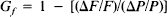Abstract
In cats anesthetized with α-chloralose, auto-regulation of blood flow (F) in the middle meningeal and common carotid arteries was assessed by bleeding and subsequently reinfusing the animals to achieve a 25% step reduction in mean arterial blood pressure (P), while maintaining the systolic blood pressure >80 mmHg. The integrity of autoregulation was assessed by calculating the gain factor Gf = 1 — [(ΔF/F)/(ΔP/P)]. Cats were examined intact, after hexamethonium (10 mg/kg), and after papaverine (6 mg/kg). Reduction of blood pressure of 25 to 60 mmHg produced equivalent drops in carotid blood flow (Gf = 0.041 ± 0.34; mean ± standard deviation, n = 12). There were only small changes in flow in the middle meningeal artery during this procedure (Gf = 0.91 ± 0.29). Hexamethonium did not block autoregulation in the middle meningeal artery (Gf = 0.92 ± 0.13, n = 4). However, papaverine almost completely abolished the ability of the artery to autoregulate (Gf = 0.10 ± 0.16, n = 7). The results suggest that the middle meningeal artery possesses an ability similar to that of the cortical circulation to autoregulate its blood flow through intrinsic, non-neuronal mechanisms. This will have important implications for the study of disturbances of dural arterial control in migraine and other headaches.
Migraine headache seems to involve perception of pain from the blood vessels of the dura, including the middle meningeal artery (Kimmel, 1961; Moskowitz, 1990; Penfield and McNaughton, 1940). Control mechanisms of cranial blood vessels, including those of the middle meningeal artery, are also disturbed (Moskowitz, 1990; Olesen et al., 1990; Totaro et al., 1992). The dural arteries are innervated by sympathetic (Keller et al., 1989), parasympathetic (Hara et al., 1989; MacKenzie and Scatton, 1987), and sensory (Mayberg et al., 1984; Moskowitz et al., 1990; Steiger et al., 1982) neurons, all of which have the potential to alter blood vessel caliber and blood flow within the arteries. There has been much speculation about the roles these three neuronal innervations might play in both the sensory and blood-flow disturbances that occur in migraine (Moskowitz et al., 1990).
Although autoregulation (the maintenance of blood flow in the face of changes in perfusion pressure) occurs to some extent in most tissues, attention has focused on tissues with high metabolic requirements or low tolerance to ischemia, such as cerebral (Paulson et al., 1990) and renal (Pollock and Banks, 1991) tissues. Autoregulation of blood flow is a prominent feature of the cortical circulation (Florence and Seylaz, 1992; Lassen, 1964; Mchedlishvili et al., 1973; Yoshida et al., 1966) and is not mediated by neuronal mechanisms, although the “envelope” of autoregulation may be modified by the sympathetic nervous system (Eklof et al., 1977; Gotoh et al., 1971; Mchedlishvili et al., 1973; Thomas and Bannister, 1980). Because the dura appears to be mainly a structural tissue without a major metabolic demand, it is often implicitly assumed that its blood supply is not critical, is dependent only on humoral or efferent neuronal mechanisms, and is not autoregulated. Two studies that have examined, inter alia, this assumption appear to confirm it (Chen et al., 1984; Linder, 1981). Embryo-logically, dural tissue is derived from mesoderm, unlike the pia mater and neural tissue, which are derived from ectoderm (Gil and Ratto, 1973). The middle meningeal artery is a branch of the external carotid artery in humans, entering the cranial cavity and invading the territory of the internal carotid supply (Padgett, 1948; Sensenig, 1957; Stattin, 1961). On these bases, the dural circulation might be expected to resemble the external carotid circulation and not have well-developed properties of autoregulation.
This study shows that this unspoken assumption is wrong and that the dural circulation of the cat is the equal of the cortical circulation in its ability to autoregulate its flow.
METHODS
Twelve cats, mean weight 2.93 ± 0.65 kg (mean ± standard deviation) were anesthetized with intraperitoneal injections of α-chloralose (60 mg/kg). The femoral artery and vein were cannulated to measure blood pressure and heart rate and to administer intravenous drugs and fluids. A side arm of the arterial catheter was used to remove and reinfuse arterial blood. Animals were intubated and ventilated with air to keep end-expiratory CO2 in the range of 3.5–4.0%.
Throughout the experiment, the animal was paralyzed with intravenous gallamine triethiodide, 20 mg/kg. The depth of anesthesia was assessed periodically during the experiment by testing for sympathetic responses to noxious stimulation (pupillary dilatation, tachycardia, elevated blood pressure) during paralysis or allowing the paralysis to wear off and testing for withdrawal reflexes. Supplementary doses of either chloralose or gallamine were given when necessary. Rectal temperature was monitored throughout the experiment with a thermistor and maintained at 37–38°C by means of a heating blanket.
Animals were mounted in a David Kopf stereotaxic frame, and craniotomies were performed using a 6-mm dental burr at low speed. The inner and outer diploe in the region of the middle meningeal artery were removed in layers, exposing an underlying translucent layer of periosteum adherent to the dura. While drilling was in progress, the surgical field was bathed in normal saline to minimize the possibility of thermal injury to the underlying tissues.
Although the hardware and software of the LaserFlo perfusion monitor is designed to measure tissue mean blood velocity and volume in a complex microvessel bed (Borgos, 1990), it is capable of measuring bulk laminar blood flow in large (0.1-mm diameter) blood vessels. A standard three-fiber probe was modified by removing it from its protective pencil and separating the fibers. One receiving fiber was disabled by cutting it back, and the remaining pair were glued together tangentially. This modification served two purposes: first, to halve the intensity of the signal received so that it fell within the working range of the LaserFlo meter; second, to change the relation between the emitter and two receiving fibers from triangular to linear between two fibers only. This ensured that the received signal was composed only of light reflected from the artery, rather than from both the artery and adjacent cortex. To ensure this latter condition, the probe was placed so that both emitter and reflector fibers were aligned along the long axis of the artery. Either the main trunk or major branch of middle meningeal artery was chosen for measurement, the criterion being that the blood velocity in the artery was ∼20% below the maximum Doppler shift displayable on the LaserFlo meter (8 KHz, although the instrument can register 16 KHz). The arrangement is shown in Fig. 1.
