Abstract
Objective:
A new configuration of closed-incision negative pressure therapy (ciNPT) dressings now covers the incision and a broader area of peri-incisional tissues. We have implemented these ciNPT dressings following simple mastectomy (SM) or skin-sparing mastectomy with implant-based reconstruction (IBR). This study assesses patient-reported outcomes of this new protocol.
Approach:
Patients underwent SM or IBR for breast cancer. ciNPT with wide-coverage dressings were placed over the entire breast, and −125 mmHg was applied for 14 days. Upon dressing removal, patients rated their experience using the Wound-Q™ Suction Device Scale and recorded their satisfaction on a Likert scale ranging 1–5.
Results:
Thirteen SM patients and 12 IBR patients were included in the study. The median age was 62 years, and SM patients were significantly older (p < 0.01). Patients rated the ciNPT device highest on items relating to its function and appearance, and lowest on noise and interference with sleep and physical activity. The overall mean score for the combined cohort was 64.8/100. The mean score for SM patients (74.8 ± 19.9) was significantly greater than for IBR patients (53.9 ± 9.6, p < 0.01). The mean overall patient satisfaction rating was 3.92 on a 5-point scale; 4.0 in the SM group and 3.8 in the IBR group.
Innovation:
This study is the first to report on the patient experience with these newly available wide-coverage ciNPT dressings.
Conclusion:
Overall, the dressing was well-tolerated by patients, and satisfaction was high. The positive reception of ciNPT with wide-coverage dressings supports continued use at our hospital.


INTRODUCTION
Closed-incision negative pressure therapy (ciNPT) is a noninvasive, automated system that covers the incision with foam dressing, creating an impermeable seal and applying a vacuum that removes exudate and supports incision healing. The use of ciNPT devices is becoming increasingly commonplace in oncoplastic breast surgery to manage incisions that may have an elevated risk of postoperative wound healing complications. 1 –4 Patients managed with ciNPT after mastectomy have been reported to have lower rates of skin necrosis, 1 dehiscence, 4 surgical-site infection, seroma formation, and overall postoperative complications. 3 As a result, the use of ciNPT has been integrated as part of the standard postoperative protocol following mastectomy at our tertiary hospital.
INNOVATION
Previously, standard ciNPT dressings were shaped into narrow foam strips, covering the incision and a few centimeters of skin on each side. A new iteration of ciNPT implements wider foam dressings that apply the benefits of negative pressure to a larger area of surrounding tissues (Fig. 1). This new design has been used effectively to manage breast, sternal, and abdominal incisions, with positive healing outcomes. 5,6 Although previous studies have examined the clinical impact of the wide-coverage dressings, none to date has documented patients’ comfort and satisfaction with the new configuration. This study is the first to report the patient experience using these newly introduced dressings.
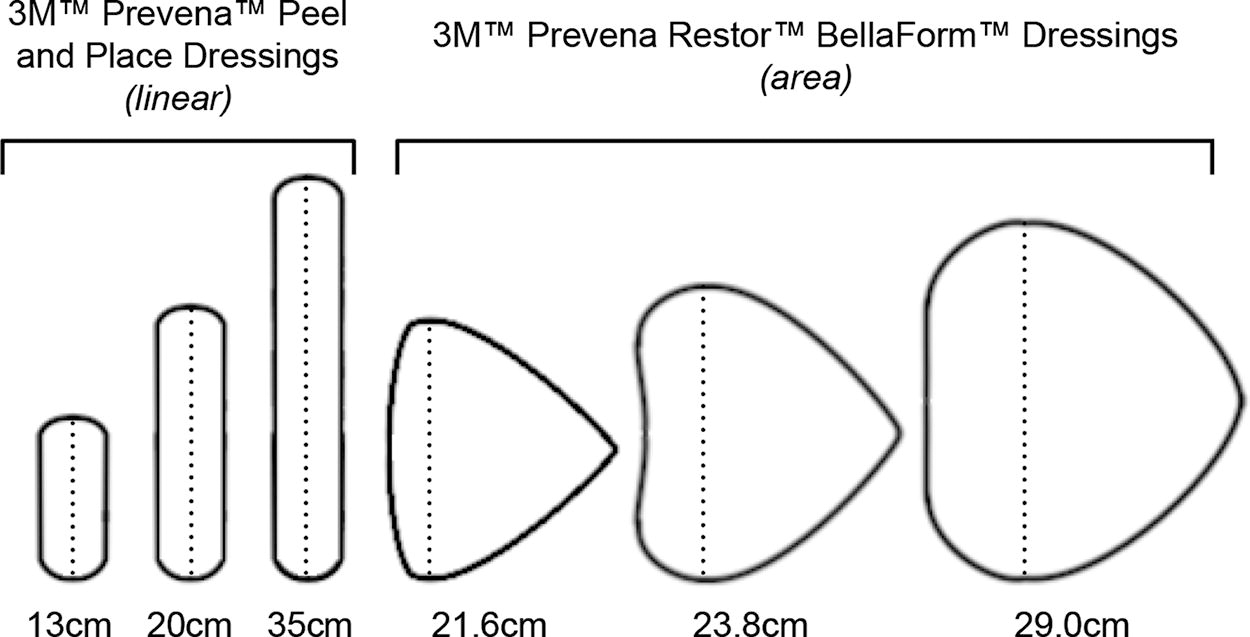
Illustration of the new ciNPT dressing sizes and shapes, comparing the conventional linear dressings (3M™ Prevena™ Peel and Place Dressing) with the new area dressings (3M™ Prevena Restor™ BellaForm™ Dressing). The dotted line represents the length measured in centimeters. ciNPT, closed-incision negative pressure therapy.
