Abstract
We have investigated whether there is a duration threshold for the effects of phenylephrine-induced hypertension on CBF, brain energy metabolism, and cerebral parenchymal specific gravity (SG) following transient forebrain ischemia in gerbils. Sixty gerbils were randomly assigned to one of the four treatment groups: one control group and three groups subjected to an increase of 25 mm Hg in MABP induced by treatment, 30 min after reperfusion, with phenylephrine for 15 min, 30 min, or 60 min. The local CBF was measured continuously, and the SG was evaluated 120 min after reperfusion. Sequential changes in brain energy metabolism, as shown by the ratio of phosphocreatine to inorganic phosphate (Pi), the β-ATP/Pi ratio, and intracellular pH, were also measured. The 15-min induced hypertension regimen was most suited to the recovery of brain energy metabolism, which was associated with an increase in local CBF and a decrease in cerebral edema. These results demonstrate that a suitable duration can be chosen to optimize the beneficial effects of phenylephrine-induced hypertension on ischemic brain injury following transient forebrain ischemia.
The recovery of brain energy metabolism following cerebral ischemia is important in maintaining cerebral function, and many attempts to achieve it have been reported (e.g., Chen et al., 1992). Induced hypertension is thought to be an attempt at the recovery of brain energy metabolism associated with an increase in CBF after cerebral ischemia (Table 1); paradoxically, it is liable to produce cerebral edema (Kogure et al., 1981). We hypothesized that a shorter duration of induced hypertension might improve delayed postischemic hypoperfusion, brain energy metabolism, and cerebral edema and that a longer duration of induced hypertension might increase cerebral edema and worsen brain energy metabolism. The aim of the present study was to evaluate the duration threshold of the effects of phenylephrine-induced hypertension on brain energy metabolism and cerebral edema following transient forebrain ischemia in gerbils.
Published effects of induced hypertension on the ischemic brain
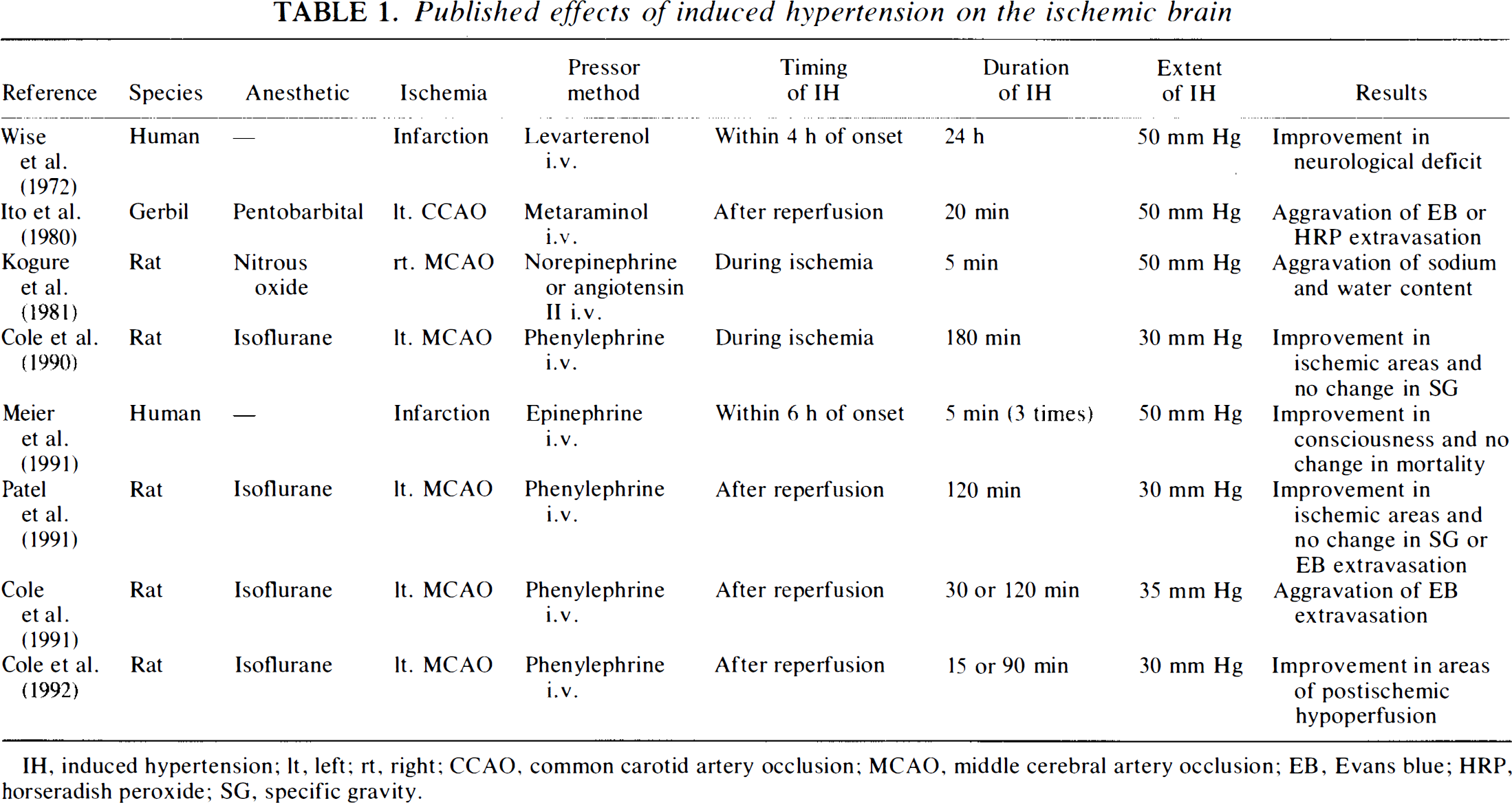
IH, induced hypertension; lt, left; rt, right; CCAO, common carotid artery occlusion; MCAO, middle cerebral artery occlusion; EB, Evans blue; HRP, horseradish peroxide; SG, specific gravity.
