Abstract
Objective:
The objective of this open-label, interventional, prospective clinical study was to evaluate the effectiveness of a multimodal wound matrix (MWM) in moving chronic, nonhealing wounds that had failed prior therapies onto a healing trajectory. The overall response rate was the proportion of subjects who had greater than 40% reduction in size after 4 weeks of treatment. Secondary objectives included the percentage area reduction (PAR) after 4 and 12 weeks, incidence of ulcer closing, and changes in quality of life.
Approach:
An open-label, interventional, prospective cohort, real-world evidence study was conducted following the STROBE criteria. Criteria included chronic nonhealing wounds of multiple etiologies in subjects with extensive comorbidities. Results were compared with data from the U.S. Wound Registry.
Results:
A total of 111 subjects entered the screening phase and 64 were treated. Fifty-three wounds were eligible for the dataset that included 18 diabetic foot ulcers, 19 venous leg ulcers, 2 pressure injuries, 1 surgical, 1 lower extremity wound, and 12 unclassified etiologies. The objective response rate was 42%. The 4-week PAR was 34%. The 12-week PAR was 66%. Eighteen wounds closed by week 12.
Innovation:
MWM is a formulation technology developed to address the major obstacles that prevent healing. Results were evaluated in a patient population with extensive comorbidities that had failed prior treatments and would be generally excluded from controlled trials.
Conclusion:
The results from this study support the contention that MWM achieves substantial clinical improvement in a complex patient population not enrolled in clinical trials and demonstrates an advancement in wound management.
Keywords
INTRODUCTION
Chronic, nonhealing wounds represent significant morbidity and mortality, especially to the elderly and those with coexisting conditions or compromised immune systems.1–4 Refractory wounds are chronic wounds that do not respond to treatment, including evidence-based modalities. A wound is considered chronic when it does not progress through an orderly and timely healing process. The health care cost of these chronic wounds can add up to 2–3% of the health care budgets in developed countries. 5
Dr. Yadwinder Dhillon, MD

We conducted a real-world study of refractory wounds treated with a multimodal wound matrix (MWM) to more accurately reflect the needs of high-risk subjects with multiple comorbidities who are typically managed in wound centers and other clinical settings. The aim was to evaluate clinical improvement in previously unresponsive wounds among a high-risk subject population that would be excluded from controlled trials, given that the clinical outcomes may more accurately translate into a greater potential impact on this population’s long-term outcomes. Our main objectives were (i) to evaluate the potential effectiveness of MWM in the treatment of chronic nonhealing wounds or ulcers across a range of etiologies in wound clinics, (ii) to evaluate the impact on the healing trajectory, specifically moving stalled wounds toward healing, and (iii) to evaluate the percentage area reduction (PAR) of wounds after 4 weeks of MWM treatment. Secondary objectives were (i) change in quality of life (QoL), (ii) enhanced activities in daily living, (iii) time to maximum closure or complete closure, and (iv) incidence of healing at week 12 as end of study (EOS). Our objective response rate was defined as the proportion of subjects who had at least a 40% reduction after 4 weeks of treatment.
MWM’s formulation was designed specifically to assist the body with the healing cascade and to address the characteristics common to all chronic wounds, regardless of etiology.6–11 These characteristics include edema, biofilm colonization, and decreased localized perfusion. The MWM comprised components derived from marine and plant sources, including omega fatty acids, medium- and long-chain fatty acids, and cold-water fish peptides, whose benefits are relevant to wound healing.8–10 Specifically designed both chemically and physically to target the major hurdles obstructing the healing process, MWM treatment aims to convert these wounds into a healing trajectory. 6 This study was conducted to evaluate the effectiveness and efficacy of MWM’s formulation on the wounds it was designed to treat.
INNOVATION
This study’s objective was to enroll and evaluate a population that represents subjects treated in the real world as opposed to those included in RCTs. Because of the extensive comorbidities of subjects with nonhealing wounds, the exclusion criteria of RCTs can eliminate the population that are treated daily in the clinic and results can be biased. Regardless of their comorbidities, age, or wound type, subjects with chronic wounds that had not responded to therapy for longer than 8 weeks were treated with MWM. Results show that MWM demonstrated clinically significant improvement in treating wounds where prior therapy has failed.