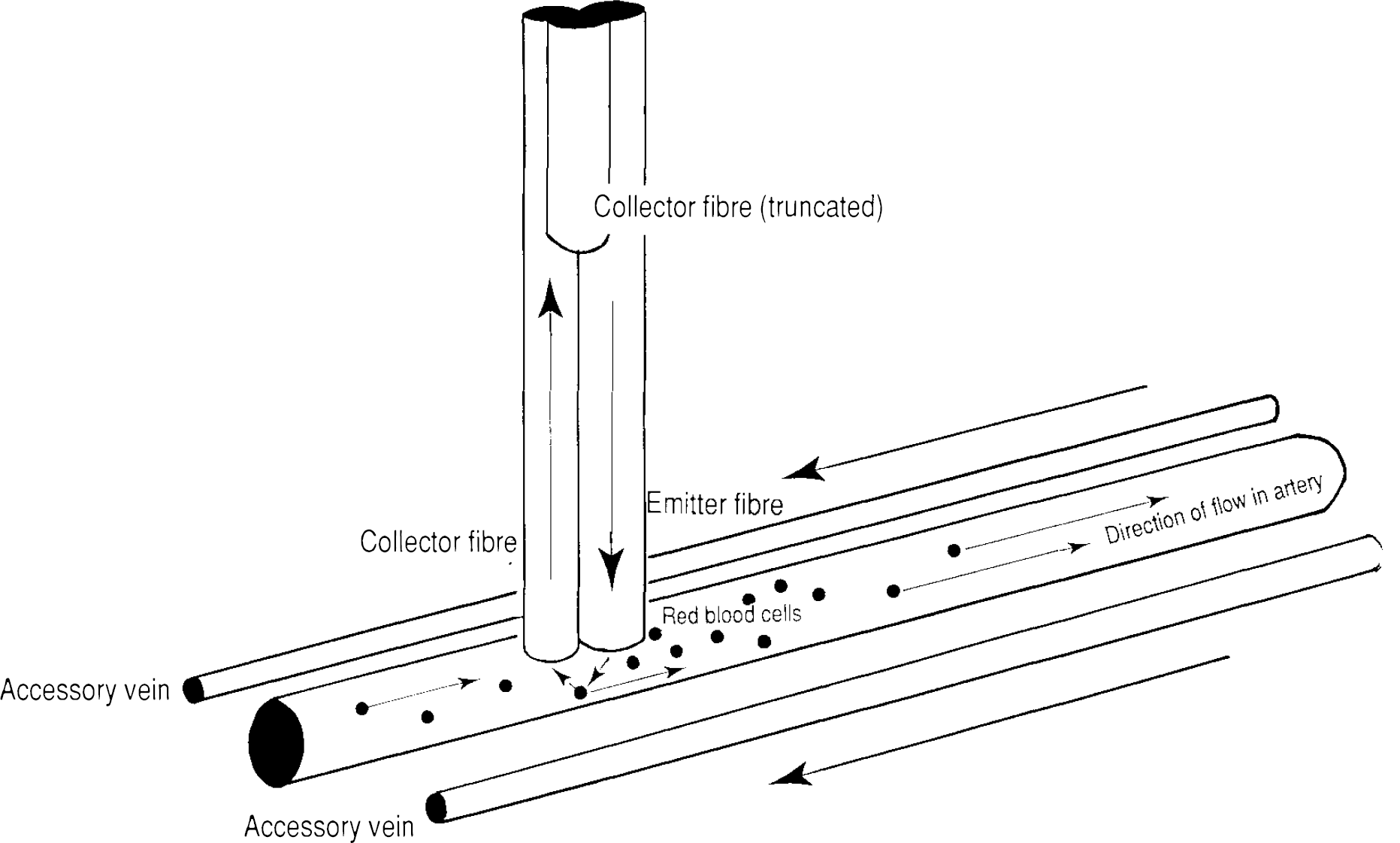
Measurement of laminar blood flow in the middle meningeal artery, using a modified TSI laser-Doppler probe system. One of the collecting fibres of the probe has been truncated. The remaining collector fibre and the emitter fibre have been aligned along the long axis of the artery. This geometry, in arteries of adiameterapproximately equal to that of the probe, implies that the average angle of incidence of light received at the collector fibre is 45°. The diagram also shows the accessory veins that accompany the middle meningeal artery.
This modified probe was calibrated as follows. The middle portion of a length of polyvinyl tubing of nominal internal diameter 0.2 mm (Dural Plastics, SV8, external diameter 0.5 mm) was dipped into a paraffin oil bath heated to 120°C, removed, and stretched by hand to produce a tapered section. Cat blood was circulated through the tubing by a Braun 1–3000 Perfusor perfusion pump, at rates of 0 to ∼35 μl/min. The laser Doppler probe was applied to this tube in the same way as it was applied to the middle meningeal artery in the in vivo experiments. Both Doppler shift and the “volume” signal were recorded for a number of different perfusion rates, up to the maximum Doppler shift recordable, and for three different tube diameters. Tube internal diameter in the tapering section was estimated as a fixed ratio (0.4) of the external diameter, measured with a micrometer.
The carotid arteries were exposed bilaterally and stripped of their fascia for a length of several centimeters. Common carotid blood flows were monitored by placing electromagnetic flow probes (1.5-mm opening) around each artery, and the signal recorded on EMI model flow-meters.
Autoregulation was assessed by withdrawing blood from a side arm of the arterial catheter to produce a >25% reduction in systemic blood pressure, while maintaining systolic blood pressure >60 mm Hg. The integrity of autoregulation was assessed by calculating the gain factor (Gf), as described by Norris et al. (1979).
where P = mean arterial blood pressure, and F = blood flow in the artery.
All signals were recorded on computer and analyzed off-line using GlobaLab data-acquisition and analysis software.
Video- or photomicroscopy was used to examine and record the anatomy and flow patterns in middle meningeal arteries of some cats. An MTI model 65 videocamera, with microscope lens was focused on the artery just upstream or downstream of the laser-Doppler probe. The camera was coupled to a NEC model NQ N9305A Videorecorder. Red blood cell velocity could be estimated by examining successive frames of the videorecording. Videomicroscopy was also used to monitor flow in the plastic tubes used to calibrate the modified laser-Doppler probe.
The following drugs were used in the experiments: α-chloralose (Calbiochem), gallamine triethiodide (May & Baker), papaverine hydrochloride (David Bull Laboratories), and hexamethonium hydrobromide (Sigma).
