Abstract
In 2022, the Alliance of Wound Care Stakeholders convened the 2-day Wound Care Evidence Summit™. The Summit brought together a multidisciplinary group of stakeholders that included payers, government agency policymakers, prominent researchers, wound care medical specialty societies, patient and clinical associations, wound care clinics, and manufacturers to discuss wound care evidence and coverage issues. The Summit focused on a wide variety of wound care topics, with an emphasis on the processes payers use to create their coverage policies and the type, quantity, and characteristics of clinical evidence payers require. The most valuable outcome of the Summit was the frank and open discourse among stakeholders, with unprecedented participation from payers and the U.S. Food and Drug Administration (FDA) on the subjects of trial design, product-approval pathways, and coverage policy determination. Stakeholders provided actionable ideas for ways to improve clinical trial research and design that will yield better evidence and ultimately better wound care. This article examines the quality, adequacy, and relevance of the existing chronic wound care research base and discusses the gaps, associated problems, and implications for clinical trial design and execution as identified by Summit participants.

INTRODUCTION
If you put the right people in the room, things can happen.
—Lynn Shapiro Snyder, JD
Partner, Epstein Becker Green
T
This 2-day Summit brought together a multidisciplinary group of stakeholders that included payers, government agency policymakers, prominent researchers, wound care medical specialty societies, patient and clinical associations, wound care clinics, and manufacturers who traditionally might not have been in the same room at the same time.
The Summit comprised panel discussions and speaker presentations that were designed to not only educate but also to promote an open exchange of perspectives among stakeholders who rarely have opportunities to meet in a private forum. To our knowledge, this meeting was the first of its kind to examine the state of scientific research in wound care from the perspectives of researchers and payers. It was designed to encourage stakeholders to tackle the most pressing evidence and coverage issues and build collaborative and sustainable solutions. As Dr. William Ennis, Chief Medical Officer, Healogics, pointed out, the Summit broke down walls so that the payer community and the provider community could listen to one another and start building a framework that will ultimately lead to improvement in patient care and outcomes.
The enthusiasm over the course of the 2 days was palpable and the collaborative nature of the Summit empowered participants to openly share their insights, even when they disagreed with one another. Everyone was engaged in discovering how we could improve clinical research so that payers will have the evidence they need to formulate positive coverage determinations for wound care products and procedures.
A full list of Summit speakers appears on the Alliance website https://www.woundcarestakeholders.org/images/Speaker_list_-_bios_and_photos.pdf We want to acknowledge the payer medical directors who spoke at the Summit: Nancy Awadallah, MD, Aetna Wyndolyn Bell, MD, FAAP, Retired, UnitedHealth Group Deneen Bowlin, MD, CareFirst BlueCross Blue Shield Robert McDonough, MD, JD, Aetna Todd Prewitt, MD, Humana James Rollins, MD, MSHA, PhD, Centers for Medicare & Medicaid Services (CMS) Jeffrey Stone, DO, MPH, FUHM, CWS, Molina Healthcare of Texas.
The goals of the Summit were ambitious:
Explore the current state of wound care research, wound care delivery, clinical trial design, and research generalizability.
Communicate with commercial and government payer medical directors and policymakers about research evidence standards, the use of clinical practice guidelines, and evidentiary standards in coverage decision-making.
Identify actionable solutions to address gaps and limitations in current wound care evidence.
Discuss the acceptability of real-world evidence (RWE) as a solution to some of the quality and adequacy problems in current wound care evidence.
Determine next steps to actualize the solutions that were discussed.
The Summit focused on a wide variety of wound care topics (Table 1), with an emphasis on the processes payers use to create their coverage policies and the type, quantity, and characteristics of clinical evidence payers require.
Wound Care Evidence Summit topics
FDA, U.S. Food and Drug Administration.