CLINICAL PROBLEM ADDRESSED
Nonhealing wounds impact subjects’ lives and result in a burdensome impact on health care costs
The Food and Drug Administration established recommendations for conducting clinical trials in wound care, and while such criteria are an important step toward consistency in wound healing research, the exclusion of many potential subjects from trials remains a concern with both direct and indirect implications when interpreting results. 7 Controlled trials may actively exclude subjects with multiple comorbidities or wounds that may otherwise be classified as complex. This can create a dichotomy between outcomes reported in clinical research and outcomes observed by clinicians who often rely on data from trials to assist them in their medical decision-making. Clinical research in wound care needs to better reflect the profile of the patient population treated in wound care centers and settings where wound management services are provided.
MATERIALS AND METHODS
Subject population
This study (NCT05921292) was an open-label, interventional prospective real-world evidence study that was conducted from April 2022 through April 2024. It was designed to evaluate the effect of MWM in a cohort of subjects with refractory wounds. A refractory wound was defined as a wound that had not responded to treatment for more than 8 weeks in a wound clinic, and after a further 2 weeks of standard care at the site during the screening phase of the study. The intent of the study design was to include subjects commonly treated daily in wound care clinics. The protocol included a broad inclusion criteria and narrow exclusion criteria with the intent to eliminate the bias that can be experienced in controlled trials. The aim was to screen approximately 111 subjects with an expectation of 100 completers.
The study was approved by the institutional review board/independent ethics committee and was conducted in accordance with the principles consistent with the Declaration of Helsinki, good clinical practice, applicable regulatory requirements, and the Belmont Principles of respect for persons, beneficence, and justice. All subjects provided written informed consent to participate in the study.
The eight clinical sites in this study were located in the United States and included hospital outpatient wound care centers, wound and research offices, and physician offices. Subjects were selected by the principal investigators (PIs) from their respective internal patient populations within their clinical sites of service. Subjects who had received treatment for at least 2 months with no significant response before the screening visit were identified as potential subjects for the study. Eligibility in the inclusion criteria included active smokers and opioid users. Subjects on dialysis or with wounds less than 2 cm2 in size and with a body mass index (BMI) >65 were excluded (Supplementary Table S1). Following the inclusion/exclusion criteria, subjects underwent the screening visit as part of a 2-week run-in period (Fig. 1).
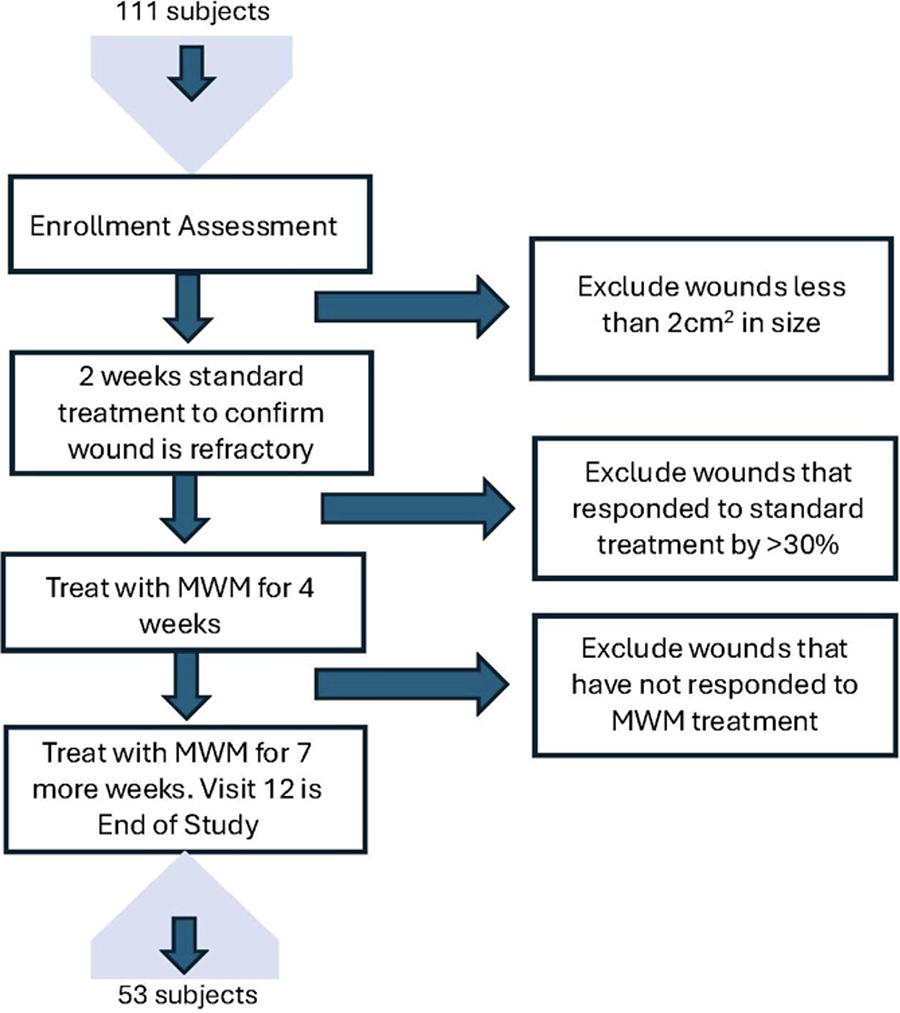
Schematic of the trial design depicting the enrollment assessment, the 2-week screening period, the treatment periods after the first 4 weeks, and at the end of study visit. Reasons for wound exclusions are shown at each stage. MWM, multimodal wound matrix.