CLINICAL PROBLEM ADDRESSED
For patients undergoing mastectomy, there is an area of traumatized tissues that extends beyond the point of incision. In our experience, the wide-coverage ciNPT dressing provides support similar to a postoperative bra, bolstering the entire breast footprint, minimizing movement of the soft tissues, and mitigating postoperative edema. While this may enhance patient comfort, broader dressing sizes could also add bulk, effect placement on the breast, and have other unintended consequences on the patient’s daily activities. As patients are typically discharged to continue ciNPT at home, persistent discomfort could negatively impact adherence to therapy, thereby slowing incision healing. Having recently introduced the device into our tertiary breast service, we aimed to assess patient experience and satisfaction after undergoing simple mastectomy (SM) and immediate IBR.
MATERIALS AND METHODS
The study was conducted in accordance with the Declaration of Helsinki and regional and institutional regulations. Included in this study were patients consecutively undergoing either SM or skin-sparing mastectomy with immediate implant-based breast reconstruction (IBR) for breast cancer. Patients provided informed consent before data collection, according to institutional protocol. Drains were placed only for patients undergoing IBR. Full-coverage ciNPT (3M™ Prevena Restor™ BellaForm™ Incision Management System; 3M, St. Paul, MN) was applied with continuous −125 mmHg for 14 days (Fig. 2). The dressings were removed on postoperative Day 14.
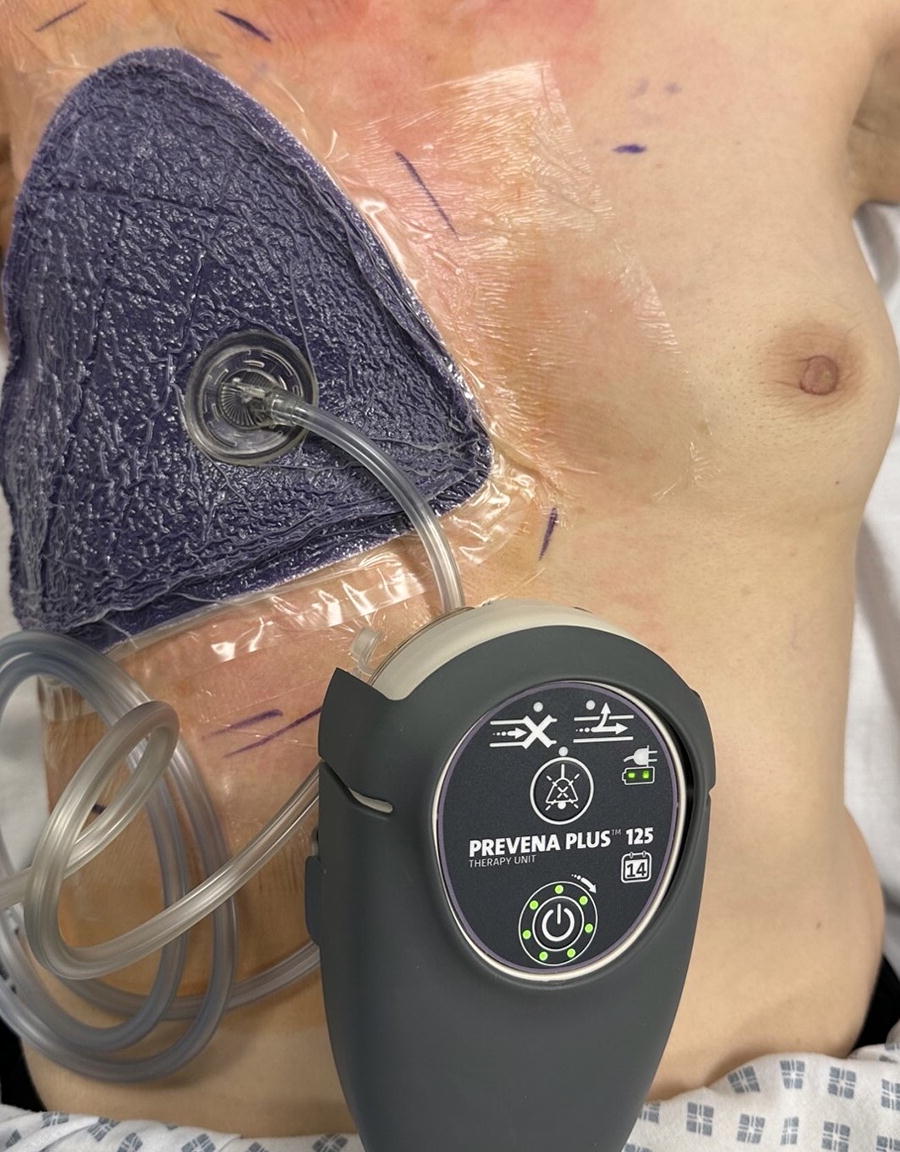
Placement of the wide-coverage ciNPT dressing over the right breast after simple mastectomy. Clear drape was used to protect skin surfaces prone to creasing. The dressing is connected to a lightweight ciNPT unit (3M™ Prevena™ Plus 125 Therapy Unit), and −125 mmHg is maintained for 14 days via a pressure-sensing connection pad (3M™ SensaT.R.A.C.™ Pad).
Patients were asked to complete the validated Wound-Q™ Suction Device Scale (Techna Institute, Toronto, Canada) consisting of 9 items. 7 Scores were summed and converted to equivalent Rasch transformed Q scores from 0–100. Additional wound experience questions were asked, alongside space for free-text comments. Patients were also asked to rate their satisfaction with the device on a 1–5 Likert scale.
RESULTS