KEY THEMES FROM THE WOUND CARE EVIDENCE SUMMIT
Several key themes emerged during the Summit (Fig. 1). In this first editorial, we'll examine the quality, adequacy, and relevance of the existing chronic wound care research base and discuss the gaps, associated problems, and implications for clinical trial design and execution as identified by Summit participants. The focus will be on commercial and government payer evidence requirements. Future editorials will address these topics:
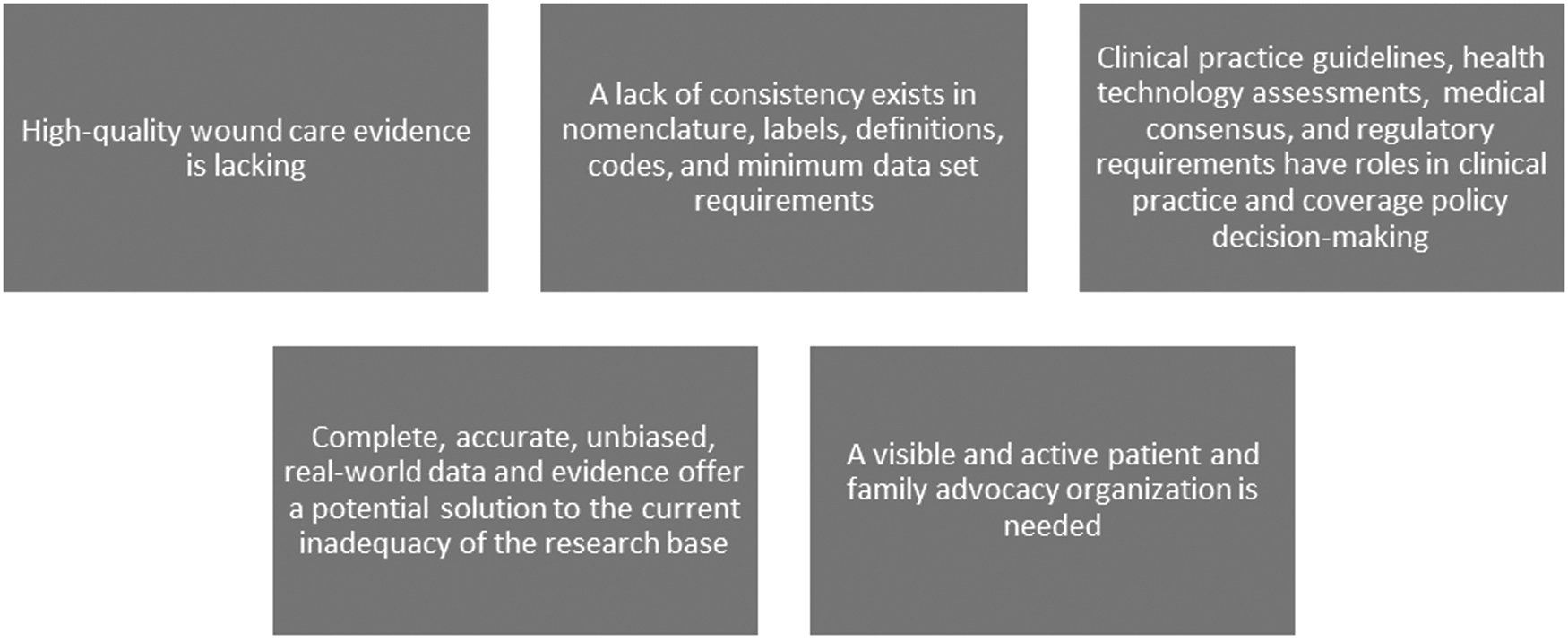
Key themes identified at the Summit.
Coding, minimum data sets, real-world data, and RWE
The role of clinical practice guidelines, health technology assessments, and medical consensus in clinical practice and coverage policy decision-making.
STAKEHOLDERS AGREE THAT WOUND CARE CLINICAL RESEARCH SHOULD BE IMPROVED
The FDA has one set of rules. The CMS has another set of rules. And quite frankly, our payers have a whole host of different rules … a pre-FDA approval pathway and a postmarket trial are different.