Assessments were taken at the initial screening visit and included a medical history, physical examination, vital signs, treatment history of the chronic wound/ulcer, and target wound determination and assessment for extrudate signs and symptoms and infection. QoL and activities in daily living (ADL), pain assessment on a visual analog scale, and percentage body surface area involvement were also collected. Enhanced activities in daily living were measured in an instrumental Activities in Daily Living (ADL) scale at scheduled visits, and the Barthel Index that measures the following criteria: chair/bed transfers, ambulation, ambulation/wheelchair, stair climbing, toilet transfers, bowel control, bladder control, bathing, dressing, personal hygiene, and feeding. Reported pain perception used a 10 mm line and subjects marked on the line from no pain to worst pain possible at each scheduled visit.
The 2-week run-in period was included in the study design to further confirm that wounds were refractory at the time of study start and that they were not responding to standard care immediately before MWM treatment. By excluding subjects who showed an improvement of PAR of more 30% during the 2-week run-in period, chronicity was further confirmed immediately before MWM treatment. The 2-week period further assessed subject adherence and the likelihood of the subject completing the trial.
The screening visit included digital imaging and measuring to further confirm that wounds were refractory and nonresponsive after the 2 weeks of standard treatment. During subject visits within the screening period, wounds were cleaned, debrided as necessary, and dressed with the dressing selection left to the discretion of the investigator. Diabetic foot ulcers (DFUs) received off-loading and venous leg ulcers (VLUs) received compression therapy/wraps during the screening period, with the type of offloading or compression utilized also at the investigators’ discretion. Ulcers that responded to this treatment and reduced in size by more than 30% over the 2-week screening period were recorded as nonrefractory and did not continue in the study.
Additional inclusion criteria for VLUs required vascular assessment to confirm adequate perfusion, confirmation of venous disease, biopsy performed on ulcers older than 6 months, and compression bandaging. Additional inclusion criteria for DFUs included confirmation of type I or type II diabetes, arterial testing, known hemoglobin type A1c of ≤12% within 6 months of the study, and off-loading.
Eligible subjects were treated once weekly with MWM. Subjects who missed more than one treatment visit were determined noncompliant and excluded from the dataset. Wounds were evaluated and measured after 4 weeks of treatment, at treatment visit 5 (TV5). At that time, if there was a positive response to treatment with MWM, the PI could choose to continue for 7 more weeks of treatment with the MWM. This decision did not require a reduction in wound size but could be determined by the PI’s judgment on the wound’s response to treatment based on, for example, status of granulation tissue, increased perfusion, tissue color, or other signs of a healing response to the treatment. Visit 12 was the EOS after 11 treatments and final measurements were compared with baseline to determine the PAR. PAR, 100%, and closure of the wounds were determined as 100% re-epithelialization with no drainage or exudate present. There was no follow-up with the subject after closure.