Twenty-five patients participated in this study: 13 SM and 12 IBR (Table 1). The median age was 62 years (interquartile range, 52–75). Patients in the SM group were significantly older (p < 0.01). The number of bilateral cases and patients that underwent axillary surgery at the same time was similar between groups. The average scores on the Wound-Q™ Suction Device Scale for each cohort are shown in Table 2. Higher scores reflect a better outcome. The average scores for patients in the SM and IBR groups were 74.8 and 53.9, respectively (p < 0.01). Optional comments provided in the free-text feedback section are shown in Fig. 3.

Free response comments offered by surveyed patients.
Cohort demographics
IQR, interquartile range.
Average score for Wound-Q items
SD, standard deviation.
For 5 (20%) patients in the IBR group, the ciNPT required a follow-up assessment from a health care professional. Two were removed prematurely due to loss of negative pressure and discomfort. Itching or erythema was reported by 9 (36%) patients (6 SM, 3 IBR), which resolved spontaneously in all patients once the dressing was removed. Nineteen (76%) patients reported that the dressing interfered with washing, but this was “just a little” for all, apart from 5 (25%) patients who reported that this was a significant problem (1 SM, 4 IBR). Upon dressing removal, only 3 (12%) patients reported feeling any pain. Twenty-two (88%) patients said that they would consent to having the dressing again (12 SM, 10 IBR). There were no serious adverse events, including infection, dehiscence, or rehospitalization, that occurred in any of the patients.
Mean overall satisfaction rating on a Likert scale from 1–5 for the overall cohort was 3.92. This score was 4.0 for the SM group, and 3.8 in the IBR group.
DISCUSSION
The use of ciNPT has been associated with improved clinical outcomes compared with standard postoperative dressings on various incision types. 8 –13 More specifically, breast incisions managed with ciNPT have correlated with lower rates of total wound complications, surgical-site infections, seroma, dehiscence, and necrosis. 13 These positive clinical outcomes reflect what we have experienced at our hospital, as we have adopted the use of ciNPT covering the entire breast into our postmastectomy protocol. Because ciNPT is continued at home after patient discharge, patient comfort, understanding, and satisfaction are important for therapy adherence and consequent effectiveness. In this study, we conducted the first patient-reported quality-of-life assessment of ciNPT with the newly designed wide-coverage foam dressings after oncological breast surgery.