Blood pressure, blood flows, laser-Doppler signals, and gain factors are presented as means, with standard deviations. Student's t test was used to test the significance of difference between means of these parameters.
RESULTS
Circulation of cat blood through polyvinyl tubes produced “velocity” (Doppler shift) signals that were linearly related to blood flow from zero to nearly the limit of Doppler shift measurable (8 KHz) (Fig. 2A). The volume signal was practically constant over the velocity range at any one tube diameter and was proportional to the cross sectional area of the tube (Fig. 2B). RBC velocity at low velocities (up to 1 mm/s) could be measured by direct timing of the passage of cells across the video camera field of view. Higher velocities could be estimated by macroscopic measurement of the velocity of a small air meniscus deliberately induced into the tubing. An RBC velocity of ∼1 mm/s was associated with a laser-Doppler signal of 1.01 KHz and an air-bubble velocity of 3.63 mm/s was associated with a laser-Doppler velocity of 3.57 KHz.
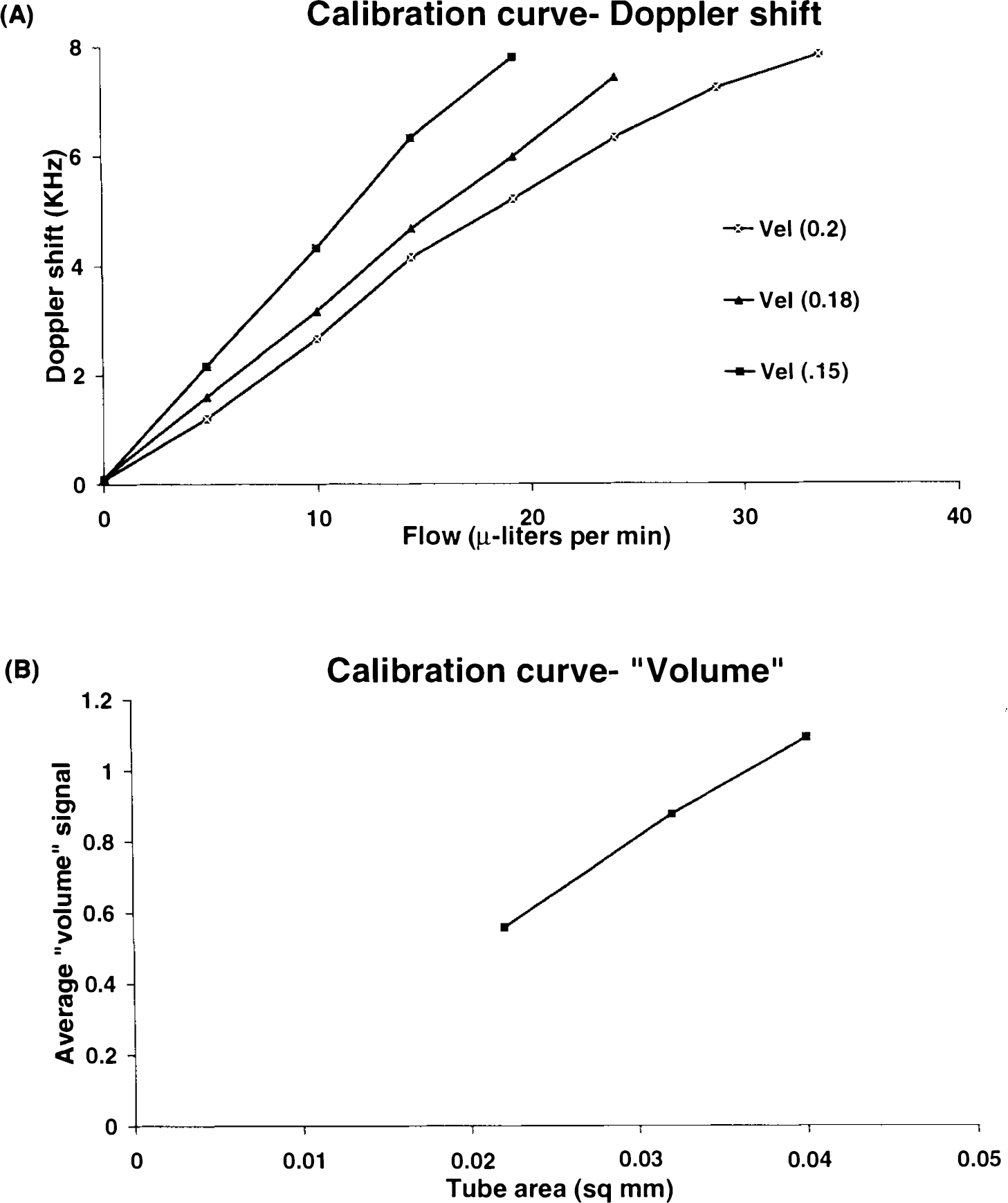
System calibration curves for blood flow in a polyvinyl tube of tapering diameter.
Video microscopy of the middle meningeal artery confirmed previous descriptions of its anatomy in humans (Kerber and Newton, 1973; Roland et al., 1987). The single artery was flanked on each side by a small vein. Blood flow in both the artery and vein was ∼6 mm/s, with flow in both the veins being opposite in direction to that in the artery (Fig. 1). In most cases, the laser-Doppler probe could be positioned, under dissecting microscope observation, so that it lay only over the artery (Fig. 1). Where this could not be achieved (—10% of experiments), the experiment was abandoned. Correct placement was possible in all twelve experiments described in this article.