–Dr John Lantis
Professor of Surgery, Mount Sinai and Morningside Hospitals
Speakers affirmed that a large majority of wound care products go through the 510(k) pathway to gain product approval. With this approach, premarket clinical trials are not usually required for proof of efficacy because many wound care products are classified as medical devices. The U.S. Food and Drug Administration (FDA) provides guidance about clinical trial designs for drugs and biologics and those wound care products not classified as medical devices, such as cellular and tissue-based products. Consequently, a majority of substantive trials are designed without FDA input and performed primarily to achieve reimbursement from the CMS and private payers and to bolster physician adoption of the technology. Only in 3% to 4% of cases does the FDA require premarket approval (PMA), meaning that clinical trials must be performed to prove efficacy and safety.
Speakers emphasized that the FDA and payers have different objectives; the path to gain regulatory approval is separate from the process for coverage and reimbursement. The FDA wants evidence showing that a product on the 510(k) pathway is safe and effective enough to gain market approval, whereas CMS and commercial payers evaluate whether a product or a procedure has proven efficacy and is reasonable and necessary to determine coverage policies. Even evidence of efficacy and safety, which may satisfy the FDA, does not always translate into proof for payers that a product is better than the existing standard of care and should be covered.
There are many wound care products on the market that are substantially equivalent in their applications. In the absence of comparative effectiveness research, commercial payers may choose to cover and reimburse one product differently than another similar product. When there is no evidence supporting product superiority, cost can be a barrier to coverage, inclusion on a product formulary, and reimbursement.
Speakers agreed that without adherence to widely accepted research design standards, such as those identified in the Alliance's Panel On Wound Care Evidence-based Research (POWER™) study, 1 the wound care evidence base is not robust. Too often studies are of insufficient duration and enroll too few patients and thus are inadequately powered. Granted, clinical trial enrollment can be challenging, and slow enrollment may result in studies with smaller than desired numbers of patients, but such challenges should not prevent researchers from designing studies with enough power to produce results that are relevant to real-world practice.
One of the most significant issues raised by the audience to payer panels was that study populations do not reflect the types of patients being treated in the real world. The trials often enroll healthier patients than those seen in clinical practice. In some cases, trial subjects are people with wounds that might very likely heal without study intervention. Studies have focused on inclusion and exclusion criteria so narrowly that sick and less-mobile patients with comorbidities—those patients most likely to have difficult-to-heal chronic wounds—are often excluded from the trials. Such narrow inclusion criteria limit the generalizability of the research. Moreover, if payers do grant coverage, the inclusion and exclusion criteria used in the clinical trial become the basis for coverage, which limits use of the product or procedure in the real world.
It is easy to understand why sponsors have taken this approach. Trials designed to treat sicker patients with more severe wounds over long periods of time—with enough follow-up to demonstrate that wounds stay healed—strain budgets and resources. Simpler inclusion criteria and minimal exclusion criteria mean larger studies and a higher risk of failure. The result, however, is a lack of innovation in the wound care industry since most 510(k) products in wound care have similar broad treatment indications and many products in a given category are substantially equivalent.
OPPORTUNITIES TO IMPROVE CLINICAL TRIAL RESEARCH AND DESIGN
We can do trials so much better. This is not rocket science.
—Dr Marissa Carter
President, Strategic Solutions, Inc.
The most valuable outcome of the Summit was the frank and open discourse among stakeholders, with unprecedented participation from payers and the FDA on the subjects of trial design, product-approval pathways, and coverage policy determination. Stakeholders provided these actionable ideas for ways to improve clinical trial research and design that will yield better evidence and ultimately better wound care.
Adopt consistent nomenclature
There was consensus among stakeholders that definitions for basic terminology in the wound care industry are inconsistent, even for the definition of a chronic wound. For example, the FDA uses the terms “nonhealing chronic wounds” and “chronic cutaneous ulcers” interchangeably because it does not make distinctions in its definition of chronicity based on time points. This disconnect in how wounds are described impedes accurate clinical documentation, negatively impacts clinical trial design, and presents limitations when researchers want to use electronic health records (EHRs), administrative claims, or registry data for RWE. EHRs often are not equipped to support the documentation requirements providers must meet to get coverage and reimbursement and create data for RWE.