MATERIALS AND METHODS
Anesthesia was induced and maintained, when the animal showed an auditory response, in 60 adult male Mongolian gerbils (Meriones unguiculatus), weighing 60–90 g (mean, 73.6 ± 7.4 g), by intraperitoneal injection of 40 mg/kg and 10 mg/kg of pentobarbital sodium, respectively. PE-10 catheters were inserted into the femoral artery to measure MABP and into the femoral vein for drug infusion. The bilateral common carotid arteries were exposed through a midline ventral cervical incision. MABP was measured before and during transient forebrain ischemia and for 120 min after reperfusion. The rectal temperature was monitored and maintained close to 37.0°C in a heated box at almost 100% humidity throughout the experiment.
After 30 min of stabilization of the MABP and rectal temperature, bilateral common carotid artery occlusion (BCCAO) was performed for 60 min using aneurysm clips. The gerbils were then assigned randomly to one of the following four treatment groups, (a) In the 15-min induced hypertension (15HT) group (n = 15), the MABP was increased 30 min after reperfusion by 25 mm Hg by phenylephrine infusion (10 μg/kg/min) for 15 min, followed by saline infusion for 45 min. (b) In the 30-min induced hypertension (30HT) group (n = 15), animals were infused with phenylephrine for 30 min, followed by saline infusion for 30 min. (c) In the 60-min induced hypertension (60HT) group (n = 15), animals were infused with phenylephrine for 60 min. (d) In the control group (n = 15), animals were infused with saline for 60 min. The CBF and specific gravity (SG) were measured in eight, phosphorus 31 nuclear magnetic resonance (NMR) spectroscopy in seven, and MABP in 15 gerbils in each group.
The local CBF was measured continuously using a laser Doppler flowmeter (Periflux PF 2B; Perimed, Stockholm, Sweden). A laser Doppler flow probe (PF 302, diameter 0.45 mm; Perimed) was placed stereotaxically in a hole (0.8 mm in diameter) made in the parietal bone 1–2 mm posterior to the coronal suture and 2 mm lateral to the midline, located microscopically to avoid large dural and pial vessels. Measurements were taken continuously before and during transient forebrain ischemia and for 120 min after reperfusion. The pre-occlusion local CBF values were taken as the 100% level, and each experimental value was expressed as a percentage of this value.