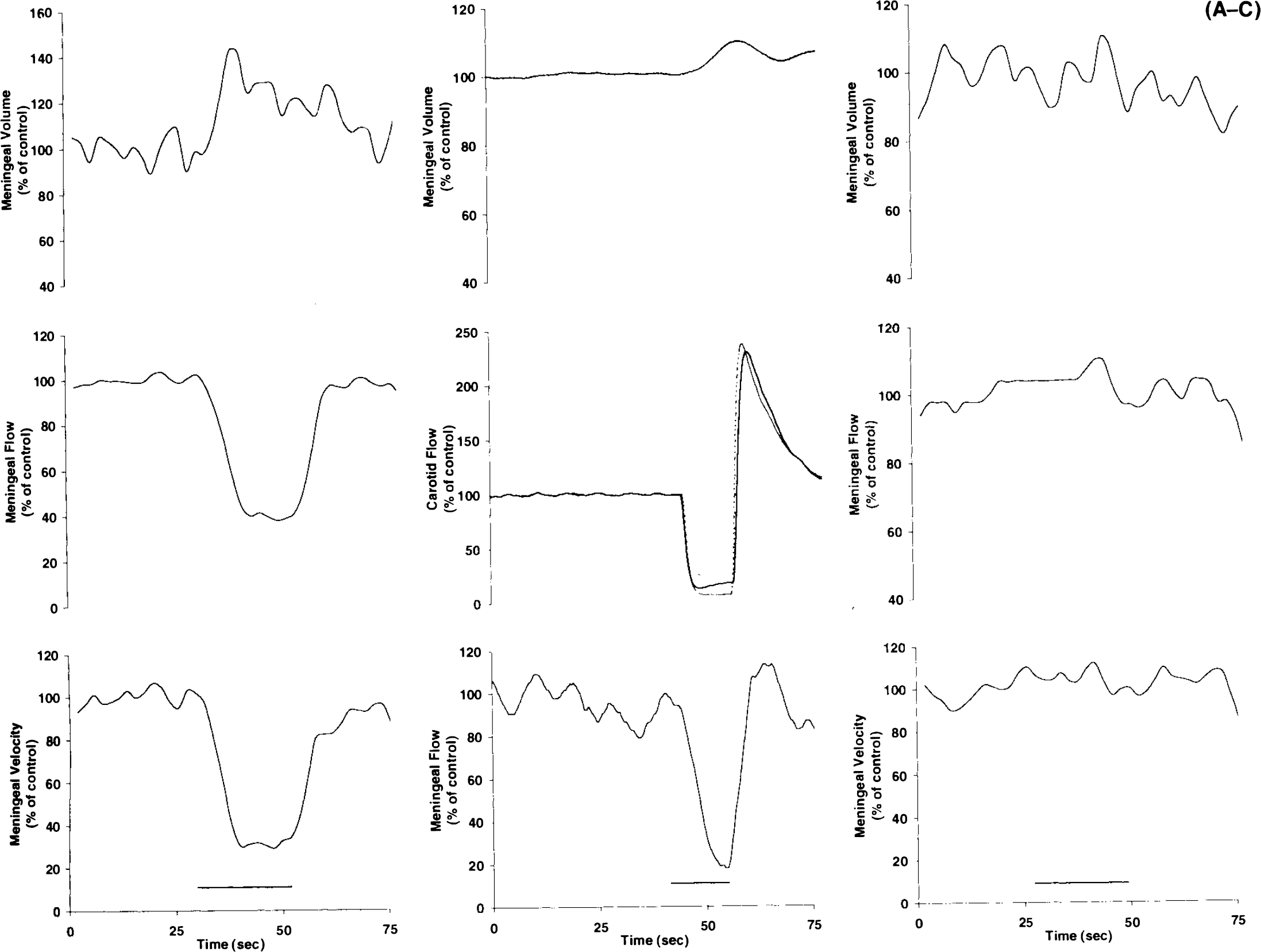
Occlusion of the middle meningeal artery with a small blunt probe just proximal to the recording site halted the visible movement of red blood cells in the artery and reduced the received velocity and flow signals to low levels (Fig. 3A). Occlusion of both carotid arteries reduced but did not halt the flow of blood in the middle meningeal artery (Fig. 3B). In some experiments, a small piece of opaque plastic was slipped underneath the dura and between the middle meningeal artery and the underlying cortex to shield the cortex from laser light emitted by the probe. None of the signal components was substantially altered by this procedure (Fig. 3C).
Mean resting levels of blood pressure and blood flows in the carotid and meningeal arteries in the 12 cats are shown in Table 1. Blood pressure and carotid flow were within normal ranges (Lambert et al., 1984). In the middle meningeal artery or its branches (diameter 100 to 300 μm), red blood cell (RBC) “velocity” was between 1.3 and 8.3 KHz Doppler shift and “volumes” in the range from 0.11 to 0.87 (instrumental units, reportedly equal to number of scattering incidents per photon and proportional to total RBC number). The resulting “flow” rate (velocity · volume · 60) ranged from 55 to 234, with a mean of 125. Although this figure is proportional to bulk arterial flow in milliliters per minute, the manufacturers assume it to be equivalent to flow expressed as ml min−1 100 g−1. The flow signal measurable in the dura adjacent to the probe was usually 10–20% of that measurable directly over the artery. Blood flow measured in the underlying cortex stripped of its dura is generally 18 laser-Doppler units (Piper et al., 1991).
Resting levels of cardiovascular parameters
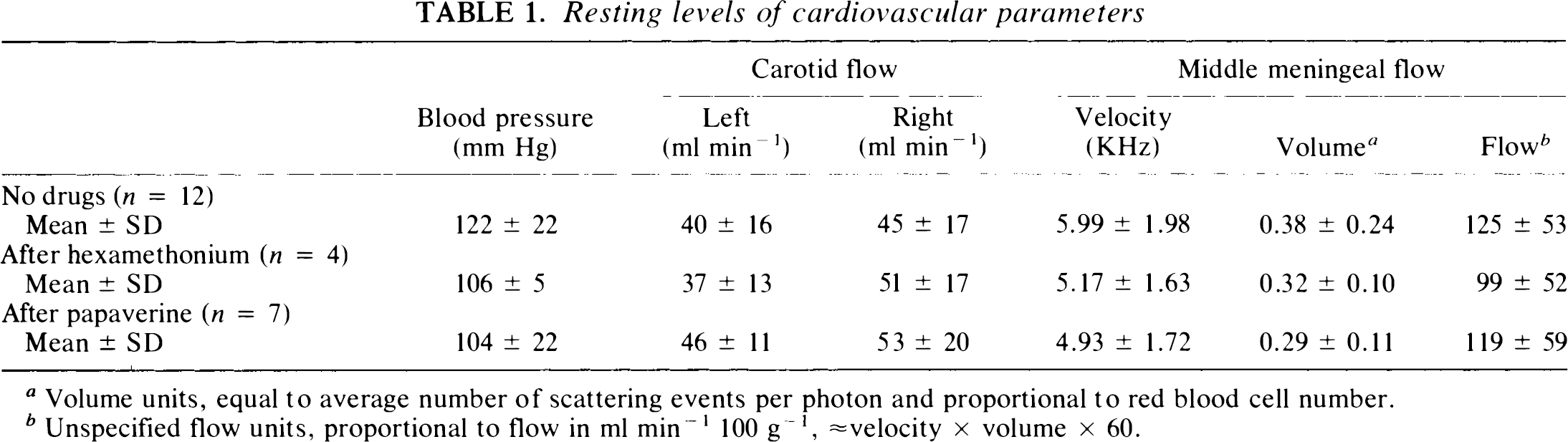
Volume units, equal to average number of scattering events per photon and proportional to red blood cell number.
