Abstract
Glut1 is a specific transporter system that mediates glucose transfer across the blood–brain barrier (BBB). Although the main location of Glut1 is in the capillary endothelium of the brain, its local distribution in different brain regions is not as well defined. In the present investigation, the local pattern of Glut1 distribution was determined in 13 brain structures using an immunoautoradiographic method developed for this purpose. A polyclonal antibody directed against the C-terminal amino acid sequence of Glut1 was applied to cryosections of rat brains. A secondary antibody was added that had been coupled to [35S]. Results show a heterogeneous distribution of Glut1 in the brain with activities of [35S] ranging from 65% below to 15% above the mean. White matter activity was lower than gray matter activity. For comparison, capillary sections were counted in corresponding cryosections by indirect immunofluorescence using fibronectin antibodies. In addition, local cerebral glucose utilization (LCGU) was analyzed in identical brain structures of conscious rats by the quantitative autoradiographic 2-deoxyglucose method. Significant correlations were found between Glut1 density and either LCGU or capillary density. Results indicate a tight coupling of Glut1 transporter density and capillary density to the LCGU of different BBB structures in adult rats.
Keywords
Cerebral oxidative metabolism requires a constant supply of glucose to meet the energy demands of the brain. Delivery of glucose from the blood to the brain involves transport of this nutrient across the endothelial cells of the blood–brain barrier (BBB) and the plasma membranes of neurons and glial cells. This process is mediated by at least two isoforms of the facilitative glucose transporter family, Glut1 and Glut3. Besides Glut3, which is primarily expressed in brain neurons (Gerhart et al., 1992; Maher et al., 1993; Nagamatsu et al., 1992), two molecular mass forms of Glut1 have been detected in brain. These two forms are encoded by the same gene but differ in their extent of glycosylation (Sivitz et al., 1989). The existence of Glut1 in adult rat brains was demonstrated in several reports (Gerhart et al., 1989; Maher et al., 1994; Kumagai et al., 1994), although its regional density is not well known. Regional variations of local cerebral glucose utilization (LCGU) and corresponding variations of capillary density have been demonstrated for the brain. The question, therefore, arose as to whether or not the glucose transporter Glut1 varies in different brain regions and, if so, whether it can be related to local variations of LCGU and capillary density. For this purpose, an immunoautoradiographic method was developed that allows analysis of the local distribution of Glut1 from autoradiographs of brain sections. Cryosections were incubated with a primary antibody and, after rinsing, with an [35S]-labeled secondary antibody. After exposure to x-ray film, optical densities (ODs) were measured in different brain structures from autoradiograms and related to a set of standards. In addition, capillary density was determined for the brain structures investigated. To this end, antibodies directed against the basement membrane constituent, fibronectin, were applied to brain cryosections followed by secondary antibodies coupled to a fluorescent dye (Göbel et al., 1990). Using a fluorescent microscope, color photographs were taken and capillary spots counted. LCGU was measured in a group of conscious rats using the quantitative 2-deoxyglucose method developed by Sokoloff et al. (1977). LCGU was measured in the same brain structures as used for the analysis of Glut1 density.
METHODS
Glut1 autoradiography
Experiments were performed on five Sprague–Dawley rats. Animals were killed by decapitation, and all brains were removed rapidly, shock-frozen in 2-methylbutane (Merck, Darmstadt, Germany), chilled to −40°C, and embedded in M-1 embedding matrix (Lipshaw, Detroit, MI, U.S.A.). Then, 10 μm thick coronal sections were cut in a cryomicrotome (Leica, Nußloch, Germany) and placed on slides coated with polylysine, diluted 1:10 with distilled water (Sigma, Deisenhofen, Germany). After air drying, sections were fixed with absolute acetone at room temperature for 2 min and then transferred into Triton X 100 (Sigma, Deisenhofen, Germany), 0.2% in phosphate-buffered saline (PBS).
