Abstract
Objective:
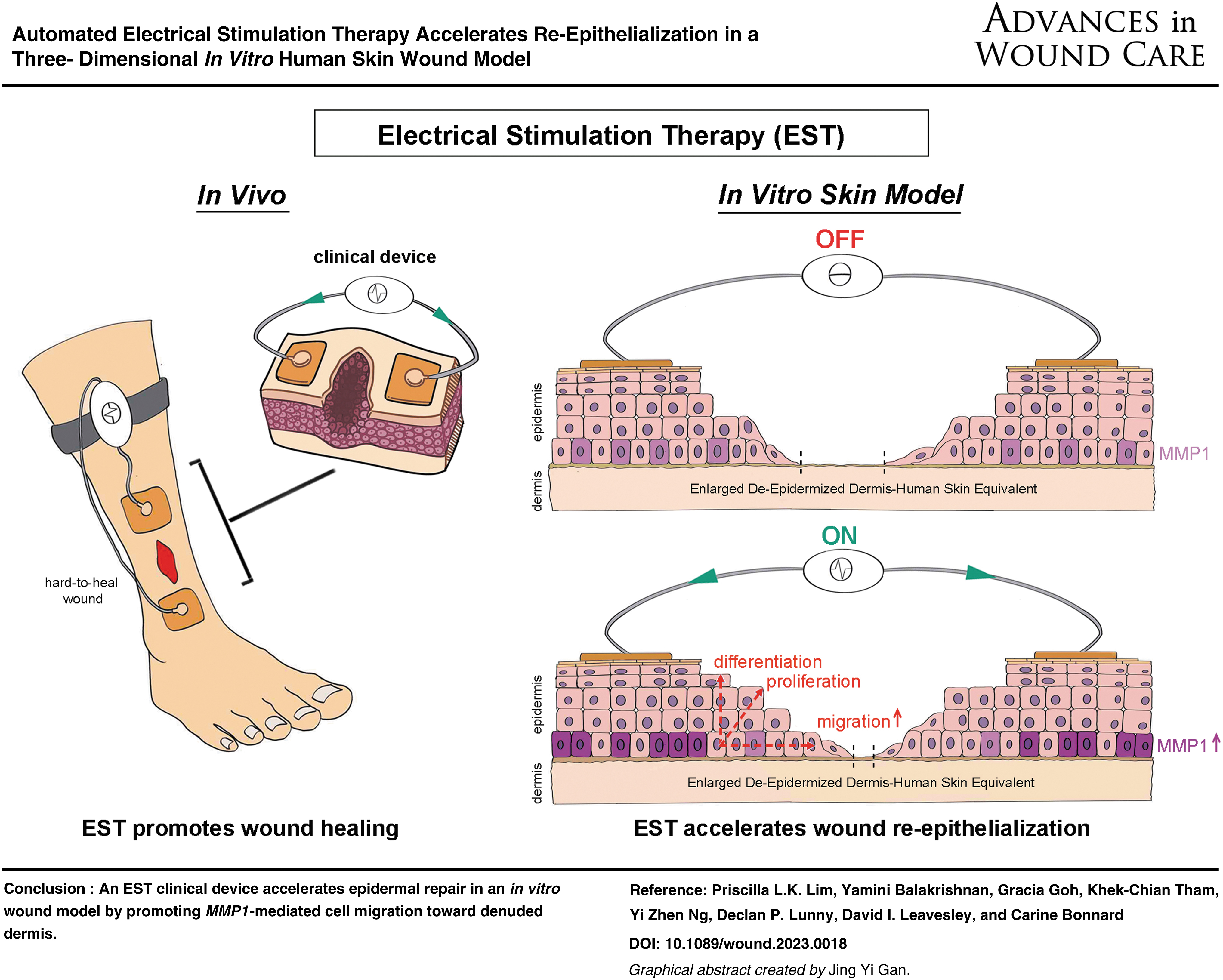
Electrical Stimulation Therapy (EST) shows promise for the purpose of accelerating wound healing, but the right electrical stimulation parameters and its mode of action remain unclear. We aim to evaluate the effect of a new EST clinical device on epidermal repair using an in vitro human skin wound model.
Approach:
We scaled up a well-established 3D De-Epidermized Dermis-Human Skin Equivalent (DED-HSE) wound model to fit a clinically used device that delivers preprogrammed microcurrent EST. The impact of EST on re-epithelialization of 4-mm circular epidermal wounds was assessed after 4 and 7 days of treatment, using metabolic activity assay, immunohistochemistry (IHC) staining, and RNA in situ hybridization.
Results:
EST was successfully applied to the wounded in vitro skin model. Large DED-HSEs retained good cell viability for up to 7 days of EST treatment. Excisional wounds subjected to EST for 4 days consistently exhibited faster closure (mean 65.8%, n = 9) compared to untreated wounds (mean 49.7%, n = 9) (p < 0.05). Wounds exposed to EST exhibited significantly longer epithelial tongues (re-epithelialization mean 50.3%, n = 9) than untreated wounds (mean 26.2%, n = 9) (p < 0.001), suggesting faster keratinocyte migration and proliferation. Increased MMP1 transcription (p < 0.05) in ES-treated periwound suggests a mechanism for enhanced keratinocyte migration. IHC staining showed advanced epidermal proliferation (p63) and differentiation (K10) in EST-exposed wounds (n = 15), as well as stronger attachment of the newly formed epidermis into the dermis compared to untreated controls (n = 15) (p < 0.001).
Innovation:
We present a novel approach to assess an EST clinical device designed to stimulate wound healing. Using a scaled-up 3D human skin wound model, we could demonstrate the positive effect of EST on epithelial cell responses and shed light on possible mechanism.
Conclusion:
Our study provides experimental evidence that microcurrent therapy accelerates wound closure and improves the quantity and quality of re-epithelialization.

INTRODUCTION
Patients with chronic wounds suffer from lesions that do not heal within a timely period (6–12 weeks), are itchy and painful, and often become infected. Chronic wounds result in functional impairment, compromised sleep quality, and depression, and reduce the quality of life. 1
Current clinical wound management interventions are designed to maintain wound bed moisture, remove excess wound exudates, and minimize infection. Depending on the type of wound, some interventions have been proposed to initiate or re-start the healing process, including debridement, hyperbaric oxygen, negative pressure, ultrasound, and electromagnetic fields. 2 –5 While clinical practitioners have access to a variety of these therapies and interventions, few achieve complete wound closure in all patients, urging the need to develop new therapeutics.
The presence of an endogenous transepithelial potential (TEP) across skin tissue has been known of, and investigated for almost 150 years. 6 The active transport of ions across the cell membrane generates the asymmetrical distribution of salt ions in the stratified epidermis, creating an endogenous TEP or “skin battery” that varies from 10 to 60 mV in mammalian skin. 7,8 Injury disrupts the membrane potential and abolishes the ion gradient. Accordingly, acute skin lesions generate a lateral electric field (EF) of 100–200 V/m, with the wound edge being more positive than the center. 6,8 Substantial evidence suggests that these bioelectric processes are critical components of natural wound healing. 7 –10
Several studies suggest that the endogenous EF is dysfunctional, or minimal in chronic wounds, contributing to delayed wound healing. 11 Therefore, strategies to restore endogenous bioelectric signals with the application of exogeneous electrical fields have been developed. 12 –16 Several prototypes, delivering direct, alternating, and pulsed currents with variable parameters (voltage, frequency, duration, and amplitude), have been tested in vitro and in animal models. These experimental studies have shown that exogeneous electrical stimulation (ES) accelerated wound healing and have underlined some aspects of the cellular and physiological responses to ES. 17
Changes in cell orientation, directed migration, cell proliferation, inflammatory cell infiltration, stimulation of peripheral nerves, increased perfusion, enhanced angiogenesis, and inhibition of bacterial growth have each been described as key mechanisms activated by ES during the wound healing process. 18 –21 A number of clinical trials have also demonstrated the positive effect of electrical stimulation therapy (EST) on wound healing, especially on patients with chronic wounds. 15,22,23
Although the application of exogenous ES is a promising therapy, the clinical adoption of EST for recalcitrant wounds remains low. This may be because EST devices have traditionally been large, costly, unportable, and most importantly inefficient. Side effects such as pain, muscle contraction, and skin irritation are factors that may discourage patients from using EST. 24,25 It is therefore essential to continue developing appropriate clinical devices, as well as improving preclinical testing methods that assess safety, efficacy, and mechanisms of action.