Eight animals in each group were killed by decapitation 120 min after reperfusion. The brain was rapidly removed and sectioned along the coronal planes, where the local CBF was measured. Seven specimens were taken from the ischemic areas of the bilateral cerebral cortex using a 2.0-mm punch needle and placed for determination of SG in a bromobenzene-kerosene density gradient column (Marmarou et al., 1978), which was calibrated daily with potassium sulfate standards. The SG of each sample was determined by linear regression analysis; only columns with correlation coefficients >0.9950 were used. The two samples with the extreme values were discounted, and the average SG was calculated on the basis of the other five samples.
In vivo phosphorus 31 NMR spectroscopy was performed using a 6.34-Tesla spectrometer (JEOL JNM-GSX270; Nihon Denshi Co., Tokyo, Japan). The areas of the inorganic phosphate (Pi), phosphocreatine (PCr), and β-ATP peaks were measured in order to calculate the PCr/Pi and β-ATP/Pi ratios, the parameters of brain energy metabolism. The intracellular pH (pHi) was calculated from the chemical shift difference between the Pi and PCr resonances (Petroff et al., 1985). Measurements were made before BCCAO and 30, 120, 150, 180, and 240 min after reperfusion. During the phosphate 31 NMR measurement, the temperature in the probe was maintained at 37°C by warm water circulating in a plastic tube.
The effects of induced hypertension on the four groups were compared using repeated-measures analysis of variance (ANOVA). Differences between the four groups at each time point were analyzed using one-way ANOVA. If the p value was <0.05 for each analysis, the Bonferroni correction for multiple comparisons was employed to evaluate the significance of differences between all pairs from the four groups. Within-group comparisons were made using the Student's t test for paired data. Differences were considered statistically significant if the p value was <0.05. All results are also given as the mean ± SD.
RESULTS
Continuous intravenous infusion of phenylephrine produced a 24 ± 12 mm Hg increase in MABP, which was statistically significant (p < 0.01) (Fig. 1A). The MABP decreased to the pre-occlusion level within a few minutes of termination of infusion. In the control group, the local CBF fell continuously for the first 45 min after reperfusion to 71.1 ± 21.7% of the pre-occlusion level (p < 0.05) and remained at this level until the end of the experiment (Fig. 1B). In all HT groups, the local CBF increased significantly until 45 min after reperfusion, at which time the respective values for the 15HT, 30HT, and 60HT groups were 132.0 ± 18.8%, 127.9 ± 13.3%, and 126.5 ± 19.9% of the pre-occlusion level (p < 0.01 in all cases); the local CBF decreased, reaching the control group level 60 min after reperfusion in the 15HT group and 75 min in the 30HT and 60HT groups.