The primary antibody used to immunostain the Glut1 protein was a polyclonal antibody (Calbiochem/Novabiochem, Bad Soden, Germany, Lot B10094), developed in rabbit, against a synthetic 13-amino acid C-terminal sequence peptide of Glut1, as previously described by Haspel et al. (1988). Brain sections were incubated for 30 min at room temperature in a humid incubation chamber, together with primary antibodies diluted 1:25 in PBS. This dilution allowed saturation of the antigens, as determined by a saturation curve. Brain sections were washed three times for 5 min each in PBS, to which 0.2% Tween 20 (Sigma) had been added, and then incubated with the secondary antibodies anti-rabbit IgG for 30 min. These secondary antibodies were a combination of [35S]-labeled (Amersham, Braunschweig, Germany) and unlabeled (Dianova, Hamburg, Germany) anti-rabbit IgG. Specific activity was set to 0.35 Ci/mM by an appropriate mixture of the two secondary antibodies. The result of this procedure was that all ODs of brain sections measured were within the range of ODs of the radioactive standard sets used. Secondary antibodies were given in a concentration of 3 nM/ml, which safeguarded saturation of the primary antibodies. Slides were then rinsed three times for 5 min each in 0.2% Tween 20 in PBS and dehydrated in ascending concentrations of alcohol (50, 75, 90%). After air drying, slices were exposed to Kodak MinR1 x-ray film for 2 weeks. Local tissue concentrations of [35S] were determined by densitometric analysis of the autoradiograms with an image analyzing system (MCID, Imaging Research Inc., St. Catharines, Ontario, Canada). The measured ODs were converted to tissue concentrations by a set of [14C] standards that were exposed together with the brain sections. The [14C] standards were adjusted to [35S] by the following procedure. Different activities of [35S] were added to brain paste, which was cut into 10 μm thick sections after freezing. Cryosections were exposed to x-ray film for 2 weeks together with the [14C] standards. Therefore, appropriate activities of [35S] could be assigned to the [14C] standard set.
The mean concentration of Glut1 protein in whole brain was determined from brain paste of adult rats that was cut and immunostained by the same procedure as was native brain. Values demonstrated for each brain structure represent a mean of 14 measurements of ODs made for that structure in three consecutive sections from one rat each.
Measurement of capillary density
Capillary density was determined in six brains of Sprague–Dawley rats. After decapitation, brains were removed rapidly and shock-frozen in 2-methylbutane (Merck) chilled to −40°C and embedded in M-1 embedding matrix (Lipshaw). Then, 5 μm thick coronal sections were cut in a cryomicrotome (Leica) and placed on slides coated with poly-l-lysine, (Sigma). After air drying, sections were fixed with absolute acetone at room temperature for 2 min and then transferred into PBS. To immunostain the basal lamina of blood vessels, polyclonal anti-human fibronectin developed in goat (Sigma) was used as primary antibody diluted 1:50 in PBS. The secondary antibody was a 5[(4,6-dichlorothriazin-2-yl)amino]fluorescein (DTAF)-conjugated polyclonal donkey anti-goat antibody (Dianova) diluted 1:1600 in PBS. Cryosections of locations similar to those used for Glut1 staining and for measurement of LCGU were observed in a fluorescent microscope (Axioplan, Zeiss, Oberkochen, Germany). The fluorescent filter combination for DTAF consisted of a 450–490 nm primary filter, a 510 nm dichroic interference mirror, and a 520 nm secondary filter. Each brain section was photographed with Kodak Elite 400 film. Color slides of the fibronectin-stained capillaries were taken from each brain structure. Slides were placed on an illuminated screen and magnified 25-fold through a microscope (Leica).
Test of antibody specificity
The specificity of the antibodies used was tested by several approaches. In one series, primary antibodies were omitted. In a second series, the Glut1 antibody was preincubated with an excess (1.6 mg/ml) of a synthetic C-terminus peptide of Glut1 (WAK-Chemie, Bad Homburg, Germany). In these experiments, negligible fluorescent staining or ODs were detected. In another setting, preimmune rabbit serum, diluted 1:25, was incubated instead of primary antibodies. Secondary antibodies were used in a concentration as usual. No specific pattern was observed. Altogether, these experiments indicated only negligible nonspecific effects of the antibodies used.