Treatment protocol
The subjects were treated weekly with MWM (Omeza Complete MatrixTM, Omeza® LLC, Sarasota, FL) following the manufacturer’s instructions for use. The wound was cleaned and debrided as per the investigator’s discretion before the MWM was applied in strips to the wound bed. Approximately 3–5 min after the application, the MWM was spread evenly throughout the wound. A nonadherent inert primary dressing was applied to the wound following the application of MWM to manage exudate, to promote a moist wound healing environment, and to protect the wound area. DFUs were off-loaded, and multilayer compression bandages were used in the treatment of VLUs.
Safety
Assessment of adverse events (AEs) and serious adverse events (SAEs), treatment-related and nontreatment-related were monitored until resolved. Concomitant medication changes were also reviewed at each visit. No safety scale was used in this trial to assess pressure ulcers or surgical-site infections. However, as defined in 21 CFR 312.32(a), site and PIs were required to notify CRO within 24 h of making such discovery and to comply with the procedures set forth in the protocol.
Data imaging
Wound images were captured at each visit. Swift® mobile application was used diagnostically to capture wound images, wound measurements and visit date at every visit to eliminate bias from manual measurements. Swift® follows industry best practices and complies with all applicable industry standards such as the Health Insurance Portability and Accountability Act and U.S. Food and Drug Administration standards. Data and images were stored securely in the Amazon Web Services cloud with daily remote data support backups. Photographs were examined to ensure elimination of subject identification. Swift® measurements of the wounds were compared at treatment visit 1, treatment visit 5 after 4 weeks of treatment, and at week 12, which was EOS visit after 11 weeks of treatment. If wound closed before week 12, that visit became EOS.
Electronic laboratory notebook platform was not used.
Statistics
An analysis was conducted to determine whether wound healing over time (baseline to week 12) was statistically significant, regardless of wound type. The methodology used a longitudinal statistical method to account for repeated measures over time. The null hypothesis stated wound size is reduced over time. The alternate hypothesis stated wound size remains the same over time. Seven different models were run for each wound type (DFU, VLU, other). The models assessed the healing trajectory and significance of changes over time, without direct comparisons between wound types. The data were analyzed using line plots for observed values, change from baseline, and percentage change from baseline. A paired t-test was performed to evaluate the reduction in wound size between Visit 1 and Visit 12.
RESULTS
One hundred eleven subjects signed informed consent forms from eight wound care sites across the United States over a 20-month period from April 2022 through January 2024. Following the 2-week screening period, 13 (12%) subjects were discontinued in the trial since their wounds decreased in size greater than 30%, and 26 (23%) screened failed for wounds being too small. Eight subjects (7%) were noncompliant during the screening period. Sixty-four wounds were treated with MWM. Evaluation at study end found 11 wounds ineligible for final data analysis. Nine subjects had inaccurate wound measurements recorded during their treatment, one subject had missing measurements, and another missed more than two treatment visits.
The study population (n = 53) included 34 males and 19 females (Fig. 2A, B). Subject ages ranged from 35 to 100 years with an average of 64.3 years (median: 63 years) (Table 1). All subjects older than 65 presented with two or more comorbidities (range: 2–26 per subject). A total of 17/25 subjects presented with five or more comorbidities (Supplementary Table S2). In subjects younger than 65, the comorbidity count ranged from 2 to 23 with 12/28 having five or more comorbidities. BMI ranged from 15.94 to 52.94 with an average of 33.5 kg/m2 (median: 31.8 kg/m2) (Supplementary Table S3).

Swimmer plots of study showing subjects’ gender, ethnicity and race, and response to MWM treatment over time. ( Male Subjects Female Subjects
Male Subjects Female Subjects  Hispanic
Hispanic  Latino
Latino  African American
African American  American Indian/Alaskan Native
American Indian/Alaskan Native  100% Percent Area Reduction (PAR).