Overall, the dressing was well-tolerated by patients, evidenced by the vast majority reporting that they would consent to its use again, and the high overall satisfaction ratings. Patients gave the highest marks on the ciNPT device appearance and ability to remove fluid from the incision. Total scores were markedly higher in the SM group compared with the group undergoing IBR. Although the cause of this difference cannot be definitively identified, the largest score differences (>1 point) between the two groups were on the device noise and interference with socializing and physical activity. Interference with normal activities may, in some part, be affected by the placement of drains only after IBR, as the negative pressure tubes or device could be difficult to manage alongside drains. Likewise, the presence of drains can make it difficult to maintain the seal necessary for device function, which could have contributed to the 2 cases in which ciNPT was discontinued early in the IBR group. Loss of seal would also have triggered an automated alarm from the device, which might explain the worse noise scores in the IBR group. No follow-up assessments or discontinuations for discomfort or loss of seal occurred in the SM group. In addition, the patients undergoing IBR were significantly younger than those undergoing SM. Patient age can impact quality-of-life scores after oncological breast surgery, with younger patients having a larger reduction in quality of life and slower recovery after treatment. 14 –17 This effect of age could have a confounding effect on the Wound-Q scores in the two cohorts.
Despite these differences in the ciNPT device assessment, the overall satisfaction scores were high and similar between the two groups. While the reasons for this are likely multifactorial, the free response answers could provide context. The negative responses primarily focused on the practical impacts of ciNPT, including the weight, the noise, and interference with showering. In contrast, the positive responses pointed to feelings of greater support, less anxiety, and optimism about body image. Such psychosocial factors are significant predictors of satisfaction with breast appearance and overall outcome postmastectomy, 18 and may explain the high scores across the patient population.
Few studies have previously assessed patient-reported quality of life or satisfaction with any type of ciNPT dressing. Manoharan et al. included quality-of-life assessments in a randomized controlled trial of patients managed with conventional dry dressings or linear ciNPT after primary knee arthroplasty. 19 The patients maintained a wound diary, reporting that ciNPT had better leakage control and wound protection than dry dressings, although quality-of-life factors (including odor, movement impairment, self-hygiene, and sleep) and overall satisfaction were scored similarly high in both treatment groups. In another randomized controlled trial, morbidly obese patients undergoing cesarean delivery were also equally satisfied with linear ciNPT or conventional dressings, with 89% willing to use ciNPT again and 91% satisfied with wound healing. 20 Ferrando et al. also observed patient-reported quality of life among patients receiving linear ciNPT or standard dressings after oncological breast surgery. 1 The patients scored both ciNPT and standard dressings 6/10 on the Body Image Scale, 21 and ciNPT significantly higher on the Patient Scar Assessment Scale. 22
This study is limited in that it is an observational study with a relatively small sample size. Due to the nature of the therapy studied, it is also not possible to rule out the impact of the placebo effect on patient-reported outcomes. The study contains no comparative therapy group, and therefore does not attempt to establish superiority over alternative incision management techniques. The Wound-Q™ Suction Device Scale was selected to specifically assess outcomes and satisfaction related to ciNPT; however, further studies that use the Breast-Q™ Scale (Techna Institute) to collect outcomes specifically related to breast surgeries, or other assessments to score pain, scarring, or additional outcomes, may be needed to further understand the quality-of-life benefits of ciNPT after mastectomy.
This small cohort study supports the use of ciNPT with wide-coverage dressings in SM patients and demonstrates that the dressing is well-tolerated by the majority of reconstruction patients. Still, areas for improvement remain to better the patient experience among those undergoing immediate IBR.
KEY FINDINGS
Recently, we have begun using newly available wide-coverage ciNPT dressings following mastectomy for breast cancer.
We retrospectively assessed the patient-reported experiences with these dressings after (SM) or mastectomy with immediate breast reconstruction (IBR).
Thirteen SM patients and 12 IBR patients were included in the study. After 14 days of ciNPT, patients evaluated the dressings using device-specific and satisfaction scales.
Patients scored the dressings highly on function and appearance, and low on noise level and interference with daily activities. The SM group gave significantly higher scores then the IBR group. However, overall satisfaction was high in both groups.
Footnotes
ACKNOWLEDGMENT AND FUNDING SOURCES
The authors thank Mikaela M. Sifuentes, PhD (3M), for article preparation support. No external funding was provided for this study.
AUTHORS’ CONTRIBUTIONS
B.G.B. contributed to the conceptualization, data curation, investigation, and writing of the article. A.P. contributed to the conceptualization, data curation, investigation, methodology, supervision, writing, and review and editing of the article.
AUTHOR DISCLOSURE AND GHOSTWRITING
A.P. is a paid consultant to 3M Company (St. Paul, MN, USA). B.G.B. has no conflicts of interest to disclose.
ABOUT THE AUTHORS