Measurement of LCGU
For measurement of LCGU five Sprague–Dawley rats were investigated. The quantitative 2-deoxyglucose method developed by Sokoloff et al. (1977) was applied to conscious rats using the standard 45-min protocol (Grünwald et al., 1993). These animal experiments were in accordance with institutional guidelines. Analyses of Glut1 densities and LCGU were performed in corresponding autoradiograms at identical locations for each structure investigated. Both Glut1 density and LCGU were determined for a total area of at least 2.5 mm2 in each brain region and animal. Great care was taken to analyze the same regions and areas for both parameters.
Statistical analysis
All data are expressed as means ±SD. Since the parameters of interest could not be determined in identical animals, different groups of animals were compared. To correlate parameters measured, a linear regression line was calculated to identify the relationship between mean Glut1 density and either mean LCGU or mean capillary density of each brain structure (Figs. 2 and 3). The level of significance for testing the value of r was set at p < 0.01. Although the experimental design does not allow for rigorous statistical estimation of the correlations, nevertheless, the approximations from the “typical animal” mean profiles do indicate positive relationships between the Glut1 density and LCGU or capillary density.
RESULTS
A prerequisite for the quantification of transporter density in brain sections is application of primary and secondary antibodies in concentrations that would saturate all epitopes. Therefore, 10 increasing concentrations of the primary antibody were tested together with an excess of secondary antibodies and vice versa. Since saturation was obtained at a dilution of the first antibody <1:25 and at a concentration of 0.3 nM/ml for the secondary antibody, these concentrations were used in all experiments.
Figure 1 shows, for comparison, typical autoradiograms of Glut1 and of 2-deoxyglucose. Distribution of radioactivity appeared to be comparable, although not completely identical, in many brain structures. Preabsorption of Glut1 antibody with synthetic peptide showed negligible nonspecific staining, as shown in Fig. 4. To verify the degree of similarity in the local pattern of Glut1, LCGU, and capillary density, their distributions were compared in 13 brain structures. Results are shown in Table 1. It is evident that the mean density of Glut1 in the brain structures investigated varies from 65% below brain paste to 15% above brain paste. The highest density of Glut1 was measured in the inferior colliculus; this is congruent with the finding of the highest LCGU and capillary density. The lowest density of Glut1 was observed in the cerebellar peduncle; this corresponds to the lowest LCGU and a low capillary density in this structure. The mean density of Glut1 is compared to the mean LCGU and mean capillary density in Figs. 2 and 3. A correlation was found between