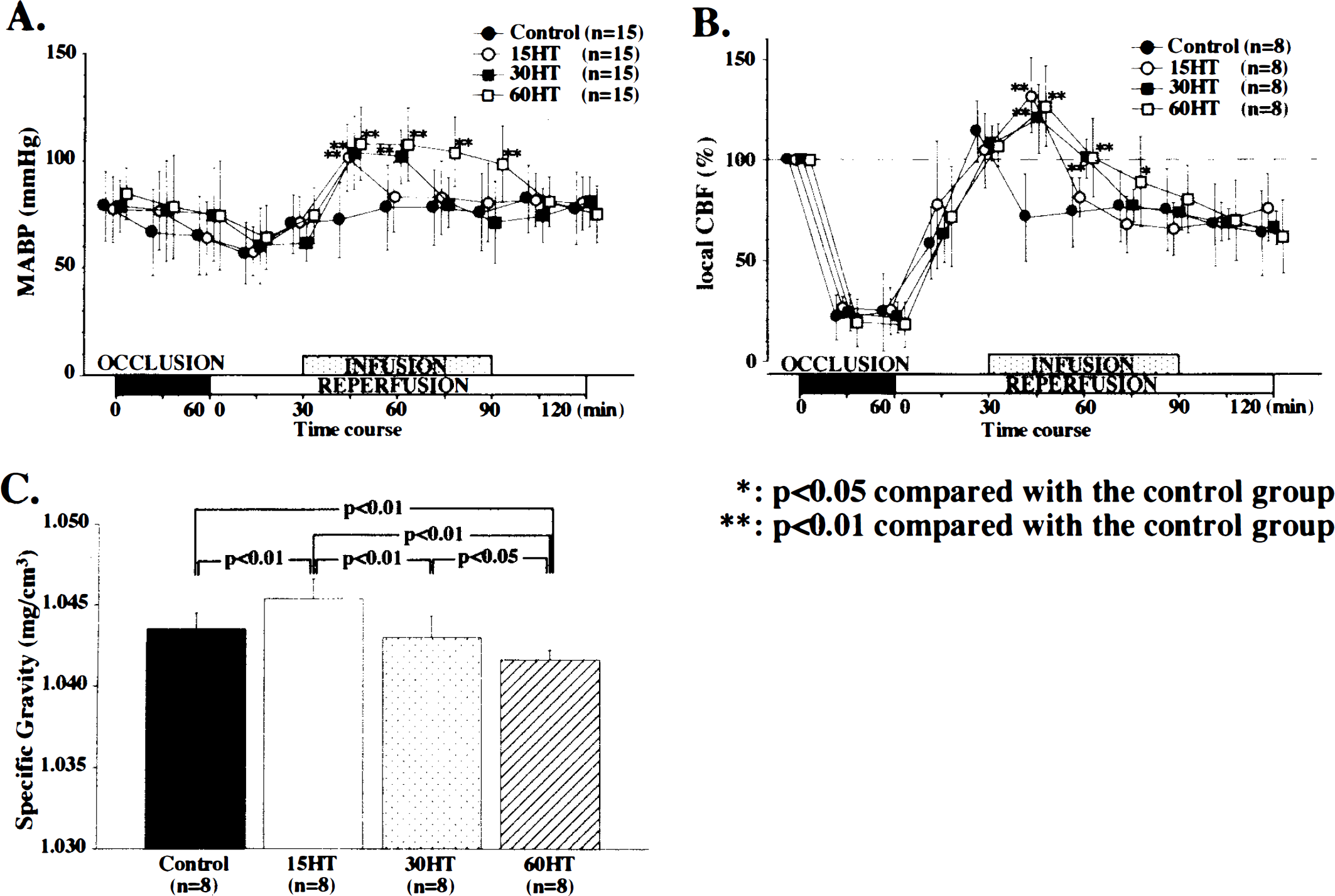
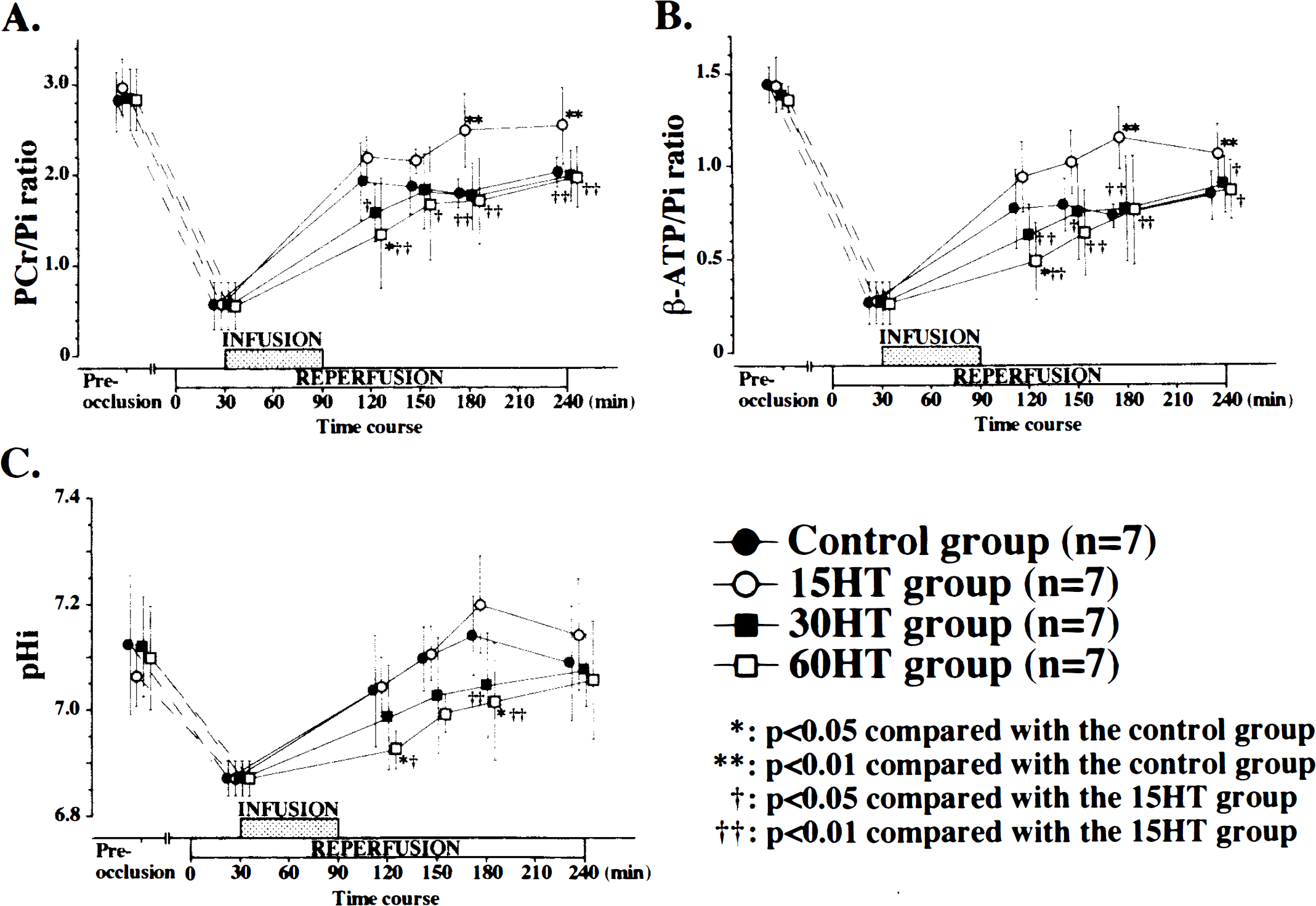
The SG of the samples from the 15HT group was greater than that from the control group (p < 0.01). However, the SG of those from the 30HT and 60HT groups showed a significant decrease, which was dependent on the duration of induced hypertension (Fig. 1C). Over the 240-min period, the recoveries of the PCr/Pi and β-ATP/Pi ratios were significantly greater in the 15HT group compared with the control group (p < 0.001) (Fig. 2A,B) or the 30HT and 60HT groups. Following BCCAO, the pHi acidified; its recovery was, at certain time points, significantly slower in the 30HT and 60HT groups (p < 0.05) (Fig. 2C).
DISCUSSION
Our results suggest that a shorter duration (15 min) of phenylephrine-induced hypertension after reperfusion may be beneficial for the recovery of brain energy metabolism associated with increasing local CBF and decreasing cerebral edema. A longer duration (30 and 60 min) of induced hypertension, on the other hand, may have a deleterious effect on the recovery of brain energy metabolism and may exacerbate cerebral edema. Two possible mechanisms are thought to be responsible for the beneficial effects of a shorter duration of phenylephrine-induced hypertension. First, the main effect of phenylephrine-induced hypertension on brain energy metabolism derives from an increase in local CBF. The noradrenaline sensitivity of the α-adrenergic receptors of the cerebral arteries is significantly less than those of many other arteries (Bevan et al., 1987), and the α1-agonist phenylephrine therefore increases cerebral perfusion pressure by causing contraction of the extracerebral tissue arteries, resulting in an increased local CBF in the ischemic brain, where cerebral autoregulation is impaired. Second, phenylephrine may also directly ameliorate ischemic brain damage via α-adrenergic receptors, if phenylephrine can cross the blood-brain barrier (BBB). Koide et al. (1986) reported that circulating catecholamines may decrease brain damage either by penetrating the BBB or by modulating the activity of afferent fibers to the brain; however, this possibility seems unlikely, since in their study MABP increased in response to infusion of catecholamines.
