Abstract
Objective:
To identify how cellular and/or tissue-based products (CTPs) relate to value in terms of cost per quality-adjusted life years (QALYs) in wound care in comparison with treatments in other medical fields.
Approach:
This is a cross-sectional study and a cost-effectiveness analysis. Payment limits for each CTP were obtained via the Healthcare Common Procedure Coding System Q codes and formulated as cost inputs into a cost-utility model published for treatment of Wagner 1 diabetic foot ulcers using dehydrated human amnion and chorion allograft versus standard of care (SOC). Additional changes to cap the number of CTP applications and adjustments for recent inflation were made. The literature was searched for other cost-utility models in other diabetes-related diseases as a comparison.
Results:
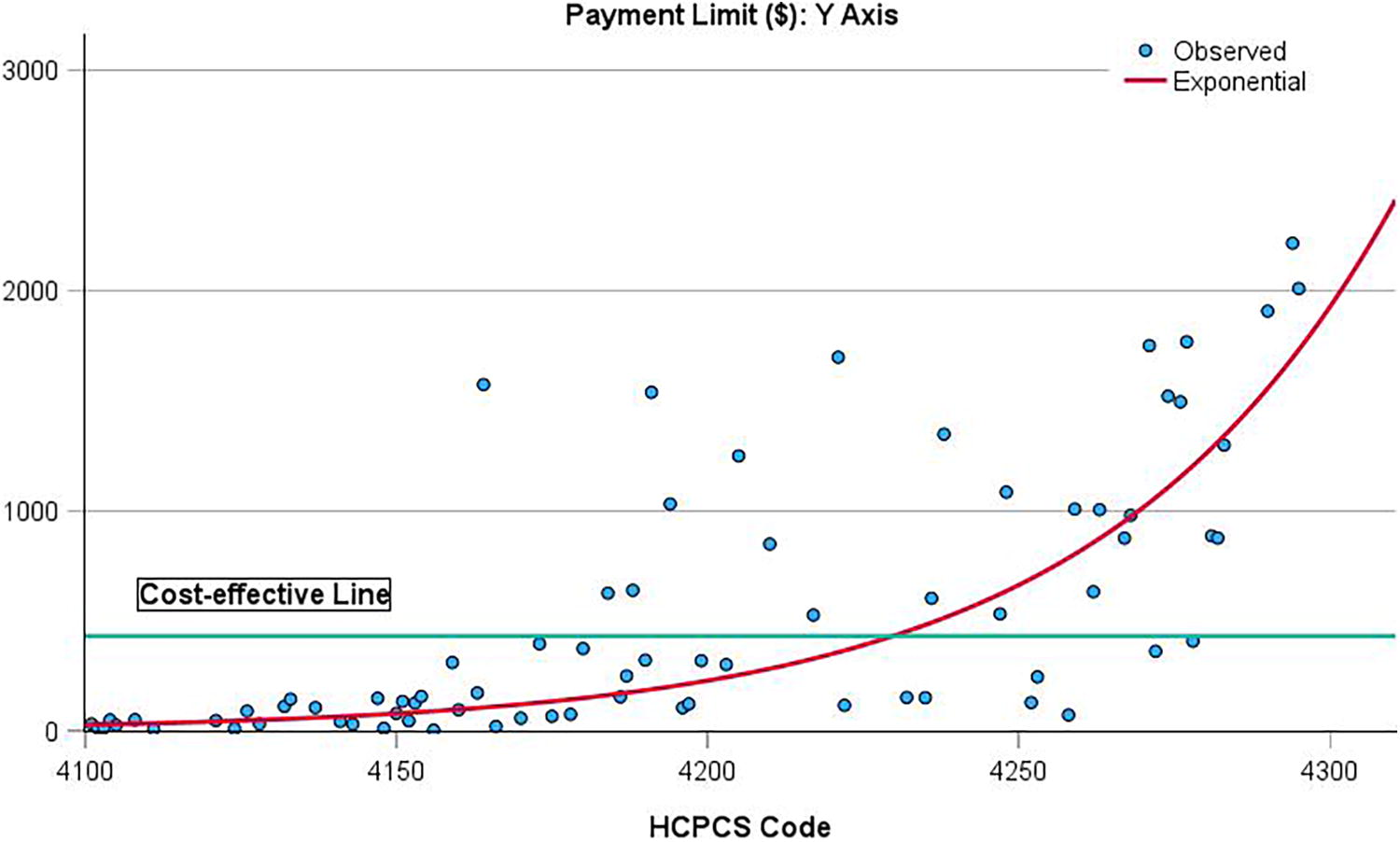
When the payment limit was ≤$140 per square centimeter, interventions were dominant (less costly, better outcomes) compared with SOC. When the limit exceeded $430 per square centimeter, the cost-effectiveness threshold of $100,000/QALY was exceeded. Newer Q codes are generally much more expensive and likely to not be cost-effective, similar to the results for many other chronic diabetes-related diseases .