LCGU and density of Glut1 for the brain structures investigated (Fig. 2). A significant correlation was also found between capillary density and Glut1 density (Fig. 3). In Figs. 1 and 2 and in the respective calculation, the epiphysis was not included because its properties differ from those of the other brain structures. As a leaky barrier structure, the epiphysis lacks Glut1 at its capillaries (Rahner-Welsch et al., 1995). In contrast to the other brain structures, which are of the tight barrier type and in which Glut1 is found predominantly at the capillaries, the epiphysis shows a diffuse distribution of Glut1.
Comparison of Glut1 density with LCGU and capillary density in various brain structures (means ± SD)
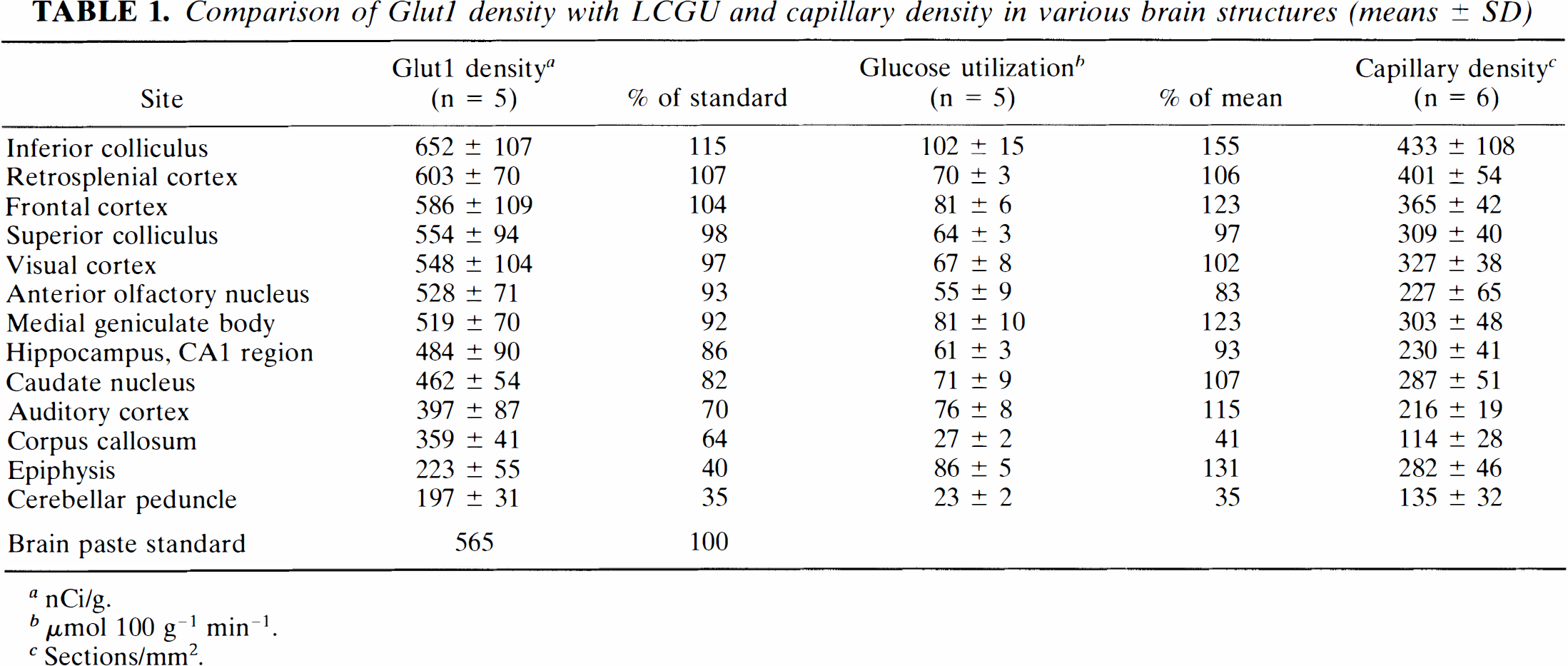
nCi/g.
μmol 100 g–1 min–1.
Sections/mm2.
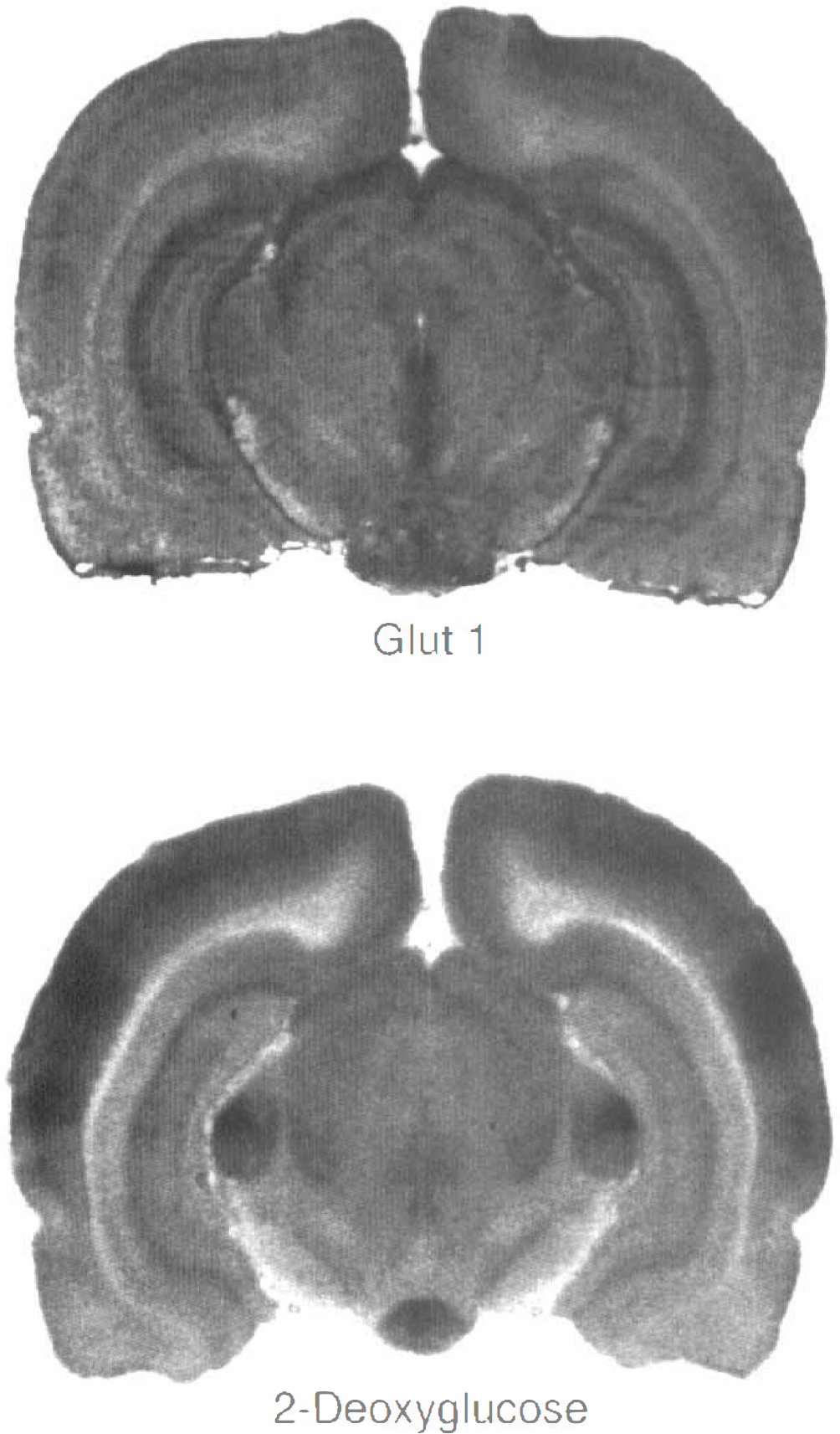
Autoradiograms of the distribution of Glut1 (upper part) and 2-deoxyglucose (lower part). A higher OD corresponds to a higher density of Glut1 or a higher glucose utilization, respectively. Both Glut1 and 2-deoxyglucose are distributed heterogeneously in the rat brain.
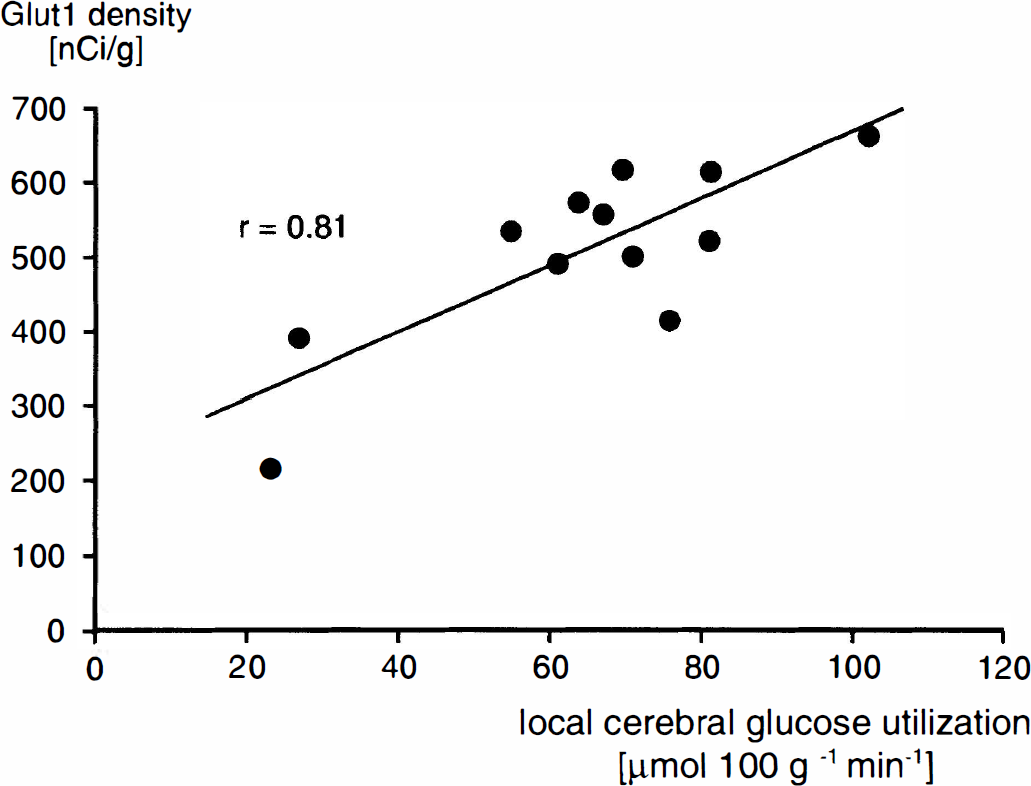
Relationship between LCGU and Glut1 density in different brain structures. Each point represents the mean value determined for one brain structure in which either glucose utilization (five rats) or Glut1 density (five rats) was measured. A significant correlation exists between glucose utilization and Glut1 density (y = 4.46x + 221).