To overcome problems with complex clinic-based EST systems, portable, pocket-sized, single-use EST devices have been developed to deliver microcurrents at levels similar to endogenous bioelectric signals. 26 –28 These devices offer a subsensory intervention that does not trigger muscle contraction or hinder patient mobility. Although clinical trials have shown positive effects of microcurrent EST devices in wound healing, there remains a significant gap in understanding their efficiency and mode of action. 21,29
To assess the effect of microcurrent ES on wound repair and explore its underlying mechanisms, simplified 2D skin cell cultures, as well as more complex 3D De-epidermized Dermis (DED) and skin explants have previously been used successfully. 30 –32 However, although these in vitro assays have demonstrated positive effects of ES, they are not designed to test EST clinical device directly. The electrical apparatus and experimental setup do not replicate the actual clinical setting, the duration of treatment is often much shorter than in vivo, and decreased cell viability in ex vivo skin explants have been observed, limiting the direct transfer of findings from the bench to the clinic. 33
In this study, we have used a different approach by testing a clinically used EST device that delivers preprogrammed microcurrents in an enlarged 3D DED-Human Skin Equivalent (DED-HSE). 34,35 Experimental design and culture conditions were optimized to observe the effects of microcurrent EST for up to 7 days. Using metabolic activity assay and immunohistochemistry (IHC) staining, we assessed the effect of EST on epidermal wound closure, focusing on the migration, proliferation, and differentiation of keratinocytes over denuded basement membrane. Further IHC staining and RNA in situ hybridization (ISH) revealed possible mechanisms of action.
INNOVATION
We have scaled up and optimized a 3D in vitro DED-HSE wound model to accommodate testing of a clinical EST device. This innovative system allowed us to evaluate the effect of microcurrent, sequentially released by a clinical device for up to 7 days, on epidermis reconstruction. The positive effect of this clinical device on wound re-epithelialization could be assessed at the cellular and molecular levels at different time points, shedding light on potential EST mechanisms of action.
CLINICAL PROBLEM ADDRESSED
Hard-to-heal wounds are a silent and growing epidemic due to the aging population and the continued threat of obesity and diabetes worldwide. Various therapies have been proposed to improve wound healing in patients suffering from recurrent and painful wounds, including exogenous EST. Clinical studies have shown that EST could, in most circumstances, reduce pain and kick-start the healing process in recalcitrant wounds. However, limited in vitro studies have demonstrated the effectiveness and mode of action of EST in wound healing. Our study presents in vitro experimental evidence that microcurrent released by an easy-to-use and painless clinical device improves tissue repair by accelerating epithelium reconstruction. These results corroborate the substantial body of anecdotal and clinical observations that indicate EST devices are effective and beneficial for the clinical management of wounds.
MATERIALS AND METHODS
Human skin tissues
Approval was obtained from the A*STAR Human Biomedical Research Office, under the IRB 2019-094, for the use of ethically sourced human skin materials from Unison Collaborative (Singapore) and Biotoskin (France). Written informed consent was obtained from patients before skin collection, and all patient identifiers were removed. Skin tissues were obtained from healthy donors, with BMI <30, undergoing elective abdominoplasty to meet the need of large-sized skin for each experiment (minimum of 150 cm2 per donor). One of the skin donations was large enough for two experiments. The presence of severe stretch marks and/or tattoo was the only exclusion criterion. Full-thickness skin tissues from four unrelated Caucasian females, 49, 55, 57, and 68 years old, were obtained.
Human epidermal keratinocyte culture
Early passage of primary human keratinocytes derived from a healthy donor (de-identified Chinese, female, 38 years old, abdomen skin) was obtained from the Asian Skin Biobank, at the Skin Research Institute of Singapore, A*STAR, and used throughout the study. Frozen cells were thawed and cultured on a layer of gamma-irradiated 3T3 feeder cells in Full Green's medium (FG) with modifications as described previously. 36 Cells were maintained at 37°C, with 5% CO2 and 95% ambient air. The study was conducted with strict adherence to the Human Biomedical Research Act 2015 of Singapore.
DED-HSE wound model
The DED-HSE is a well-established 3D in vitro human skin model often used for the evaluation of novel wound therapies. 34,37,38 To assess the effect of microcurrent that is automatically delivered by a clinically used EST device, the DED-HSE was scaled up and the culture conditions were optimized. The DED-HSEs were prepared from four full-thickness abdomen skin tissues using the method previously described by Xie et al., with some adjustments and modifications. 35
Briefly, large format DEDs (3 cm by 5 cm for day 0, and 5 cm by 10 cm for assessment after 4 and 7 days) were prepared to allow the attachment of adhesive electrodes from the clinical device (Supplementary Fig. S1a). After de-epidermization, three stainless-steel rings (Aix-Scientifics, Aachen, Germany) were carefully aligned and centered onto the papillary side of each DED, and 2 × 104 human keratinocytes were seeded in each ring (Supplementary Fig. S1b). After 2 days in culture, the rings were removed and the DEDs were placed on sterile polymer mesh in a 15-cm Petri dish with FG medium. DEDs were kept in air-liquid interface for 9 days to facilitate full epidermal growth and stratification (Supplementary Fig. S1c, d).
After 9 days in culture, three 4-mm partial-thickness wounds (n = 3) were created, with a biopsy punch (Stiefel, Castle Hill, NSW, Australia) excising through the epidermis. The three wounds were aligned in the center of each DED-HSE, parallel to the EST electrodes (Supplementary Fig. S1e, f). The adhesive electrodes were sterilized with Mikrozid AF (Schülke & Mayr GmbH, Norderstedt, Germany) before use in vitro. The wounded DED-HSEs were cultured in FG medium (with phenol red) and kept in a temperature-controlled incubator at 37°C, with 5% CO2 and 95% ambient air. A total of five independent experiments were performed using four different donors' skin. Three experiments were assessed after 4 days of EST (n = 9 treated compared to n = 9 untreated wounds), and two experiments after 7 days of EST (n = 6 treated compared to n = 6 untreated wounds).
EST clinical device
In this study, the EST clinical device tested was Accel-Heal (Cat# K560-6; Accel-Heal Technologies Limited, Hever, United Kingdom), a single-use, wearable device with two adhesive electrodes (654, Carbon, 25 Gel, in sealed pouch, sized 5.1 × 5.1 cm) that automatically delivers microcurrent over 2 days. After 2 days, the device was changed to a new device, while the electrode pads stayed on the skin. Patients with chronic wounds are usually treated for 12 days; therefore, a total of six EST devices (6 × 2 = 12 days) are used over a full treatment program. Clinical studies have demonstrated that treating diabetic foot ulcers, venous leg ulcers, and pressure ulcers with the Accel-Heal device significantly reduces wound pain and stimulates healing to wound closure. 26 –28,39 In this study, we used two sequential devices to deliver therapy for 4 days, and 4 sequential devices to deliver therapy for 7 days.
The Accel-Heal EST consists of cycles of active sessions of ES, interspersed by rest periods. For each device, there is an active session every 2 h on the first day, while there is an active session every 4 h on the second day. During each active session, Accel-Heal delivers a preprogrammed sequence of low-voltage monophasic and biphasic pulsed current (LVBMPC) in three stages, which are described in Table 1.
Parameters of electrical stimulation delivered by the EST device during the biphasic stages 1 and 3 and monophasic stage 2 of the active session
In the first biphasic stage, an electrical current is applied in a modulated sequence of 20 steps of varying amplitude and frequency. Each step applies electrical pulses for 1.6 s followed by 1 s of rest. The cycle of 20 steps is repeated 8 times. In the second monophasic stage, the electrical current is pulsed at an amplitude of 40 μA at a frequency of 10 Hz for a total of 20 min. In the third biphasic stage, the same cycle of 20 sequential steps as in the first stage is repeated twice. As a result, the three stages of an active session last 28 min and 40 s. The device adjusts voltage to ensure the programmed level of current is delivered.