The Alliance has been a driving force behind efforts to update outdated terminology and harmonize nomenclature between FDA and CMS. For nearly a decade, the Alliance has engaged with FDA staff and advocated for updates to its 2006 Chronic Cutaneous Ulcer and Burn Wounds Guidance for Industry. Summit participants noted that the guidance document still includes the outdated and clinically inaccurate terminology “skin substitutes,” which are now referred to as cellular and/or tissue-based products (CTPs) for skin wounds and should be aligned with the ASTM Standard Guide for Categories and Terminology of CTPs. The Alliance helped to lead the effort for this terminology change. In addition, the FDA classification for 510(k) and PMA biological “cellular and tissue-based products for skin wounds” as “wound dressings” is outdated and does not represent the technology or mechanism of action of these products.
The Alliance will continue to have ongoing conversations with the FDA to promote consistent wound care nomenclature and proper classification of wound care products.
Create consensus around the standard of care
The lack of consensus in clinical guidelines was made explicitly clear during the Summit. Since wound care is multidisciplinary, physician specialty societies, clinical associations, and wound care clinics often develop their own clinical practice guidelines that may or may not be in alignment. Kara Couch, Director of Wound Care Services, George Washington University, noted that a PubMed search limited to wound care guidelines published within the past 5 years identified 27 different current guidelines. Guidelines published before that time period still appear in the literature, even those that are outdated or expired.
Such variability in clinical practice guidelines impedes research since there is a lack of consistency in treatments provided to the “usual care” control arm. Furthermore, it negatively impacts the quality of wound care, as clinicians could be following outdated guidelines in practice. The Wound Care Collaborative Community (WCCC), a collaboration with the FDA whose initiatives aim to improve product safety and patient access to cutting-edge medical technology, took on the challenge and established a work group to create consensus around standards of care.
Explore innovative trial designs
Dr. David Armstrong, professor of surgery at the University of Southern California, remarked that the wound care population is changing; patients are getting sicker and living longer with complications. “Our studies largely have not been changing rapidly enough to accommodate all of that. That has left us with fewer and fewer patients that will benefit from many of these very, very helpful trials.” He suggested an approach used in some countries, where all patients treated at wound or limb preservation clinics are automatically considered for clinical trials. Using a similar approach in the United States would require robust consortia and clinical trial networks, as well as a patient registry, but would ensure that study populations better reflect the real world.
A multidisciplinary panel of stakeholders discussed the potential value of adaptive methodologies in wound care research. Adaptive clinical trial designs, as defined by the FDA, are those that allow for prospectively planned modifications to one or more aspects of the design based on accumulating data from subjects in the trial while the trial is ongoing. 2 Adaptive design requires an a priori well-defined interim analysis with metrics that allow for adaptation during the trial; adaptations must be planned and agreed upon before data are reviewed. Adaptive designs enable investigators to identify the optimal clinical benefit of the study intervention without undermining the integrity and validity of the study. In contrast to a fixed trial design, adaptive designs often require less time and fewer participants, and ultimately may be less costly. 3
While the panel agreed that adaptive designs have value, Dr. James Rollins, Acting Deputy Director, Division of Policy and Evidence Review, CMS, warned that changes in a number of variables, such as definitions or measurements, could threaten the internal validity of the study. Dr. Robert McDonough, Senior Director for Clinical Policy Research and Development, Aetna, provided cautionary advice about what is and what is not adaptive design: “So you have a planned clinical study where you're going to have some interim analysis, whereby you make adaptations … But it has to be done carefully. What we don't want to see is people calling it adaptive design where they sort of make ad hoc decisions about changing the trial protocol on the fly and calling that an adaptive design. Because that is a lot more difficult to interpret, that kind of study.”
Panelists encouraged researchers and manufacturers to confer with the FDA, CMS, and commercial payers about innovative designs before engaging in these approaches to ensure the research will meet the expectations and requirements of these entities for approval and coverage.