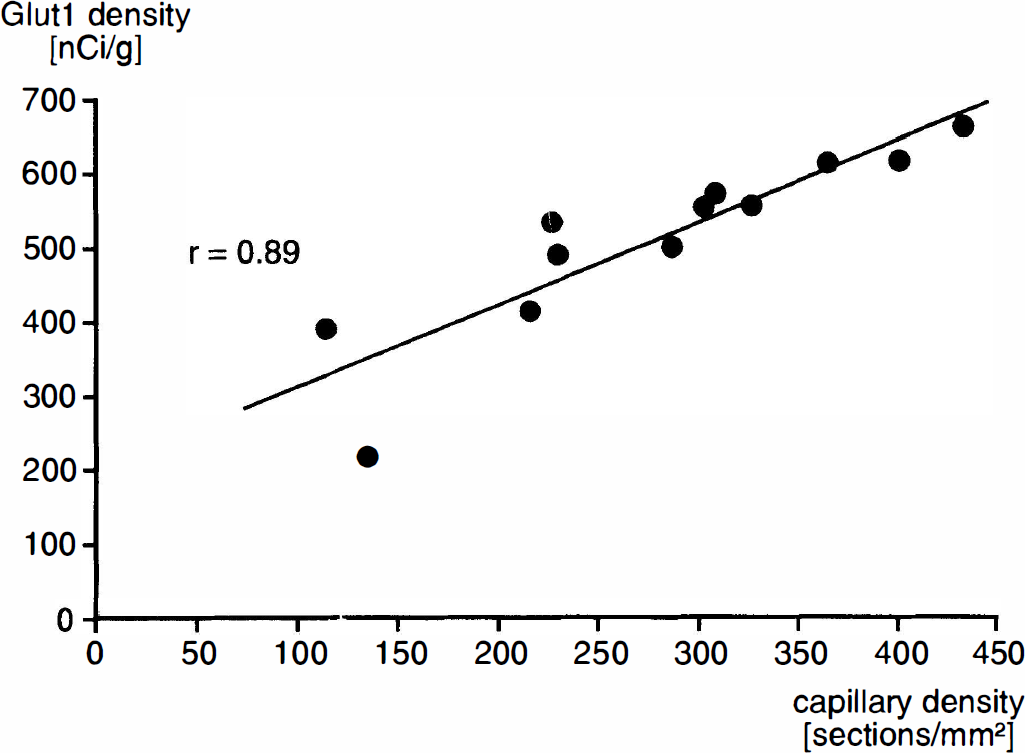
Relationship between local capillary density and Glut1 density in different brain structures. Each point represents the mean value determined for one brain structure in which either capillary density (six rats) or Glut1 density (five rats) was measured. A significant correlation exists between capillary density and Glut1 density (y = 1.10x + 203).