The proprietary therapeutic regimen was first described in a patent (GB2432320) published in 2012 based on the empirical experiences of the inventor, and later incorporated into a clinical device. 40 Constantly varying the amplitude and frequency of the current is believed to reduce the chance of habituation and the higher frequencies (shorter pulse durations) are thought to offer deeper tissue penetration.
Once the device is activated, an electrical circuit is established across the skin and through the wound. To ensure proper function of the device and to monitor the current flow through the circuit, a green LED on the ES device flashes every 2 or 5 s (Supplementary Video S1). No LED flashing or fast flashing (every second) is a sign of low battery or disconnected electrical circuit (Supplementary Video S2). DED-HSEs were treated for 4 and 7 days to examine the process of re-epithelialization at the early stage of healing. A new device was replaced every 2 days and daily assessment showed proper LED flashing throughout the treatment period, validating our optimized DED-HSE wound platform.
Measurement of wound closure and toxicity by MTT assay
On 0, 4, and 7 days post-treatment, each wounded DED-HSE was harvested and placed in 50-mm Petri dish containing 0.5 mg/mL of 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) solution.
41
The samples were incubated in 5% CO2 at 37°C for 90 min to allow the formation of formazan. Viable cells with active metabolism react with the tetrazolium ring of the MTT producing a purple-colored insoluble formazan product. Images of the DEDs were captured using Nikon SMZ745T Stereo Dissecting microscope, to observe cell viability across the wounded tissue, and to measure wound closure. Using Image J software (https://imagej.nih.gov/ij/), each open wound area (X) was measured and subtracted from the original wound area on day 0 (mean Y) to calculate the percentage of wound closure (Supplementary Table S1), using
Hematoxylin and eosin staining
After MTT assay, each DED-HSE was bisected into two equal halves where re-epithelialization looked the most advanced. The tissue was fixed in 10% neutral buffered formalin and processed into formalin-fixed paraffin-embedded (FFPE) blocks. They were subsequently sectioned and stained with hematoxylin and eosin (H&E) according to standard histological protocols. 42
Full sections were scanned using Zeiss Axio Scan.Z1 system (Carl-Zeiss, Oberkochen, Germany). Wound edge images were taken with Olympus System Microscope BX43 (Olympus LS, Tokyo, Japan), at objective 20 × or zoomed in from the scanned images.
H&E images were used to quantify the extent of re-epithelialization, by calculating the length of the newly formed epidermis (or epithelial tongues) emerging from each wound edge, across the wound bed. The percentage of re-epithelialization was calculated on day 4 for each DED-HSE, based on the ratio of sum of both epithelial tongues (right and left wound edges) to the total wound length (Supplementary Table S2).
Immunohistochemistry
FFPE blocks were sectioned and processed for IHC using standard protocols. 43 Four markers, Ki-67, tumor protein 63 (p63), keratin 14 (K14), and keratin 10 (K10), were selected to assess the effects of treatment on cell proliferation and differentiation. The following primary antibodies were used: mouse monoclonal anti-K10 [DE-K10] (1:200) from Dako (Agilent Technologies, Santa Clara, CA); mouse monoclonal anti-K14 [LL001] (1:25) was a supernatant gift from E. Birgitte Lane's laboratory (Skin Research Institute of Singapore, A*STAR); mouse monoclonal anti-p63 [4A4] (1:50) from Abcam (Ab735; Cambridge, United Kingdom); and mouse monoclonal anti-Ki-67 [MIB-1] (1:50) from Dako. Heat-induced epitope retrieval of the FFPE sections used 90°C heated 0.01M sodium citrate buffer pH6 for 15 min.
Optimal cutting temperature (OCT) blocks were prepared for two batches of DED-HSEs (one on day 4 and one on day 7). OCT blocks were sectioned at 7 μm for immunofluorescent staining. Collagens COLIV, COLVII, and COLXVII, laminin V (LAM V), and integrins α3, β1 and α6, β4 were revealed to assess the dermal-epidermal junction, as well as keratin 16 (K16) expressed in migrating keratinocytes.
The following primary antibodies were used: mouse monoclonal anti-K16 [LL025] (neat) and mouse monoclonal anti-COLVII [LH7.2] (neat) were supernatant gift from E. Birgitte Lane's laboratory (Skin Research Institute of Singapore, A*STAR); mouse monoclonal anti-COLIV [CIV22] (1:50) from Agilent; rabbit monoclonal anti-COLXVII [EPR18614] (1:1000) from Abcam ab184996; mouse monoclonal anti-LAM V [P3E4] (1:50) Millipore MAB1949; mouse monoclonal anti-ITGA3 [F35 177-1] (1:50) from Abcam ab20140; rat monoclonal anti-ITGA6 direct FITC conjugate NKI-GoH3 (1:50) from AbD Serotec; mouse monoclonal anti-ITGB1 [P5D2] (1:50) from Santa Cruz; and rat monoclonal anti-ITGB4 [439-9B] (1:50) from Abcam ab110167. All IHC results were confirmed in at least three independent experiments using different donors' skin.
RNA ISH
FFPE blocks were sectioned at 5 μm. RNA ISH was performed using RNAscope™ 2.5 HD Red Assay kit from ACDBio following the manufacturer's protocol. RNAscope probe against human MMP1 (#569311) and MMP3 (#403421) from ACDBio was used in the protocol. Using ImageJ software, the Color Deconvolution function was applied on the RNAscope image. After setting the threshold to include only stained cells on the image, “Analyze Particle” function was used to obtain the total area of stained cells, which was normalized against the total length of the tissue measured (Supplementary Table S3).
Histological quantification
This method of quantification indicates the extent of keratinocyte migration. Ki-67 and p63 immunostaining images were used to count the total number of cells in the newly formed epidermis, which was normalized to the total length of the wound (distance between two wound edges) to assess epidermal growth. The percentage of Ki-67 cells was calculated using (number of Ki-67-positive cells/total number of cells on Day 4 or Day 7) × 100 (Supplementary Table S4). The percentage of p63 cells was calculated using (number of p63-positive cells/total number of cells on Day 4 or Day 7) × 100 (Supplementary Table S5). The percentage of epidermal detachment was calculated using (the total length of epithelial tongues detached/total length of epithelial tongues) × 100 (Supplementary Table S6).
Statistical analysis
Data were analyzed and compared using Student's independent t-test using GraphPad Prism (GraphPad Software, La Jolla, CA). Statistical significance was assumed with a p-value ≤0.05 (indicated by *), p-value ≤0.01 (indicated by **), and p-value <0.001 (indicated by ***). Data are plotted in column graph with standard error of the mean (SEM).
Electronic laboratory notebook was not used.
RESULTS
Scaled-up DED-HSE wound model is optimized for EST device assessment
To assess the effect of a clinically used EST device, a well-established DED-HSE wound model was enlarged and the culture conditions were optimized. DED-HSE size was increased from the usual 1.2 × 1.2 cm to 5 × 10 cm to mimic the clinical situation where two adhesive electrodes are placed on either side of the wounds (Fig. 1 and Supplementary Fig. S1). 35
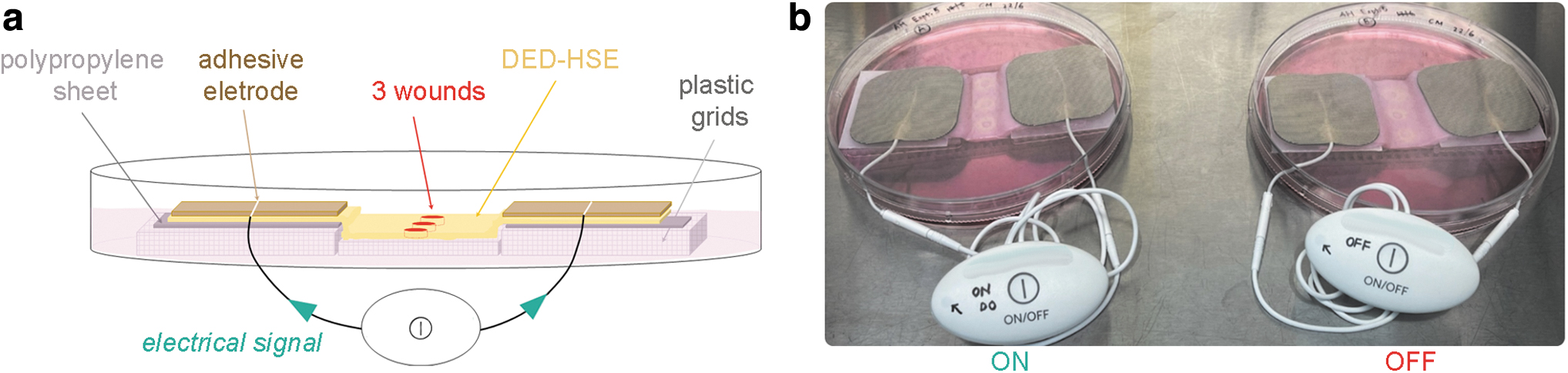
To avoid circumvention of electric current and to restrict ES to the DED-HSE, nonelectrical conducting components were used in the setup, replacing the usual stainless-steel grid with a plastic support (Fig. 1a and Supplementary Fig. S1c, d). Both sides of the DED were lifted by placing additional layers of plastic mesh (5 cm by 5 cm) and polypropylene sheets below the DED to block culture medium and mimic the clinical use on intact skin. The sterilized adhesive electrodes were then placed on the papillary side of DED-HSE (Fig. 1 and Supplementary Fig. S1e, f).