Unspecified flow units, proportional to flow in ml min−1 100 g−1, ≈ velocity × volume × 60.
An average of 35 ml of blood was removed in graded steps, until blood pressure had decreased 25 to 70 mm Hg. The mean decline in blood pressure was 48 mm Hg. The mean blood volume removed, expressed as a percentage of estimated total blood volume, was 20% (Fig. 4).
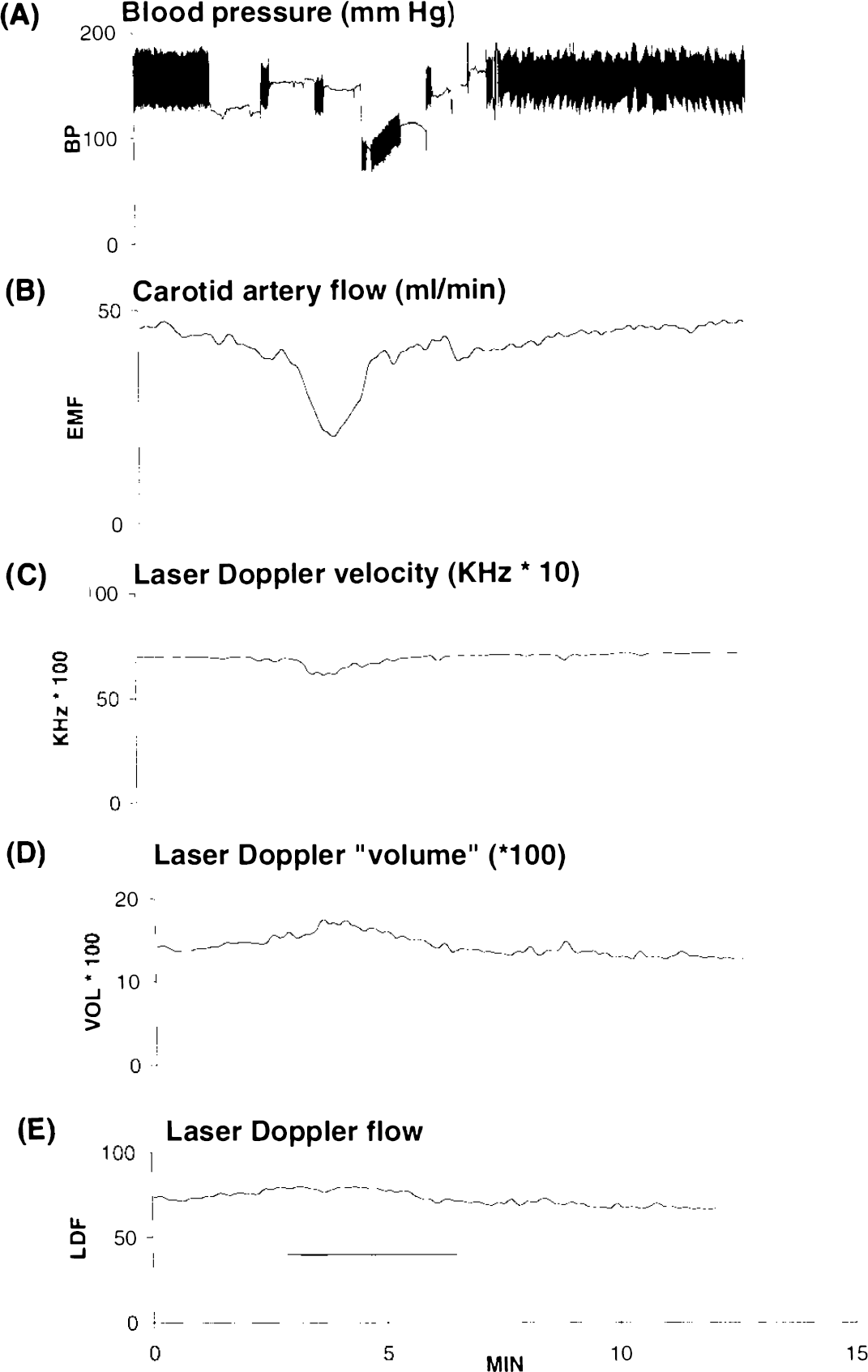
Results from one cat, showing the effect of hemorrhage on blood pressure
Reduction of blood pressure by this amount produced equivalent drops in carotid blood flow (Fig. 4). In the middle meningeal artery, laser-Doppler velocity decreased and laser-Doppler volume increased in most cats. There was either no decrease or only a small decrease in the flow signal from the middle meningeal artery during this procedure. The changes produced by blood withdrawal are summarized in Table 2. Blood pressure and carotid artery blood flows returned to normal on reinfusion of the withdrawn blood (Fig. 4).
Changes in cardiovascular parameters produced by graded blood withdrawal

n = 12.
NS, not significant.
Volume units, equal to average number of scattering events per photon and proportional to red blood cell number.
Unspecified flow units, proportional to flow in ml min−1 100 g−1, ≈ velocity × volume × 60.
The calculated gain factors were 0.04 ± 0.34 for the carotid artery (12 cats, 23 arteries) and 0.91 ± 0.29 for the middle meningeal artery (n = 12; Fig. 5). The former is not significantly different from zero, which represents absence of autoregulation (t = 0.5). The latter is significantly different from zero (t = 10.83; p < 0.001) but is not significantly different from 1.0, which represents perfect autoregulation (t = 1.1). There was no relation between the size of the decrease in blood pressure produced by bleeding and the measured gain factor in the middle meningeal artery (Fig. 6).