Innovation:
This study presents decision makers with tools, by which they can determine as to whether a given CTP is likely to be cost-effective for patients.
Conclusion:
Over a third of all CTPs will very likely result in noncost-effective interventions. This number is likely to be higher when wounds are larger or used in other wound types where they are less efficacious. The recent trend in much higher costs for CTPs is worrisome.
INTRODUCTION
Chronic, nonhealing wounds affect more than 16% of Medicare beneficiaries and conservatively cost Medicare almost $25 billion per year, although the real cost may be three times that. 1 The increasing prevalence rate of nonhealing wounds, even among patients younger than 65, their refractory nature, and their associated morbidity, drives medical innovation. A major area of innovation is that of “bioengineered products.” Since the introduction of a bilayered living cellular construct more than a quarter century ago, at least 183 products have become commercially available to facilitate the healing of various types of chronic wounds. Originally referred to somewhat inaccurately as “skin substitutes,” since they are not actually a substitute for skin, though the Dermal Regeneration Template does regenerate the dermis and forms a neo-dermis, this increasingly diverse group of materials has been variously referred to as “regenerative tissues,” cellular and/or tissue-based products (CTPs), which is the name officially recognized by the American Society for Testing and Materials, and more recently cellular, acellular, and matrix-like products (CAMPs). 2 A suitable acronym has been elusive because the products are diverse combinations of living and nonliving, autologous and nonautologous cells or tissues, human placental components, xenograft tissues or tissue components, and synthetic materials.
A detailed review of these products, including their various pathways to clearance or approval from the Food and Drug Administration (FDA), is available elsewhere 3 and is beyond the scope of this brief report. The availability of data from prospective randomized controlled trials (RCTs) is highly variable and for some products, nonexistent. When prospective RCTs are performed, the trials target relatively superficial diabetic foot ulcers (DFUs) or venous leg ulcers (VLUs) in only the healthiest of subjects. Thus, the results of RCTs are nongeneralizable to the typical patient. 4 Little is known about the efficacy and the real-world effectiveness of these products in the typical patient who has multiple serious ulcers and many comorbid diseases, a knowledge gap that may only be addressed by the expanded use of real-world evidence.
These once novel materials were initially available only in the provider-based outpatient department (PBD) of hospitals, often referred to as hospital-based outpatient departments (HOPDs). However, since 2015, they have been under a reimbursement method in the PBD called “package pricing,” by which the cost of the product is included in the overhead costs of the hospital service for its application. There are two payment rates, one for the “low-cost tier” and one for the “high-cost tier” products. In 2024, the payment rate for high-cost products for most applications is $1,739.33. 5 The package price represents the limit that a hospital can afford to pay for a given piece, regardless of its price per square centimeter. Many PBDs report significant financial losses when using these products since individual pieces are often priced above the payment threshold. The package price threshold generally limits PBDs to smaller pieces of any given CTP, limiting their use to smaller wounds in the PBD setting. Thus, package pricing has driven the use of CTPs into the office-based setting where the product can be reimbursed separately, usually based on the average selling price (ASP) plus 6%. However, a variety of methods exist enabling practitioners to retain a significant percentage of the product cost (e.g., discounts far below the ASP, or bonus payments for “early payment” or “research”). With such arrangements, potential practitioner revenue for the use of CTPs increases with the increasing cost of the product, costs that are subsequently passed on to Medicare, less the 20% patient responsible portion, which may be paid by the patient or by a secondary insurance.
The value proposition of this growing suite of products is a critical question since their cost continues to increase at a nearly exponential rate. Currently, pricing per square centimeter, based on either the ASP or the wholesale acquisition price (WAC), varies from $30/cm2 to nearly $3,000/cm2 for a recent product. The reasons for these cost escalations cannot be related to the cost of manufacturing when nearly identical products can vary dramatically in ASP. This brief report aims to identify how the CTP cost relates to value in terms of quality-adjusted life years (QALYs) using a model of DFUs and to supply some context in understanding the relationship not only in this area of wound care treatment but also in other areas of diabetes-related diseases.
CLINICAL PROBLEM ADDRESSED
Health care providers and payers need to know whether CTPs are a good value proposition for their patients in the office setting. This study enables individuals whether this is likely to be so from the relevant Q code and/or ASP for nonsevere DFUs.