100% Percent Area Reduction (PAR).
Subjects’ age distribution and ulcer sizes by subgroups
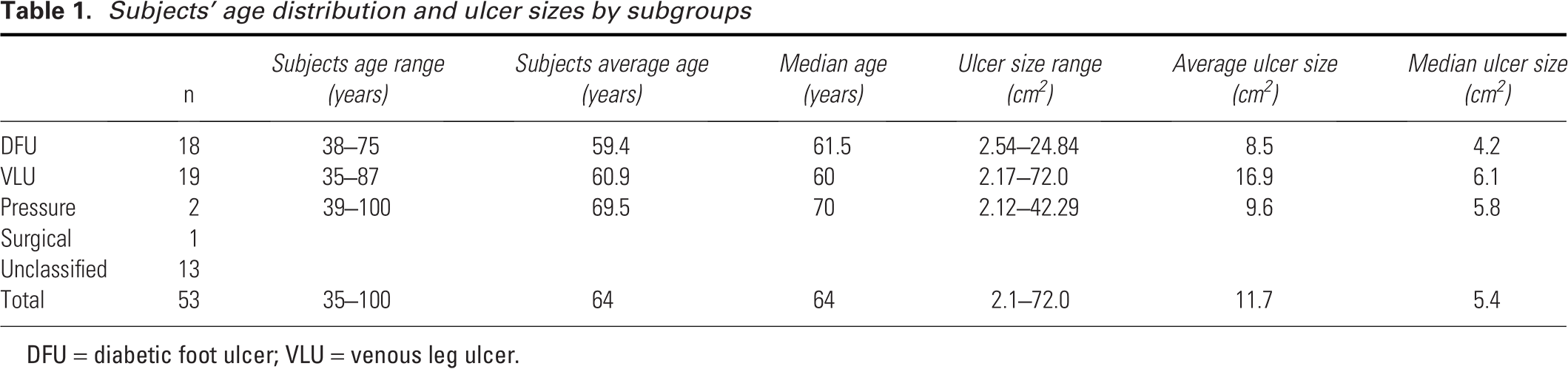
DFU = diabetic foot ulcer; VLU = venous leg ulcer.
The wounds treated included 18 DFUs, 19 VLUs, 2 pressure injuries, 1 surgical wound, 1 lower extremity ulcer, and 12 unclassified ulcers. Wound sizes ranged from 2.1 cm2 to 72.0 cm2 with an average of 11.8 cm2 (median: 5.9 cm2). The age of the wounds ranged from 10 to 304 weeks with an average of 38 weeks (median: 22 weeks) (Table 1). Six wounds were older than a year, with three wounds older than 3 years.
The overall 4-week PAR was 34% and the overall 12-week PAR was 66%. The objective response rate was 42% with 22 of the 53 wounds decreasing in wound size by more than 40% after 4 weeks of treatment. Four wounds closed by 100% after 4 weeks of MWM treatment (Fig. 3A). Thirty-four percent (18/53) of the wounds saw 100% re-epithelialization by week 12 (Fig. 3A), which included six DFUs, eight VLUs, and four other. Thirty-two percent (16/53) of the wounds did not respond at all to treatment, eight of which were labeled “unclassified.” Fifty-five percent (29/53) of the wounds decreased by more than 40% by week 12.

The PAR of subjects’ study ulcers at 4 and 12 weeks. ( DFU 4 week PAR
DFU 4 week PAR  VLU 4 week PAR
VLU 4 week PAR  Other wounds 4 week PAR
Other wounds 4 week PAR  DFU 12 week PAR
DFU 12 week PAR  VLU 12 week PAR
VLU 12 week PAR  Other wounds 12 week PAR. (
Other wounds 12 week PAR. ( PAR at 4 weeks
PAR at 4 weeks  PAR at 12 weeks. (
PAR at 12 weeks. (
Statistical analysis focused on healing progression from baseline (visit 1 to week 12) regardless of wound type. A longitudinal statistical method was used to account for repeated measures over time using seven different models to assess the healing trajectory and significance of change over time, without direct comparison between wound types. Results were consistent across the three models for each wound type, which strengthened the robustness of the findings. Using line plots, the data of all wound types were analyzed for observed changes, change from baseline, and percentage change from baseline. Using a paired t-test to assess the reduction in the wound size across all wound types from visit 1 to visit 5, and from visit 1 to visit 12, the reduction in wound size is statistically significant. The p value for reduction in wound size from visit 1 to visit 5 in DFUs is 0.0385, and in VLUs is 0.0238. The p value for reduction in wound size from visit 1 to visit 12 is 0.0065 in DFUs, and 0.0106 in VLUs.