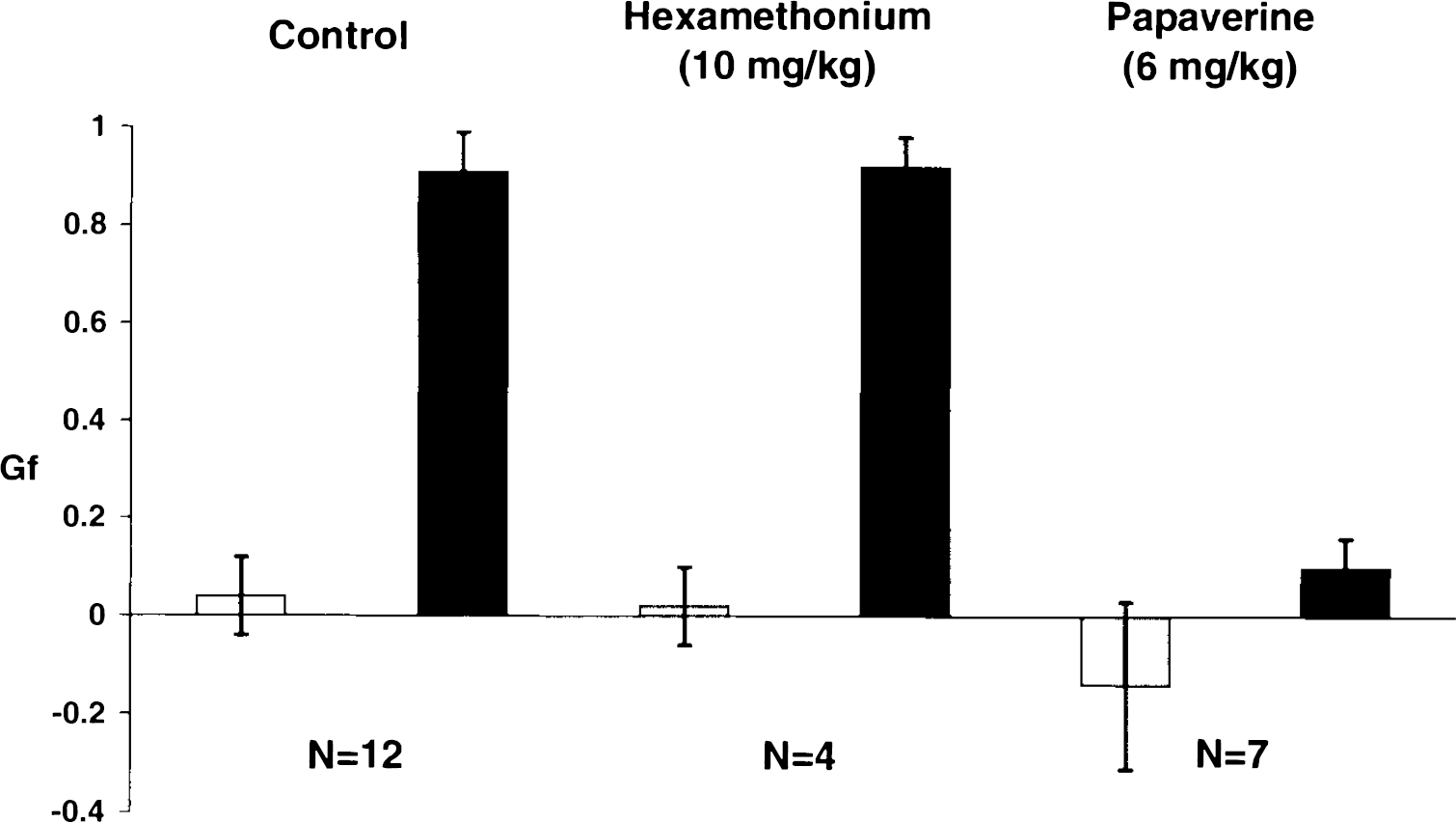
Measured gain factors (Gf, see “Methods”) for carotid □ and middle meningeal arteries (▪) in response to hemorrhagic shock under control situations and after intravenous injections of hexamethonium or papaverine. Autoregulation in the middle meningeal artery was abolished by papaverine but was not affected by hexamethonium. G, = 1 — (ΔF/F)/(ΔP/P). F, flow; P, blood pressure.
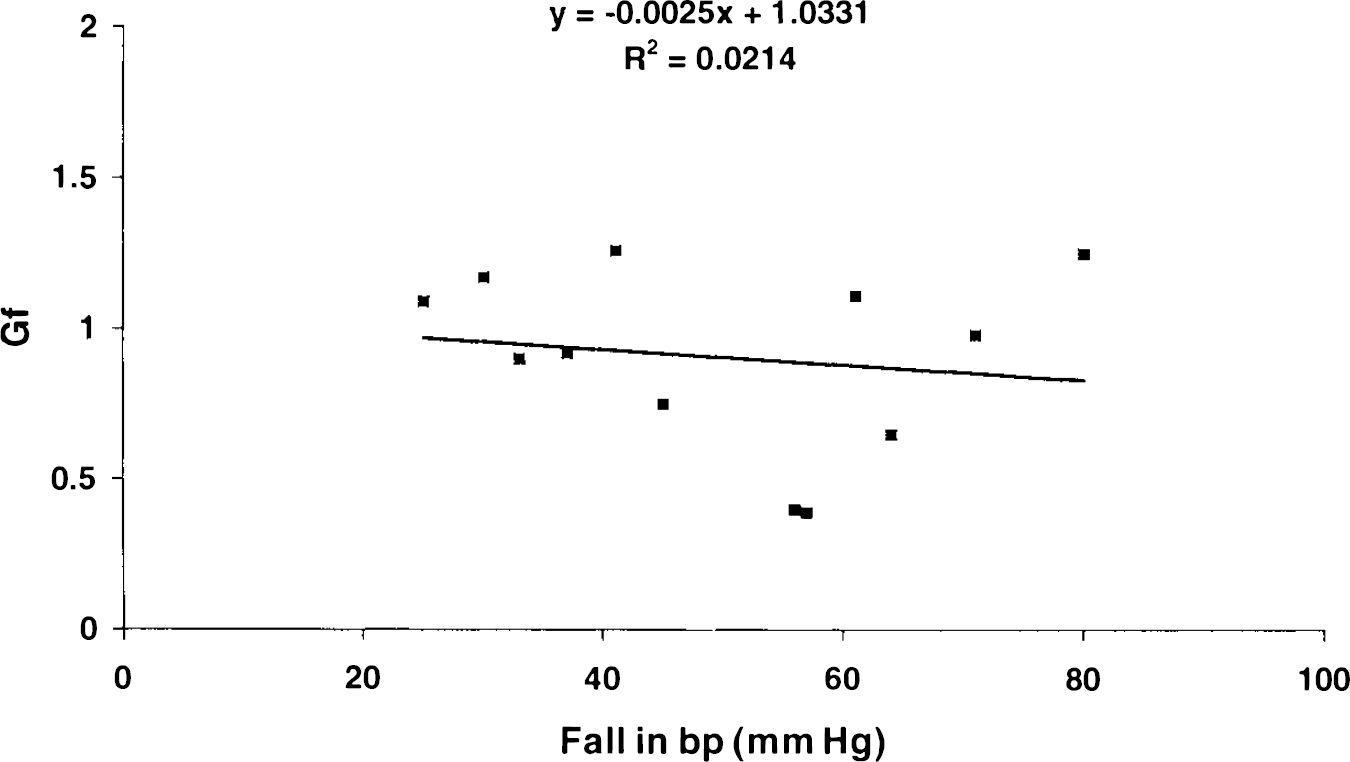
Scattergram showing the lack of relation between the size of the blood pressure decrease (Δbp) produced by bleeding and the gain factor (Gf) calculated from the consequent change in flow in the middle meningeal artery. There is no evidence that the autoregulatory range was exceeded in any of the experiments. The calculated regression equation linking the two parameters is shown. The value of R2 is not significant.
