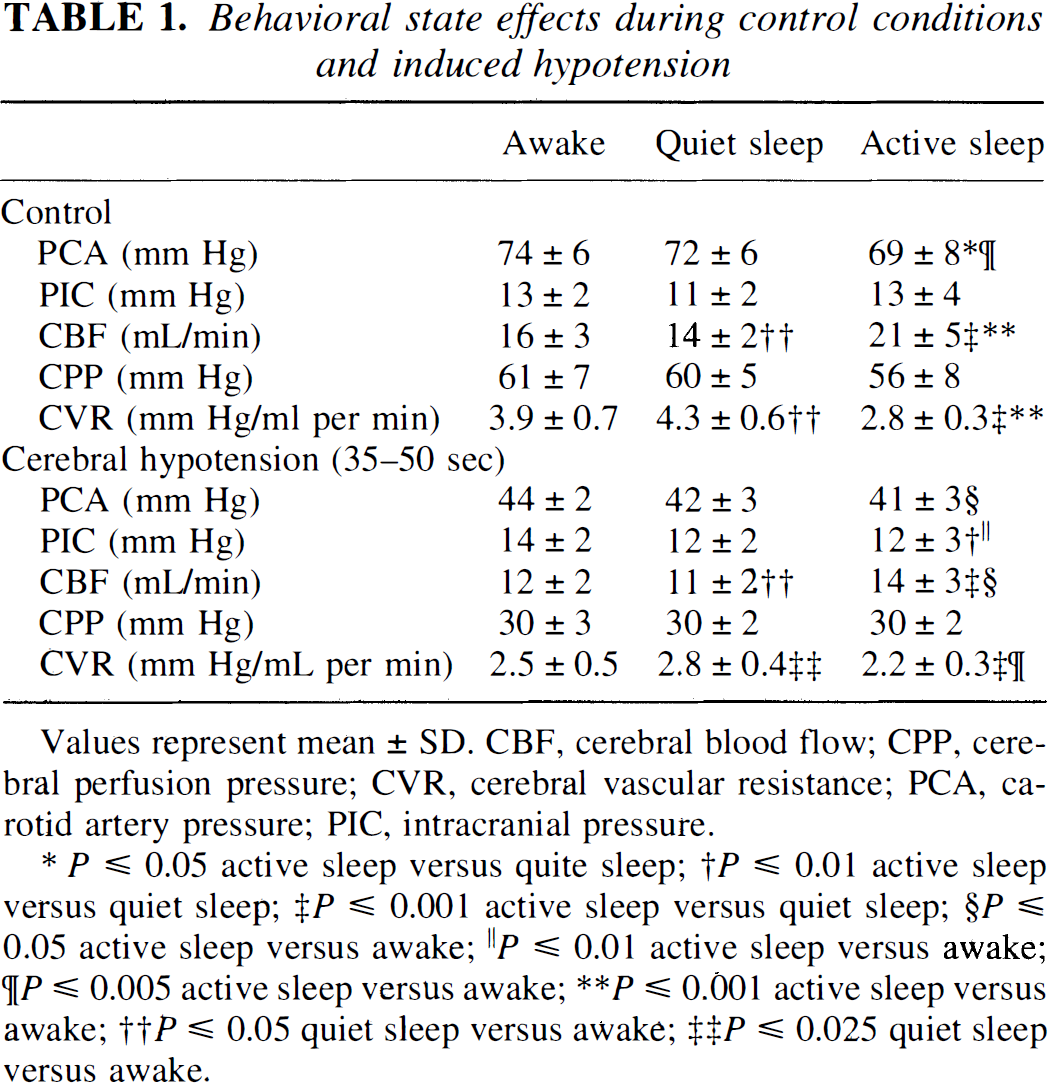
Values represent mean ± SD. CBF, cerebral blood flow; CPP, cerebral perfusion pressure; CVR, cerebral vascular resistance; PCA, carotid artery pressure; PIC, intracranial pressure.
P ≤ 0.05 active sleep versus quite sleep
P ≤ 0.01 active sleep versus quiet sleep
P ≤ 0.001 active sleep versus quiet sleep
P ≤ 0.05 active sleep versus awake
P ≤ 0.01 active sleep versus awake
;P ≤ 0.005 active sleep versus awake
P ≤ 0.001 active sleep versus awake
P ≤ 0.05 quiet sleep versus awake
P ≤ 0.025 quiet sleep versus awake.