Include endpoints beyond wound healing
FDA expectations and the lack of consistency in research methodology and design standards have led researchers to overlook important clinical trial endpoints. Speakers acknowledged that it is inappropriate and sets unrealistic patient, provider, and payer expectations to maintain a uniform goal of complete healing across the treatment of all wounds.
Dr. Dev Verma, Medical Officer, Division of Dermatology and Dentistry, Center for Drug Evaluation and Research, FDA, noted that even though 100% wound closure is the most common efficacy endpoint in wound care research, other co-primary or multicomponent endpoints may be acceptable if such endpoints are validated as being clinically meaningful to patients and can be reliably measured. The inclusion of alternative, patient-reported endpoints beyond total wound healing would allow for coverage of care in an expanded patient population. When complete wound healing is not possible, investigators should consider other meaningful endpoints such as 4
Increased patient activity
Reduced social isolation
Reduced pain
Reduced infection
Time to heal
Time to recurrence
Improved ambulation
Increased independence
Limb preservation.
Of these endpoints, stakeholders must prioritize those that are most viable. Summit participants noted that most recently the FDA has been working collaboratively with the WCCC to identify scientifically achievable and clinically relevant endpoints that would serve as primary outcomes in clinical trials.
Create an updated clinical trial guidance document
The Alliance assembled a panel of prominent, respected wound care researchers (POWER) and in 2012 published an article on criteria for conducting methodologically sound randomized controlled clinical trials. 1 Based on what we learned at the Summit, it is time to update these criteria and create a user-friendly guidance document that provides direction in designing trials that will produce generalizable and replicable results and meets approval and coverage criteria. The aim would be to create guidance that is easy to use and becomes widely adopted by manufacturers and researchers. The document would address study duration, number of subjects, blinding, randomization, inclusion and exclusion criteria, endpoints, and data analyses. Ideally, such a clinical trial guidance document would be developed with input from all stakeholder groups via a consensus conference.
Leverage alternate sources of evidence
Speakers agreed that minimum data sets, patient registries, and real-world data can complement data obtained through randomized clinical trials and overcome some of the inadequacies of the existing research base. Dr. Verma shared the FDA's perspective about RWE: “real-world data and real-world evidence may be used to help inform the nature of nonhealing chronic wounds and this can help inform the appropriate length of clinical trials and how they're designed and who are enrolling.” However, when it comes to coverage, payers have not used RWE as part of their process of making coverage determinations. There are drawbacks to using real-world data, which we'll discuss in more detail in a future editorial.
CONCLUSION
The best way to understand what we need to do to improve wound care research and clinical trial design is to ask payers and policymakers directly about the types of evidence they need to approve products and formulate positive coverage policies. The Wound Care Evidence Summit offered an ideal setting for stakeholders to interact with one another, discuss their mutual challenges and concerns, and brainstorm actionable solutions (Table 2). Stakeholders made it clear that it is time to rethink clinical trial design in wound care research to position wound care for future innovation and success.
Proposed solutions to improve evidence quality and potential organizational lead
Marcia Nusgart, Founder and CEO of the Alliance and the driving force behind the Summit, offers this conclusion. “We achieved what we set out to do, and that was to bring together payers, policymakers, providers, researchers, manufacturers, and leaders in the wound care industry to tackle these tough issues. We learned that, as Lynn Shapiro Snyder so aptly stated, who is in the room matters. To achieve the best outcomes for the patients we serve, the wound care community needs to be involved and have a seat at the table when decisions are being made and policies are being crafted by the payers and regulatory agencies. We need to stand up, show up, and volunteer when presented with opportunities to serve. The Alliance will continue to be the united voice for the wound care community.”
ACKNOWLEDGMENTS AND FUNDING SOURCES
The authors wish to thank Cynthia L. Kryder, MS, MWC, and Karen Ravitz for their editorial assistance. Funding for this conference was made possible, in part by 1R13AG072862-01A1 from the National Institute on Aging.
AUTHOR DISCLOSURE AND GHOSTWRITING
There are no competing financial interests. The contents of this article were expressly written by the authors listed. No ghostwriters were used to write this article.
ABOUT THE AUTHORS