Intravenous injections of hexamethonium (10 mg/kg) decreased blood pressure from a mean of 132 to 112 mm Hg. There was no significant effect on carotid or meningeal blood flow. After hexamethonium, autoregulation in the middle meningeal artery was not blocked (Gf = 0.92 ± 0.13; n = 4). The gain factor for autoregulation in the carotid arteries was not significantly altered from control by hexamethonium pretreatment (Fig. 5).
Intravenous injections of papaverine (6 mg/kg) significantly increased flow in the carotid arteries from 42 ± 19 to 49 ± 16 (Student's paired t test, p < 0.05; n = 7) but had no significant effects on blood pressure or flow in the middle meningeal artery. Papaverine almost completely abolished the ability of the middle meningeal artery to autoregulate (Gf = 0.10 ± 0.16; n = 7). The gain factor for autoregulation in the carotid arteries was not significantly altered from that of control by papaverine pretreatment (Fig. 5).
DISCUSSION
These experiments demonstrate that a laser-Doppler system designed specifically for perfusion measurements can be modified to render it useful for measurements of blood flow in small-diameter arteries such as the middle meningeal artery. Calibration of the system with a small plastic tube showed that the volume signal was proportional to the cross-sectional area of the artery being measured. The velocity signal was directly proportional to the laminar flow velocity of RBCs in the lumen. The resultant “flow” signal gives an uncalibrated but reliable measure of flow in the artery.
For several reasons, we consider that both the velocity and volume signals (and hence the flow signal) from the probe arise primarily from the lumen of the artery and not from the underlying cortex. First, the velocity and volume signals are both significantly higher than those normally recorded from the cortex (Piper et al., 1991; Shepherd and Oberg, 1990) by factors of 2 to 5. Second, moving the probe to a position just adjacent to the artery significantly reduced both components of the signal by about the same factor. Third, selective occlusion of the artery just upstream of the probe reduced the velocity signal but not the volume signal, indicating cessation of flow but no change in “diameter.” Fourth, insertion of a black plastic sheet behind the artery, which eliminated both illumination of, and returned signal from, the underlying cortex, did not significantly attenuate the signal.
With this arrangement, the mean flow in the artery, as displayed on the instrument meter, was “125.” For most perfusion measurements, TSI (the manufacturers of the Laser-Flo) claim that this “flow” meter reading is proportional to tissue perfusion in ml 100 g−1 min−1. The manufacturers have chosen the proportionality factor (60) to make the reading as near as possible to be numerically equal to perfusion in ml 100 g−1 min−1. It is also worth noting another coincidence—that the Doppler shift in KHz for 780 nm light (that used in the TSI meter), when scattered at an average angle of 45° (see Fig. 1) is almost numerically equal to the laminar flow in millimeters per second. This coincidence of the two scales was confirmed in our calibration experiments with plastic tubing.
By using this information, we were able to calculate that the blood flow in the feline middle meningeal artery of 300 μm diameter, carrying RBCs at an average velocity of 6 mm/s, is ∼25 μl/min. Dural blood flow in dogs is reported to be 38–45 ml min−1 100 g−1 (Chen et al., 1984; Faraci et al., 1989) and, if this figure is accepted, the middle meningeal artery in our cats was supplying ∼10 cm2 of dura.
The results of the experiments that we report here demonstrate that the middle meningeal artery possesses the same ability as the cortical circulation to autoregulate its blood flow through intrinsic, nonneuronal mechanisms. In this respect, the middle meningeal artery resembles the cerebral rather than the extracerebral circulation. This result differs from those of Chen et al. (1984) and Linder (1981), who reported that dural blood flow does not display autoregulation. Differences in species or anesthetics esthetics might account for the discrepancy. Chen et al. carried out their experiments in pentobarbitone-anesthetized dogs, and Linder used rabbits anesthetized with fluanison-fentanyl, whereas we used chloralose-anesthetized cats. A more likely explanation is that Chen et al. and Linder assessed blood flow in the dural tissue en masse, using the microsphere technique. This approach may overlook changes within subsections of the vascular tree, especially if arteriovenous shunting occurs. It appears that arteriovenous anastomoses (AVAs) are a significant feature of the dural circulation and can divert significant proportions of blood flow (den Boer et al., 1992). Linder assessed autoregulation under conditions in which blood pressure was reduced to as low as 35 mm Hg, which may be below the normal limit of autoregulation.
Autoregulation of blood flow in the meningeal vascular bed appeared to involve active vasodilatation of the main trunk of the artery, shown by an increase in the volume signal at the same time as there was a decrease in RBC velocity. If autoregulation had been mediated only by mechanisms involving smaller resistance vessels or AVAs, neither volume, velocity, nor flow signals from the main meningeal trunk would have been altered. This is not to say, however, that these other components do not also participate in the dilatation. As indicated previously, the apparent conflict between our results and those of Chen et al. (1984) and Linder (1981) can best be explained by assuming that the opening of AVAs is an important mechanism in dural autoregulation.