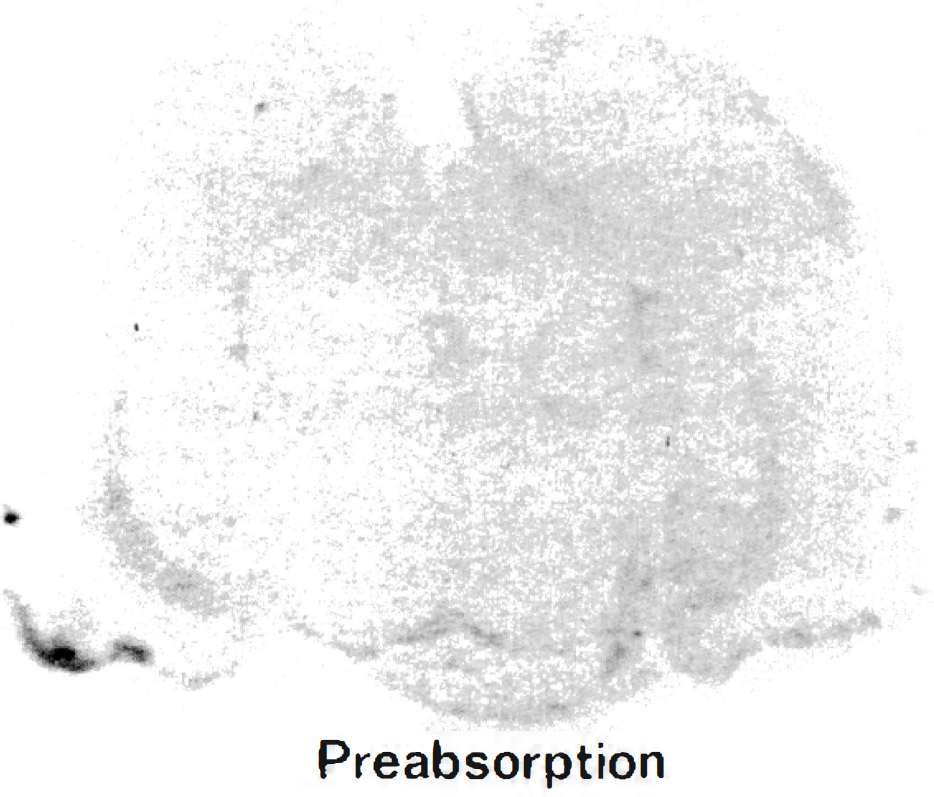
Control autoradiogram of a brain section stained by anti-Glut1 antibody after preabsorption of the antibody by the synthetic peptide. Negligible nonspecific staining was found.
DISCUSSION
In the present study, the local distribution of Glut1 has been quantified in the rat brain by an immunoautoradiographic method. Local heterogeneities were found in the pattern of Glut1 staining between different brain structures. The local density of Glut1 correlated with LCGU and capillary density in the brain structures investigated.
Discussion of method
The local resolution of the immunoautoradiographic method used for the measurement of the density of Glut1 is comparable to that of the 2-deoxyglucose method. Therefore, structures such as corpus callosum or cerebral peduncle, which appear to be too small to be dissected and analyzed accurately by blotting methods, may be investigated. Autoradiograms of Glut1 and LCGU can be directly compared, although they were obtained from different types of experiments. In 2-deoxyglucose studies, the tracer was injected i.v. into the intact animal 45 min before decapitation, whereas in Glut1 experiments, it is topically applied to brain cryosections after decapitation. Therefore, Glut1 autoradiograms showed the morphology of the glucose transporter protein whereas the 2-deoxyglucose autoradiogram represented metabolic activity of the respective brain structure as related to brain function. The third parameter measured in the present study, capillary density, was linked to both other parameters, since Glut1 is located mainly at the capillaries (Maher et al., 1994) and LCGU is correlated to capillary density (Klein et al., 1986).
Brain paste was used as a reference for the measured densities of Glut1. Since brain paste contains an average concentration of Glut1, the measured tissue concentrations could be related to this average concentration. For calculation of tissue concentrations of [35S] from the measured ODs in the autoradiograms, [14C] standards were calibrated to yield [35S] activities. [14C] standards were used because of the long lasting half-life of [14C] and its closeness in radiation and energy to [35S].
Primary and secondary antibodies were applied in saturating concentrations as verified by saturation curves. Concentrations of antibodies used in the present study were high enough to saturate the antigenic epitopes and low enough to yield low nonspecific binding.