Three subjects were treated with antibiotics during the study. One subject with an undiagnosed lower extremity ulcer received 10 treatments of MWM and saw a 70% reduction in ulcer size. The subject’s comorbidities included osteoarthritis, osteopenia, hypothyroidism, and rheumatoid arthritis. At TV11 the PI at the site prescribed doxycycline for 7 days as a second ulcer had developed. Both ulcers looked infected, and the target ulcer size had increased to its original size. At TV12 there was no further change in ulcer size from TV11. A second subject with an undiagnosed ulcer at the right foot was prescribed a single IV course of piperacillin and tazobactam for osteomyelitis for a foot fracture on the opposite leg after treatment visit 9. The subject suffered from pyoderma gangrenosum. The third subject was prescribed 7 days of doxycycline for an infection not related to the ulcer at treatment visit 8. There was no change in the subject’s ulcer size throughout treatment, but the subject missed two treatment visits. No other subjects received antibiotics during the trial.
Subpopulations
Statistically, the PAR from baseline to week 12 was found to be consistent across the three models for each wound type, but due to the physiological differences between wound types, the complexity of the comorbidities of the subject population, and the protocol requirements for off-loading for DFUs and compression wraps for VLUs, the wounds were grouped by wound types for further evaluation.
Diabetic foot ulcers
Eighteen subjects (17 male, 1 female) with an average age of 59.4 (median: 61.5) and ranging from 38 to 75 years were treated with the MWM (Table 1). The ulcer sizes ranged from 2.59 to 24.84cm2 with an average size of 8.47cm2 (median: 4.21cm2). The age of the ulcers ranged from 10 to 34 weeks, with the average age at 20 weeks (median: 18). The 4-week average PAR was 39% (Fig. 3B). Thirteen of the ulcers were treated up to 12 weeks and had an average PAR of 63%, with six wounds closing completely (Fig. 3E).
Venous leg ulcers
Ninteen subjects (10 male, 9 female) with an average age of 60.9 (median: 70) and ranging from 35 to 87 years were treated with MWM (Table 1). The ulcer sizes ranged from 2.17 to 72.02cm2 with an average of 16.86cm2 (median: 6.08cm2). The age of the ulcers ranged from 13 to 32 weeks, with the average age at 19 weeks (median: 19). The 4-week PAR was 38.2% (Fig. 3C). Fifteen of the ulcers were treated up to 12 weeks and had an average PAR of 69.4% with eight VLUs closing completely (Fig. 3F).
Wounds of other etiologies
Sixteen subjects (seven male, nine female) were treated with MWM with an average age of 69.5 (median: 70) in a range of 39–100 years (Table 1). The ulcer sizes ranged from 2.12 to 42.29cm2 with an average of 9.55cm2 (median: 5.75cm2). The age of the ulcers ranged from 16 to 304 weeks, with the average age at 105 weeks (median: 48). The 4-week PAR was 38% (Fig. 3D). Fifteen of the ulcers were treated up to 12 weeks and had an average PAR of 69% with four wounds closing completely (Fig. 3G).
Safety results
Twenty-four subjects experienced a total of 43 AEs and 7 SAEs. The SAEs were unrelated to the study drug. There were eight AEs considered probable, three possible, and one definitively linked to the investigational product. Thirty-one AEs were not linked to the product.