Two DED-HSEs prepared from the same donor's skin were used for each experiment: one DED-HSE where the device was constantly switched ON (treated), and a duplicate where the device was kept OFF to serve as an untreated control (Fig. 1b). Daily assessment of the device LED showed flashes every 2 or 5 s throughout the treatment period, confirming proper current flow and validating our EST testing platform.
Cell viability is maintained, and epithelial wound closure is improved by EST
To assess the effect of EST on cell viability and re-epithelialization, a colorimetric MTT assay was performed on DED-HSEs on day 0, and after 4 and 7 days of treatment. Purple formazan crystals, produced by viable cells with active metabolism, were observed in all treated DED-HSEs, with no obvious difference from untreated controls (Fig. 2a, b).
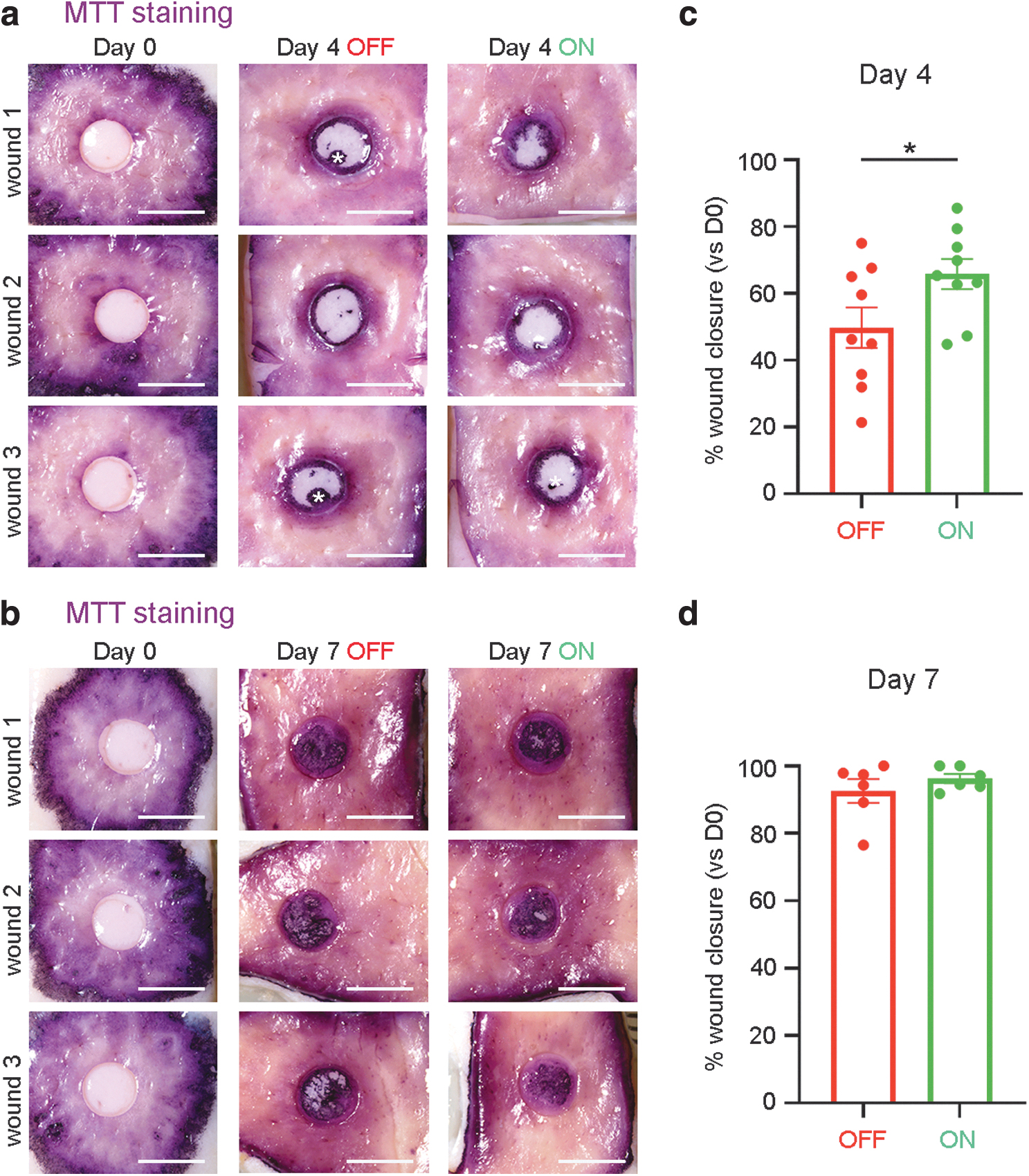
To evaluate the effect of EST on re-epithelialization, the percentage of wound cover was measured in each of DED-HSE wound on days 4 and 7 and compared to day 0. After 4 days of ES treatment, wounds had closed significantly faster (mean 65.8% ± 4.5 closure, n = 9) compared to untreated wounds (mean 49.7% ± 6.0 closure, n = 9), in three independent experiments using different donors' skin (p < 0.05) (Fig. 2c and Supplementary Table S1). Wound closure was almost complete by day 7 and was consequently not significantly different between treated and untreated wounds (mean 96.2% ± 1.3, n = 6 vs. mean 92.5% ± 3.5, n = 6). However, visually re-epithelialization looked more advanced in wounds exposed to ES compared to controls (Fig. 2b, d). To confirm these observations, we next examined the development and structure of the newly formed epidermis using histology.
Microcurrent EST promotes keratinocyte migration
The effect of EST on wound re-epithelialization was assessed at the tissue and cellular levels using H&E, ISH, and IHC staining on days 0, 4, and 7. H&E images revealed significantly longer epithelial tongues in wounds treated with ES for 4 days (mean 50.3% ± 4.7, n = 9) compared to untreated wounds (mean 26.2% ± 3.3, n = 9) (p < 0.001) (Fig. 3a–c, Supplementary Fig. S2a and Supplementary Table S2), suggesting faster migration of keratinocytes from the wound edges across the denuded dermis upon EST.