Brachiocephalic arterial occlusions decreased CPP, CBF, and CVR in all behavioral states (Figs. 2 through 4; Table 1). Cerebral perfusion pressure reached a minimum (30 mm Hg) within 5 to 10 seconds and remained unchanged throughout the occlusion period in each behavioral state. Cerebral blood flow fell rapidly and significantly with the onset of hypotension (P ≤ 0.001), reaching a minimum value within 5 to 10 seconds of the onset of occlusion. In each behavioral state, CBF tended to return toward control within 10 to 15 seconds of the onset of cerebral hypotension (Fig. 3). In quiet sleep and wakefulness, CBF reached a plateau within 15 seconds of the onset of occlusion. During these plateaus, CBF was significantly higher than the minimum value (P ≤ 0.001), but significantly lower than the control level (P ≤ 0.001). In active sleep, the values of CBF observed during the plateau did not significantly exceed the minimal value until much later in the period of hypotension (50 seconds, P ≤ 0.05).
In all behavioral states, CVR decreased significantly (P ≤ 0.001) in response to reductions in CPP (Fig. 4). In quiet sleep and wakefulness, CVR decreased promptly during hypotension (within 5 seconds, P ≤ 0.05) and continued to decrease significantly for only 10 to 15 seconds after the onset of cerebral hypotension. In active sleep, the fall in CVR was delayed, and CVR did not decrease significantly from the control values until 10 to 15 seconds after the onset of hypotension. Moreover, in active sleep, CVR continued to decrease significantly later into the period of cerebral hypotension (20 to 25 seconds). The time required for maximum vasodilation to occur was assessed for each individual hypotensive period in each behavioral state and was found to be significantly longer in active sleep (35±11 seconds) than in wakefulness (27±11 seconds, P ≤ 0.001) and quiet sleep (20±6 seconds, P ≤ 0.05).
Within the plateau phase of hypotension (35 to 50 seconds after the onset of occlusion), CPP did not differ between behavioral states (Table 1). The plateau value of CBF in active sleep was significantly greater than that in both wakefulness (P ≤ 0.05) and quiet sleep (P ≤ 0.001, Table 1). In addition, the plateau value of CBF in wakefulness was significantly greater than in quiet sleep (P ≤ 0.05). Similarly, the plateau value of CVR was significantly less in active sleep than in both wakefulness (P ≤ 0.005) and quiet sleep (P ≤ 0.001), and less in wakefulness than in quiet sleep (P ≤ 0.025).