It is possible that an increase in perfusion pressure may create an additional risk of exacerbating vasogenic cerebral edema (Kogure et al., 1981) and may also result in BBB changes (Ito et al., 1980). Many previous studies have shown that induced hypertension causes an increase in Evans blue extravasation from the cerebral vessels to the tissues (Table 1). In the present study, we found that a shorter duration (15 min) of phenylephrine-induced hypertension caused a decreased incidence of cerebral edema, while a longer duration (30 and 60 min) caused an increase (Fig. 1C). It therefore seems that there is a duration threshold for the effects of phenylephrine-induced hypertension on production of cerebral edema.
Some investigators have used the 60-min BCCAO model in gerbils in physiological and pharmacological studies (Kobayashi et al., 1977; Avery et al., 1984; Sasaki et al., 1989). In our preliminary study (unpublished data), using phosphorus 31 NMR spectroscopy, we investigated differences in recovery of brain energy metabolism following different durations (30, 60, and 90 min) of BCCAO under pentobarbital anesthesia. Disturbances in brain energy metabolism in the 30-min ischemia group were so slight that values returned almost to pre-occlusion levels 120 min after reperfusion, while in the 90-min ischemia group, they were so severe that the high mortality rate made it impossible to work with this group. These results show that it is necessary to choose a suitable period of ischemia such that brain energy metabolism plateaus below the pre-occlusion level while at the same time ensuring a high survival rate. We did not investigate the neurological outcome of these animals in the absence of anesthesia. However, the simultaneous occurrence of several biochemical and functional abnormalities investigated with phosphorus 31 NMR spectroscopy following ischemic insult suggests a causal relationship between membrane and mitochondrial development and neurological function (Nioka et al., 1993). For anesthesia we used pentobarbital, which is known to cause hypotension and to reduce perfusion pressure in the brain. There is a possibility that the prolonged ischemia we used in the pentobarbital-anesthetized gerbil creates an artificial condition in which transient induced hypertension appears to improve outcome. Thus, further investigation of the effects of experimental conditions seems to be required.
In the present study, in which we produced phenylephrine-induced hypertension 30 min after reperfusion, reactive hyperemia occurred soon after reperfusion and continued for 30 min (Fig. 1B). When phenylephrine-induced hypertension is produced during the period of reactive hyperemia, it is possible that the increased perfusion pressure may produce vasogenic cerebral edema (Kuroiwa et al., 1985). Previous studies (Levy et al., 1979) have shown that postischemic hypoperfusion occurs following reactive hyperemia and that during postischemic hypoperfusion there is a discrepancy between CBF and the demand for energy metabolism. Thus, the initial period of postischemic hypoperfusion, that is, 30 min after reperfusion, is considered the most effective time to apply phenylephrine-induced hypertension (Cole et al., 1992).
In the clinical context, our forebrain ischemia reperfusion model might be similar to the situation after percutaneous transluminal recanalization in cerebral embolism or transient carotid artery occlusion in cases of cerebral aneurysm and carotid-cavernous fistula. If the appropriate duration of induced hypertension could be determined, induced hypertension might be a safe and useful therapy for ischemic stroke in the future. However, the appropriate duration of induced hypertension in the gerbil forebrain ischemia model may not be directly applicable to man, and such therapy should be used with caution until the appropriate duration has been determined.
Footnotes
Abbreviations used
Acknowledgment:
We thank Tomohiro Matsuyama,