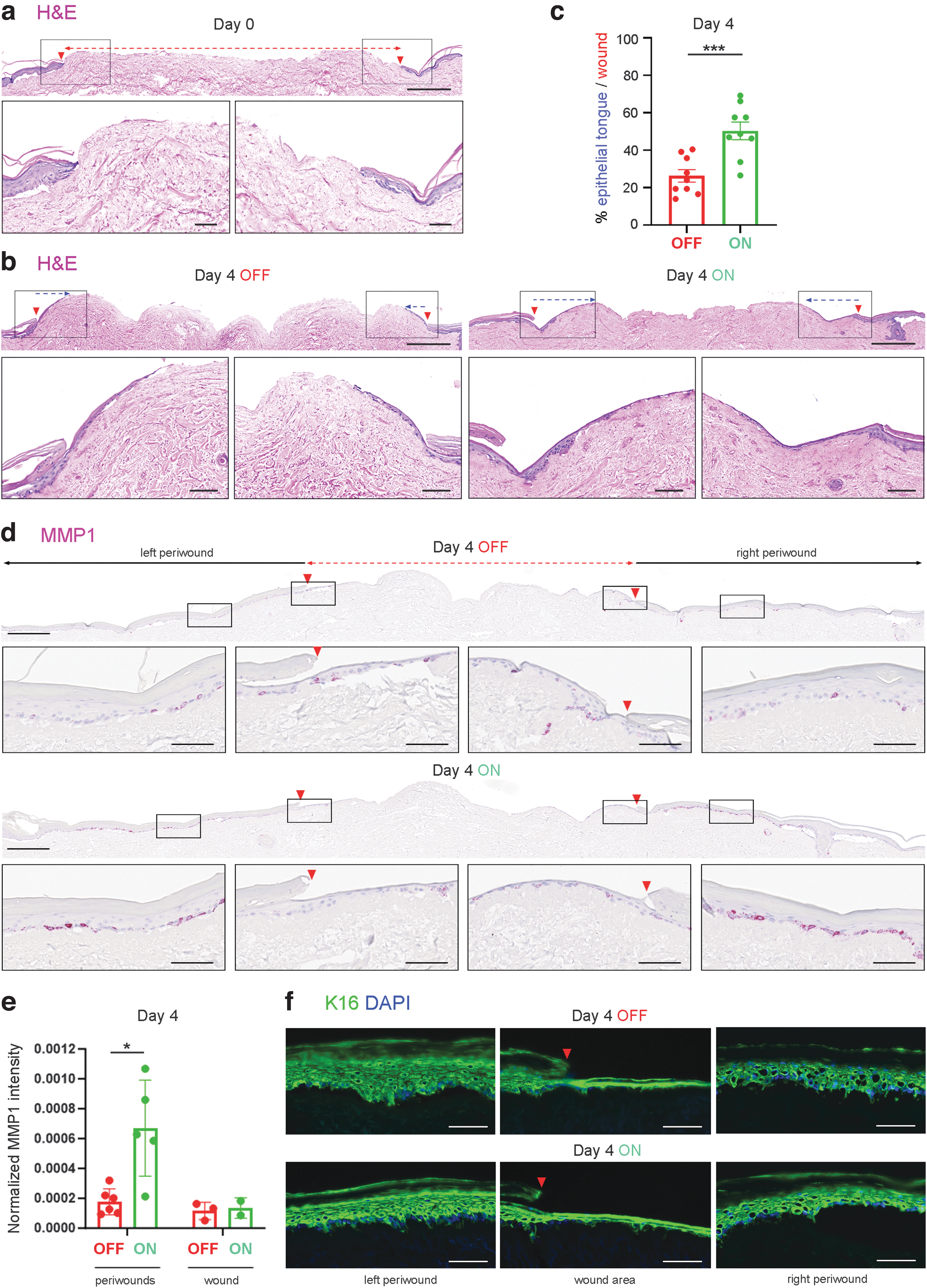
To further investigate the mechanism of EST promoting cell migration, the distribution of matrix metalloproteinases MMP1 and MMP3 was assessed as they are known to facilitate cell migration by remodeling dermis and cell attachment. 44,45 IHC for MMPs showed weak signals (data not shown), so RNA-scope ISH was used to detect MMP expression at the transcript level. After 4 days of EST, MMP1 expression was significantly increased in basal keratinocytes located in the periwound area (mean 0.00067, n = 5) compared to untreated skin (mean 0.00018, n = 6) (p < 0.05) (Fig. 3d, e and Supplementary Table S3). Interestingly, increased MMP1 expression upon EST exposure was not observed in epithelial tongues, and at a later stage of epidermal development on day 7 (Fig. 3d, e and Supplementary Fig. 2b). MMP3 expression was not increased (Supplementary Fig. 2c).
Keratin 16 (K16), also known to be expressed in activated keratinocytes upon wounding, 46 showed stronger fluorescent signals in both periwounds and wounds of 4-day treated DED-HSEs compared to untreated wounds (Fig. 3f) (not quantified), supporting the finding of enhanced cell migration upon ES exposure.
Microcurrent EST improves the quantity and quality of re-epithelialization
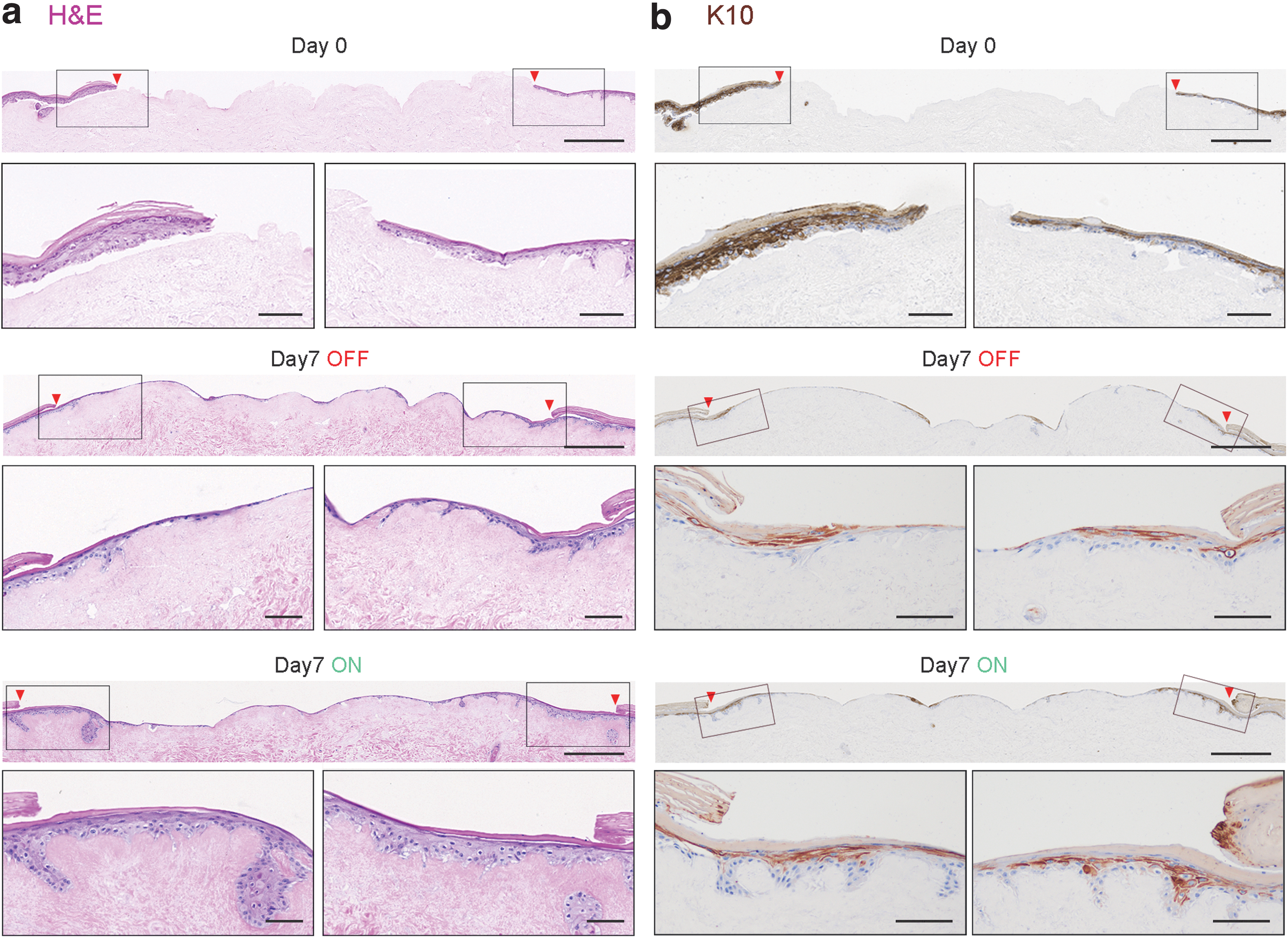
Examination of H&E staining of ES-treated DED-HSEs on day 7 revealed a thicker neo-epidermis compared to untreated controls, consisting of a greater number of cells with a more advanced epidermal stratification and cornified layer (Fig. 4a and Supplementary Fig. S3a). This observation was confirmed by IHC staining of K10, a differentiation marker expressed in postmitotic keratinocytes localized in the suprabasal layers of the epidermis (Fig. 4b and Supplementary Fig. S3b). 47
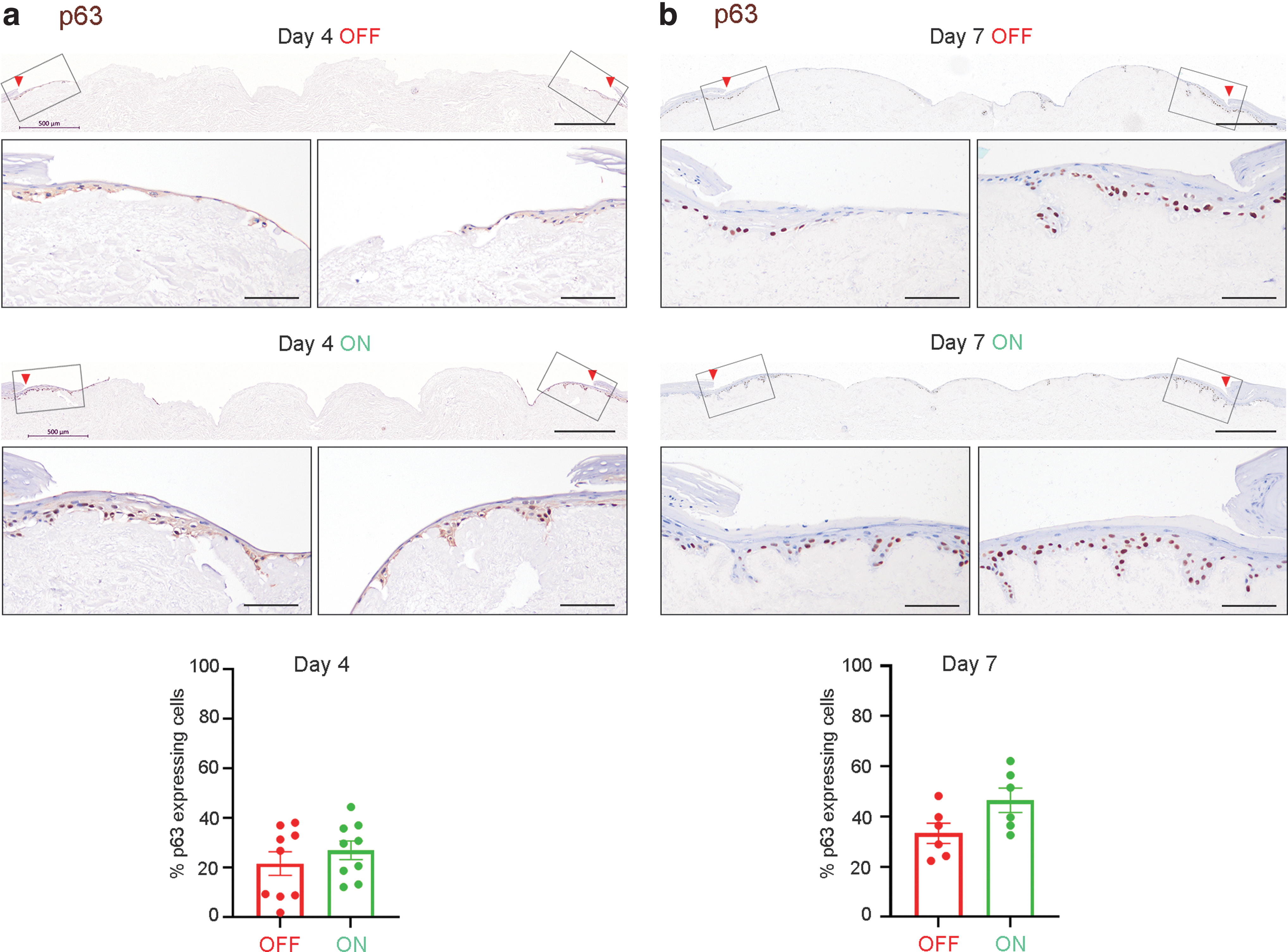
To further investigate the greater cellular growth of the newly formed epidermis in ES-treated wounds, IHC was used to assess the level of Ki-67, a proliferation marker expressed in mitotic cells, and p63, a well-characterized transcription factor involved in both epidermal proliferation and differentiation. 