The seven nonrelated SAEs included a death from pneumonia, myositis, hyperglycemia, and hospitalization for infection (2), fatigue, and heart failure. There were three AEs considered “possible” that included infection with Pseudomonas aeruginosa (2), and pain (1); eight considered “probable” that included erythema (2), exudate (2), pain (2), yellow discharge, and inflammation. The one AE recorded definitively linked to the investigational product was a subject who reported pain when MWM was applied to the ulcer.
Subject-reported outcomes
QoL, ADLs, and pain assessment were collected for this trial. The data were inconclusive, and results did not consistently correlate with wound outcome or healing status.
DISCUSSION
The healing rates presented in this study are improved over reported data. Fife, et al, reported that randomized controlled trial (RCTs) of uncomplicated, small ulcers of different wound types among relatively healthy subjects consistently reported mean healing rates around 40%, although these rates varied widely across trials (range: 7.7–90.6%). 12 , 13 Based on real-world data and RCTs, healing rates over 90% as publicly reported can be achieved only by creating extreme censoring rules, which are not likely to fall within acceptable standards of data management.8–13
In this study of nonhealing refractory wounds, we enrolled subjects at specialty wound clinics who had demonstrated no clinical improvement despite at least 8 weeks of standard treatment at the site and an additional 2 weeks of high-quality wound care after entering the screening phase. Subjects had failed advanced therapies and modalities before this trial, including gentian violet/methylene blue foam, alginates, collagens, silver, Manuka honey, cellular tissue products, and negative pressure wound therapy. Treatment with the MWM resulted in moving 70% of these refractory wounds toward healing.
We chose to perform a prospective real-world assessment of the MWM technology to study how this multimodal product is effective in addressing the clinical needs of high-risk subjects who are managed in wound centers and other settings where wounds are treated. MWM demonstrated clinical improvement among subjects despite a variety of issues and comorbidities that are known to pose obstacles to healing. Evaluating a positive response marked by either progress toward or complete healing in previously unresponsive wounds among a high-risk population was deemed to have greater translational value and proof of concept of MWM’s intended technology.
The aim of this study was to enroll 100 subjects. We screened 111 but only treated 64 because of the inherent challenges with this subject population. The study included subjects with complex medical conditions, including active smokers, poorly or uncontrolled diabetics, opioid users, BMIs above 30 kg/m2, and advanced age (Supplementary Table S3). Thirty-one of the comorbidities may have direct, adverse effects on wound healing, such as diabetes, hypertension, hyperlipidemia, coronary heart failure, and venous insufficiency. The correlation of multiple comorbidities on life expectancy has been well documented. 14 Increased age, BMI, opioid use, and smoking status, for example, are known negative predictors of wound healing.15,16 Other measures such as hemoglobin type A1c for metabolic control, ankle brachial index for perfusion, or skin temperature for inflammation are correlated with poor wound healing.14,16–19 However, Cho, Mattke, et al, examined the impact of comorbidities and wound characteristics as factors pertaining to healing. Their conclusion was consistent with prior research which revealed that wound-level characteristics are better predictors of wound healing than subject-level characteristics, such as demographic information and comorbidities.13,16,18–20
The current study is limited by the variability of prior wound treatment and the presence of a comparison group. However, our cohort could be compared with the prior clinical condition since the subjects had wounds that were refractory to treatment from wound care specialists and had an additional 2 weeks of high-quality care. Only 12% of the enrolled cohort of subjects did not progress past the screening phase because of a >30% improvement in wound size by following the study protocol. This supports the content that those remaining wounds were refractory to conventional treatment.
This study had a high number of refractory wounds that were unclassified. Of these, 8 of the 12 did not respond to treatment. Some wound etiologies may mimic others, for example, a malignancy can be misdiagnosed as a venous ulcer, therefore leading to inappropriate treatment. Not having an accurate diagnosis may negatively impact the outcome of any wound treatment, whether in a controlled study, or in an everyday clinical setting.
In this trial, regardless of wound outcome, 14 subjects did not complete the treatment phase. They were withdrawn for lack of improvement or withdrew for various reasons, such as subject relocation, transportation issues, or simply no-show. Dropouts can be a problem in wound care clinical trials. A subject with more complex wounds and extensive comorbidities can find it more challenging to complete a 12-week trial with a 2-week screening period, compared with a potential subject whose health is less tenuous and wound less complex. Basic concerns such as subject reliability in presenting for weekly assessment represent common obstacles in the recruitment of subjects who are more accurately representative of the wound population at large.