MATERIALS AND METHODS
The payment limits for CTPs (Q codes) were obtained by first downloading the April 2024 Centers for Medicare and Medicaid Services (CMS) ASP list, 6 and focusing on the CTP Q code range starting with Q4100. All products that are not priced by square cm (e.g., priced by weight or volume; meaning they are powders or injectables) were removed and the remaining list sorted by numerical order. The First Coast part B fee database 7 was then queried for codes missing from the list and/or having higher number codes than the ASP list currently contains. Each potential Q code was queried to determine if a price was listed, and if so was included. The process was continued with ascending numbers until no more Q codes could be found. Recent CMS Q code decisions were also checked. Table 1 shows the final list.
HCPCS code (Q code) and associated payment limit ($)
Bolded payment values come from First Coast.
HCPCS, Healthcare Common Procedure Coding System.
A microsimulation HE model developed for Wagner 1 DFUs in which standard of care (SOC) versus dehydrated human amnion and chorion allograft (dHACA) (MTF Biologics, Edison, NJ) plus SOC 8 was utilized under the following conditions: (1) all costs except CTPs were increased by 6% 9 to account for inflation from 2019 to early 2024; (2) the number of CTP applications was capped at 6 as a reasonable compromise based on reimbursement; (3) it was assumed that different sizes available for CTPs are still the same cost per square cm; and (4) it is assumed that hypothetical CTPs would have approximately the same performance (healing rate) as dHACA (dehydrated human amnion and chorion allograft). Models were run using various ASPs from $30 to $3,000 per cm2. In addition, for the $30 per cm2 microsimulation, the influence of not having a full range of sizes was explored by assuming that only a size of 2 × 4 cm was available.
The Q2 2024 payment limits were also plotted (Y axis) versus sequential code numbers (oldest codes generally have the lowest numbers 7 ) on the X axis, along with a “cost-effective” line, meaning that cost/QALY for this payment limit value is estimated at $100,000/QALY. 10 –12
RESULTS
CTPs were the dominant treatment option obtained when the payment limit was approximately ≤$140 per cm2, meaning less costly and with better outcomes (more QALYs) compared with SOC (Table 2). The cost-effectiveness threshold of $100,000/QALY was exceeded when the payment limit was ≥$430 per cm2 (Table 2; Fig. 1). Although the X axis in Figure 1 does not represent linear time, the newest Q codes were generally much more expensive and exceed the $100,000/QALY threshold. The fit line was best represented by an exponential function (R 2 ∼0.5).
Innovation diagram algorithm.
Cost utility obtained for cellular and/or tissue-based product versus standard of care at different values of payment limits per square centimeter
CTP strategy (versus standard of care [SOC]) is dominant, with cost savings and better outcomes (higher quality-adjusted life years [QALYs]).
CTP, cellular and/or tissue-based product.
DISCUSSION
The overall goal in this study was not to demonstrate a specific benchmark for payers or decision makers at which prices for CTPs mean that they are no longer cost-effective per se (although we do calculate it), but to highlight the relationship between cost and value, and demonstrate the alarming trend in the upward pricing of CTPs.
Publicly available data show that the ASP and/or WAC for newly marketed CTPs are generally higher than products that have been on the market for several years. More than one-third of CTPs with a Q code now exceed the cost-effectiveness threshold of $100,000/QALY in our model and although there is debate as to what the upper limit should be, it is likely within the $100,000 to $150,000/QALY range. 10 –12 The increasing price of CTPs suggests that the price pressures that help control the drug/biological market do not work in the CTP market. CTP manufacturers generally do not use wholesalers or otherwise make commercial use of a published WAC such as drug/biological companies. Also, Medicare is the largest customer for CTPs in the United States, so unlike drugs, the pricing pressures from other payers are limited or nonexistent. Another observation is that a vast majority of the high-cost CTPs are amnion-based products. No clinical trials to date have demonstrated consistent superiority of one product over another in this subcategory, which begs the question why there should be such large price differentiation. The model that we used is based on an RCT 13 that yielded one of the largest differences observed between intervention (CTP) and SOC groups in nonsevere DFUs: 85% versus 33% over a period of 12 weeks. One could argue that if some of the more highly priced, newer CTP entrants added more value (i.e., more effectiveness and/or even more accelerated wound-healing trajectories) it might offset the increased cost. However, this is not the case. After exhaustive searching in the literature for these products, few were found to have any clinical trial data let alone an RCT that had superior characteristics to the dHACA trial. Thus, this value proposition would seem specious, which is why we did not attempt to model scenarios in which healing was faster. The flip side to this point is that if healing trajectories are in fact worse than the model we used, the cost utility values will be worse.