48,49 As expected, Ki-67 was mainly detected in keratinocytes localized in the basal layer of the epidermis, demonstrating maintenance of cell proliferation in the scaled-up DED-HSE wound model (Supplementary Fig. S4a, b). Although the total number of cells in the newly formed epidermis exposed to EST was higher than untreated skin, the percentage of Ki-67-expressing cells was similar to that of untreated samples on day 4 (mean 60.8% ± 5.7, n = 6 vs. mean 55.6% ± 4.4, n = 6) (Supplementary Fig. S4c and Supplementary Table S4).
On day 7, a similar trend was observed. The total number of cells in ES-treated epidermis was higher than untreated, while the percentage of Ki-67-expressing cells was similar in both conditions (mean 22.4% ± 6.6, n = 6 vs. mean 18.5% ± 2.1, n = 6) (Supplementary Fig. S4d). We noted that the proliferative rate, as assessed by the % of Ki67-positive cells, decreased from day 4 to day 7 by almost three-fold, showing that the new epidermis was progressing toward a differentiated state by day 7.
IHC images of p63 showed nuclear staining in basal keratinocytes, confirming proper epidermal development in the in vitro DED-HSE model. The percentage of p63-expressing cells in ES-treated wounds appeared to be higher (mean 26.9% ± 3.7, n = 9) compared to untreated DED-HSEs (mean 21.6% ± 4.7, n = 9) on day 4 (Fig. 5 and Supplementary Table S5). The same trend was observed on day 7 where the percentage of p63-positve cells was higher in ES-treated wounds (mean 46.6% ± 4.8, n = 6) compared to untreated controls (mean 33.3% ± 4.0, n = 6), but the difference was not statistically significant (p = 0.0645) (Fig. 5, Supplementary Fig. S4e, f and Supplementary Table S5).
K14, a structural protein expressed in keratinocytes localized in the basal and epibasal layers of the epidermis, was also examined by IHC. 50 Although the level of K14 expression was similar in all epidermis layers of DED-HSEs, larger proliferative epidermis was observed in wounds treated with ES after 4 and 7 days, as previously described (Fig. 6a, b and Supplementary Fig. S5). Longer and thicker stratum corneum was also observed in samples exposed to EST for 7 days, confirming faster differentiation and maturation of the epidermis, coherent with K10 staining.