A comparison of the decrease in CBF and CVR from control to the plateau phase of hypotension revealed that the vasodilatory response to hypotension was smaller in active sleep than in either wakefulness or quiet sleep (Fig. 5). Although CPP decreased the same amount (P > 0.05) during the occlusions in active sleep and quiet sleep (27±7 mm Hg and 30±6 mm Hg, respectively), the decrease in CBF was greater in active sleep (6.9±3.2 mL/min) than in quiet sleep (3.1±1.0 mL/min, P ≤ 0.001). The decreases in CVR reflected these flow differences, decreasing much less in active sleep (0.6±0.3 mm Hg/mL/min) than in quiet sleep (1.5±0.3 mm Hg/mL/min, P ≤ 0.001, Fig. 5). Whereas the decrease in CPP observed in wakefulness (31±6 mm Hg) slightly exceeded that in active sleep (P ≤ 0.05), the changes in CBF were smaller (3.9±1.4 mL/min) and the changes in CVR were larger (1.3±0.3 mm Hg/mL/min) in wakefulness than active sleep (P ≤ 0.001, Fig. 5). The changes in perfusion pressure, CBF, and CVR were equal in wakefulness and quiet sleep.
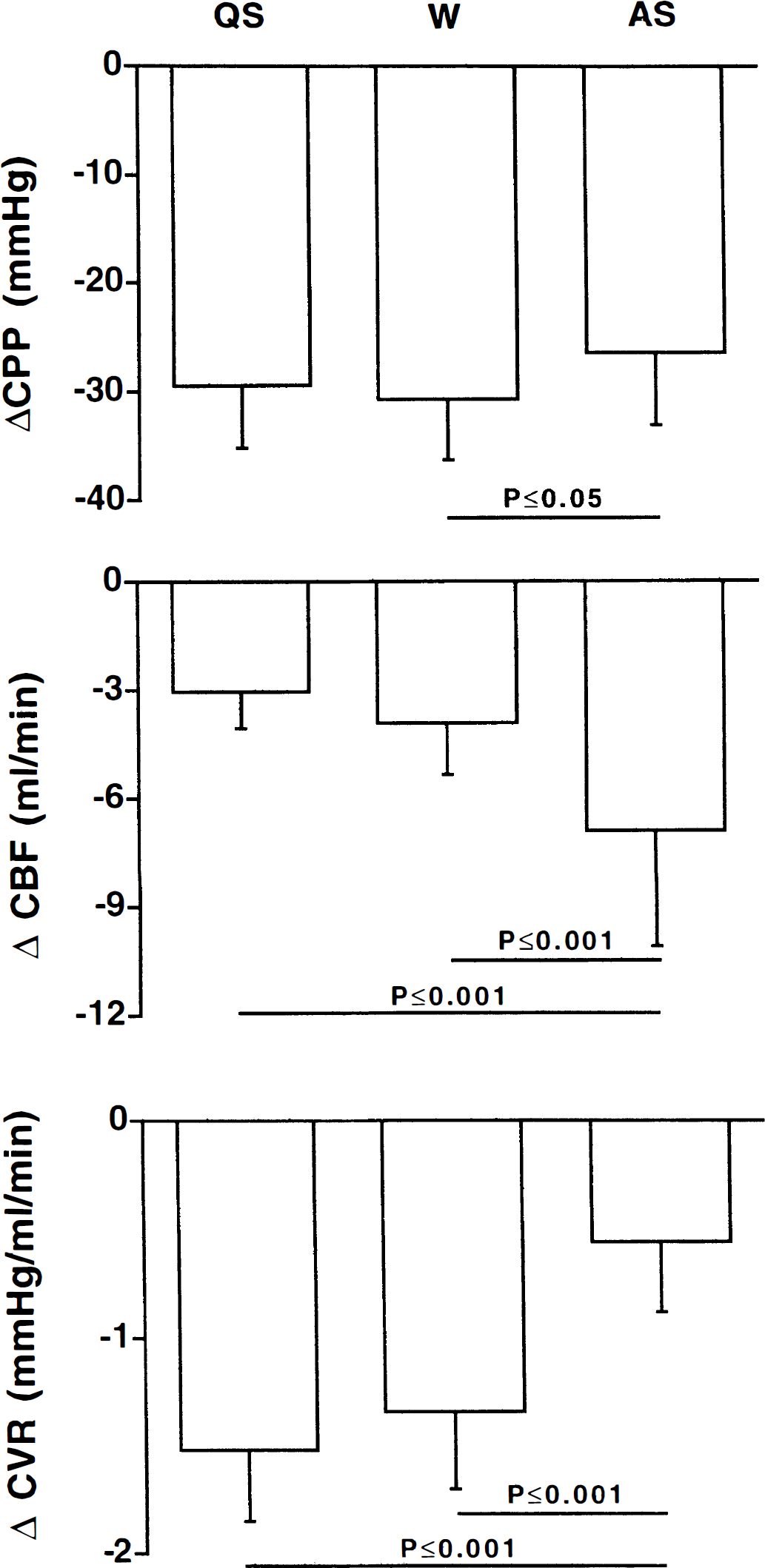
Occluding the brachiocephalic artery decreased cerebral perfusion pressure (CPP) by equal amounts during quiet sleep (QS) and active sleep (AS), and only slightly more in wakefulness (W) than AS (ΔCPP, upper panel). This decrease in perfusion pressure produced a significantly greater decrease in cerebral blood (ΔCBF) flow in AS than in QS and W (middle panel). In addition, the decrease in cerebral perfusion pressure was accompanied by a significantly smaller decrease in cerebral vascular resistance (ΔCVR) in AS than in QS and W (lower panel). Values represent mean±SD of the difference between the control values and the values recorded in the interval between 35 and 50 seconds during the inflation.
Control blood gas and pH values in wakefulness were equivalent to those obtained within our laboratory in newborn lambs (pH = 7.43±0.03, Pao2 = 100±6 mm Hg, Sao2 = 94±2%, Paco2 = 40±3 mm Hg, Hb = 8.4±0.7 g/dL, and base excess = 2±2 mmol/L). No significant differences were observed in blood gas and pH data between the control and hypotensive periods in any sleep state. Control values of carotid artery pressure, intracranial pressure, CBF, CPP, and CVR are presented in Table 1.
DISCUSSION
Our study is the first to demonstrate that the vasodilatory responses underlying autoregulation of the cerebral circulation function during sleep. Cerebral vasodilation occurred during acute hypotension in all behavioral states. Interestingly, the rate of vasodilation was slower in active sleep than in wakefulness and quiet sleep. In addition, as we discuss later, active sleep is distinguished by having less vasoactive reserve than wakefulness and quiet sleep, and thus by having a lesser ability to maintain CBF during hypotension.
No previous study has examined the process that underlie cerebral autoregulation in sleep, possibly because of the difficulty of making rapid, continuous measurements of CBF in sleep. We have used a new technique that continuously records the blood flow in the superior sagittal sinus. This technique provides a simple quantitative measurement of CBF that is linearly related to arterial inflow (Grant et al., 1995b). Importantly, this measurement is continuous and responds rapidly (within one heart beat) to variations in CPP (Grant et al., 1995b).