According to Paulson et al. (Paulson et al., 1990), autoregulation is rather poorly developed in the external carotid circulation of humans. As we show in these experiments, the common carotid vascular bed of the cat does not, taken as a whole, show evidence of autoregulation. It is possible, but unlikely, that this lack of autoregulation was a consequence of the removal of the fascia from the main trunk of the common carotid arteries to facilitate electromagnetic flow–probe placement. Although the common carotid artery is innervated at this point (Mineta et al., 1992), the greater bulk of the innervation of its downstream vascular tree arises from sympathetic (Matthews and Robinson, 1980) and parasympathetic (Ruskell, 1971) nerve trunks in the head. Removal of fascia does not prevent vasoconstriction in the carotid territory after sympathetic stimulation (Bogduk et al., 1981). Thus it is unlikely that there was any functional denervation of the arterial beds that were the subject of this study.
Based on our own previous results (Lambert et al., 1984; Lambert et al., 1988), ∼35–45% of the common carotid flow in cats supplies cerebral tissue. This vascular bed definitely displays autoregulatory properties (Paulson et al., 1990), so the failure of our experiments to show any degree of auto-regulation in the common carotid can be explained only by the summation of cerebral tissue autoregulatory dilatation with vasoconstriction elsewhere, probably in the external carotid territory.
Meningeal arteries arise from a variety of major arterial trunks, with the middle meningeal being a branch of the maxillary artery in humans. Therefore it lies in the external carotid territory. It cannot be so easily classified in the cat, in which it arises before the point at which the external carotid artery ramifies to supply the rete mirable and, ultimately, the brain. Other dural and meningeal vessels arise from the ophthalmic artery (Kuru, 1967), the vertebral artery (Newton, 1968), and the internal carotid artery (Stattin, 1961). Thus they might be expected to share the autoregulatory properties that the internal carotid vascular bed possesses.
The dura mater is commonly regarded principally as a structural membrane, serving functions of protection and support for the brain, for which it exerts a “steadying influence” (Williams and Warwick, 1980). On this basis, the metabolic demands of the dura should be small and easily met without recourse to complicated vascular control mechanisms such as autoregulation.
Although the blood vessels of the dura are well described, their functional role, particularly of the arteries, has not often been addressed. According to standard texts (Williams and Warwick, 1980), the middle meningeal artery, the subject of this study, principally supplies bone, with only small branches supplying the dura itself. This view is at variance with the description of Kerber and Newton (1973), who described a very complex arterial vascular system in the dura. The intriguingly complex anatomy of the dural arteries and veins has also been described in some detail by Roland et al. (1987). The dural veins and sinuses, of course, form a major drainage mechanism for the brain (Roland et al., 1987; Williams and Warwick, 1980).
Kerber and Newton described six classes of arterial structures within the dura: (a) the larger dural arteries such as the middle meningeal; (b) “primary anastomotic arteries” (PAA), vessels of constant diameter, which course over all of the dura, uniting the circulations of the dura of the two hemispheres; (c) arteries to the skull, which supply its metabolic needs; (d) secondary anastomotic arteries (SAA), which form a mosaic network within the dura; (e) penetrating vessels, which course at right angles to the PAA and SAA and terminate in an “unexpectedly rich capillary network,” and (f) arteriovenous anastomoses, which arise from the PAAs. The PAAs apparently unite the entire dura as a single vascular unit, ensuring its blood supply is maintained at a relatively constant head of pressure. The work of Roland et al. (1987) confirmed most of these findings and described an even greater elaboration of the venous system.
This great elaboration of dural blood supply implies a more important role for the dura than merely a structural one. Kerber and Newton suggested a role for the dura in the regulation of the blood–brain barrier and in the production and regulation of cerebrospinal fluid. Roland et al. speculated that the complexity may be of significance in the vascularization of pathogenic dural structures.
Autoregulation of blood supply is a common feature of many tissues and can be mediated through metabolic (Paulson et al., 1990), neural (Gotoh et al., 1971), and myogenic (Paulson et al., 1990; Pollock and Banks, 1991) mechanisms. Our results with hexamethonium suggest that neural mechanisms, at least those depending on functional nicotinic autonomic ganglia, do not play a part in auto-regulation of blood flow in the middle meningeal artery. Although only four animals were used in the hexamethonium experiments, the chances of making a type II error are quite small, and the power of the test is high (1 — β = 0.93). The dose of hexamethonium used (10 mg/kg) is sufficient to block parasympathetic and sympathetic outflow initiated by electrical activation of the central nervous system (Borchard, 1990). We have shown previously that this dose of hexamethonium is sufficient to block neurogenic vasodilatation of the cranial circulation (Lambert et al., 1984).
The dural vessels are accompanied by perivascular sensory fibers with their cell bodies in the trigeminal ganglion (May berg et al., 1981; May berg et al., 1984). It has been postulated that local axon reflexes in these nerves may play a role in metabolically mediated regulation of cranial flow (Shiokawa et al., 1992), although no equivalent role in pressure-regulated autoregulation has been proposed. Our experiments cannot rule out the possibility that some form of antidromic trigeminal activation accounts for the autoregulatory vasodilatation, however.
In our experiments, papaverine was able to block totally autoregulation of blood flow in the middle meningeal artery. Papaverine produced relatively small decreases in resting levels of the velocity and volume signals. It is thus unlikely that the block of autoregulation produced by papaverine was due to production of total relaxation of the middle meningeal artery. Papaverine blocks autoregulation in various vascular beds, including the cerebral and renal circulations (Ogawa and Ono, 1986, 1987), possibly through interaction with calcium channels or second messengers.
If autoregulation is truly a characteristic of the dural circulation, this will have important implications for the study of disturbances of dural arterial control in migraine, subarachnoid hemorrhage, stroke, dural lesions, and other conditions in which there are disturbances of dural blood flow.
Footnotes
Acknowledgment:
We gratefully acknowledge the discussion and criticism of this work offered by Professor James W. Lance and the technical assistance of Mr. Mark Hellier and Mrs. Jane Peralta. This research was supported by grants from the National Health and Medical Research Council of Australia, the Prince Henry Hospital Centenary Research Fund and Medical Research Council of Australia, from the Duncan Flockhart Division of Glaxo Australia, by the J. A. Perini Family Trust, the Australian Brain Foundation, and the Neurological Research Fund.