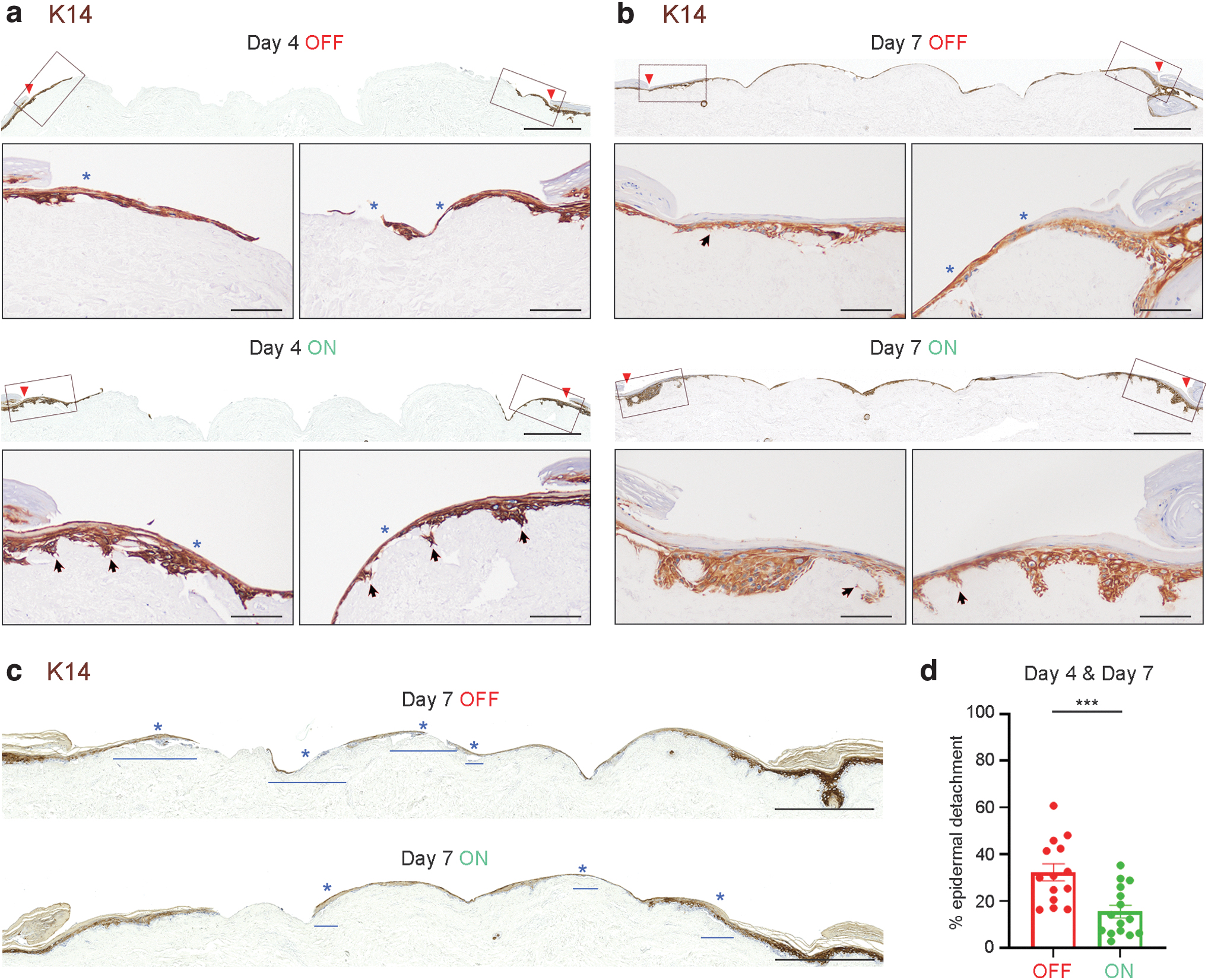
Microcurrent EST increases attachment to the wound bed
Immunohistochemical staining for K14 also highlighted a phenomenon that was observed throughout the histological work: the newly formed epidermis appeared to be more strongly attached to the dermal matrix of DED-HSEs when the ES device had been switched ON. To validate this observation, we measured epidermal detachment and normalized this to the total length of newly formed epidermis on each wound on days 4 and 7 (Fig. 6c). This revealed that the percentage of epidermal detachment was significantly higher in untreated wounds (mean 32.3% ± 3.6, n = 15) compared to treated wounds (mean 15.5% ± 2.7, n = 15) (p < 0.001) (Fig. 6d and Supplementary Table S6). Changes in cell shape and deeper epidermal projections anchoring onto the dermis were also more pronounced in ES-treated DED-HSE wounds (Fig. 6a, b), suggesting that EST may enhance cell-cell and cell-basement membrane attachment (Fig. 6 and Supplementary Fig. S5).
To explore this observation, we selected several biomarkers that are expressed in the dermal-epidermal junction and that contribute to cell-cell and cell-matrix adhesion, and cell migration and proliferation during wound healing. 51 Integrins α3β1 and α6β4, and LAM V, as well as collagens (COLIV, VII, and XVII) deposited in the basement membrane were detected by IF in all DED-HSEs; however, there was no obvious difference between ES-treated and untreated samples (Supplementary Fig. S6). Since most of these proteins were already present on the surface of the DEDs, it was not possible to differentiate any newly synthetized proteins activated by EST and quantify any change in expression using IHC.
DISCUSSION
Several in vivo and in vitro studies have shown that natural endogenous electrical potentials arise upon injury and promote normal wound healing. 6,8 These electrical signals become disrupted or abolished in patients with chronic wounds, providing a rationale for applications of exogenous ES to rescue this physiological defect and resolve aberrant wound healing. 11 –15
Although a growing number of case studies report beneficial effects of EST in a wide range of hard-to heal indications, the adoption of EST is not widespread. 12 This could be attributed to prevailing problems with the design of devices (less-than optimal ES, large or heavy devices, and not user-friendly), as well as side-effects that patients may experience while using EST such as sensory or motor discomfort, skin irritation, and aggravation of pain. 24,25 A coherent scientific rationale and unequivocal demonstration of benefits of EST to wound healing is needed to convince clinicians, academics, and industry to adopt the technology.
In our study, we aimed to investigate the effect of a new, easy-to-use, and painless EST clinical device on wound healing. Our approach was to use an in vitro human skin wound model that allowed us to (1) place the electrodes of the clinical device near the wounds, (2) assess the efficiency of microcurrents released by this device on wound re-epithelialization, and (3) investigate biological mechanisms that may be triggered by this EST.
Several investigations of ES on skin wound healing have been reported using diverse in vitro models. Two-dimensional cell scratch assays have shown directional migration of keratinocytes when exposed to pulsed direct current EFs, random migration of fibroblasts upon short ES, and macrophage migration upon oscillatory EFs. 30 –32 Compact bioelectronic platforms with unidirectional ES demonstrated improved motility of diabetic patients' keratinocytes. 52
Alternating current, direct current, or degenerate waves tested on keloid fibroblasts were shown to downregulate collagen I expression. 53 Enhanced cell migration by activation of Src and inositol–phospholipid signaling was reported from monolayer epithelial cultures exposed to EF. 54 Electrostatic chambers delivering electric signals in cells on planar culture or tissues in 3D gels also showed great potential for observing cellular responses to ES. 55 DED-HSE and skin explants have additionally been used to test ES as they recapitulate differentiated features of epidermal layers that 2D cell cultures do not. 56,57 While these in vitro models provide valuable insights on the effects of ES, none of them is suitable to directly test clinical devices that continuously deliver microcurrent EST over a period of 7 days.
We choose to scale up a well-established DED-HSE wound model previously used to evaluate topical formulations, therapeutics, and dressings. 34,37,38 In contrast to ex vivo skin explants, the DED-HSE constructs continue to grow and remain metabolically viable for up to 3 months. Importantly, they retain skin-like morphology, physiology, biochemistry, biophysics, and native skin-specific functionality. 35,58 This is the first time that the DED-HSE has been successfully redesigned in a larger format, to place adhesive electrodes on either side of the wounds and test EST in a similar way to the use of a EST device in patients. The experimental setup was optimized to restrict the flow of current to the wounded skin and keep the ES device dry, sterile, and functioning for up to 7 days at physiological temperature.
Using this optimized skin wound model, we evaluated the effect of an automated microcurrent EST, which has been previously shown to impart positive clinical responses across a wide range of hard-to heal indications. 26 –28,39
Using MTT staining, we first observed consistent cell viability in all ES-treated DED-HSEs, with no obvious difference with untreated controls. This result showed that neither electrical signals nor chemicals released by the electrodes adversely affected the metabolic activity of epidermal cells, confirming that the EST device is biocompatible and safe for patient's skin.
MTT staining also showed that after 4 days of treatment with this EST device, injured in vitro skin tissue healed significantly faster compared to untreated wounds. The acceleration of wound closure upon exposure to EST was in concert with longer epithelial tongues emerging from the wound edges, as revealed by histological images.
Investigations into the mechanisms of action accounting for the acceleration of re-epithelialization showed that MMP1, which encodes a collagenase that degrades collagen 1 and 3 to facilitate keratinocyte migration, was significantly upregulated in the basal layer of the epidermis surrounding the wounds (periwound) when exposed to EST. 59 –61 Interestingly, an increased MMP1 expression in cells behind the leading edges of wound regions was observed only at the early phase of re-epithelialization when cells were mainly migrating toward the wound bed.