We reviewed ulcer response based on wound size to see if there was a difference between the rates of smaller and larger wounds closing. When we grouped all the ulcers treated in the study as less than 6 cm2 in size (n = 31) versus larger than 6 cm2 (n = 23), the average 4-week PAR was 84% for the smaller ulcers compared with 43% average 4-week PAR for the larger ulcers. When we compare the rate of healing (cm2/week) between the groups, the larger ulcers healed at 0.97 cm2/week, which is 50% faster than the smaller ulcers that close at a rate of 0.48 cm2/week. When we compared the ulcers that closed more than 40% over 12 weeks, the rate of area reduction at 4 weeks is four times faster for the larger wounds. Further clinical studies are needed to evaluate these results and to investigate the mechanistic effects of MWM at the cellular level.
In addition, anticipating an overall improvement in subject-reported measures that would parallel wound improvement was a hopeful finding. The results correlating wound improvement as a direct impact on activities of daily living, QoL, and pain appeared to be inconclusive. However, in as much as these measures may be quantifiable, they are nonetheless, subjective. The presence of multiple comorbidities could also diminish the correlation that subjects’ wounds had on each of these measures, so wound improvement directly impacting these measures is potentially complicated by the effects of other existing conditions. The U.S. Food and Drug Administration has recently qualified a patient-reported QoL chronic wound scale/assessment intended for wounds existing longer than 3 months. 21 The intention is to incorporate this scale in future real-world evidence trials with the MWM.
Despite the experience of our investigators, there was wide variability in the products they each had used before this trial to manage wounds of the same etiology. Some investigators had used products predominantly that contain silver, while others had preferred topical antibiotics and other antimicrobials, while still others had used amniotic grafts, collagens, and alginates. (MWM was the only treatment used during this trial.) This information demonstrates the fact that despite best practices, no two providers will necessarily approach the management of a wound by using the same products as their colleagues. While treatment algorithms and guidelines were previously followed by the investigators, no true standard of care was observed in terms of products applied to wounds. The wound care specialists who participated in this study chose the subjects enrolled in the study because previous or current therapy was not effective. The additional 2-week run-in period before initiation of MWM was additional confirmation of the refractory status of the wounds studied. Once MWM was introduced through the study protocol, improvement in wounds that were previously unresponsive was evident, regardless of prior products used, wound etiology, or investigator. Besides the clinical data regarding PAR as well as weeks to closure, such improvement supports the claim that MWM demonstrated significant clinical improvement, especially in instances where prior products used had not been effective.
KEY FINDINGS
MWM moved 37/53 nonhealing wounds that had failed prior therapy toward healing.
MWM had responses in all types and age duration of nonhealing wounds.
By week 12, MWM had reduced the wounds in size by an average 66%.
By week 12, MWM closed 18/53 wounds (100% reepithelialization with no drainage).
Footnotes
ACKNOWLEDGMENTS
The authors thank Vickie R. Driver, DPM, MS, FACFAS, for her guidance on and contribution to the study design; Shawanda Daniel, DrPH, and Julie Primo, RN, for their contributions in support of the clinical trial, and Lisa Kang, MSPH, for biostatistical support.
FUNDING STATEMENT
The Clinical Trial was sponsored by Omeza® LLC, Sarasota, FL, USA.
AUTHOR DISCLOSURE AND GHOSTWRITING
D.P.B. and S.J.B. are employees of the sponsor, Omeza LLC. No other authors have conflicts of or competing interest to disclose with the sponsor.
PREPRINT
An earlier version of this article was posted as a preprint at medRxiv
ABOUT THE AUTHORS
The following authors were principal investigators at the trial sites:
The following authors were sub-principal investigators or wound care specialist health care providers providing treatment for the trial:
All principal investigators equally contributed, reviewed, and edited the article.
Supplemental Material
Supplemental Material
Supplemental Material
Abbreviations and Acronyms
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