We tried to compare our results in the context of other areas of medicine, for example, diabetes-related complications as suggested by one reviewer, but the published results are limited so could mean a biased attempt to understand whether the situation we have is unique to wound care. For example, we found 11 cost-utility studies 14 –25 after thorough literature searching that mostly involved comparisons of costly head-to-head anti-VEGF intravitreal drug injections or comparisons with other, older treatments for diabetic retinopathy or macular edema. Out of 41 comparisons, only 15 (37%) would be considered cost-effective using the $100,000 per QALY benchmark. If anything, this limited analysis suggests that our wound care situation might not be unique in medicine.
A potential problem for the health economics of CTPs when framed as cost-utility is that the maximum theoretical QALY gain is also relatively small: as an example, Redekop et al. found a change of 0.09 for healing a noninfected DFU (0.75 to 0.84). 26 Based on this change (though including results from other complications) in the original model, the actual QALY gain over 1 year per patient (one wound per patient) was estimated at 0.033, which is even smaller. In the context of other areas of medicine, Wisløff et al.’s 27 analysis of all cost-utility studies published in 2010 (n = 370) showed that the median incremental QALY gain was 0.06, which suggests that healing nonsevere DFUs with CTPs may be at the low end of effectiveness in the scheme of medical interventions. In simplistic terms, trying to demonstrate that a CTP offers value in nonsevere chronic wounds is a fool’s errand when its cost is exorbitant.
In terms of limitations, the exact results are only strictly valid for the model used, which is for Wagner 1 DFUs. For other nonsevere wound types, such as VLUs or stage 2 pressure injuries, the results are not likely to be substantially different if addition of a CTP to SOC increases healing rates by a good margin (e.g., at least 25% at 12–16 weeks) although the ASP at which the CTP is unlikely to be cost-effective will be lower than the benchmark found in this study. In addition, the mean area of wounds at randomization for the clinical trial on which the model was based is small: dHACA: 2.1 cm2; SOC: 3.1 cm2. Much larger wounds (e.g., >10 cm2), on average, take longer to heal than smaller wounds and CTP costs would be higher because larger sizes would be needed. Consequently, the cost-effectiveness will be considerably lower (see also Table 2, second entry). A similar situation pertains to CTPs only available in large sizes when they are used to treat much smaller wounds. For more severe wounds, the results are uncertain because the clinical effectiveness of most CTPs for such wounds is yet to be established.
Key Findings
For Wagner 1 DFUs and CTPs with ASPs >$430 per square centimeter, the intervention is unlikely to be cost-effective.
ASPs for new CTPs have increased very substantially in the past few years.
For other nonsevere wound types or larger wounds, the impact on cost-effectiveness compared with Wagner 1 DFUs will mean products are less cost-effective.
In conclusion, based on publicly available data, the cost of CTPs per square centimeter has continued to increase over time. Although the price per square centimeter of these products can vary by nearly 100-fold, there are no data to demonstrate that CTPs at high end of cost produce better outcomes than those at the low end of cost. Even assuming the outcome of a healed DFU, there is a limit to the justifiable cost of a CTP from the standpoint of the Medicare trust fund, to say nothing of the financial contribution required by the patient. Among superficial Wagner Grade 1 DFUs, a reasonable quality-of-life economic model suggests that products <$140/cm2 yield a better outcome than SOC when defined by QALYs. As costs increase, however, cost-effectiveness approaches then exceed established cost-effectiveness thresholds. Absent a biblical impact on underlying disease, the use of very high-cost CTPs cannot be justified.
INNOVATION
CTPs are becoming increasingly more expensive in the office setting. However, it impossible to know whether more expensive CTPs are cost-effective, meaning that for the price they deliver positive benefits. By knowing the Healthcare Common Procedure Coding System Q code, we show the associated ASP per square centimeter and whether use of a particular CTP is likely to be cost-effective for shallow DFUs. For other wound types, the results will be similar if certain criteria are met. We also present data that indicate pricing trends are increasing exponentially. The study algorithm is shown in Figure 2.
Plot of payment limit ($/cm2) (X axis) versus CTP Q code (arranged from lowest to highest HCPCS code number) (y axis). CTP, cellular and/or tissue-based products; HCPCS, Healthcare Common Procedure Coding System.
Footnotes
AUTHORS’ CONTRIBUTIONS
M.J.C.: Conceptualization (lead with C.E.F.), methodology (lead with C.E.F.), formal analysis (lead), writing—original draft (equal), and review and editing (equal). C.E.F.: Conceptualization (lead with M.J.C.), data curation (lead), methodology (supporting), and writing—review and editing (equal).
AUTHOR DISCLOSURE AND GHOSTWRITING
M.J.C. is the President of Strategic Solutions and C.E.F. is the Chief Medical Officer of Intellicure and the Executive Director of the USWR. There was no ghostwriter used in the preparation of this article.
FUNDING STATEMENT
The original dHACA study was funded by MTF Biologics.
ABOUT THE AUTHORS