Upon ES, higher production of MMP1 may enhance digestion of underlying matrix, facilitating cell detachment and migration to fill the opened wounds. When keratinocytes reached the wound, stronger attachment to the matrix is needed for cell proliferation, explaining decreased MMP1 expression. This is in line with previous reports showing that MMP1 expression was confined to specific locations in the wound, and rapidly turned off once re-epithelialization was complete and the basement membrane began to reform. 59 –61
Keratin 16, known to be expressed in migrating keratinocytes upon wounding, also showed increased expression in ES-treated DED-HSEs compared to untreated samples. These results are aligned with a previous study showing that decreased keratinocyte migration correlated with downregulation of MMP1 and K16 expression in a high-glucose environment. 62,63 We could therefore hypothesize that poor wound healing in patients with hyperglycemia may benefit from EST by stimulating MMP1/K16 expression.
When EST-treated keratinocytes reached the DED matrix, MMP1 expression was decreased to similar intensity as in controls, slowing down cell migration and initiating epidermal growth. Although Ki-67 staining did not show significant increase in cell division, IHC analyses revealed advanced epidermal proliferation (p63) and differentiation (K10 and K14) in ES-treated wounds compared to untreated controls, suggesting that microcurrent EST stimulated epidermal growth. Although Ki-67 staining did not show significant increase in cell division, IHC analyses revealed advanced epidermal proliferation (p63) and differentiation (K10 and K14) in ES-treated wounds compared to untreated controls, suggesting that microcurrent EST stimulated epidermal growth.
While analyzing the histological images, we observed a striking phenomenon found throughout all experiments. When fixed or cryopreserved DED-HSEs were processed for IHC work, we noted firmer attachment and deeper integration of the epidermis onto the acellular dermal matrix exposed to EST, suggesting that cell-cell and cell-basement membrane attachment may be enhanced by EST. The more robust epidermal attachment seen in vitro would undoubtedly be a beneficial outcome in the clinical treatment of chronic wounds.
This finding is in line with previous reports demonstrating that during electrotaxis, cells actively extend membrane protrusions, as well as overexpress and relocate membrane receptors and cell junction proteins. 64,65 Enhanced attachment of keratinocytes to the underlying DED matrix seen in ES-treated wounds may be attributed to an increased expression of p63, a transcription factor known to regulate genes involved in cell adhesion and survival in epithelial cells. 66 Other studies have shown that genes involved in cell adhesion, tissue remodeling, and wound healing process were differently expressed in human dermal fibroblasts exposed to ES, enhancing cell growth and adhesion on heparin-bioactivated conductive scaffold. 67
Based on these findings and the comprehensive review of Pastar et al., different biomarkers essential in cell-cell and cell-matrix adhesion during re-epithelialization in wound healing were tested by IF. 68 Immunofluorescent images confirmed different morphology and orientation of basal keratinocytes toward the dermis when exposed to EST. Unfortunately, there were no obvious difference in the expression of integrins (α3, β1, α6, and β4), basement membrane collagens (COLIV, VII, and XVII), and LAM V when compared to untreated wounds.
As previously described, besides matrix remodeling processed by proteases, attachment of keratinocytes onto the extracellular matrix of the wound and to their neighboring cells (controlled by focal contacts and hemidesmosomes) is also central to mechanisms that regulate collective cell migration and cell growth. 44,45,51 Further investigation using transcriptomics (RNA sequencing) may be used to identify genes that are responsive to ES and reveal deeper biological mechanisms underlying this form of EST for wound healing.
In summary, we have developed a large-format in vitro 3D human wound model, adapted to assess a clinically used EST device, enable preclinical evaluation, and support on-going mechanism of action studies. Using this optimized model, we demonstrate at the molecular, cellular, and tissue levels that the preprogrammed sequences of LVBMPC accelerate re-epithelialization by promoting keratinocyte migration on denuded basement membrane, and subsequent processes of cell proliferation and differentiation. We also observed enhanced attachment of the neostratified epidermis to the dermis when exposed to EST.
These in vitro data provide support FOR anecdotal and clinical observations of improved healing using this device. 26 –28,39 Our study reinforces the notion that EST is an undervalued treatment modality with substantial clinical potential to jumpstart the wound healing process in patients with hard-to-heal wounds and to expedite their recovery.
KEY FINDINGS
We present a modified 3D in vitro human skin wound model that facilitates testing of ES medical devices using existing and/or novel clinical protocols.
This proof-of-concept study demonstrates the advantages of using 3D in vitro human skin wound models for preclinical evaluations.
This study presents experimental evidence that EST accelerates re-epithelialization by enhancing keratinocyte migration and attachment to wound bed.
This in vitro study corroborates the clinical observations that indicate EST devices are effective and beneficial for the management of patient with chronic diabetic foot ulcers, venous leg ulcers, and pressure ulcers.
Footnotes
ACKNOWLEDGMENTS
The authors would like to thank the Asian Skin Biobank (ASB) at the Skin Research Institute of Singapore (SRIS), especially Alimat A., Lee J., and Yap A., for generating and providing primary epidermal keratinocytes, as well as Assoc Prof Tan BK. from the Department of Plastic, Reconstructive and Aesthetic Surgery, Singapore General Hospital, Dr Chua A. from Singapore General Hospital, Prof Chan Jerry KY. from KK Women's and Children's Hospital, and the SingHealth Tissues Repository's team for providing waste surgical skin tissues to the ASB for developing in vitro skin models (IRB 2020-209).
The authors also thank Grometto L. for editing the movies, and to Gan JY. for creating the graphical abstract. This study was funded by Accel-Heal Technologies Limited (UK), the manufacturer of the electrical stimulation devices used in this study. We also thank Dr Robin Martin (Robin Martin PhD Scientific Consulting) for his advice and guidance. The development of the DED-HSE platform was funded by Singapore's Agency for Science, Technology & Research (A*STAR) under the Industry Alignment Fund Pre-Positioning Fund (IAF-PP HBMS) H1701a0004 and H1701a00B9.
AUTHOR CONFIRMATION
As submitting author, I attest that I have written consent from all authors to submit the article, and that all authors accept complete responsibility for the contents of the article. This article has not been previously published. It is not currently under consideration elsewhere. The work reported in this study will not be submitted for publication elsewhere until a final decision has been made as to its acceptability by Advances in Wound Care. Finally, I attest that the article is independent and original work.
AUTHORs' CONTRIBUTION
P.L.K.L.: Conceptualization (supporting); data curation (equal); formal analysis (equal); investigation (lead); methodology (equal); project administration (equal); supervision (supporting); validation (equal); visualization (equal); writing–original draft (supporting); and writing–review and editing (supporting); Y.B.: Data curation (equal); formal analysis (equal); investigation (equal); project administration (supporting); validation (equal); visualization (equal); writing–original draft (supporting); and writing–review and editing (supporting); G.G.: Data curation (supporting); formal analysis (supporting); methodology (supporting); and visualization (supporting); T.K.C.: Data curation (supporting); formal analysis (supporting); methodology (supporting); and visualization (supporting); Y.Z.N.: Conceptualization (supporting); funding acquisition (supporting); project administration (supporting); and writing – review and editing (supporting); D.L.: Investigation (equal); methodology (equal); validation (equal); visualization (equal); and writing–review and editing (supporting); D.L.: Conceptualization (equal); funding acquisition (supporting); resources (supporting); supervision (supporting); and writing–review and editing (supporting); C.B.: Conceptualization (equal); formal analysis (supporting); funding acquisition (lead); project administration (lead); resources (lead); supervision (lead); visualization (lead); writing–original draft (lead); and writing–review and editing (lead).
AUTHOR DISCLOSURE AND GHOSTWRITING
This study was funded by the manufacturer of the electrostimulation technology used in this study. The funder had no role in the study design, process, data generation, and analysis. No ghost writers were employed.
FUNDING STATEMENT
The study was funded by Accel-Heal Technologies Ltd, Hever, UK.
ABOUT THE AUTHORS
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Figure S5
Supplementary Figure S6
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
Supplementary Table S6
Supplementary Video S1
Supplementary Video S2
Abbreviations and Acronyms
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