We measured cerebral vasodilatory responses to acute hypotension as a first step toward assessing the presence and effectiveness of cerebral autoregulation during sleep. The rapid step changes in arterial blood pressure that we induced lowered perfusion pressure below the break point of the autoregulatory curve that has previously been defined for the awake lamb (Purves and James, 1969; Szymonowicz et al., 1990). We anticipated, therefore, that if cerebral autoregulation functions in sleep, it would be revealed within our study as vasodilation of the cerebral circulation. Although it has been suggested that rapid changes in perfusion pressure (in excess of 0.4 mm Hg/s) may not be accompanied by cerebral autoregulation (Barzó, 1993), vasodilation of precapillary cerebral vessels occurs when blood pressure is reduced at rates in excess of 0.4 mm Hg/s (Kontoset al., 1978). Moreover, the more quickly blood pressure is changed, the more quickly precapillary vasodilation occurs (Kontoset al., 1978). Our results clearly demonstrate that cerebral vasodilation occurs even when CPP is lowered at a rate 10 times the “critical rate” of 0.4 mm Hg/s. More importantly, our results demonstrate the occurrence of vasodilation in sleep and in wakefulness and so provide evidence that the mechanisms required for autoregulation of the cerebral circulation function in all behavioral states.
The time course of cerebral vasodilation has not been previously assessed during sleep. It has, however, been shown in anesthetized and awake animals and humans that cerebral autoregulation begins within 1 to 2 seconds of a decrease in arterial pressure and is complete within 20 to 90 seconds (Aaslid et al., 1989; Florence and Seylaz, 1992; Newell et al., 1994; Rapela and Green, 1964). Our new information that details these features of cerebral autoregulation during sleep is largely compatible with these earlier studies. In our study, the onset of cerebral vascular vasodilation was essentially coincident with the imposed fall in CPP during quiet sleep and wakefulness but was delayed (about 10 to 15 seconds) in active sleep (Figs. 2 and 3). Vasodilation was complete within 20 seconds in quiet sleep, 27 seconds in wakefulness, and in 35 seconds in active sleep.
The fundamental mechanisms that control autoregulation of the cerebral circulation may differ between behavioral states. Our observations that the time for maximum cerebral vasodilation was shortest in quiet sleep supports this possibility. It also is possible that the mechanisms involved in the vasodilatory process are similar in each state and that the differing vasodilation times reflect a linearity in the time course of the vasodilatory process. Thus, the vasodilation time in quiet sleep may be shorter because vasodilation begins in a vascular bed that has greater resting tone (i.e., partially vasoconstricted), whereas in active sleep the vessels are already largely dilated. Further studies are required to explain the varying times required for maximal vasodilation.
Although CVR fell to the lowest absolute level during hypotension in active sleep, the vasodilatory response was limited in this state. Because control levels of CVR were substantially lower in active sleep than in quiet sleep or wakefulness, the magnitude of the reduction induced by an equal change in CPP beginning from equal control pressure (i.e., the vasodilatory reserve) was smallest in active sleep (Figs. 4 and 5). As a consequence, the ability of the cerebral circulation to auto-regulate in response to hypotension is limited in active sleep.
The limited vasodilatory reserve in active sleep is significant in that it may place the brain at risk for ischemic hypoxia during acute hypotension. This suggestion may account for the observation that the risk for ischemic stroke is greatest in the morning, late in sleep, the time period when active sleep is most prevalent (Marshall, 1977; Shapiro and Dement, 1993).
Variations in arterial blood gas or pH influence both the position of the autoregulatory curve (Paulson et al., 1990) and the rate at which autoregulation occurs during hypotension (Aaslid et al., 1989). However, they did not contribute to the differences in control circulatory variables that we observed between states, or to the differing responses to hypotension in this study, since there were no significant differences in pH, Paco2, Pao2, and Sao2 between states in the control periods, and no differences within states during the occlusions. It is possible, however, that local tissue oxygenation, Pco2, and pH may differ between behavioral states and that changes in these factors may have occurred during the occlusion, which is consistent with a metabolic pathway underlying autoregulation of the cerebral circulation (Edvinsson et al., 1993).
In summary, our studies provide the first evidence that the vasoactive mechanisms that underlie autoregulation of the cerebral circulation function during sleep. Moreover, our data reveal that the vasodilatory reserves available for autoregulation are significantly less in active sleep than in both quiet sleep and wakefulness. The reduced vasodilatory reserve of active sleep may reflect differences in the mechanism of autoregulation in this sleep state, since the time course of vasodilation was slower in active sleep than in quiet sleep. The reduced vasodilatory reserve, coupled with a slower vasodilatory response to hypotension, suggests that the cerebral circulation and, therefore, the functional integrity of the brain, may be particularly vulnerable to hypotension during active sleep.
Footnotes
Acknowledgment
The authors thank Mr. V. Brodecky for technical assistance.