Discussion of results
In previous studies, the existence of Glut1 had been investigated with respect to barrier properties in a more qualitative way, using immunocytochemical methods. Glut1 protein was found at capillaries in tissue with BBB function and could not be detected at capillaries with leaky barrier properties (Harik et al., 1990; Young and Wang, 1990). This finding has been confirmed on the level of single brain structures by fluorescent double staining of capillaries (Rahner–Welsch et al., 1995). A more quantitative approach was chosen by Brant et al. (1993). Using quantitative immunoblot analysis, those authors found differences in the expression of Glut1 protein between brain regions. The finding of a heterogeneous distribution of Glut1 is in agreement with the present study.
Distribution of Glut1 in rat brain is correlated with distribution of glucose utilization, as shown in Fig. 1. This correlation appears to be specific for Glut1, since, in a previous study, no correlation could be found between LCGU and density of the neuronal glucose transporter, Glut3. This difference in patterns of Glut1 and Glut3 compared to that of LCGU might be explained by the divergent locations of Glut1 and Glut3. Whereas Glut1 is predominantly confined to the membranes of brain capillaries. Glut3 exists in the large compartment of parenchymal, at least neuronal, membranes. On the basis of receptor binding assays, Pardridge (1994) has calculated the concentrations of glucose transporters at brain capillary and cell membranes. He concluded that an ∼50-fold greater mass of glucose transporters exists in the brain parenchyma than at the BBB. This could mean that Glut3 may exist in abundance in the brain, thus accounting for the lack of correlation between Glut3 and LCGU (Zeller et al., 1995), whereas Glut1, although existing in high density at the capillary membranes, may yield a much lower average tissue concentration because of the limited fraction of brain tissue comprised by capillaries. The tight association between capillary density and Glut1 (Fig. 2) demonstrates the intimate interrelationship of capillaries and Glut1.
Recent studies using immunoblot analysis have shown that Glut1 exists in two molecular mass forms: a 55-kDa form, which is expressed in high concentrations predominantly at the endothelium of the BBB (Maher et al., 1994; Pardridge et al., 1990; Devaskar et al., 1991), whereas the 45 kDa form appears to be located either diffusely over the nonvascular parts of the rat brain (Kumagai et al., 1994; Maher et al., 1993; Vannucci, 1994) or, more specifically, at gray matter astrocytic foot processes in human and monkey brains (Morgello et al., 1995). Although the present study, did not allow differentiation between both mass forms of Glut1, results obtained from the epiphysis may give some hint concerning their distribution. The epiphysis, a leaky barrier structure, lacks Glut1 at its capillaries, as is known also for other circumventricular organs (Rahner–Welsch et al., 1995; Harik et al., 1990). Therefore, activity of Glut1 measured in this structure appears to reflect the nonvascular tissue concentration corresponding to the 45 kDa form. The low Glut1 content measured in the epiphysis in the present study is in accordance with the existence of the 45 kDa form in a lower concentration than the 55 kDa form (Pardridge et al., 1990). These results are also in accordance with those of Maher et al. (1992), who detected, using Western blot analysis, a lower amount of Glut1 in the epiphysis than in any other gray matter structure.
The heterogeneous distribution of Glut1 protein observed in the present study and by Brant et al. (1992) is not paralled by comparable heterogeneities of Glut1 mRNA. In situ hybridizations on mouse (Nagamatsu et al., 1992) and rat brain (Bondy et al., 1992) have shown a diffuse homogeneous pattern of Glut1 mRNA in gray and white matter. This discrepancy between Glut1 mRNA and Glut1 protein distribution indicates that the levels of glucose transporter mRNA do not directly reflect the amount or location of the protein (Maher et al., 1994). This conclusion is in accordance with findings obtained for Glut3 that have shown the existence of regional discrepancies between Glut3 mRNA and Glut3 protein (Bondy et al., 1992).
In summary, the present study demonstrates a heterogeneous distribution of Glut1 density in the rat brain. This heterogeneity is related to both LCGU and local capillary density.
Footnotes
Acknowledgment:
Supported by the Deutsche Forschungsgemeinschaft (W. K.) and Graduate Program in Molecular and Cellular Neurobiology, University of Heidelberg (K. Z.).
